Abstract
Background
Periodontitis is an inflammatory disease. The aim of this study was to investigate whether the soluble form of interleukin-6 receptor (sIL-6R) and calprotectin concentrations in gingival crevicular fluid are useful biomarkers in the evaluation of periodontitis.
Methods
First, a cross-sectional study was performed. A total of 34 periodontitis patients were enrolled and the gingival crevicular fluid samples were collected from the healthy and inflamed sites of periodontal pockets in each patient. The relationship between periodontal condition and gingival crevicular fluid sIL-6R and calprotectin concentrations was analysed statistically. The cut-off values of gingival crevicular fluid sIL-6R and calprotectin concentrations for the evaluation of periodontitis were determined using a receiver operating characteristic curve. Next, by using enzyme-linked immunosorbent assay, it was examined whether calprotectin induces sIL-6R production in THP-1 macrophages.
Results
Both gingival crevicular fluid sIL-6R and calprotectin concentrations were significantly higher in the inflamed sites than in the healthy sites (P < 0.0001). The cut-off values of gingival crevicular fluid sIL-6R and calprotectin concentrations for the evaluation of periodontal inflammation were as follows: sIL-6R: 43.5 pg/site; calprotectin: 134.3 ng/site. In the in vitro study, calprotectin significantly induced sIL-6R production in THP-1 macrophages (P < 0.01).
Conclusions
Both gingival crevicular fluid sIL-6R and calprotectin concentrations are significant biomarkers in the evaluation of periodontal inflammation.
Introduction
Periodontitis is a chronic inflammatory disease characterized by attachment loss of periodontal tissue and bone destruction. 1 Recently, it has been generally recognized that the disease is associated with increased prevalence of several systemic diseases such as cardiovascular diseases, diabetes and preterm birth.2,3 Therefore, early detection and exact diagnosis of periodontal inflammation are important for general health. Clinically, periodontal inflammations are evaluated using several clinical parameters such as probing pocket depth (PPD), bleeding on probing (BOP), tooth mobility, etc., and these clinical tests are useful in the evaluation of organic changes of periodontal tissues; 4 however, it cannot be denied that there is a difference among dentists in the examination results because of the complicated techniques. Many dentists hope that the objective examination for periodontitis based on biological inflammatory responses should be proposed and evaluated by appropriate laboratory clinical tests that focus on local microinflammation.
In periodontal lesions, the cross-talk between periodontal pathogens and host responses is regulated by several cytokines secreted by immune cells such as neutrophils, macrophages and lymphocytes, 5 and the cytokine balance regulated by immune responses has an important role in the stability or progression of inflammation. The cytokines and chemokines produced by host responses have been suggested as reliable markers in the prediction of the progression of periodontitis. A soluble form of interleukin 6 receptor (sIL-6R), known as IL-6 agonist, is released by proteolytic cleavage from the cell surface such as lymphocytes and macrophages. 6 Kotake et al. 7 reported previously that sIL-6R concentration was significantly higher in the synovial fluids of patients with rheumatoid arthritis than with osteoarthritis. In addition, our in vitro studies have shown that IL-6 induces the production of several inflammatory mediators such as matrix metalloproteinase-1, vascular endothelial growth factor (VEGF), monocyte chemoattractant protein-1 and basic fibroblast growth factor in the presence of sIL-6R in human gingival fibroblasts (HGFs).8,9 Considering the inflammatory responses, sIL-6R might be an attractive biomarker in the evaluation of periodontal inflammation. On the other hand, calprotectin, an abundant heterodimeric cytosolic protein of neutrophils, has a variety of functions such as antimicrobial activity and chemotactic factor of neutrophils. 10 Calprotectin is also considered a useful inflammatory marker of periodontitis. 11 Kaner et al. 12 reported previously that calprotectin concentrations in gingival crevicular fluid (GCF) correlate with periodontal disease severity and treatment outcome. Becerik et al. 13 also reported that elevated GCF calprotectin concentrations play a role as a reliable inflammatory marker in the pathogenesis of periodontal disease. Therefore, the evaluation of GCF calprotectin might be effective as a diagnostic marker for periodontitis clinically.
Supportive periodontal therapy (SPT) is an integral part of periodontal treatment and is essential in the prevention of the progression of the disease because periodontitis recurs frequently even after intensive treatment. 14 Determining the risk for periodontitis progression during SPT phase would help dentists control the frequency and contents of SPT visits. Therefore, we focused on the SPT phase, in order to evaluate the risk of periodontitis recurrence. In the present study, we propose the clinical usefulness of sIL-6R and calprotectin as biomarkers in the evaluation of periodontal inflammation. These biomarkers of periodontal inflammation would help dentists to choose the appropriate approach for periodontitis patients individually to prevent periodontitis progression.
Materials and methods
Subjects and oral examination
Patients and periodontal characteristics.
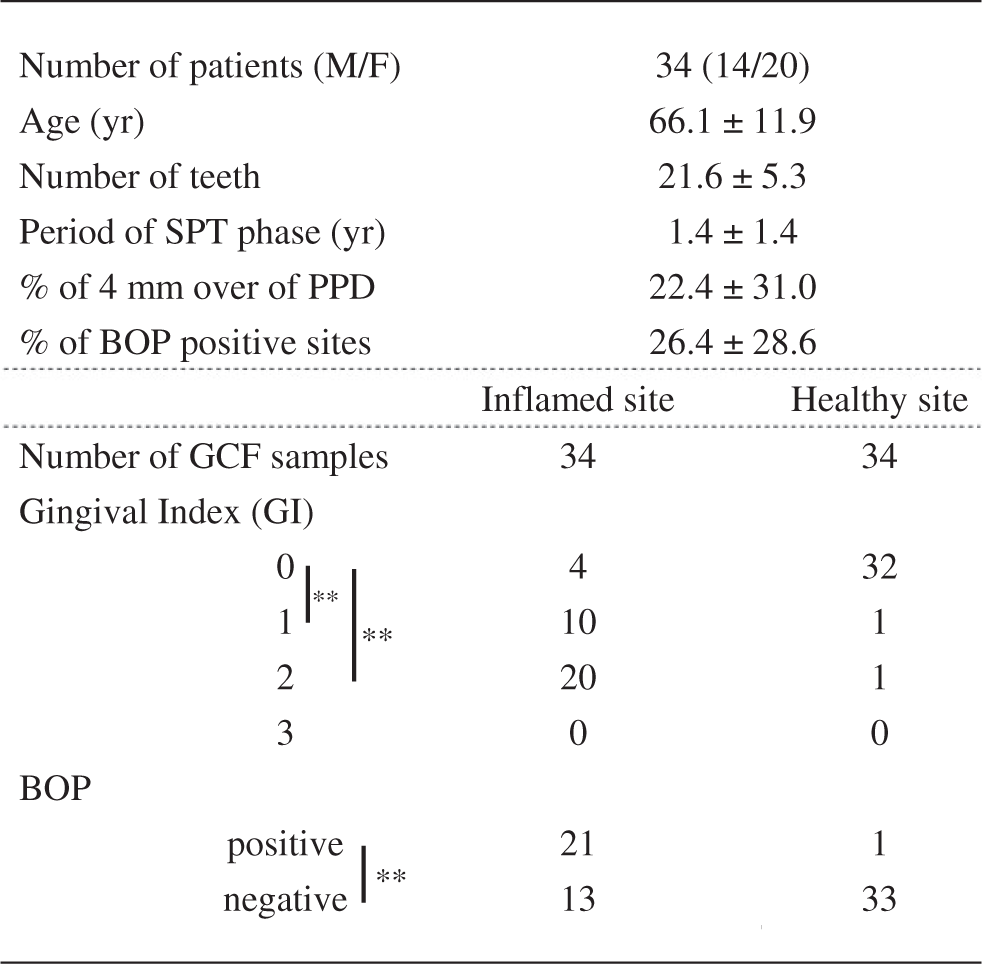
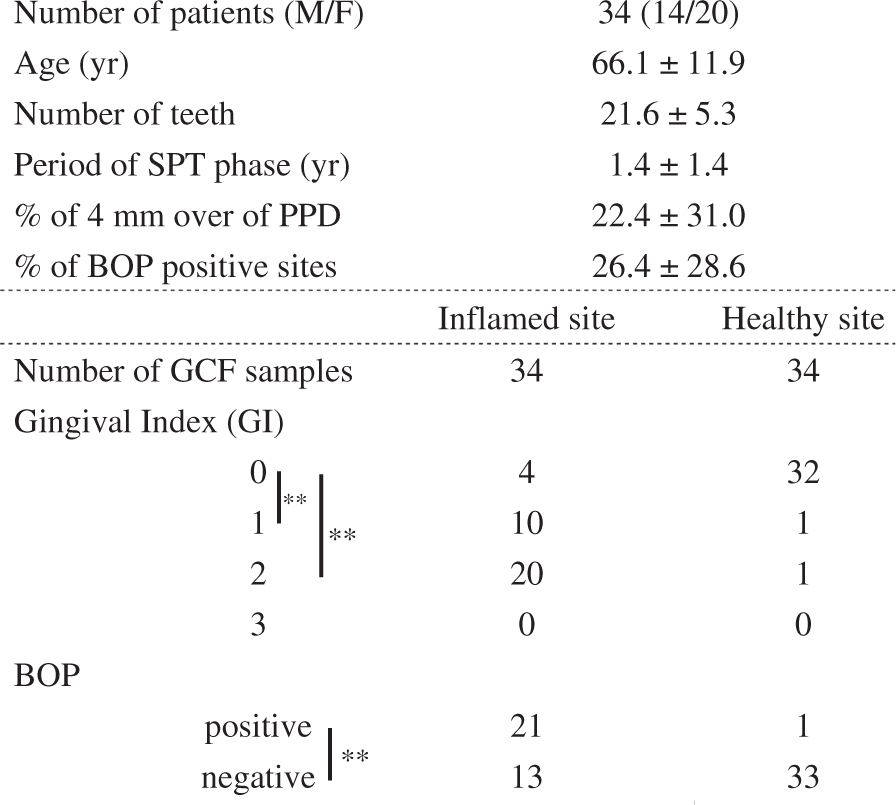
Note: Data are described by the mean ± standard deviation (SD).
M: male; F: female; PPD: periodontal pocket depth; BOP: bleeding on probing; GCF: gingival crevicular fluid; SPT: supportive periodontal therapy.
P < 0.01, chi-square test.
Inclusion criteria:
Adult patients with chronic periodontitis during SPT phase after intensive periodontal therapy.
15
Patients with three or more deepening PPD with a depth of 5 mm or more.
Exclusion criteria:
Patients with pregnant women or in lactation. Patients with systemic antibiotic intake. Frequent use of anti-inflammatory drugs. Patients with other habits, such as smoking and bruxism were recorded by a directed interview.
Trained dentists performed the examination of clinical findings (age, number of teeth, BOP, gingival index [GI] and PPD), and a supervisory doctor verified it so that there was no difference in technique among the attending dentists. All the participating subjects signed an informed consent. This study was approved by the ethics committee of Tokushima University Hospital (no. 2325).
GCF sampling and processing
GCF samples were obtained from both 1 inflamed and 1 healthy sites by inserting sterile 2 paper points (absorbent paper points #45, Dentsply/Maillefer, Ballaigues, Switzerland) in each patient and kept it in the place for 30 s as described in previous report.16,17 Healthy site was selected from the periodontal sites with PD of <3 mm. After careful removal of supra-gingival plaque with sterile gauze, GCF samples were collected and the samples were immediately placed in sterile Eppendorf tubes containing 100 μL PBS with proteases inhibitors cocktail (Roche Diagnostics, Basel, Schweiz) on ice, transported to the laboratory and stored at −80℃ until use, and the samples visibly contaminated with blood were discarded.
Concentrations of sIL-6R and calprotectin in GCF
Both sIL-6R and calprotectin concentrations in GCFs were measured using commercialized enzyme-linked immunosorbent assay Kit (ELISA) (sIL-6R: R&D Systems, Minneapolis, MN; calprotectin: Hycult Biotech, PB Uden, the Netherlands) according to the manufacturer's instruction.
Reagents
Recombinant human (rh) calprotectin was obtained from R&D Systems (Minneapolis, MN). Phorbol 12-myristate 13-acetate (PMA) was obtained from Sigma (St. Louis, MO). Porphyromonas gingivalis (P.g) LPS was purchased from Invitrogen (Carlsbad, CA, USA).
Concentrations of IL-6 and sIL-6R released from THP-1 macrophages
The human monocytic leukaemia cell line THP-1 cells (ATCC) were maintained in RPMI 1640 (GIBCO-BRL, Rockville, MD) supplemented with 10% heat-inactivated fetal bovine serum and antibiotics (Invitrogen). THP-1 cells were differentiated by 100 nM PMA. To measure the concentrations of IL-6 and sIL-6R release from macrophages, differentiated THP-1 macrophages were treated for 24 h with or without calprotectin (0, 5, 50 nM), PMA (100 nM) and P.g LPS (1 μg/mL). Supernatants were collected and stocked at −80℃ until use. The amount of IL-6 and sIL-6R was measured using sandwich ELISA kits (R&D Systems) according to the manufacturer's instruction.
Statistical analysis
In clinical study, the differences in GI or appearance of BOP between inflamed sites and healthy sites were analysed with chi-square test. Since both GCF sIL-6R and calprotectin concentrations were not normally distributed, the Mann-Whitney U test was used for the comparison of the two groups: (1) inflamed/healthy site in PPD and (2) with or without BOP, ANOVA with Tukey HSD analysis for comparison of the three groups: GI score (0, 1, 2) and Pearson's correlation coefficient test were used for statistical analysis. The cut-off values of both GCF sIL-6R and calprotectin concentrations to evaluate the presence or absence of periodontal inflammation were analysed with receiver operating characteristic (ROC) curve and their respective areas under the curve (AUC), in which sensitivity is plotted as a function of 1-specificity. In the in vitro study, the statistical significances of IL-6 and sIL-6R concentrations among several stimulation groups were determined by ANOVA Tukey-HSD. Statistical analyses were performed with SPSS Statistics version 20 (Armonk, NY). In all statistical analyses, P values of less than 0.05 were considered statistically significant.
Results
Characteristics of oral conditions in periodontitis patients
The characteristics of oral conditions in subjects are shown in Table 1. As expected, there were significant differences between the inflamed sites and healthy sites in GI (GI-0 vs. GI-1, P < 0.0001; GI-0 vs. GI-2, P < 0.0001, chi-square test). In addition, there was a significant difference between the inflamed sites and healthy sites with regard to the appearance of BOP (P < 0.0001, chi-square test).
Comparison of sIL-6R/calprotectin concentrations in GCF between inflamed sites and healthy sites of periodontitis patients
Both GCF sIL-6R and calprotectin concentrations were significantly higher in the inflamed sites than in the healthy sites of the periodontitis patients (sIL-6R: P < 0.0001; calprotectin: P < 0.0001, Mann-Whitney U test) (Figure 1(a) and (b)). GCF sIL-6R concentrations correlated statistically to the GCF calprotectin concentrations (r=0.650, P < 0.0001, Pearson's correlation coefficient test) (Figure 1(c)).
GCF sIL-6R concentrations (a) and GCF calprotectin concentrations (b) in patients with periodontitis. Statistical differences of GCF sIL-6R/calprotectin concentrations in healthy and inflamed sites were analysed using Mann-Whitney U test. Bar shows median. (c) Correlation between GCF sIL-6R concentrations and GCF calprotectin concentrations were analysed using Pearson's correlation coefficient test. r: correlation coefficient; H: healthy site; P: inflamed site; **P < 0.01.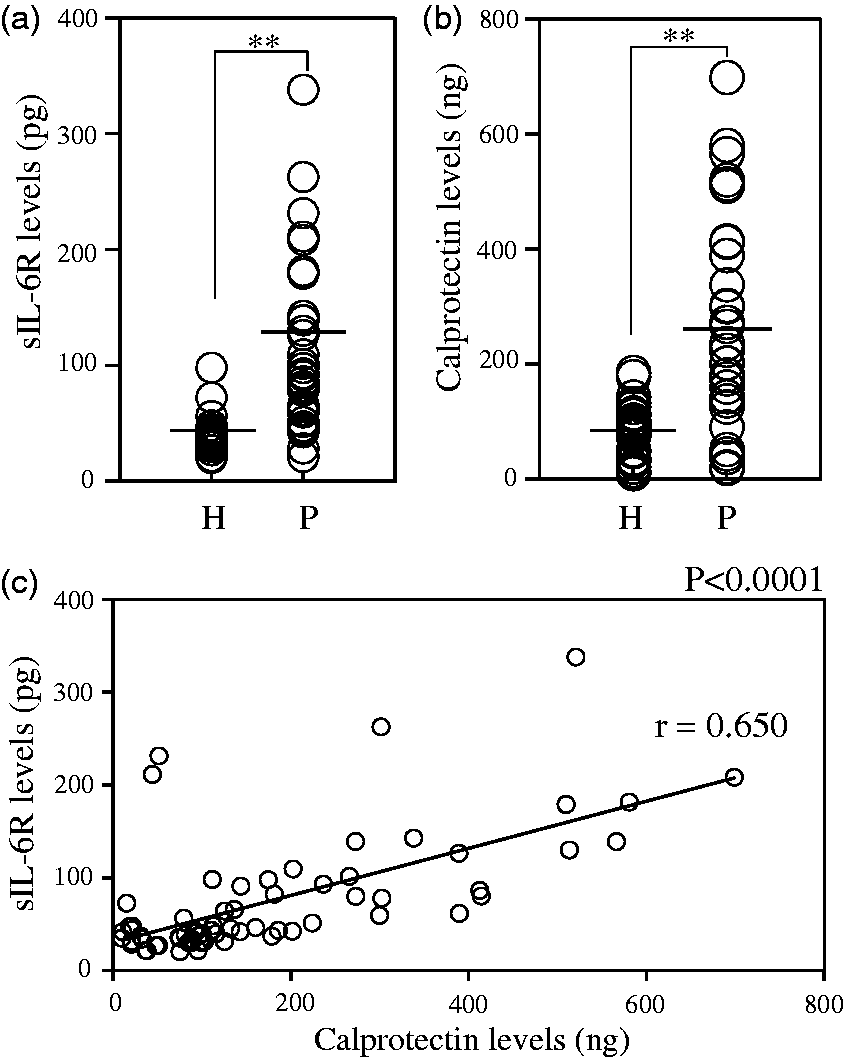
Relationship between sIL-6R/calprotectin concentrations in GCF and clinical parameters of periodontitis
The relationship between GCF sIL-6R and calprotectin concentrations and clinical parameters of periodontitis was investigated. GCF sIL-6R concentrations in both the GI-1 and GI-2 groups were significantly higher than in the GI-0 group (GI-0 vs. GI-1: P = 0.031, GI-0 vs. GI-2: P < 0.0001, ANOVA with Tukey HSD) (Figure 2(a)). GCF sIL-6R concentrations in BOP-positive group were significantly higher than in BOP-negative group (P = 0.001, Mann-Whitney U test) (Figure 2(b)). In addition, GCF calprotectin concentrations in both the GI-1 and GI-2 groups were significantly higher than in the GI-0 group (GI-0 vs. GI-1: P < 0.0001, GI-0 vs. GI-2: P < 0.0001, ANOVA with Tukey HSD) (Figure 2(c)). GCF sIL-6R concentrations in BOP-positive group were also significantly higher than in BOP-negative group (P = 0.018, Mann-Whitney U test) (Figure 2(d)). Importantly, there were no significant differences between GI-1 and GI-2 groups in both sIL-6R and calprotectin concentrations in GCF (sIL-6R: P = 0.153; calprotectin: P = 0.986, ANOVA with Tukey HSD) (Figure 2(a) and (c)).
Relationship between GCF sIL-6R/calprotectin concentrations and periodontal clinical parameters. Statistical differences between GCF sIL-6R concentrations and GI (a), appearance of BOP (b) were analysed using Mann-Whitney U test. Statistical differences between GCF calprotectin concentrations and GI (c), appearance of BOP (d) were also analysed using Mann-Whitney U test. Box plot shows median, the lower and upper quartiles of all the data. Error bar shows SD. *P < 0.05; **P < 0.01.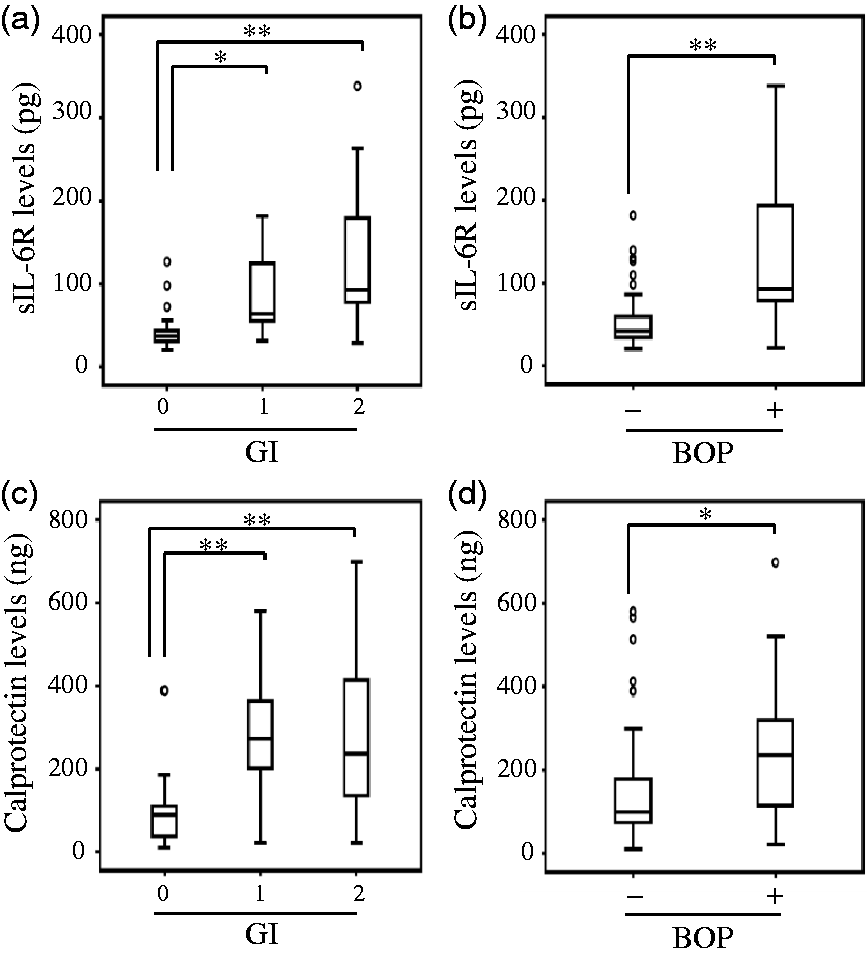
ROC analysis for cut-off values of sIL-6R/calprotectin in GCF in the evaluation of periodontitis
Figure 3 shows the ROC curves determining both sIL-6R and calprotectin concentrations in GCF to evaluate the presence or absence of periodontitis. The AUCs were 0.70 and 0.83 for sIL-6R and calprotectin, respectively. The most appropriate cut-off values were 43.5 pg/site (sensitivity: 0.895, specificity: 0.592), and 134.3 ng/site (sensitivity: 0.765, specificity: 0.912) for sIL-6R and calprotectin, respectively. All curves provided high sensitivity and specificity in evaluation for periodontitis.
ROC curves of GCF sIL-6R concentrations (a) and GCF calprotectin concentrations (b) to determine presence or absence of periodontitis in patients with periodontitis. The cut-off values indicated by the arrow were calculated and provided in each panel.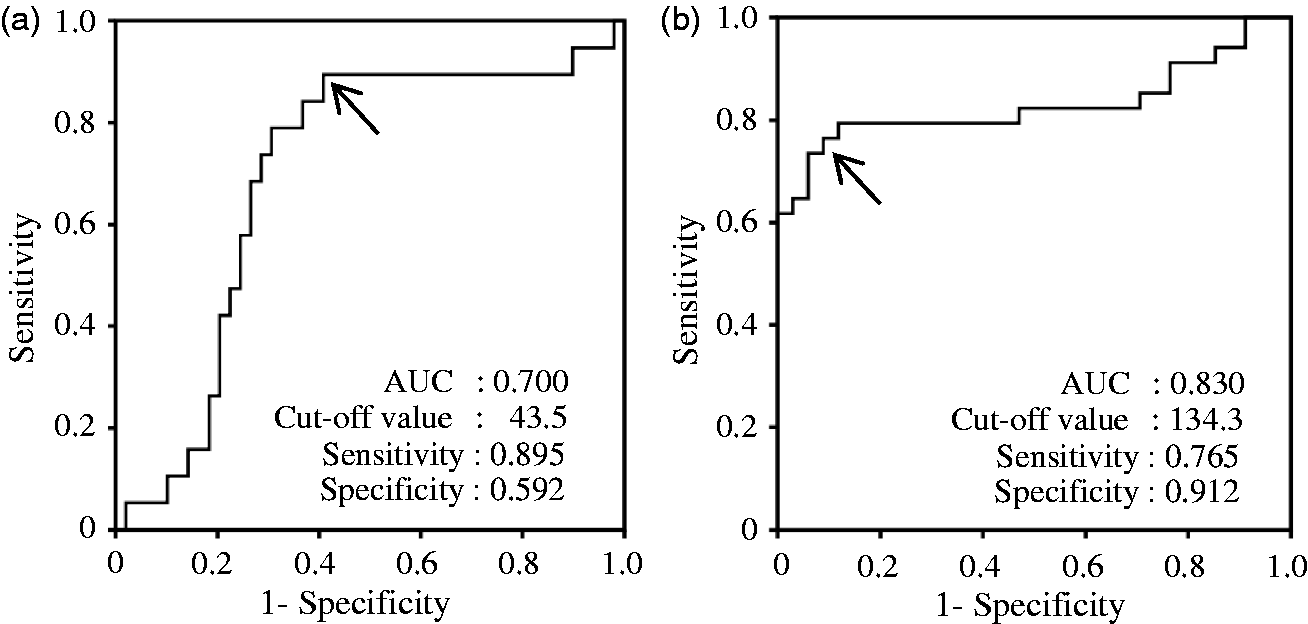
Induction of sIL-6R production in THP-1 macrophages by calprotectin
First, it was examined whether calprotectin induces IL-6 in THP-1 macrophages. P.g LPS was used as a positive control. As shown in Figure 4(a), calprotectin did not induce the IL-6 production in THP-1 macrophages (ANOVA-Tukey HSD). P.g LPS increased the IL-6 significantly in the cells compared with other stimulation (P < 0.0001, ANOVA-Tukey HSD). Next, it was examined whether calprotectin induces sIL-6R in THP-1 macrophages. As shown in Figure 4(b), calprotectin increased sIL-6R production significantly in THP-1 macrophages (P < 0.001, 0 vs. 50 nM, ANOVA-Tukey HSD). P.g LPS did not increase the sIL-6R production in the cells. In addition, we could not detect sIL-6R in culture supernatants of undifferentiated THP-1 monocytes (data not shown).
IL-6 and sIL-6R production in THP-1 macrophages. Concentrations of (a) IL-6, (b) sIL-6R production in THP-1 macrophages. Cells were treated by calprotectin (0, 5, 50 nM), PMA (100 nM) and P.g LPS (1 μg/mL) for 24 h and collected the supernatants. Both IL-6 and sIL-6R concentrations were measured described in ‘Materials and Methods section’. Data are represented as the mean ± SD from three independent experiments. **P < 0.01 as compared with control (ANOVA Tukey-HSD).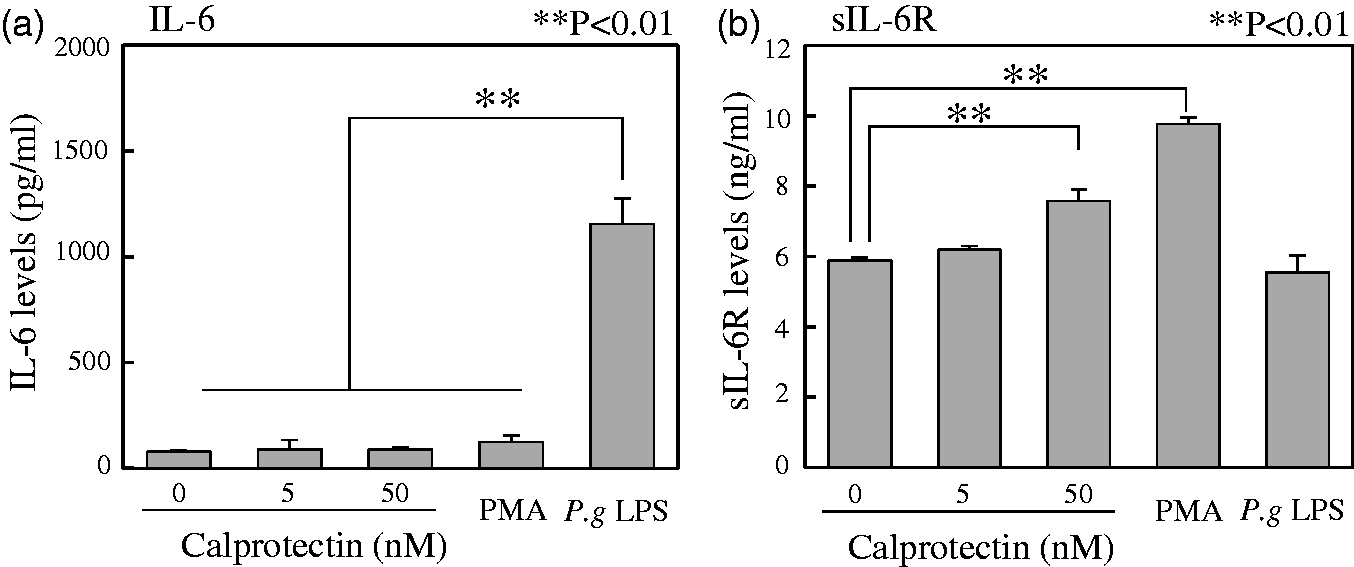
Discussion
Periodontitis is an inflammatory disease caused by periodontal bacteria such as P.g. 1 Clinically, since the recurrence of periodontitis is frequently associated with poor oral health even after intensive periodontal treatment, SPT is important for the maintenance of periodontal health. 14 Poor oral health may have a profound effect on not only the progression of periodontitis but also on the deterioration of general health. 2 Therefore, the presence or absence of persistent low-grade infection by periodontitis is recognized as a significant focus for physicians, and objective evaluation for periodontal inflammation based on biological responses might be essential for general health.
It has been well known that GCF in patients with periodontitis contains inflammatory cells, serum proteins, bacteria and numerous immune/inflammatory mediators. 18 Since collection of GCF is relatively non-invasive, it should be a convenient tool for the clinical evaluation of periodontal inflammation. As shown in Figure 1, we found that both GCF sIL-6R and calprotectin concentrations are significantly higher the in inflamed sites than in the healthy sites. Although the elevation of GCF calprotectin of periodontitis patients corresponds to other previous reports,11–13 we examined periodontitis patients during SPT phase after periodontal treatment as subjects in the present study. Our results have shown that both sIL-6R and calprotectin are useful biomarkers in the evaluation of periodontal inflammation. Furthermore, toward the clinical use as a laboratory test to distinguish the periodontal health and inflammation, we calculated standard values of GCF sIL-6R and calprotectin concentrations using ROC curve and their respective AUC, in which sensitivity is plotted as a function of 1-specificity (Figure 3). The AUC of ROC curve is a summary measure of the discriminative ability of the model, with values between 0.90 and 1.00 indicative of excellent predictive discrimination, and between 0.70 and 0.90 indicative of moderate predictive discrimination. 19 We found that the curve for GCF sIL-6R and calprotectin concentrations provided moderate predictive discrimination (AUC, sIL-6R: 0.70, calprotectin: 0.83). Based on the ROC analysis, since the sensitivity in GCF sIL-6R concentrations was a bit higher than that in GCF calprotectin, GCF sIL-6R is a better choice as a suitable biomarker to screen the periodontal inflammation (sensitivity, sIL-6R: 0.895, calprotectin: 0.765). In any case, the cut-off values obtained in the present study may be useful set points for screening the periodontal inflammation, and might contribute to the development of convenient tools for the diagnosis of periodontitis.
The periodontal examinations that dentists perform routinely may not be useful for the evaluation of the progression of periodontitis. Our previous results have shown that the predictive values of routine periodontal parameters (plaque control record, BOP and PPD) were relatively low because of the complicated techniques. 20 In the present study, therefore, we focused on biomarkers, sIL-6R and calprotectin in GCF, to evaluate the periodontal inflammation. We examined whether GCF sIL-6R and calprotectin concentrations correlate to the clinical findings of patients with periodontitis. GI and BOP were examined as routine clinical parameters for periodontitis. Since GI does not consider PPD and degrees of bone loss, it is entirely evaluated by the inflammatory appearance of gingival tissues. 21 By evaluating gingival inflammation using GI, we found that there were statistical differences between GI-0 and GI-1 in both GCF sIL-6R and calprotectin concentrations, whereas there were no significant differences between GI-1 and GI-2 in both GCF sIL-6R and calprotectin concentrations. These findings might indicate that both GCF sIL-6R and calprotectin concentrations are suitable biomarkers to distinguish periodontal health and low-grade inflammation. Furthermore, GCF sIL-6R and calprotectin concentrations are higher in the BOP-positive group than in the BOP-negative group. BOP, which is induced by gentle manipulation in the gingival sulcus, is known as a sign of extant inflammation. Pathologically, the BOP-positive sites indicate the erosion/ulceration or destruction of gingival epithelium and the infiltration of capillary vessel induced by VEGF. 9 Many researchers have reported the elevation of GCF IL-6 concentrations in inflamed periodontal sites.22,23 Since the complex of IL-6 and sIL-6R induces production of VEGF in HGFs,8,9 significant elevation of sIL-6R concentrations in the BOP-positive sites might induce the VEGF release from HGFs.
Interestingly, we demonstrated that GCF sIL-6R concentrations correlated statistically to the GCF calprotectin concentrations (Figure 1(c)). To investigate the biological relationship between sIL-6R and calprotectin, we examined whether sIL-6R production is induced by calprotectin in cultured macrophages, because macrophage is one of important inflammatory cells that regulate periodontal inflammation. 24 As shown in Figure 4, we have shown for the first time that PMA-differentiated THP-1 macrophages treated with calprotectin, but not P.g LPS, induced sIL-6R production significantly. In addition, P.g LPS, but not calprotectin, induced IL-6 production significantly in macrophages. Although it is difficult to understand the whole picture of cytokine network in periodontitis lesions, at least in part, calprotectin-mediated macrophages might be a candidate of sIL-6R-producing cells and have agonistic effects of IL-6 to the surrounding cells such as fibroblasts. Our clinical data have shown that GCF calprotectin concentrations are significantly higher in the inflamed sites than in the healthy sites (Figure 2(d)). Although it is still unclear whether calprotectin induces VEGF production directly in several periodontal cells, we have considered that calprotectin might induce VEGF production indirectly through the sIL-6R induction from macrophages, resulting in clinically positive BOP when examined by the dentists. The series of findings proved that both GCF sIL-6R and calprotectin concentrations are clinically significant to distinguish between inflamed and healthy sites of periodontal tissues in patients with periodontitis.
This study had several limitations. First, because all patients were assessed at our hospital, this study is limited inherently by the single-centre selection bias. Second, no adjustments were made for other factors which may impact progression of periodontitis during SPT phase such as age and comorbidities of other diseases. Third, since our clinical study is analysed by a cross-sectional design, we cannot discuss the relation of cause and effect among several parameters.
Periodontitis is characterized as a region-specific disease. Recently, the increase of patients with systemic diseases requiring non-invasive minimal procedure during periodontal treatment has been recognized as a severe problem in the dental field. 25 Development of diagnostic skill is required to distinguish periodontal health and inflammation non-invasively. Taken together, both GCF sIL-6R and calprotectin concentrations are useful biomarkers in the evaluation of periodontal inflammation in patients with periodontitis. We believe that our approach contributes to create a new tool for the maintenance of periodontal health in the future.
Footnotes
Acknowledgements
We thank Omar Rodis (Tokushima University) for critical reading of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by Grant-in-Aid for Scientific Research (B) (No. 15H05054), Scientific Research (C) (No. 16K11832) and Grant-in-Aid for Young Scientists (B) (No. 16K20673) from the Japan Society for the Promotion of Science.
Ethical approval
This study was approved by the ethics committee of Tokushima University Hospital (No. 2325) and written informed consent was obtained from each subject.
Guarantor
KN.
Contributorship
YK performed in vitro assay, statistical analysis and wrote the article. JHL, TI, YN recruited patients, collected samples and measured cytokine concentrations. JK, TN participated in data interpretation. KN planned the study design, supervised the analysis and revised the article.