Abstract
Background
Lecithin:cholesterol acyltransferase (LCAT) is a plasma enzyme that esterifies cholesterol. Recombinant human LCAT (rhLCAT) is now being developed as an enzyme replacement therapy for familial LCAT deficiency and as a possible treatment for acute coronary syndrome. The current ‘gold standard’ assay for LCAT activity involves the use of radioisotopes, thus making it difficult for routine clinical use.
Methods
We have developed a novel and more convenient LCAT activity assay using fluorescence-labelled cholesterol (BODIPY-cholesterol), which is incorporated into proteoliposomes as a substrate instead of radiolabelled cholesterol.
Results
The apparent Km and Vmax were 31.5 µmol/L and 55.8 nmol/h/nmoL, rhLCAT, respectively, for the 3H-cholesterol method and 103.1 µmol/L and 13.4 nmol/h/nmol rhLCAT, respectively, for the BODIPY-cholesterol method. Although the two assays differed in their absolute units of LCAT activity, there was a good correlation between the two test assays (r = 0.849, P < 1.6 × 10−7, y = 0.1378x + 1.106). The BODIPY-cholesterol assay had an intra-assay CV of 13.7%, which was superior to the intra-assay CV of 20.8% for the radioisotopic assay. The proteoliposome substrate made with BODIPY-cholesterol was stable to storage for at least 10 months. The reference range (n = 20) for the fluorescent LCAT activity assay was 4.6–24.1 U/mL/h in healthy subjects.
Conclusions
In summary, a novel fluorescent LCAT activity assay that utilizes BODIPY-cholesterol as a substrate is described that yields comparable results to the radioisotopic method.
Introduction
Lecithin:cholesterol acyltransferase (LCAT) is primarily produced by the liver and has a well-established role in esterifying free cholesterol (FC) in plasma.1,2 LCAT resides on high-density lipoprotein (HDL) particles and to a lesser degree on low-density lipoprotein(LDL) and other lipoprotein fractions. It is activated by apolipoprotein A-I (apoA-I), the main protein component of HDL. 3 Mutations in the LCAT gene cause two rare autosomal recessive diseases, namely familial LCAT deficiency (FLD) and fish-eye disease (FED). FLD is characterized clinically by severe hypoalphalipoproteinaemia, corneal opacities, haemolytic anaemia and renal disease.4,5 Renal disease, which presents in FLD patients as early as the third or fourth decade of life, is the main cause of morbidity and mortality. 6 FED patients have a less severe form of the disease, characterized mainly by corneal opacities and low HDL-C.
Currently, there is no specific therapy for LCAT-deficient patients. Animal studies have demonstrated that recombinant human LCAT (rhLCAT) replacement therapy rapidly restored the normal lipoprotein phenotype in LCAT knockout mice, 7 suggesting that rhLCAT replacement therapy may be useful as a therapeutic strategy for LCAT deficiency. Recently, rhLCAT (ACP-501) infusion was able to safely raise plasma HDL-cholesterol in a phase I clinical trial 8 and corrected the lipid abnormalities when tested in a single FLD patient. 9 For the further clinical development of rhLCAT into a therapy, a simple assay for the measurement of plasma LCAT activity is needed.
The most common assay for measuring plasma LCAT activity involves the use of proteoliposomes containing radiolabelled cholesterol as the substrate. 10 A simple colorimetric enzymatic assay has also been described. 11 The method is based on the measurement of the change in the concentration of the substrate (unesterified cholesterol), but it has relatively low sensitivity and requires a large sample volume (200 µL). A more sensitive assay, using the stable isotope 13C-cholesterol and a mass spectrometer, has recently been reported and can be used to detect both free and esterified cholesterol (EC). 12 This assay, however, does not have a high throughput and requires complex instrumentation. Homan et al. 13 recently reported a novel assay for LCAT activity, using the fluorescent sterol, dehydroergosterol (DHE). They quenched the fluorescence of unesterified DHE by cholesterol oxidase and detected the fluorescence of only the esterified DHE. The method works well for purified LCAT preparations, but for plasma samples, the method first requires the depletion of apolipoprotein B-containing lipoproteins, 13 which can contain as much as 25% of total LCAT activity in plasma. 13
In the current study, we developed a novel assay for measuring LCAT activity in whole plasma, using fluorescence-labelled cholesterol (BODIPY-cholesterol) instead of radiolabelled cholesterol as a substrate. Esterified BODIPY-cholesterol and free BODIPY-cholesterol were separated by thin layer chromatography (TLC), and fluorescence from the scanned spots was used to quantitate LCAT activity. Our results demonstrate that the BODIPY-cholesterol method for plasma LCAT activity is a relatively simple assay compared with the more labour-intensive radioiosotopic assay and yields comparable results.
Materials and methods
Materials
A synthetic apoA-I mimetic peptide containing the LCAT activation region of apoA-I (P-642, PVLDLFRELLNELLEALKQKLK; M. W. 2623.17), which forms stable complexes with phosphatidylcholine and sterol, 14 was used to make the proteoliposome substrate. 10 3H-labelled cholesterol (1,2-3H-cholesterol, 49.0 Ci/mmoL, 1.0 mCi in 1.0 mL of ethanol or 1.0 mCi/mL [20.4 pmol/µL]) and 23-(dipyrrometheneboron difluoride)-24-norcholesterol (BODIPY-cholesterol, 810255P) were purchased from PerkinElmer (Boston, MA, USA) and Avanti Polar Lipids, Inc. (Alabaster, AL, USA), respectively. Bovine serum albumin (BSA, essentially fatty acid free grade, A7511-25 G) and 2-mercaptoethanol (2-ME, M3148-25ML) were purchased from Sigma-Aldrich (St. Louis, MO, USA). LCAT assay buffer is 10 mmol/L Tris, 140 mmol/L NaCl and 1 mmol/L ethylenediaminetetraacetic acid (EDTA) (pH 7.4). rhLCAT was isolated from conditioned cell culture medium of CHO cells stably transfected with human LCAT gene, aliquoted and stored at −70℃. 7 Freshly thawed aliquots were used for each experiment.
Preparation of proteoliposome substrate
Proteoliposome substrate solution sufficient for 40 assays was prepared by mixing 21.8 µL of 131 mmol/L phosphatidylcholine and 10.5 µL of 8.7 mmol/L BODIPY-cholesterol in a glass tube and evaporating under N2. To the tube containing the dried substrates, 665 µL of LCAT assay buffer, 250 µL of 1 mg/mL P-642 peptide and 85 µL of 0.72 mol/L cholic acid in LCAT assay buffer were added. After mixing by vortexing at maximum speed for 1 min, the solution was dialysed (dialysis tubing MWCO of 6000–8000) (Spectrum Laboratories, Inc., Rancho Dominguez, CA, USA) against LCAT assay buffer overnight at 4℃. The proteoliposomes (1 mL) were added to an equal volume (1 mL) of LCAT assay buffer containing 2% BSA and 10 mmol/L 2-ME and then incubated in a water bath for 20 min at 37℃. Finally, 0.4 mL of LCAT assay buffer, containing 35 mmol/L 2-ME, was added to the proteoliposome solution (final concentration of cholesterol in proteoliposome: 2.11 nmol/60 µL for one sample). The proteoliposome solution was then divided into small aliquots and stored in a liquid nitrogen tank until use.
For making the proteoliposome substrate with 3H-cholesterol, 7.1 µL of 12.931 mmol/L cold cholesterol in ethanol and 1.25 µL of 20.4 µmol/L (1.0 mCi/mL) 3H-cholesterol in ethanol were used instead of BODIPY-cholesterol, along with 21.8 µL of 131 mM phosphatidylcholine and evaporated under N2. Subsequently, the same procedure used to make the BODIPY-cholesterol proteoliposomes was used to make 3H-cholesterol proteoliposomes. 10
Sample incubation for LCAT activity assay
Five microlitres of human EDTA-plasma or five-fold diluted plasma from human LCAT transgenic mice 15 or various concentrations of rhLCAT (0–1.0 mg/mL) were placed into 2.0-mL microcentrifuge tubes (Corning Inc., Corning, NY, USA). Next, 60 µL of proteoliposome were added to each sample, and the mixture was incubated in a shaking water bath for 30 min at 37℃. To stop the LCAT reaction, 1 mL of 99.5% cold ethanol (The Warner-Graham Company, Cockeysville, MD, USA) was added to the samples. Samples were then centrifuged at 12,000 × g for 10 min at 4℃, and the supernatants were transferred to a new microfuge tube. Supernatants were then completely evaporated, using a SpeedVac concentrator for 2 h. Dried samples were stored at −20℃ until further analysis. All animal studies were approved by the National Heart, Lung, and Blood Institute Animal Care and Use Committee.
TLC and detection
For the BODIPY-cholesterol method, the dried samples were dissolved in 30 µL of chloroform, whereas 30 µL of chloroform containing 0.1 mg/mL cholesterol and 0.7 mg/mL cholesterol oleate were used for the 3H-cholesterol method. Samples were then vortexed for 1 min followed by a brief centrifugation. The entire volume of each sample was applied to a TLC plate (Flexible plates for TLC, PE SIL G; Millipore, Billerica, MA, USA). 10 The TLC plate was placed into a closed glass tank, saturated by a developing solvent (petroleum ether, diethyl ether and acetic acid in mole portions of 230:60:3). 10 After 25 min, the plate was removed, the solvent front was marked and the plate was air-dried for at least 10 min. For the BODIPY-cholesterol method, the cholesterol and EC spots were detected using a Typhoon 9400 Variable Mode Imager (GE Healthcare, Piscataway, NJ, USA) set at 488 nm for excitation and 520 nm for emission. The fluorescent intensity of the spots was analysed by software ImageQuant Version 5.2 (Molecular Dynamics, Sunnyvale, CA). To determine the optimal incubation time for the BODIPY-cholesterol method, the samples were incubated for 0.5, 1 or 4 h at 37℃. Various amounts of rhLCAT (0, 0.025, 0.05, 0.1, 0.2, 0.4, 0.6, 1.0 mg/mL) were added to 60 µL of BODIPY-cholesterol proteoliposomes.
For the 3H-cholesterol method, the TLC plates were air-dried for 10 min and then placed into another closed glass tank containing crystals of iodine on the bottom. After cholesterol and EC were stained as brown spots, the spots were marked by pencil and then immediately cut out with scissors. The cut out spots were placed into a scintillation vial filled with 10 mL of CytoScint (MP Biomedicals, Irvine, CA, USA) and radioactivity was counted in a scintillation counter (LS 6500, Beckman Coulter, Fullerton, CA, USA) for 1 min.
The relative mobility on the TLC plate of EC (Position B) compared with FC (Position A) was calculated. The maximum extent that the solvent reached was defined as Position C, and the relative mobility was calculated with the following formula: (distance from A to B)/(distance from A to C). Specific LCAT activity (nmol/mL/h) was expressed as plasma FC (nmol) × (ratio of EC:FC obtained from spots by TLC) per sample volumes (mL) per incubation time (h).
Kinetics of LCAT activity assays
To determine the Km and Vmax of the two LCAT activity assays, proteoliposome solutions with various amounts of substrate (3H-cholesterol or BODIPY-cholesterol) were prepared by the same procedure described above. For the 3H-cholesterol method, the molar proportion of cholesterol and 3H-cholesterol was the same for each proteoliposome. Typically, 0.04 µg of rhLCAT was routinely incubated with 60 µL of proteoliposome for 30 min at 37℃. The values for the apparent Km and Vmax were determined from Michaelis-Menten curves, as calculated by Prism 6 software (GraphPad Software, Inc., San Diego, CA, USA).
Reproducibility of the LCAT activity assays
To evaluate the reproducibility of the LCAT activity assays, plasma obtained from two healthy human volunteers were measured six times to determine within-run variation and five times on five consecutive days to determine the between-run variation. Pooled plasma from hLCAT transgenic mice 15 (diluted five-fold) was used as a control/standard for LCAT activity in each assay. To obtain plasma from these mice, blood was collected from the retro-orbital sinus with a heparinized capillary tube (250 µL) and placed into tubes with EDTA (final concentration 4 mM) and the tubes were then centrifuged at 1000 × g for 20 min at 4℃. The specific LCAT activity for the 5 × diluted hLCAT-Tg mice was approximately 5000 nmol/mL/h in the 3H-cholesterol method. The LCAT activity in the 5 × diluted plasma of the same mice was determined to be 5000 U/mL/h in our BODIPY-cholesterol method. Specific LCAT activities of human plasma samples in the BODIPY-cholesterol method were calculated by the same formula as with the 3H-cholesterol method and then normalized to the value obtained with the control hLCAT-Tg mouse plasma.
Human plasma samples
Blood was drawn from 20 healthy volunteers (mean age: 43.9 ± 15.6 years old; male:female = 9:11) with normal lipids (total cholesterol: 162.8 ± 19.3 mg/dL [mean ± SD]) or from patients diagnosed with FLD. Plasma samples were obtained by centrifugation at 2000 × g for 10 min at 4℃ and stored at −80℃ until use. FC was measured using Free Cholesterol E (Wako Pure Chemicals, Osaka, Japan). The study was approved by the Institutional Review Board (IRB) at the National Heart, Lung, and Blood Institute.
Statistical analysis
The correlation of LCAT activity between the 3H-cholesterol and BODIPY-cholesterol methods was calculated by the Pearson’s correlation coefficient (r value). The discordance between the two assays was analysed by Bland–Altman analysis. A P < 0.05 was considered to be statistically significant. Reference range was estimated as central 95% range, using non-parametric statistics. All samples were routinely measured in duplicate unless otherwise indicated.
Results
Assay design
Other sterols besides cholesterol have been shown to serve as substrates for LCAT, as long as they contain a hydroxyl group on the A-ring, which is the site for the LCAT esterification reaction.
16
Based on this finding, we hypothesized, as shown in Figure 1, that BODIPY-cholesterol would also serve as a substrate for LCAT. Furthermore, the BODIPY group, which is attached to the hydrophobic alkyl side chain of cholesterol, is also relatively hydrophobic. It has been shown that BODIPY-cholesterol was able to insert into phospholipid membranes similar to cholesterol
17
and recently has been used as an alternative for radiolabelled cholesterol in plasma cholesterol efflux assays.
18
Scheme of LCAT activity assay using BODIPY-cholesterol.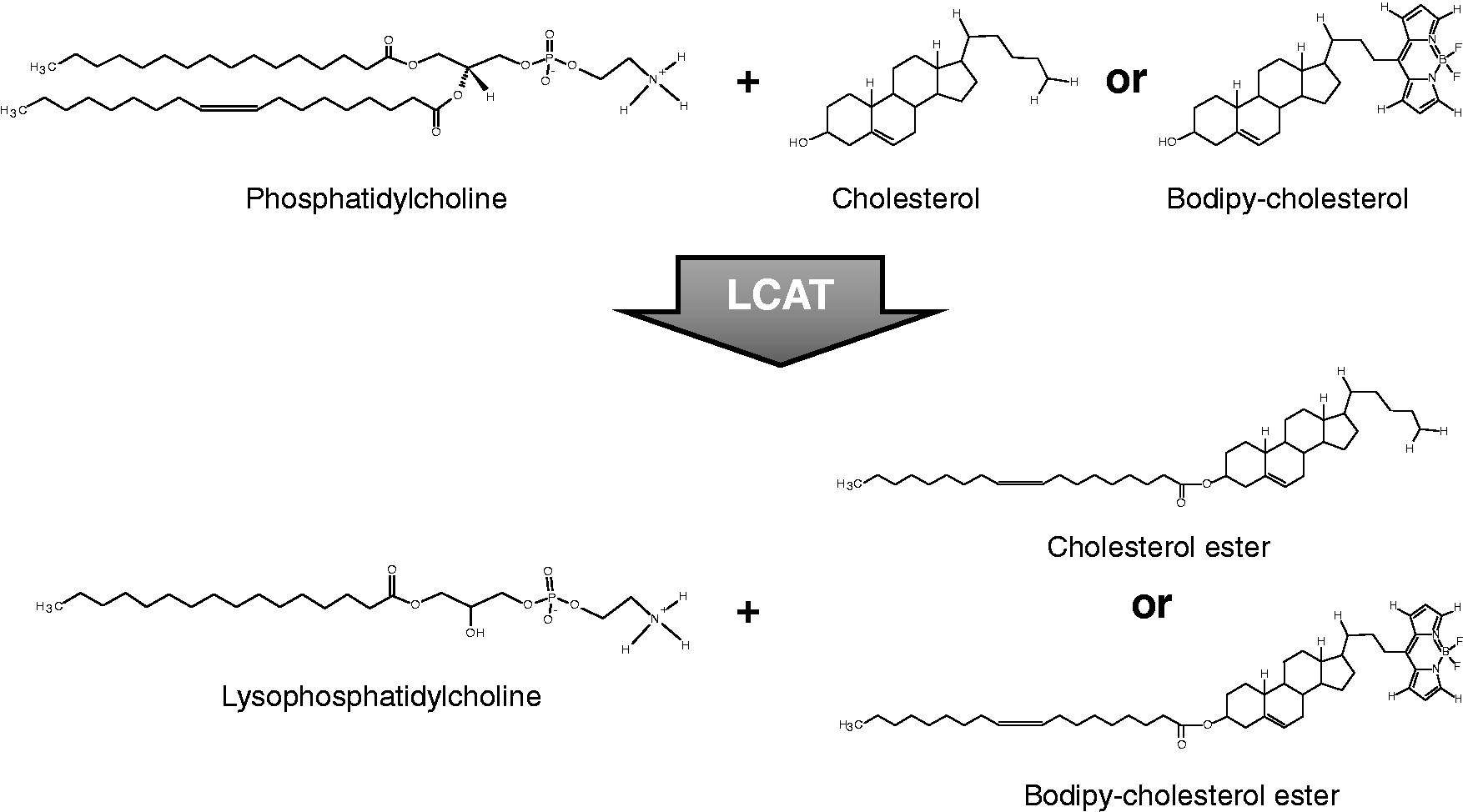
After the incubation of the proteoliposome substrates containing either radiolabelled cholesterol or BODIPY-cholesterol, the extent of the LCAT reaction was quantified after separation of the free sterol from the esterified sterol by TLC. For the 3H-cholesterol method, the relative mobility was 0.709 ± 0.013 (mean ± SD, n = 10) for cholesteryl ester, whereas the esterified BODIPY-cholesterol, in the fluorescent assay, had a relative mobility of 0.331 ± 0.016 (n = 10). The free and esterified fractions for the 3H-cholesterol method were determined after cutting the spots out and measuring radioactivity by liquid scintillation counting. For BODIPY-cholesterol, the amount of fluorescence in the free and esterified spots was directly quantified from the plates by fluorescent scanning (Figure 2).
Separation of free and esterified BODIPY-cholesterol on a TLC plate.
Assay reaction kinetics
Using rhLCAT, the apparent values of Km and Vmax for the 3H-cholesterol method were 31.5 µmol/L and 55.8 nmoL/h per nmoL rhLCAT, respectively (Figure 3). For the BODIPY-cholesterol method, the apparent Km and Vmax were 103.1 µmol/L and 13.4 nmoL/h per nmol rhLCAT, respectively (Figure 3). These results indicate that BODIPY-cholesterol can be a substrate for LCAT but is less effective as a substrate than cholesterol.
Michaelis-Menten curve for LCAT assay.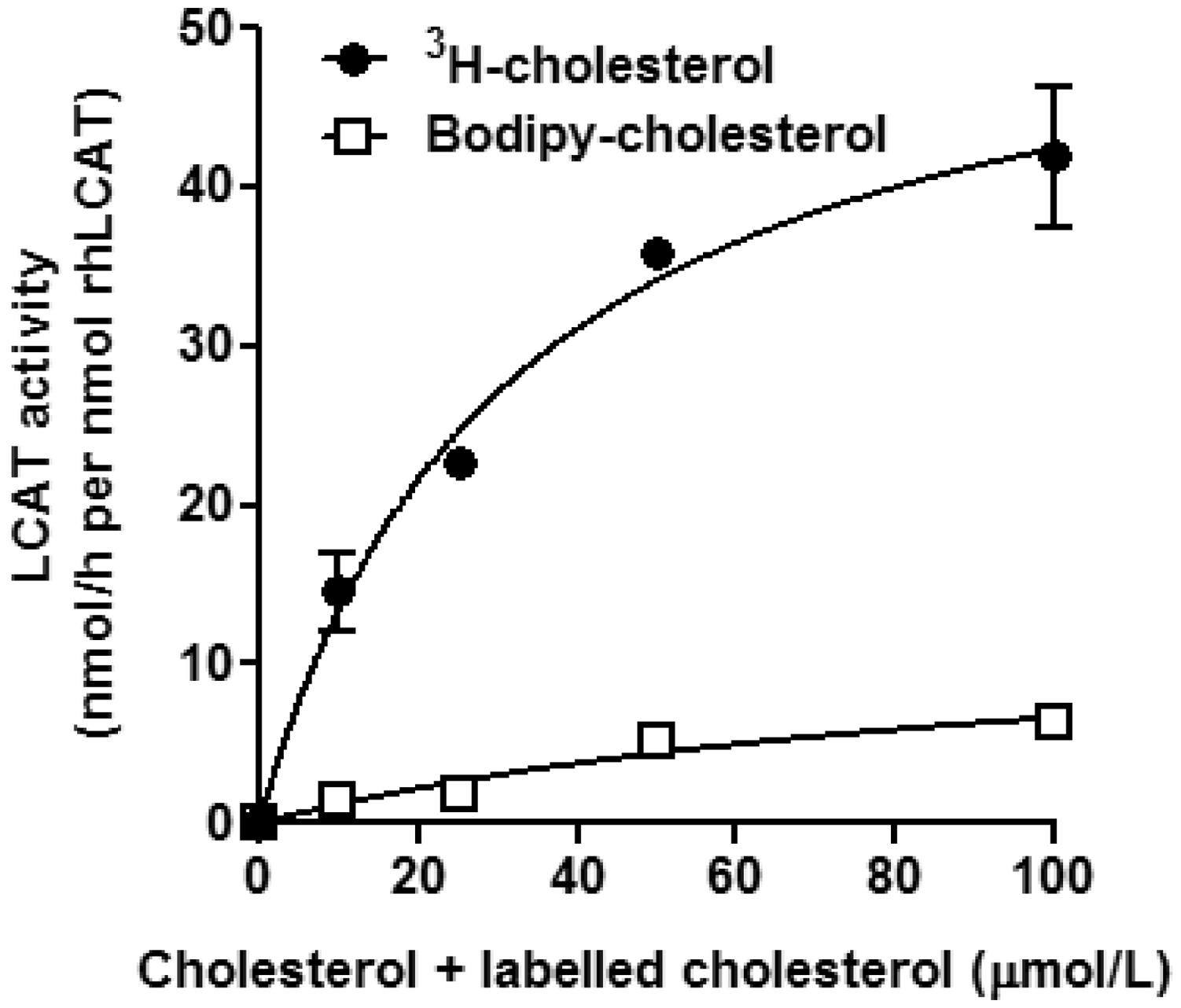
Assay incubation time
When various amounts of rhLCAT were incubated for 0.5, 1 or 4 h at 37℃, a time-dependent increase in LCAT activity was observed (Figure 4). For all LCAT doses tested, the 0.5-h incubation curve showed the best linearity and was chosen as the optimal assay time. As shown in the inset, LCAT concentrations as low as 25 µg/mL were within the linear range.
Test for optimal incubation time.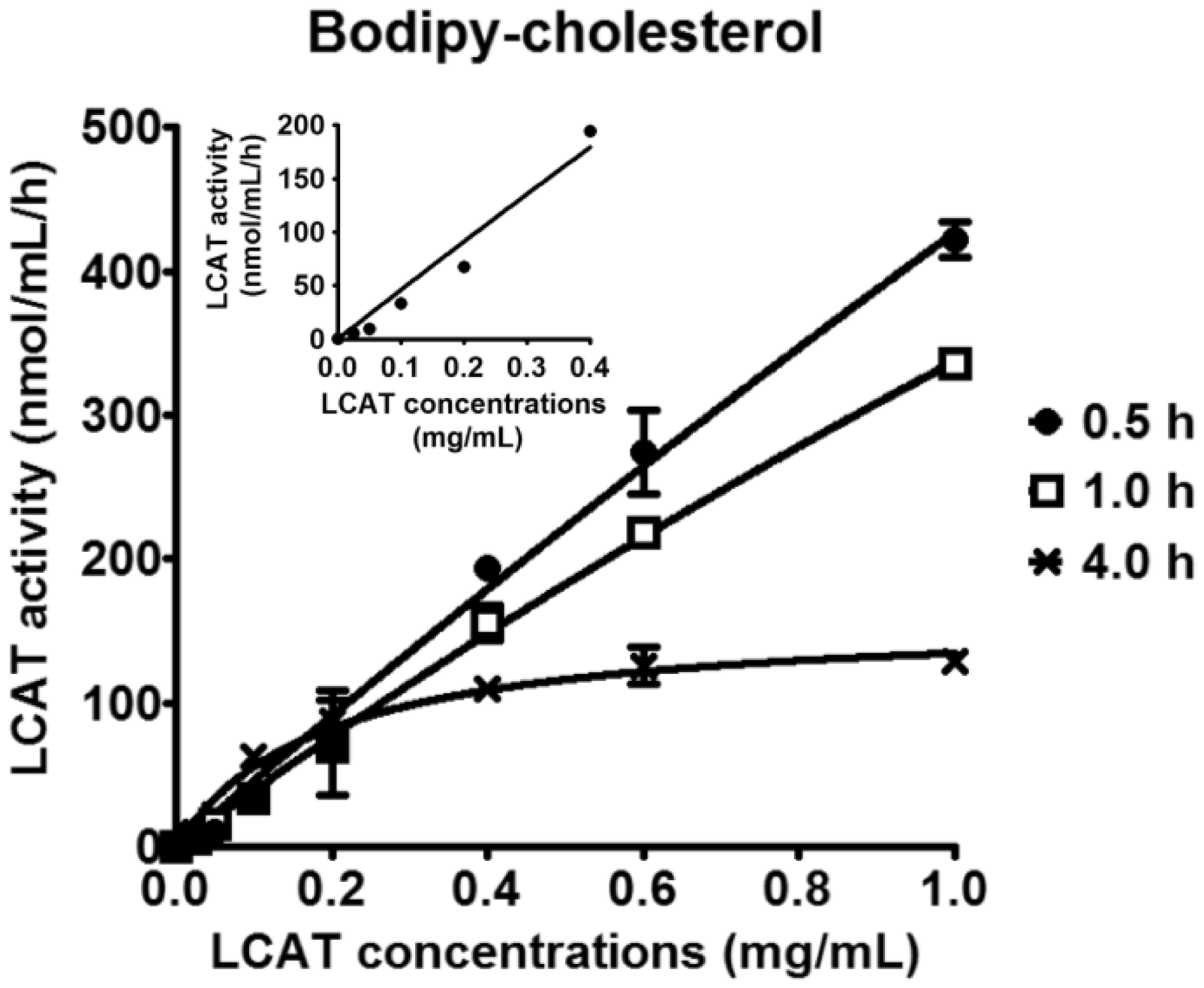
Assay precision
We next tested the reproducibility of both LCAT activity assays. In the 3H-cholesterol method, the coefficient of within-run variation (n = 6) and between-run variation for five days were 8.5% (101.8 ± 8.7 nmol/mL/h [mean ± SD])) and 20.8% (105.2 ± 21.9 nmol/mL/h), respectively. For the BODIPY-cholesterol method, the coefficient of within-run variation (n = 6) and between-run variation for five days were 10.8% (16.7 ± 1.8 U/mL/h) and 13.7% (20.8 ± 2.9 U/mL/h), respectively. In addition, proteoliposomes substrates stored in liquid nitrogen were stable for at least 10 months for both assays (Figure 5).
Test for stability of proteoliposomes stored in liquid nitrogen.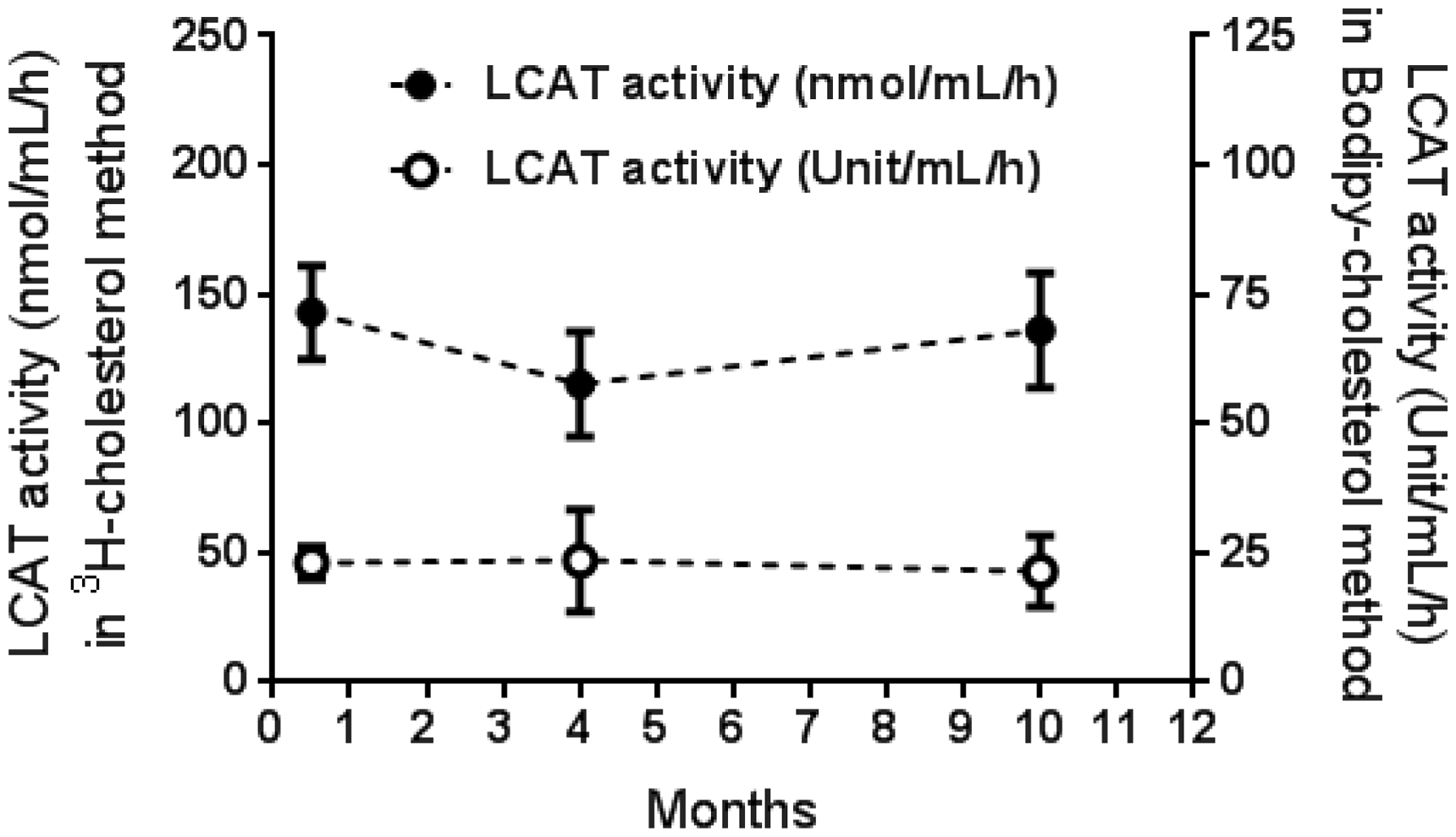
LCAT activity in human plasma samples
To determine whether the BODIPY-cholesterol method is suitable for measuring LCAT from human plasma, we determined the correlation between the BODIPY-cholesterol and 3H-cholesterol methods on normal human plasma samples (Figure 6(a)), as well as a few plasma samples from a single FLD patient.
9
Although the two assays yielded different absolute units, there was a reasonably good correlation between the two methods (Figure 6(a), r = 0.849, P < 1.6 × 10−7, y = 0.1378x + 1.106) from normal plasma samples. The mean value of LCAT activity in normal plasma samples for the BODIPY-cholesterol assay was 15.2 ± 5.6 U/mL/h, with an estimated reference range of approximately 4.6–24.1 U/mL/h. The reference range (n = 20) for the LCAT activity assay using 3H-cholesterol was 56.3–148.8 nmol/mL/h in healthy subjects. In the Bland–Altman plot, the mean ratio between the two assays was 0.164 and ranged between 0.041 and 0.288 (Figure 6(b)).
Correlation between 3H-cholesterol and BODIPY-cholesterol methods in human plasma. (a) The mean values of LCAT activity in the 3H-cholesterol and BODIPY-cholesterol methods were 102.7 ± 25.2 nmol/mL/h (mean ± SD) and 15.2 ± 5.6 Unit/mL/h, respectively. There was a good positive correlation between LCAT activities of the 3H-cholesterol and BODIPY-cholesterol methods (r = 0.849, P < 1.6 × 10−7, y = 0.1378x + 1.106). (b) The mean relative biases of LCAT activity between BODIPY-cholesterol method and 3H-cholesterol method were analysed by Bland–Altman analysis. In Bland–Altman plot, the mean of the ratio was 0.164 with the range between ± 1.96 SD from 0.041 to 0.288.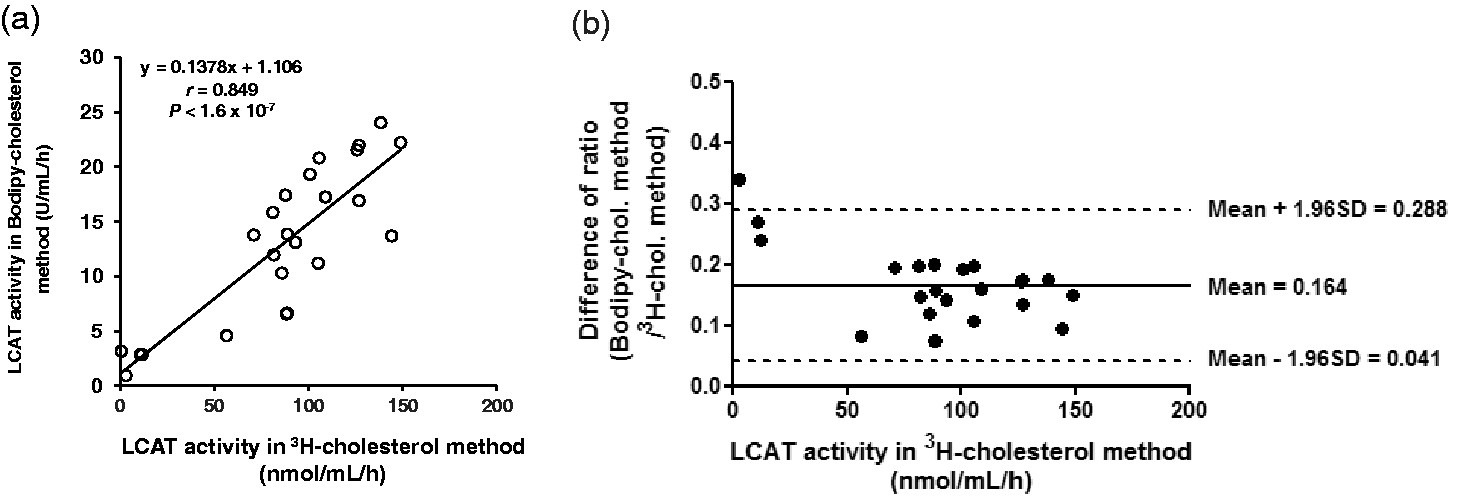
In Figure 7, we analyse the samples by both assays from a previous study,
9
in which recombinant LCAT was intravenously infused into an FLD patient. In case of the radioisotopic assay, LCAT activity shortly peaked after the end of the 1 h infusion of recombinant LCAT and then quickly decayed to near baseline after 24 h. The BODIPY-cholesterol assay showed nearly identical results, thus indicating that this assay should also be suitable for monitoring recombinant LCAT therapy in the future.
LCAT activity by the 3H-cholesterol and BODIPY-cholesterol methods after infusion of recombinant LCAT into an FLD patient.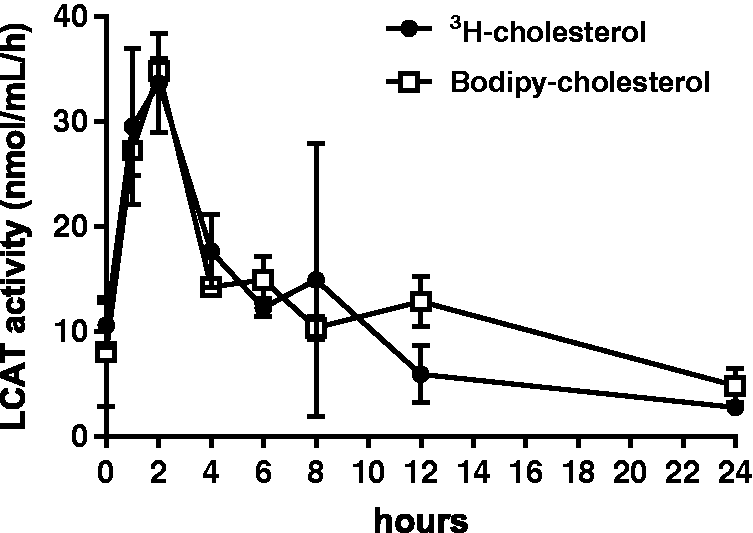
Finally, we tested the suitability of the assay for making the diagnosis of FLD by measuring LCAT plasma activity by both assays in a family with three affected siblings. All three FLD subjects had no detectable LCAT activity by either assay, whereas the obligate heterozygote father had 41% residual LCAT activity by the BODIPY-cholesterol assay versus 42% residual activity for the radioisotopic assay when compared with plasma from a normal subject.
Discussion
Although LCAT is a relatively abundant plasma enzyme and several ELISA kits for measuring LCAT mass are available, it is known that some FLD patients have mutations that affect LCAT activity but have a relatively normal LCAT mass, 19 thus making LCAT activity the preferred method for diagnosing LCAT deficiency. In the present paper, we describe a novel fluorescent assay that has several advantages over the commonly used radioisotopic assay for measuring LCAT activity.
The values for the apparent Vmax and Km for BODIPY-cholesterol were approximately four-fold lower and three-fold higher respectively, than those for 3H-cholesterol (Figure 3). The results for 3H-cholesterol are similar to what has been previously reported. 20 This indicates that BODIPY-cholesterol is a less preferred substrate for LCAT than 3H-cholesterol, which probably accounts for the apparent lower sensitivity of the fluorescent assay. The reason for this is unknown given that both sterol substrates are identical in the A-ring where esterification occurs. BODIPY-cholesterol is frequently used as a cholesterol analogue and behaves like cholesterol in a wide variety of other biochemical and cellular assays, 17 but depending on the application the results can sometimes differ from radiolabelled cholesterol, such as for the cell-based cholesterol efflux assay. 18 It is possible that because of differences in its physiochemical properties, the side chain of BODIPY-cholesterol may interact differently than cholesterol with lipid membranes. 17 LCAT has recently been crystalized 21 and cholesterol has been shown to fit in a relatively small hydrophobic pocket near the active site. Based on this, the other possibility for the difference between the two assays may relate to how BODIPY-cholesterol fits into the substrate binding pocket of LCAT. It is also important to point out that the relationship between the two assays shown in Figure 6 may vary depending on the sensitivity of the fluorescent scanner used for reading the TLC plate.
The BODIPY-cholesterol method was linear over a relatively broad range of rhLCAT concentrations (Figure 4), and sufficiently sensitive in that LCAT activity was detected in all tested human plasma samples from normal individuals. Furthermore, the precision of our method was relatively good and was superior to the radioisotopic assay in terms of the between-run precision. Importantly, the BODIPY-cholesterol assay also showed a relatively good correlation to the radioisotopic assay (Figure 6) and can be used to distinguish human FLD heterozygotes from homozygotes. Like the radioistopic assay, the BODIPY-cholesterol assay showed no detectable LCAT activity in the three FLD subjects tested. Given the similar LCAT activity profile of the two assays after infusion of recombinant LCAT into an FLD patient, the BODIPY-cholesterol assay should be suitable for monitoring recombinant LCAT therapy. 9 One advantage of the fluorescent assay is that the proteoliposome fluorescent substrate containing BODIPY-cholesterol can be stored frozen for at least 10 months without significantly altering activity (Figure 5). Similarly, because LCAT activity is stable to freezing,1–5 fresh frozen plasma samples should be suitable for preparing both high and low LCAT activity controls for the fluorescent assay.
A major advantage of the new fluorescent assay compared with the radioisotopic assay is that it requires less time, particularly for the quantitation of the free and esterified sterol spots on the TLC. Besides the extra care it takes to handle radioactive specimens, the spots corresponding to the free and EC on the TLC plates have to be manually removed and quantified by liquid scintillation counting. In contrast, fluorescence in the spots from the entire plate can be rapidly and accurately determined by fluorescent scanning, which probably accounts for the improved precision of the assay. While the fluorescent assay is still labour intensive because of the need for TLC separation, in the future, this step could possibly be performed instead by HPLC to reduce the amount of time and manual labour needed to perform this part of the assay.
In conclusion, we describe a novel fluorescent LCAT activity method that has several advantages over the currently commonly used radioisotopic assay and will likely be useful in ongoing efforts to develop drugs or recombinant proteins that modulate LCAT activity.
Footnotes
Acknowledgment
We are grateful to Dr Angel Aponte, Proteomics Core, National Heart, Lung, and Blood Institute, National Institutes of Health for support in measurements with the Typhoon scanner.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by intramural National Institutes of Health funds from the National Heart, Lung, and Blood Institute.
Ethical approval
The study was approved by the IRB at the National Heart, Lung, and Blood Institute. All animal studies were approved by the National Heart, Lung, and Blood Institute Animal Care and Use Committee.
Guarantor
ATR.
Contributorship
TS and ATR researched literature and conceived the study. TS, AS, BLV, TN, EBN and SJD were involved in LCAT activity assays and data analysis. MLS and RDS were involved in collection of human plasma and patient recruitment and MLS was involved in measurement of plasma-free cholesterol. TS wrote the first draft of the manuscript. TS, LAF, BLV and ATR edited this manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
