Abstract
Tumour necrosis factor inhibitor therapy has drastically changed the management of chronic inflammatory diseases. Some important drawbacks that can cause loss of response during treatment with these drugs are related to their large individual variability, the disease burden and the formation of antidrug antibodies that increase its clearance. Therapeutic drug monitoring of these drugs is not yet recommended by all scientific societies, and if so, only in patients with inflammatory symptoms. Proactive therapeutic drug monitoring represents a new strategy with many potential clinical benefits, including the prevention of immunogenicity, a reduction in the need for rescue therapy and greater durability of tumour necrosis factor inhibitor treatment. The review is based on a systematic search of the literature for controlled trials, systematic reviews, experimental studies, guideline papers and cohort studies addressing the best practice in tumour necrosis factor inhibitor therapeutic drug monitoring. Although there is ample evidence supporting the use of therapeutic drug monitoring in clinical practice to achieve better outcomes, some challenges have been detected. Many studies are focused on finding solutions for the lack of standardization of analytical methods to measure tumour necrosis factor inhibitor and antidrug antibodies concentrations. Other challenges are development of effective cost-saving proactive algorithms to identify optimal drug concentrations and the research on the role of antidrug antibodies, especially in the management and prevention of loss of response. Therapeutic drug monitoring of tumour necrosis factor inhibitor offers a rational approach to the optimization of the treatment of chronic inflammatory disease. Although prospective controlled trials yield little conclusive evidence of its benefits, there is growing acceptance of its value in clinical practice.
Keywords
Introduction
The therapeutic use of tumour necrosis factor inhibitors (TNFis) such as infliximab (IFX), adalimumab (AD), etanercept (ETA), golimumab (GOL) and certolizumab pegol (CER), which target tumour necrosis factor-α (TNFα), has dramatically changed the management of chronic inflammatory diseases (CIDs) like rheumatoid arthritis (RA), inflammatory bowel disease (IBD) and psoriasis. However, primary non-response or loss of response during treatment may occur in approximately 40% of the patients. 1 There are several reasons for this lack of response. An important drawback of TNFi is the formation of antidrug antibodies (ADAs), which bind to the epitope of the drug and form immune complexes 2 that increase its clearance, resulting in diminished TNFi trough concentrations and inferior clinical outcomes. Furthermore, lack of response can also be caused by non-immune-related factors such as high body mass index or high disease burden, which increases drug clearance. Other pharmacodynamics factors may also lead to non-response. 3
In relation to the formation of ADA, it is widely accepted that the structures of TNFi influence their immunogenicity. 4 IFX is a mouse/human chimera antibody that joins the variable regions of a mouse antibody to the constant region of human IgG1 whereas ADA and GOL are a fully human IgG1 antibody. Both drugs bind to TNFα and neutralize its activity. On the other hand, ETA is a dimeric fusion protein that joins the human p75 TNF receptor to the fragment crystalline domain of human IgG1 and CER is a humanized Fab′ monoclonal antibody fragment conjugated with polyethylene glycol. Because the amino acid sequence of mouse proteins is usually different from their human counterparts, mouse proteins like those of IFX tend to be more immunogenic than fully human antibodies while dimeric fusion proteins hardly develop immunogenicity.
In clinical practice, dose intensification of TNFi was often the first choice of treatment when loss of response occurred. Other options are adding immunosuppressive co-medication, changing to a different TNFi, changing to a different class of immunosuppressive drug or surgical intervention. There is also evidence that therapeutic drug monitoring (TDM) could be useful in these circumstances. TDM refers to the individualization of dosage by maintaining plasma or blood drug concentrations within a target range. 5 The characteristics of drugs which make TDM useful are a marked pharmacokinetic variability, therapeutic and adverse effects related to drug concentration, a narrow therapeutic index and difficulty of clinical effect measurement. TNFi meet these requirements. In fact, it has been demonstrated that IFX concentrations correlate well with clinical response in patients with IBD and RA 6 and individual treatments can be optimized by dose adjustment based on TNFi trough concentrations and the presence of ADA.
Despite these findings, proactive TDM of TNFi drugs is not yet recommended in the guidelines of all scientific societies. In fact, The National Institute for Health and Care Excellence of United Kingdom, 7 The American Gastroenterological Association 8 and The Gastroenterological Society of Australia 9 consider TDM to be useful mainly in case of lack of response. The purpose of this paper is to provide a literature review of available research evidence on TDM of TNFi.
Analytical methods for measuring TNF inhibitors and antidrug antibodies
There are several techniques for the measurement of TNFi and ADA, 10 such as fluid-phase radioimmunoassay (RIA), solid-phase enzyme-linked immunosorbent assay (ELISA), reporter gene assay (RGA), enzyme immunoassay (EIA) or homogeneous mobility shift assay (HMSA) as is shown in Table 1. However, the lack of a reference standard entails the need to establish cut-off concentrations and therapeutic intervals for each type of assay.
Summary of assays for measuring TNF inhibitor drug and antibody concentration.

ADA: antidrug antibodies; TNFi: tumour necrosis factor inhibitor.
aAll assays can measure infliximab, adalimumab and etanercept as well as their ADAs.10–13
Steenholdt et al. 10 compared four techniques to measure IFX concentrations (RIA, ELISA, RGA and EIA). All tests showed linear correlation (R2 = 0.97 to 0.99) although statistically significant differences between the assays were revealed when different IFX serum concentrations were tested on the same day, on different days and for different individuals. The maximum difference observed for each pair of assays was 1.55 μg/mL for RIA and RGA, 1.41 μg/mL for ELISA and RIA, and 0.48 μg/mL for ELISA and RGA (P < 0.05). This study concludes that ELISA, RIA, RGA and EIA are comparable in terms of basic analytical properties, but the lower sensitivity of RGA and the inability of bridging ELISA to detect antidrug antibodies of IgG4 isotype should be considered since they could be of clinical importance. 13 The assumption is that the systematic differences in IFX concentrations and ADAs to infliximab (anti-IFX) that have been observed among the techniques could be due to serum factors and/or matrix effects which affect each of the four assays differently. 10
Among the available techniques, the most commonly used technique to measure TNFi is ELISA. Schmitz et al. 11 evaluated the analytical performance, agreement and clinically significant differences of three commercially available IFX ELISA kits: Theradiag (Lisa Tracker IFX), Progenika (Promonitor IFX) and apDia (IFX ELISA) using an automated processing system. Although they all have acceptable analytical performance and could thus be used for TDM, the apDia assay had the best precision and agreement to target values. Therefore, it was concluded that the performance of an IFX ELISA should be assessed and cut-off values for TDM should be used with caution.
An important disadvantage associated to ELISA techniques and other immunoassays is that free IFX could cause a cross-reaction that would prevent optimal detection of anti-IFX. 11 Residual drug concentrations may interfere through competitive inhibition or by forming immune complexes. Studies assessing AD and ADAs to adalimumab (anti-AD) yielded similar findings and the measurement of anti-AD in the presence of (high) drug concentrations is difficult due to drug interference in most assays. In fact, anti-AD is only quantified by ELISA in the absence of detectable amounts of circulating AD. 12 Neither is this technique suitable for measuring anti-AD in complexes with the drug.
To address the issue of false negatives in the detection of ADA, Wang et al. 14 developed a non-radiolabeled HMSA to measure IFX and anti-IFX concentrations in serum samples. Unlike ELISA assays, this test is not subject to free IFX interference, and it includes an acid dissociation step. This procedure makes it possible to separate IFX–anti-IFX immune complexes before conducting the analysis. The technique can also be used to monitor antidrug antibody formation, even in presence of high serum drug concentrations. In relation to AD, the HMSA has also proved effective in overcoming many of the limitations encountered in the solid-phase ELISA and RIA methods, 15 which suggests that it could be useful in TDM.
Llinares-Tello et al. 16 proved as well the usefulness of acid pretreatment for anti-AD detection using Promonitor (ELISA). Although the drug inhibits anti-AD detection by this technique, and progressively higher concentrations of AD cause increasing signal inhibition, the assay showed that this pretreatment led to a significant response increase, particularly at lower free AD concentrations.
The acid pretreatment has also been used before RIA assays. Van Schouwenburg et al. 17 used pH-shift-anti-idiotype antigen binding testing to analyse samples from RA patients. After the dissociation, antibodies were captured using protein A sepharose and anti-ADs were detected using 125I labelled F(ab′)2. This technique allowed the detection of anti-AD in the presence of AD and offered insight on the immune response against AD.
Recent studies focus on more specific ELISA tests. Hock et al. 18 developed a specificity ELISA-based format capable of competitively inhibiting the in vitro binding of drug to solid-phase TNF for detecting specifically ADA in patients receiving either AD or IFX; furthermore, Kopylov et al. 19 implemented an alternative sandwich ELISA using antihuman lambda chain antibody for anti-IFX in the detection step which may be less amenable to IFX interference, taking advantage of the exclusively kappa chain composition of IFX.
The development of different analytical techniques with different calibrators allows ADA results to be expressed as arbitrary units towards purified polyclonal mouse antibodies or as a titration value. Gils et al. 20 and Van Stappen et al. 21 each developed a monoclonal antibody to quantify the concentrations of anti-AD and anti-IFX, respectively. This antibody could be used as a calibrator in ADA assays to determine the binding and neutralizing effect of the ADA and contribute to the harmonization of different methods of ADA response analysis. Moreover, these authors suggest that this may also facilitate correlations between the magnitude of ADA response and the clinical outcome of TNFi therapy. However, the main disadvantage of these new calibrators is that the affinity of ADA developed in TNFi-treated patients can be of a different nature to that developed in a mouse.
In view of the difficulty of determining ADAs, some authors have proposed that their persistence should be confirmed through repeated measurements before considering them for therapeutic decisions.
Therapeutic drug monitoring of TNF inhibitors
When TNFis were introduced in clinical practice, routine TDM was not recommended and empiric dosing became the norm. However, deeper understanding of the highly variable pharmacokinetic behaviour of these drugs, combined with growing emphasis on the need for further improvements in clinical outcomes, has led to increasing interest in the role of TDM in treatment optimization.
Although a high percentage of patients (70–90%) initially respond to the treatment, remission rates after induction are still low (20–50%) and, over time, patients are at risk of loss of response to the drug. 5 This inter-individual variability in response is likely to be influenced by the observed inter-individual variability in pharmacokinetics. 5 The pharmacokinetics of TNFi can be due to many factors, including gender, body weight, route of administration and systemic inflammation. In fact, elevated markers of inflammatory activity (C-reactive protein [CRP], fecal calprotectin [FCP], erythrocyte sedimentation rate) have been inversely associated with clinical response. According to Rosen et al., 22 high serum and mucosal TNF concentrations, and stool IFX loss through the inflamed colon could explain increased drug clearance and, thus, the requirement of higher doses for induction during the first weeks of treatment. Finally, increased proteolytic degradation by the reticuloendothelial system has also been suggested as a possible mechanism that may significantly increase IFX clearance. 22
On the other hand, it has also been demonstrated that patients with high serum albumin concentrations show lower IFX clearance and, thus, maintain higher IFX concentrations than patients with lower albumin. 23 However, the main factor that affects the pharmacokinetics and efficacy of TNFi is the development of ADA which accelerates TNFi drug clearance.5,22–26
Measurement of drug concentrations and ADA are typically performed only in patients with active inflammatory symptoms or during a potential immune-mediated reaction to TNFi. However, proactive TDM of TNFi concentrations with titration to a therapeutic window represents a new strategy with many potential clinical benefits, including prevention of immunogenicity, less need for rescue therapy and greater durability of TNFi treatment. 27
TNF inhibitor concentrations
The relationship between TNFi concentrations and clinical outcomes has been reported in the Trough Concentration Adapted Infliximab Treatment trial, 28 which included 483 IBD patients treated with IFX. IFX trough concentrations > 2.79 μg/mL (AUC = 0.681; 95% CI 0.632–0.731) and anti-IFX concentrations < 3.15 μ/mL (AUC = 0.632; 95% CI 0.589–0.676) were associated with a higher remission rate, defined as a CRP of ⩽0.5 mg/dL. In fact, in anti-IFX-negative samples stratified according to IFX concentration, the median CRP concentration was significantly lower when the serum IFX concentration was ⩾3 μg/mL compared with < 3 μg/mL (2.0 versus 6.0 mg/L respectively; p < 0.001).
In patients with Crohn's disease (CD) and perianal fistulas, higher concentrations to reach a good clinical response could be necessary. In fact, Yarur et al. 29 showed that in this group of patients IFX ⩾ 10.1 μg/mL correlated with higher rates of fistula healing, defined as the absence of drainage, and complete fistula closure and mucosal healing.
The results of the Study of Biologic and Immunomodulator Naive Patients in Crohn's Disease (SONIC) trial, 30 carried out in patients treated with IFX, showed that those with higher concentrations experienced better therapeutic outcomes than those with lower concentrations. Rates of corticosteroid-free clinical remission at week 30 were greater among patients with increased serum IFX trough concentrations. The findings were similar at week 46.
Vaughn et al. 27 conducted a retrospective observational study examining the use of proactive TDM and IFX titration to target concentration in patients with IBD in clinical remission. The main aim was to describe the clinical course of TDM in patients, and the results showed that the probability of remaining on IFX therapy over the study period was greater for patients who achieved trough concentrations > 5 μg/mL (HR:0.03; 95% CI 0.001–0.1; p < 0.0001).
Although most studies focus on establishing the lower limits of the target range for trough TNFi concentrations, patients could benefit from dose de-escalation and, therefore, upper limits should also be defined. Vande Casteele et al. 31 proposed TNFi dose de-escalation to reach a target range of 3–7 μg/mL in patients with clinical remission and high IFX trough concentrations. In fact, blind de-escalation of IFX therapy in IBD patients in clinical remission based on symptoms and CRP concentrations has been associated with a high risk of relapse. On the other hand, TDM-based management has been shown to be a more accurate strategy to avoid further relapse of the disease after de-escalation in patients on long-term IFX therapy. 32 It must be borne in mind that, together with the reduction in drug costs, this could also provide additional benefits, considering the potential increased risk of adverse events related to high concentrations. Recently, Van Steenbergen et al. 33 have shown that 65% of patients with IBD who de-escalated to AD 40 mg every three weeks remained in clinical remission for a median of 24 months. In 53% of patients, AD-related adverse events disappeared after dose de-escalation.
Likewise, in patients with RA, the IFX trough serum concentrations determined when inadequate response was detected correlated well with clinical activity. Moreover, a better response was observed in patients with higher trough serum concentrations, and the presence of anti-IFX correlated with disease activity using Disease Activity Score in 28 joints (DAS28) at baseline. 34 Patients treated with detectable concentrations of IFX had a better European League Against Rheumatism Response (EULAR) (p = 0.002), DAS28 score (p = 0.002) and Simple Disease Activity Index score (p = 0.001). On the other hand, Van den Bemt et al. 35 proved that the combination of disease activity, defined as good EULAR response, and IFX serum trough concentrations > 2.5 μg/mL, determined after six weeks of treatment, could be a fair predictor for the early identification of patients who will probably show good response after six months of therapy.
Although most of the studies have been performed using IFX, there is also evidence of the usefulness of TDM in patients treated with AD. A meta-analysis 36 enrolling 14 studies with 1941 patients with IBD shows the presence of anti-AD is associated with a higher risk of loss of clinical response to AD, whereas high AD trough concentrations are associated with greater clinical response rates in patients with CD.
For TNFi in patients with psoriasis, no therapeutic ranges have been established yet. Takahashi et al. 37 reported the association between clinical efficacy and IFX and AD trough concentrations during long-term treatment. In 32 AD-treated and 20 IFX-treated psoriasis patients, the authors demonstrated that TDM was positively associated with clinical response, and they established > 0.92 and > 7.84 μg/mL as optimal cut-off values of IFX and AD concentrations, respectively, to achieve ⩾ 75% reduction of the psoriasis area and severity index score (PASI).
Table 2 summarizes the main outcomes obtained in different studies addressing TDM of TNFi and the proposed cut-off or target ranges for serum trough concentrations of IFX, AD and ETA. The selection of studies is based on a systematic search of the literature in PubMed performed up to September 2017 combining various alternative search terms for ‘anti-TNF’, ‘infliximab’, ‘adalimumab’, ‘etanercept’ and ‘drug monitoring’. Studies were included according to the most relevant controlled trials and studies reporting results of clinical outcomes.
Summary of trials associating anti-TNF drug concentration with clinical outcomes.
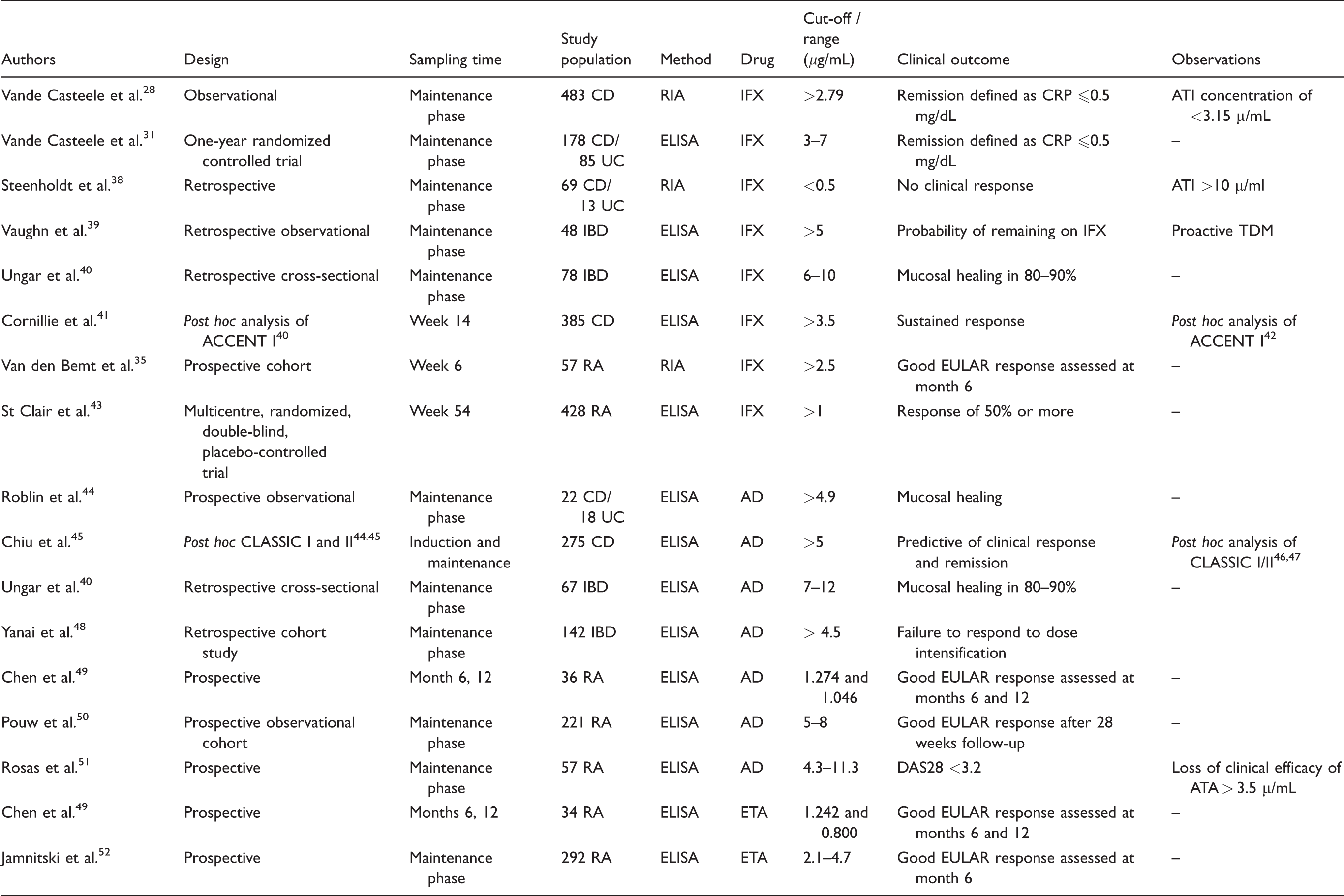
AD: adalimumab; ATA: antibodies to adalimumab; ATI: antibodies to infliximab; CD: Crohn's disease; CRP: C-reactive protein; DAS28: Disease Activity Score in 28 joints; ELISA: solid-phase Enzyme-Linked Immunosorbent Assay; ETA: Etanercept; EULAR: European League Against Rheumatism score; IBD: inflammatory bowel disease; IFX: infliximab; RA: rheumatoid arthritis; RIA: fluid-phase Radioimmunoassay; TDM: therapeutic drug monitoring; UC: ulcerative colitis.
The selection of studies is based on a systematic search of the literature in PubMed performed up to September 2017 combining various alternative search terms for ‘anti-TNF’, ‘infliximab’, ‘adalimumab’, ‘etanercept’ and ‘drug monitoring’. Studies were included according to the most relevant controlled trials and studies reporting results of clinical outcomes.
Even though some analytical methods have been developed for the measurement of GOL53,54 and CER55,56 and their ADAs, more evidence about TDM of these drugs is required to determine its usefulness and no consensus has been reached on a possible therapeutic level or cut-off associated with clinical response, remission or any other outcome.
TNF inhibitor antibodies
Antibodies to TNFis interfere with the binding of the drug to TNF and form immune complexes that hasten drug clearance via the reticuloendothelial system, 57 therefore, placing patients at risk for decreased clinical response.
Although it has been demonstrated that detectable TNFi serum concentrations could reveal circulating ADA,26,58 it is generally accepted that ADA measurement is not necessary in all patients treated with these drugs. Even so, since a notable percentage of gastroenterology and rheumatology patients (85%) with positive anti-IFX have IFX concentrations ⩽ 1 μg/mL, 59 this determination is currently carried out in the context of undetectable or low IFX concentrations. Under these circumstances, concentrations of anti-AD > 4 μg/mL or anti-IFX > 9 μg/mL could be used to identify patients who will probably not respond to an increased drug dosage. 48
The measurement of anti-IFX could also be of interest in the re-induction of IFX therapy after a long period of drug discontinuation in IBD. In this situation (length of discontinuation = 15 months), adequate drug concentrations and absence of anti-IFX early after restarting therapy have been shown to correlate well with both short- and long-term mucosal healing. 60 In fact, the presence of anti-IFX was identified as a negative predictive factor (HR:0.14; 95% CI 0.026–0.74; p = 0.021).
Testing for anti-IFX may also warn about the risk of potentially dangerous infusion-related reactions. The development of antibodies to the TNFis may lead to an effect or mechanism involving complement activation and production of anaphylatoxins, which may be the cause of severe side effects.61,62 In CD patients, the presence of anti-IFX > 8 μg/mL has been associated with a relative risk of 2.40 infusion-related reactions 60 (95% CI 1.65–3.66; p < 0.001).
In patients with RA treated with IFX, the presence of anti-IFX correlates to disease activity using DAS28 score at baseline. 35 Moreover, in a study carried out by Mazilu et al., 34 35 and 10% of the patients had undetectable and subtherapeutic IFX trough concentrations, respectively. Anti-IFX was detected in all of them and none showed EULAR response.
Chen et al. 49 assessed the presence of ADA at months 6 and 12 in 36 AR patients treated with AD and in 34 patients treated with ETA. The presence of anti-ADA was associated with lower EULAR response and lower drug concentrations compared with those without anti-ADA (p < 0.001). It is worth noting that, among the TNFi drugs, ETA has the lowest immunogenicity. In this study, none of the patients experiencing inadequate response had ADAs to etanercept (anti-ETA).
Takahashi et al. 37 also showed that the presence of anti-IFX and anti-AD contributes to loss of response to these drugs in patients with psoriasis. In their study, anti-AD and anti-IFX were detected in five out of 32 and in six out of 20 patients with loss of response determined by using PASI75 at weeks 24 and 48, respectively.
Another study based on 77 psoriasis patients receiving biologic therapy also proved that clinical severity scores were significantly higher in the antibody-positive patients. 63 ADAs were identified in the plasma of 25% of the IFX-treated patients and of 29.6% of the AD-treated patients, but in none of the patients in the ETA group. The mean PASI score at the time of blood sampling was significantly lower in ADA-negative patients (1.6 ± 1.6 versus 9.3 ± 11.2 and 2.4 ± 3.5 versus 12.5 ± 8.6 for IFX and AD, in the ADA negative versus ADA positive patients, respectively).
The suggested anti-IFX and anti-AD cut-off concentrations are shown in Table 2. Different approaches for immunogenicity reduction have been proposed. It has been observed that increasing the dose of IFX leads to reduced immunogenicity, suggesting that high drug concentrations might induce tolerance in these patients. However, an alternative explanation for these results could be that, in patients receiving high doses of the drug, the presence of residual drug concentrations might interfere with anti-IFX detection. 64
Another appropriate therapeutic approach to reduce immunogenicity to the TNFi agents and potentiate response to therapy is the use of combination therapies of these drugs with immunosuppressants. There is evidence that IFX concentrations are influenced by the concomitant use of methotrexate (MTX). In fact, the Combination Of Maintenance Methotrexate-Infliximab trial 58 conducted on patients with CD showed that those treated with MTX were less likely to develop anti-IFX than those receiving IFX only (4% versus 20%; p = 0.01). The SONIC trial yielded similar results for azathioprine (AZA) in CD patients. 30 In this study, anti-IFX were detected at week 30 in one out of the 116 patients (0.9%) receiving combination therapy and in 15 out of the 103 patients (14.6%) treated only with IFX.
On the other hand, co-treatment with immunomodulators in patients restarting after a prolonged period of drug discontinuation with low IFX concentrations has been identified as a positive predictive factor of mucosal healing (HR: 6.0; 95% CI 1.3–27; p = 0.019). 60 In view of this fact, and of the increased risk of developing severe infusion-related reactions after re-initiation (19.5% of the patients), 60 concomitant immunosuppressant therapy should always be considered in IFX retreatment cases.60,62
This strategy also seems to be useful in RA patients treated with AD. In fact, in a prospective observational cohort study including 221 RA patients, 50 median AD concentrations were higher in those taking MTX concomitantly than in those on monotherapy, 7.4 μg/mL (95% CI 5.3–10.6; p < 0.001) and 4.1 μg/mL (95% CI 1.3–7.7; p < 0.001), respectively. Similar results were obtained with ETA. 65 In a study carried out with 274 RA patients, those receiving combined treatment with MTX achieved clinical remission, evaluated by DAS28 index and radiographic non-progression (effect difference 22.05%, 95% CI 13.96–30.15; p < 0.0001).
Practical issues in therapeutic drug monitoring
In most of the studies, TNFi concentrations were determined after the loading dose and before an administration (trough concentration), once the steady state had been reached. However, there is little evidence regarding the best time for TDM. Clinical algorithms using TDM monitoring have been proposed as a tool in the management of loss of response to TNFi drugs28,37,51 to facilitate decision-making.
TDM could also be useful at the beginning of the TNFi treatment. In fact, most patients who develop anti-IFX do so within the first 12 months of IFX therapy. 66 This strategy is also supported by the findings that IFX trough concentrations at day 14 were lower in patients with acute severe UC compared to moderately severe UC, possibly due to a higher inflammatory burden and/or increased drug clearance 67 which therefore increases the probability of occurrence of anti-IFX. Furthermore, Vermeire et al. 68 showed that four weeks after starting treatment, patients with lower IFX concentrations were at increased risk for developing high titre of anti-IFX (>8 μ/mL), during the follow-up. Moreover, IFX concentrations below 4 μg/mL at week 4 had a positive predictive value of 81% to detect the ensuing development of high titre of anti-IFX.
The usefulness of the initial TDM was also demonstrated in a post hoc analysis 41 of the randomized controlled trial to assess the benefit of maintenance infliximab therapy in patients with active CD who respond to a single infusion of infliximab (ACCENT I study). 42 Cornillie et al. 41 proposed to carry out IFX TDM after 14 weeks in order to detect the need for early dose escalation, especially in patients with CD. In this group, trough IFX concentrations ⩾3.5 μg/mL and decrease CRP concentrations ⩾60% at week 14 have been associated with better response at follow-up week 54 (OR: 3.5; 95% CI 1.1–11.4) and (OR: 7.3; 95% CI 1.4–36.7), respectively.
On the other hand, there is no clear evidence in the literature concerning the development of proactive TDM algorithms to individualize therapy in patients in clinical remission. 69 After the initial dose optimization, continued concentration-based dosing has not been proved to be superior to clinically based dosing for maintaining remission after one year in patients with CD or UC. In fact, Vande Casteele et al. 31 found that the percentage of patients who achieved remission with clinically based and with concentration-based dosing was similar: 66 and 69% (p = 0.686), respectively. However, continued concentration-based dosing was associated with fewer flares during treatment. Moreover, in a retrospective analysis of 264 patients with IBD receiving IFX maintenance therapy, 70 drug titration to a target concentration (proactive monitoring) was associated with better clinical outcomes, including greater drug durability, less need for surgery and/or hospitalization, and lower risk of ADAs than TDM after patient loss of response (reactive monitoring).
A rather controversial issue is that of discontinuation of TNFi therapy in cases of deep remission. Papamichael and Vermeire 71 showed that after stopping TNFi treatment because of sustained remission under combination therapy with immunosuppressant, 85% of the patients had to restart treatment again. Retreatment is usually well tolerated and the success rate is high, but attention should be paid to the fact that patients have an increased risk of developing infusion reactions and delayed hypersensitivity during IFX reintroduction. Some authors have proposed the discontinuation of IFX or AD in IBD only in patients in remission with undetectable drug concentrations. Ben-Horin et al. 72 conducted a retrospective cohort study examining the duration of relapse-free survival after TNFi cessation, finding that relapse occurred in 16/20 (80%) of the patients who stopped TNFi while having measurable drug concentrations, against 9/28 (32%) of the patients with undetectable drug concentrations (OR: 8.4; 95% CI 2.2–32; p = 0.002). Moreover, the duration of relapse-free survival was significantly longer in patients with absence of the drug compared to those with detectable drug concentrations (p < 0.001, log-rank test). These authors suggested that the finding of an undetectable TNFi drug level in a patient with stable, long-term, deep remission may identify a subset of patients whose clinical remission is no longer dependent on TNFi treatment.
Another controversial issue is the suitability of switching between different TNFi drugs in the event of loss of response. Although it is common clinical practice, several studies have shown that patients who develop anti-IFX are more likely to develop anti-AD in comparison with TNFi naïve patients (33% versus 18%; p = 0.039). 73 Moreover, in this study, response to AD was limited even in switchers with no anti-IFX. On the other hand, patients with an immunogenic response against a first TNFi drug (IFX or AD) had a better clinical response to ETA compared to patients without ADA. 74 These findings raise the question of whether a second TNFi treatment should be offered to patients with RA when, in the absence of ADA, an initial treatment with a TNFi fails. In these patients with therapeutic drug concentrations and no clinical response, probably, it is not worth trying other TNFi because the lack of efficacy is associated with the mechanism of action. Therefore, a drug addressed to other therapeutic target should be selected.
Analogous results have been found in IBD patients treated with TNFi when they were switched to another therapy. 75 Yanai et al. 48 carried out a retrospective study in a cohort of 247 patients, demonstrating that patients with IFX trough concentrations above 3.8 μg/mL or AD trough concentrations above 4.5 μg/mL showed no clinical response to dose escalation or switch to another TNFi with 90% specificity. On the other hand, these groups of patients responded better to symptomatic therapy or to non-TNFi treatments (immunomodulators and/or different biological agents).
Finally, although postmarketing global safety experience with biosimilar IFX is continuously growing and ensures that switching is both safe and well tolerated, some physicians still advise caution when switching from an innovator IFX. There are still few studies comparing drug concentrations and effectiveness before and after the switch. A study carried out by Smits et al. 76 demonstrated that switching from innovator IFX to CT-P13 (biosimilar IFX) in a real-life cohort of 83 IBD patients did not have a significant impact on short-term clinical outcomes. Median IFX trough concentrations increased from 3.5 μg/mL (range 0–18) μg/mL to 4.2 μg/mL (range 0–21) μg/mL at week 16 (p = 0.010) with no significant changes in the medians of CRP and FCP concentrations during follow-up. Similar results have been obtained in the NOR-SWITCH trial, 77 a study where switching from originator IFX to biosimilar CT-P13 was compared with maintained treatment with originator IFX in IBD patients. Jørgensen et al. 77 demonstrated that switching from innovator to CT-P13 was not inferior to continued treatment with innovator IFX. Changes in CRP: (−0.07; 95% CI −0.17, 0.04) and (−0.04; 95% CI −0.18, 0.10) and FCP: (−0.08; 95% CI −0.27, 0.10) and (0.21; 95% CI −0.03, 0.44) in CD and UC, respectively. Comparable results were also seen for trough serum concentrations and presence of TNFi.
Cost-effectiveness of therapeutic drug monitoring
The use of biological drugs in many CID has led to a great improvement in clinical results, but it is also associated with an important impact on health care costs, driven mainly by medication costs. TDM is increasingly used to optimize TNFi therapy to improve its efficacy and security, but it could also be justified based on economic reasons. Actually, several studies suggest that the cost-effectiveness of TDM-based strategies is higher than that of empirical dose management.
One controlled trial 3 included 69 CD patients with secondary IFX failure. They were randomized to receive empirical dose intensification or TDM-based dose adjustment. At week 12, the clinical response was similar in both groups. However, in the intention-to-treat analysis, costs proved significantly lower (€3,140) in the TDM group, mainly due to discontinuation of ineffective treatments.
The study carried on by Vande Casteele et al. 31 included 263 CD or UC treated with IFX and with stable responses. During the optimization phase, doses had been adjusted to reach trough concentrations between 3 and 7 μg/mL. After this period, they were randomized to being subjected to a clinically based strategy or to a TDM-based strategy. Although one year after the optimization phase the percentage of patients who achieved clinical and biochemical remission was similar, the TDM group proved significantly cost saving (28%). This strategy was more cost effective as a result of dose reductions in patients with trough concentrations > 7 μg/mL.
There are currently no randomized controlled trials comparing both strategies in RA patients treated with AD or IFX. Nevertheless, two modelling approaches, both of them based on the Markov chain model, included this population78,79 and concluded that TDM was cost saving. Nonetheless, it should be noted that the use of this kind of modelling could yield biased results.
Despite the difficulties to extrapolate the results of these studies to other health care systems, given the high costs of TNFi drugs and the comparative lower costs associated to its determination, the use of TDM as a cost-effective strategy in clinical practice is gaining acceptance.
On the other hand, a recent review of clinical effectiveness and cost-effectiveness of TDM of TNFi using different ELISA kits versus standard care for CD carried out by the National Institute for Health Research 80 concludes that testing is not cost effective for IFX. However, the authors emphasize that the results should be viewed cautiously in view of the limited evidence. In addition, they recommend clinicians to be mindful of variation in performance of different assays and of the absence of standardized approaches to patient assessment and treatment algorithms.
Discussion
There is currently ample evidence supporting the use of TDM as a tool for therapy optimization in IBD, RA and psoriasis patients with the purpose of achieving better outcomes at lower costs. However, not all scientific societies recommend its use. In fact, scientific societies7–9 only recommend TDM in two scenarios: patients whose disease loses response to treatment and may need a higher dose of TNFi to try to recover clinical response, and patients whose disease responds to treatment with TNFi and may continue at the same level of treatment.
The implementation of TDM of TNFi is one of the great challenges in everyday clinical practice, largely due to the lack of standardization of analytical methods for the measurement of TNFi and ADA concentrations.10,20,21 ELISA is the most widespread analytical technique,11,18 despite the significant disadvantages it entails. Free TNFi concentrations could cause a cross-reaction that would prevent optimal detection of ADA,11,12,18 which is why it only quantifies the antibodies in the absence of detectable amounts of circulating drug. In order to avoid these false negative problems, Wang et al., 14 Llinares-Tello et al. 16 and Van Schouwenburg et al. 17 developed an acid pretreatment to allow the separation of TNFi–ADA immune complexes before conducting the analysis. Another handicap associated with ELISA is the inability to detect IgG4 anti-IFX, which could constitute up to 89% of IgG anti-IFX in patients treated with IFX over prolonged periods of time.12,13
The relationship between the IFX concentrations and clinical outcomes in IBD patients has been reported in several studies.27–29,38,39 Some authors go even further, recommending a target range of 3–7 μg/mL and proposing TNFi dose de-escalation in patients with clinical remission and high IFX trough concentrations. 31 Similar conclusions have been obtained from studies that included RA patients, and a target > 2.5 μg/mL has been proposed for this population. 35
Although different studies have been carried out in patients with IBD, RA and psoriasis treated with AD37,43,44,46,51 and rheumatologic patients treated with ETA47,50 establishing a therapeutic efficacy window, there is weaker evidence relative to the usefulness of TDM of these drugs.
The measurement of ADA in daily clinical practice is only considered in the context of undetectable or low drug concentrations,48,49 since their presence has been associated with disease activity.28,38,51 In this scenario, concomitant use of an immunosuppressant (MTX or AZA) enables immunogenicity reduction and may be a useful strategy to increase TNFi concentrations.30,58,69,81
Although early optimization of IFX during or immediately after the induction phase seems to improve clinical remission rates, especially in CD patients,41,68 most of the accepted algorithms based on TNFi and ADA trough level measurements have been developed for loss of response management.28,53 There is evidence for determining drug concentrations at week 1441,81 and during the maintenance phase.27,39,53 However, in patients with symptoms suggestive of lack of response, TDM of IFX and anti-IFX would be advisable during the induction phase41,67 (two or four weeks after its first administration).
Figure 1 shows our strategy proposal for comprehensive patient management during the entire treatment with TNFi drugs. In addition, a new algorithm for TDM-guided proactive decision-making that includes early optimization in patients treated with IFX is proposed in Figure 2.
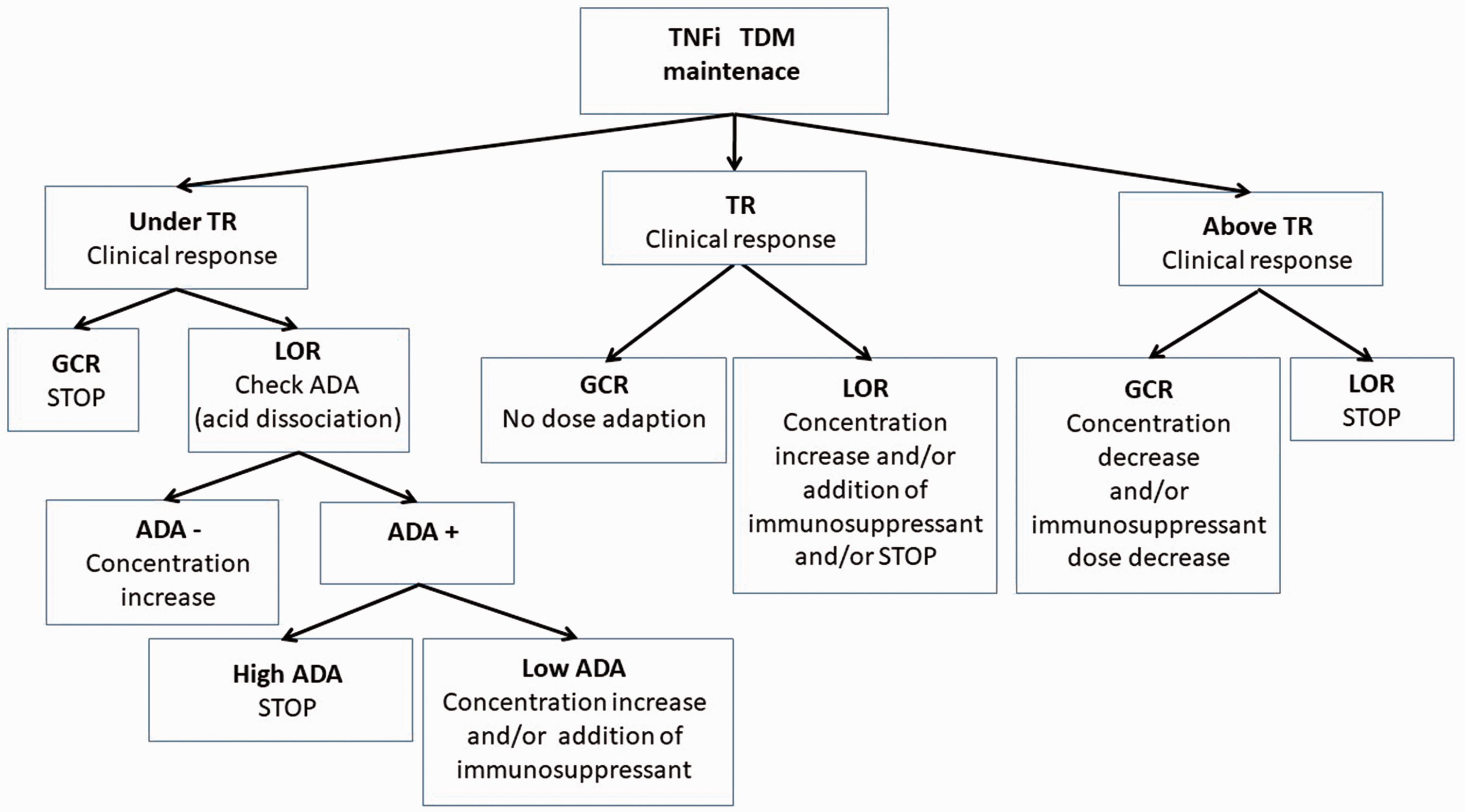
Algorithm for TDM-guided decision-making over the treatment with TNF inhibitors in patients with CIDs. ADA: antidrug antibodies; GCR: good clinical response; LOR: loss of clinical response; TDM: therapeutic drug monitoring; TNFi: tumour necrosis factor inhibitor; TR: therapeutic range.
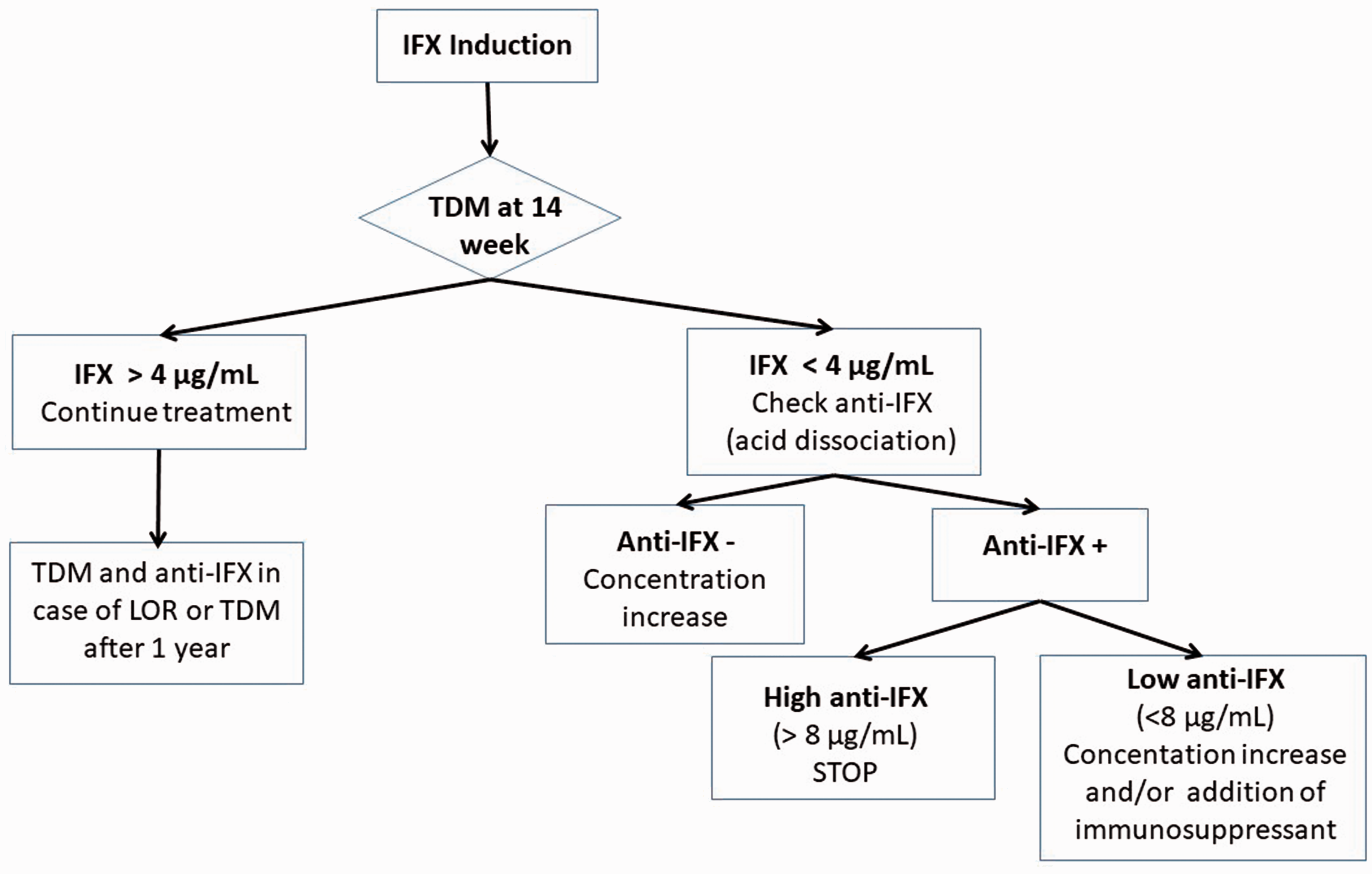
Algorithm for TDM-guided decision-making for early optimization of infliximab treatment in patients with CIDs. Anti-IFX: antidrug antibodies to infliximab; IFX: infliximab; LOR: loss of clinical response; TDM: therapeutic drug monitoring.
Future challenges should include refinement of the optimal concentration range in each patient and the ideal moment for TDM. Although knowledge of mechanisms that regulate TNFi distribution and clearance is still limited, population PK/PD modeling 24 will contribute to the definition of underlying covariates that may explain the observed intersubject and inter-occasion variability in drug concentrations.
Conclusions
TDM of TNFi offers a rational approach to the optimization of IBD, RA and psoriasis therapies. There is increasing evidence that drug concentrations correlate with clinical outcomes, and therapeutic algorithms integrating clinical response with TDM have been developed. Although there is little evidence from prospective controlled trials to definitively demonstrate the benefits of TDM, its value in clinical practice is gaining increasing acceptance.
Footnotes
Acknowledgements
This article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not required because this work is a review of biomedical literature.
Guarantor
JGS-H.
Contributorship
JGS-H, NR and MVC conceived and designed the study. JGS-H, NR and FM selected studies and researched literature. JGS-H, NR, MVC and AM-S wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
