Abstract
Background
C-reactive protein is an acute phase response marker and, in an epidemiological context, a predictor of cardiovascular risk. 25-Hydroxy-vitamin D is the best indicator for vitamin D status, but it can be altered by the presence of acute phase response. Our aim was to evaluate the association between serum concentrations of 25-hydroxy-vitamin D and C-reactive protein to assist the interpretation of vitamin D status in a clinical context.
Methods
We evaluated retrospectively 5076 patients (n = 4087 women) assessed for 25-hydroxy-vitamin D and C-reactive protein simultaneously. Subjects were classified according to the origin as hospitalized patients (n = 410) and outpatients (n = 4666). Outpatients included patients from specialized (n = 3943) and primary (n = 723) care. Serum 25-hydroxy-vitamin D was determined by using liquid chromatography and serum C-reactive protein by using immunoturbidimetry.
Results
Concentrations of 25-hydroxy-vitamin D and C-reactive protein were significantly different between hospitalized subjects and outpatients but not for specialized and primary care settings. Serum concentrations of 25-hydroxy-vitamin D decreased as C-reactive protein increased. Hospitalized patients with C-reactive protein concentrations >30 mg/L showed a significant reduction of 25-hydroxy-vitamin D. In outpatients with C-reactive protein within the reference range (≤10 mg/L), C-reactive protein concentrations were not significantly different for serum 25-hydroxy-vitamin D concentrations >37.5 nmol/L.
Conclusions
Our data question the reliability and usefulness of assessing 25-hydroxy-vitamin D status as a biomarker of nutritional status in patients displaying acute phase response, especially at concentrations of C-reactive protein >30 mg/L. In addition, the present study shows that in subjects displaying C-reactive protein values within the reference range, serum concentrations of 25-hydroxy-vitamin D >37.5 nmol/L were not associated with lower concentrations of cardiovascular risk (as assessed by C-reactive protein concentrations).
Introduction
The acute phase response (APR) is a complex systemic reaction triggered against infection, tissue injury, neoplastic growth or immunological disorders, in order to maintain body homeostasis. During the APR, more than 200 plasma proteins are modulated, of which approximately 50% are involved in nutrient transport or regulation of nutrient status. 1
C-reactive protein (CRP) is a positive acute phase reactant and a biomarker for inflammation 1 that may be used as an early marker of infection (24–48 h) and remains elevated in the presence of a continued, chronic inflammatory process.1,2 In this context, CRP concentrations have been shown to be a strong predictor of future cardiovascular disease in apparently healthy persons,3,4 and relative cardiovascular risk categories have been assigned to CRP tertiles (low <1 mg/L; average, 1 to 3 mg/L; high >3–10 mg/L), 5 while values >10 mg/L are considered to be indicative of inflammation beyond ‘low-grade’. 2
25-Hydroxy-vitamin D (25-OH-D) constitutes the major circulating form of vitamin D in serum, and it is considered the best indicator of vitamin D status. 6 It has been shown that 25-OH-D may display anti-inflammatory effects possibly by modulating inflammatory cytokines, 8 and low vitamin D concentrations have been associated with common chronic diseases and an increased risk for micro and macrovascular events.6,7
APR might alter levels of micronutrient status markers, thus leading to a misestimation of nutrient deficiency. A point that should be taken into account in order to achieve effective nutritional interventions. 9 In this context, we evaluated the association between serum concentrations of 25-OH-D and CRP to assist the interpretation of vitamin D status in a clinical context.
Subjects and methods
We evaluated retrospectively a four-year period, during which a total of 20,465 patients were assessed for 25-OH-D status (North-West Health Area, Madrid, Spain). From this population, 8207 adults also had CRP values measured simultaneously. To avoid the potential use of prescribed and over-the-counter supplements, only blood samples corresponding to the first visit were considered (n = 5371), while patients with 25-OH-D concentrations >160 nmol/L (n = 75) were excluded as suspected of being supplemented. Additionally, patients with established acute or chronic kidney disease (n = 220) were also excluded. Thus, a total of 5076 patients (n = 4087 women) were finally included and classified according to the origin of the samples as hospitalized patients (n = 410; 229 women; median age 76 years [IQR 59–83]) and outpatients (n = 4666, 3858 women; median age 60 years [interquartile range, IQR 48–70]), who in turn were further divided into specialized (n = 3943) and primary health-care settings (n = 723). The study protocol was approved by the Research Ethics Committee of the Hospital Universitario Puerta de Hierro-Majadahonda (Madrid, Spain).
Analytical methods and cut-offs
Samples were processed according to routine preanalytical and quality-controlled analytical procedures. Serum 25-OH-D was determined by an ultra-fast liquid chromatography 10 and serum CRP was measured by using immunoturbidimetry using a wide-range assay (Advia 2400, Siemens Healthineers, Spain). Analytical performance, tested by using internal controls in every run and our participation in external quality programs (monthly for CRP and quarterly for 25-OH-D), was satisfactory. For 25-OH-D: The Vitamin D External Quality Assurance Survey (DEQAS) (Charing Cross Hospital, London, UK) and the Vitamin D Metabolites Quality Assurance Programme (Vit D QAP) (NIST; Gaithersburg, MD, USA). For CRP: The External Quality Programme from the Sociedad Española de Medicina del Laboratorio (SEQCML).
Although there is no consensus regarding the cut-offs for vitamin D deficiency, in clinical practice, deficiency is generally accepted for values <50 nmol/L (<20 nmol/L as severe, 20–37.5 nmol/L as moderate and 37.6–50 nmol/L as mild), while concentrations of 50.1–75 nmol/L were considered insufficient, 75.1–100 nmol/L adequate or sufficient and >100 nmol/L desirable. To evaluate the effect of APR, serum concentration of CRP were categorized as absence of APR (CRP<3 mg/L), low APR (CRP 10.1–30 mg/L), medium APR (30.1–80 mg/L) and severe APR (>80 mg/L). Additionally, within the reference range, CPR levels were further divided according to the cut-off points proposed for cardiovascular risk (CDV): low CDV risk (CRP <1 mg/L), average CDV risk (PCR 1–3 mg/L) and high CDV risk (PCR 3.1–10 mg/L).5
Statistics
Data distributions were expressed as medians and IQR. Differences of 25-OH-D according to CRP categories were evaluated using Kruskal-Wallis H-test; the post hoc pairwise multiple comparisons were evaluated by Conover test. A value of P < 0.05 was considered statistically significant. Furthermore, pairwise comparisons of subgroups were performed using U Mann-Whitney test for non-parametric variables. For this analysis, the level of significance was adjusted according to the Bonferroni correction for multiple comparisons. Calculations were performed using Medcalc for Windows, v.11.2.1.0 (MedCalc Software, Mariakerke, Belgium).
Results
For all samples evaluated (n = 5076), median concentrations of 25-OH-D and CRP were 47.4 nmol/L (IQR [30.1–64.4]) and 1.7 mg/L (IQR [0.4–6]), respectively. Patients were categorized according to the presence/absence of APR. A clear inverse relationship was observed between both analyte concentrations with significant decrements in 25-OH-D as the magnitude of the APR/inflammation (using CRP as surrogate) increased. Serum 25-OH-D concentrations corresponding to patients showing APR were significantly decreased as compared with patients without APR (25-OH-D: 49.3 nmol/L IQR [32.8–65.8]). The effect was statistically significant (P < 0.008, according to the Bonferroni correction) in low, medium and severe APR (25-OH-D concentrations: 38.9 nmol/L IQR [23.2–58.7]; 30.5 nmol/L IQR [17.7–51.4] and 26.1 nmol/L IQR [14.8–44.9], respectively). Furthermore, concentrations of 25-OH-D in medium and severe APR were significantly reduced as compared with those found in low APR (P < 0.008, according to the Bonferroni correction).
Given that there is a marked sex bias in the population collected (4087 women/989 men), we have performed a statistical analysis of separated female (Figure 1(a)) and male (Figure 1(b)) subgroups. The inverse association between serum concentrations of 25-OH-D and CRP observed for all patients was also observed in both subgroups.
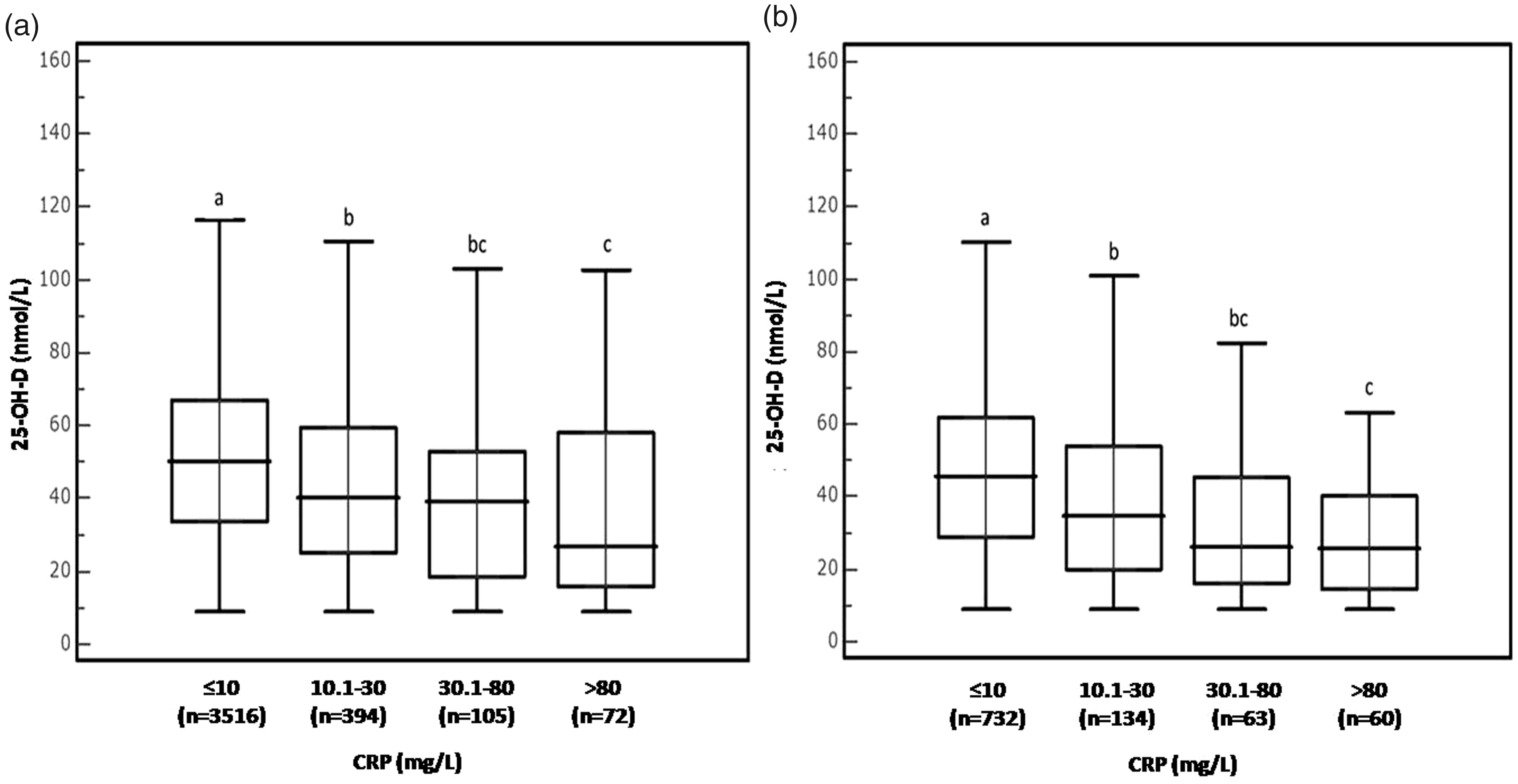
Distribution of serum 25-OH-D according to the presence/absence of acute phase response (APR) in women (a) and men (b). Absence of APR (CRP ≤10 mg/L), low APR (CRP 10.1–30 mg/L), medium APR (CRP 30.1–80 mg/L) and severe APR (CRP >80 mg/L). Boxes not sharing the same superscript are significantly different for the median values (Kruskal-Wallis test, P < 0.05).
Concentrations of both 25-OH-D and CRP were significantly different (P < 0.001) between hospitalized subjects (n = 410; median [IQR]: 26.6 nmol/L [16.1–44.7] for 25-OH-D and 24.3 mg/L [7.6–81.9] for CRP) and outpatients (n = 4666; median [IQR]: 48.9 nmol/L [32.3–65.7] for 25-OH-D and 1.4 mg/L [0.4–4.7] for CRP). Outpatients were separated into two groups according to the origin: Primary and specialized care and, as shown in Figure 2, both subgroups showed higher concentrations of 25-OH-D and lower concentrations of CRP than those observed in hospitalized subjects (P < 0.01). It is of note that 25-OH-D and CRP concentrations were not significantly different for primary (n = 723: median [IQR]: 49.4 nmol/L [33–67.9] and 1.4 mg/L [0.4–4.6], respectively) and specialized care (n = 3943; median [IQR]: 48.8 nmol/L [32.1–65.5] and 1.4 mg/L [0.4–4.8], respectively). Thus, further statistical comparisons were performed separately for outpatients (primary plus specialized care) and hospitalized subjects.
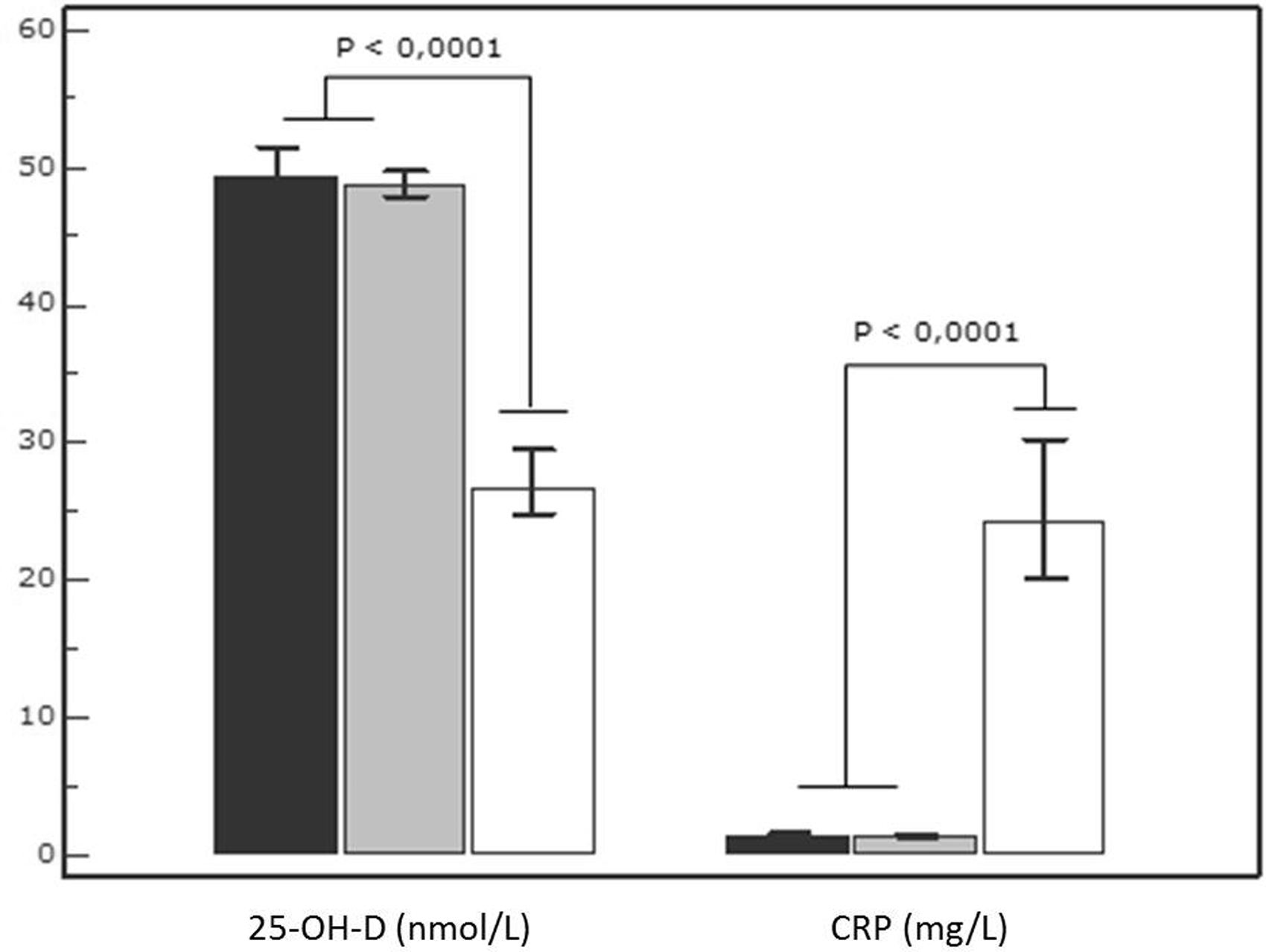
Distribution of serum 25-OH-D and CRP concentrations in outpatients and hospitalized patients. Patients were classified according to the origin of the samples. White bar denotes hospitalized patients (n = 410). Outpatients were divided into subjects from primary care (black bar; n = 723) and specialized care (grey bar; n = 3943). Bars indicate median and 95% CI. Differences were assessed using U Mann-Whitney test. For this analysis, the level of significance was adjusted to P < 0.016 according to the Bonferroni correction for multiple comparisons.
Hospitalized patients
For the whole group (n = 410), only 23 patients (5.6%) displayed a sufficient vitamin D status (25-OH-D > 75 nmol/L). In hospitalized patients, the concentrations of 25-OH-D also decreased as CRP increased. Concentrations of 25-OH-D (median, [IQR]) in the absence of APR and for low, medium and severe APR are shown in Figure 3; median concentration of 25-OH-D fell significantly (25–30%) at CRP concentrations >30 mg/L as compared with the absence APR group (P < 0.05). Consequently, the vitamin D deficiency increased according to the studied intervals of CRP.
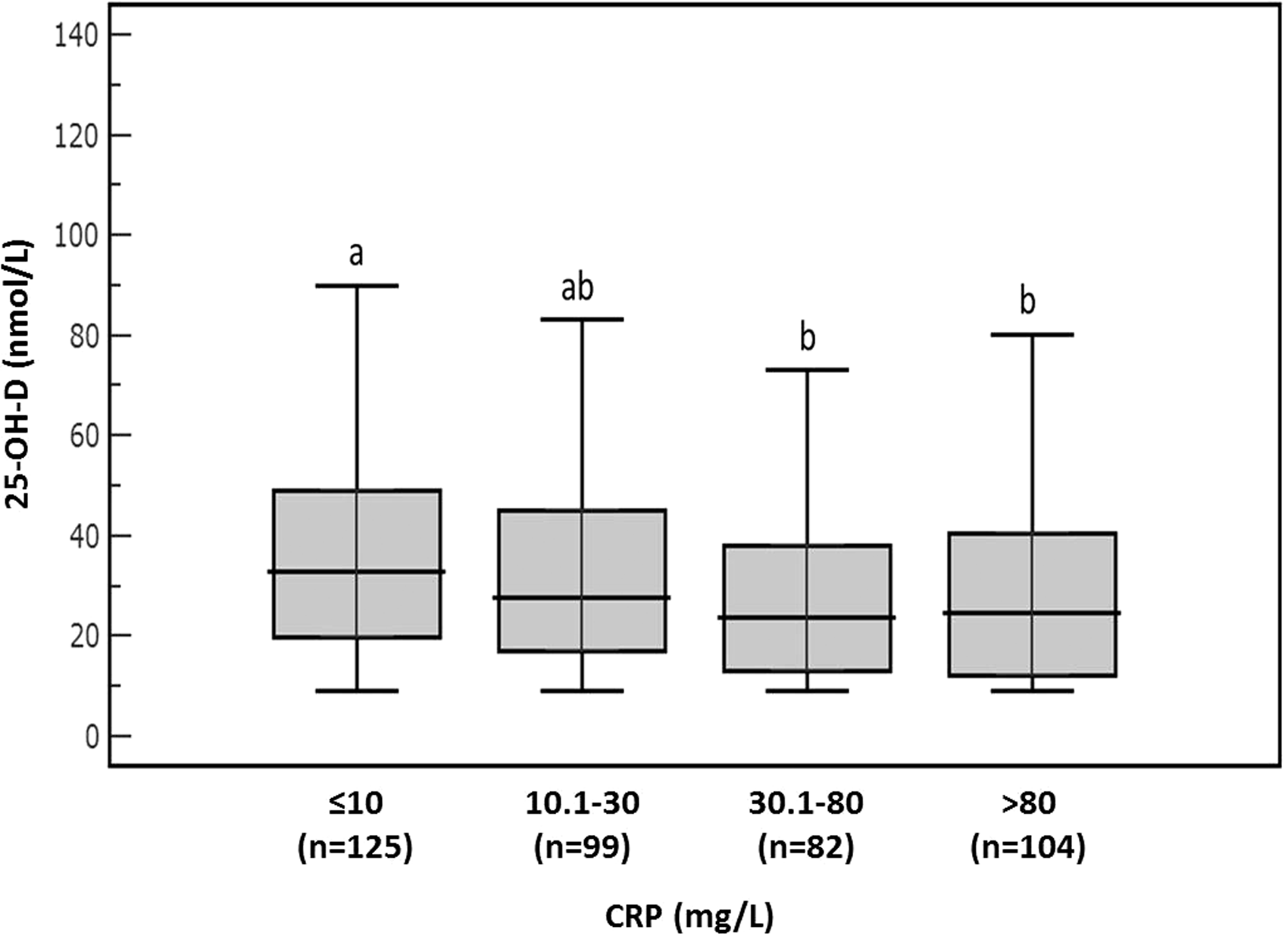
Distribution of serum 25-OH-D in hospitalized patients (n = 410) according to CRP concentrations. The absence of APR (CRP ≤10 mg/L), low APR (CRP 10.1–30 mg/L), medium APR (CRP 30.1–80 mg/L) and severe APR (CRP >80 mg/L). Boxes not sharing the same superscript are significantly different for the median values (Kruskal-Wallis test, P < 0.05).
Outpatients
For the whole group (n = 4666), 747 patients (16%) displayed a sufficient vitamin D status (25-OH-D > 75 nmol/L). As in the case of hospitalized patients, concentrations of 25-OH-D significantly decreased as the CRP increased (Figure 4(a)). After excluding subjects with inflammation beyond ‘low grade’ (CRP >10 mg/L, n = 543), CRP values were significantly different between groups with severe and moderate deficiency of 25-OH-D versus those with mild deficiency, insufficiency, adequate and desirable levels (Figure 4(b)). Interestingly, median values of CRP were not significantly different for serum 25-OH-D concentrations >37.5 nmol/L, suggesting that the cardiovascular risk, as assessed by CRP concentrations, was not modified by increasing serum 25-OH-D concentrations.
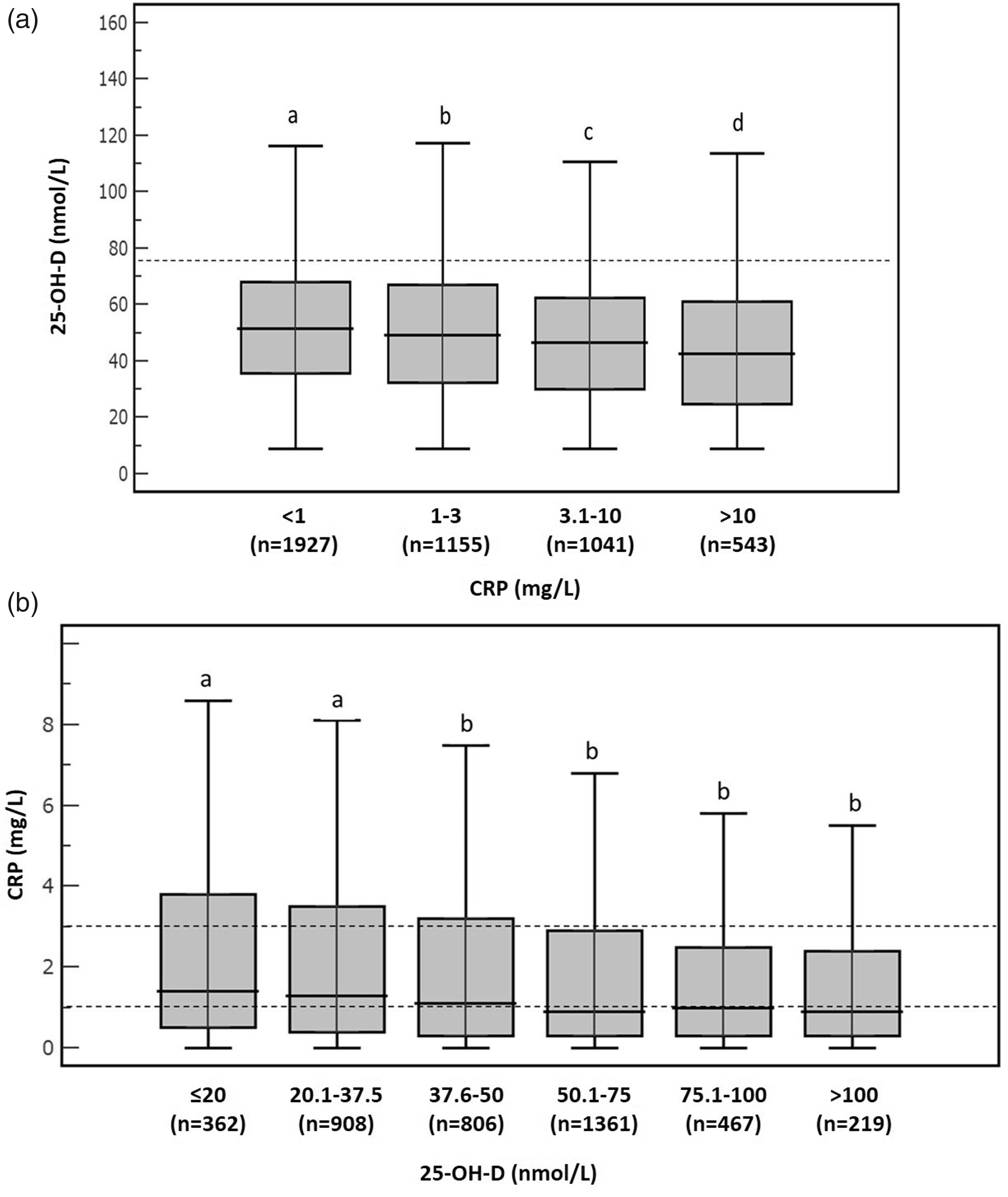
(a) Serum concentration of 25-OH-D in outpatients according to the following CRP categories: Inflammation beyond ‘low grade’ (CRP >10 mg/L), high cardiovascular risk (CRP 3.1–10 mg/L), average cardiovascular risk (CRP 1.0 – 3.0 mg/L) and low cardiovascular risk (CRP <1.0 mg/L). Boxes not sharing the same superscript are significantly different for the median values (Kruskal-Wallis test, P < 0.05). (b) Concentration of serum C-reactive protein according to 25-OH-D serum status in outpatients group excluding those showing ‘inflammation beyond low grade’ (CRP > 10 mg/L). Boxes not sharing the same superscript are significantly different for median values (Kruskal-Wallis test, P < 0.05).
Discussion
It has been suggested that it may be difficult to distinguish between changes in nutrients biomarkers due to modification of nutritional status or to an acute phase-related response, 1 and thus interpretation of plasma micronutrients should be made only with knowledge of the degree of inflammatory response. 11 In the present study, serum concentrations of 25-OH-D and CRP displayed an inverse association, with a significant decrease in serum 25-OH-D at serum CRP concentrations considered to be indicative of inflammation beyond ‘low-grade’. It is of note that patients showing medium or severe APR (CRP concentrations 30.1–80 and >80 mg/L, respectively) displayed a marked decrease (approx. 28%) in serum 25-OH-D concentrations ranging from moderate (20–37.5 nmol/L) or severe (<20 nmol/L) deficiency levels. These findings are consistent with previously observed significant reductions in plasma 25-OH-D at CRP concentrations >80 mg/L,11,12 a rapid decrease in serum 25-OH-D after acute inflammatory processes 13 and the lower concentrations of 25-OH-D in the presence of APR.9,14,15 Thus, the presence of inflammation beyond ‘low grade’ (CRP >10 mg/L) appears to be clearly associated with lower serum concentrations of 25-OH-D, perhaps related to alterations in liver function and enterohepatic cycle, protein status (specially albumin and Vitamin D binding protein concentrations), which raises uncertainties about its interpretation and questions its clinical utility in hospitalized patients with APR.
In an apparently healthy population, low 25-OH-D and high CRP concentrations may play a direct role in atherosclerosis and cardiovascular events by modulating cytokines and circulating adhesion molecules. 8 In this context, vitamin D supplementation might exert a beneficial anti-inflammatory effect, although intervention trials have shown inconsistent results. 4 Furthermore, recent studies suggest a biphasic relationship between vitamin D concentrations and cardiovascular conditions, since both low and high concentrations (<30 nmol/L and 100–150 nmol/L) have been associated with an increased risk of cardiovascular disease and proinflammatory effects.16,17 In this sense, despite our data suggesting that higher concentrations of 25-OH-D were associated with slightly, but significant, lower CRP concentrations among outpatients, no significant differences in cardiovascular risk (according to CRP categories) were observed for vitamin D concentrations >37.5 nmol/L.
This is a retrospective study including data from patients that were assessed for 25-OH-D status in our Hospital’s Health Area (which covers a population of about half-million people) during a four-year period. There is a marked sex bias in the population collected that is especially evident in non-hospitalized patients (outpatients). That is, the assessment of 25-OH-D concentrations is much more frequent in female subjects, probably in relation to the control of bone status during the peri- and menopausical period. To avoid this possible sex bias, we have performed a statistical analysis of subgroups (male and female) and we have found that the inverse association between 25-OH-D and CRP observed for the whole population studied is also found in both subgroups, thus indicating that the reported effect is not sex dependent. In this context, it should be pointed out that Clinical Practice Guidelines and recommendations from Scientific Societies do not established different 25-OH-D and CRP reference values for males and females.18,19
Finally, despite the relatively large number of subjects included, the present study displays several limitations including the retrospective design, the heterogeneity in the clinical status within and between groups and the bias of age and possibly season across the groups assessed that reflect the current scenario in our clinical practice. However, while these potential confounding factors were somehow intrinsically acknowledged during data collection, we aimed to assess the presence and magnitude of an APR/inflammation state as a ‘determinant’ of vitamin D status rather than trying to assess the effect of clinical conditions. From the present study, it is not possible to establish the mechanisms involved in the negative correlation observed between 25-OH-D concentrations and APR. Further research, including determination of other markers of vitamin D status, could help to clarify this point.
In conclusion, despite the retrospective approach of our study, our data question the reliability and usefulness of assessing 25-OH-D status as a biomarker of nutritional status in patients displaying APR, especially at concentrations of CRP >30 mg/L.
In addition, the present study shows that, at CRP values within the reference range, adequate and desirable serum concentrations of 25-OH-D (>75 and 100 nmol/L, respectively) were not associated with lower concentrations of CRP as a cardiovascular risk marker.
Interestingly, median values of CRP were not significantly different for serum 25-OH-D concentrations >37.5 nmol/L, suggesting that the cardiovascular risk, as assessed by CRP concentrations, was not modified by increasing serum 25-OH-D concentrations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by Ministerio de Economía y Competitividad (Spain) (AGL2015-68006-C2-2-R).
Ethical approval
The study protocol was approved by the Research Ethics Committee of the Hospital Universitario Puerta de Hierro-Majadahonda, Madrid, Spain (Acta n. 09.16).
Guarantor
EDN.
Contributorship
EHA, EDN, RAS and FGL conceived and designed experiments. EHA, CPB, IBN, BPS, EDN, RAS and FGL performed the analytical study. EHA, EDN, RAS, FGL analysed the data and wrote the paper. All authors reviewed and edited the article and approved the final version of the article.
