Abstract
Background
Squamous cell carcinoma antigen is used as a tumour marker and is routinely measured in clinical laboratories. We validated two different immunoassays and found three cases in which the squamous cell carcinoma antigen concentrations deviated greatly between the two immunoassays. Here, we aimed to elucidate the mechanisms responsible for these deviations.
Methods
The squamous cell carcinoma antigen concentrations were determined using the ARCHITECT SCC (CLIA method) and the ST AIA-PACK SCC (FEIA method). We performed polyethylene glycol precipitation and size exclusion chromatography to assess the molecular weight and spike recovery and absorption tests to examine the presence of an autoantibody.
Results
Both methods exhibited good performances for the measurement of squamous cell carcinoma antigen, although a correlation test showed large differences in the squamous cell carcinoma antigen concentrations measured using the two methods in three cases. The results of polyethylene glycol treatment and size exclusion chromatography indicated the existence of a large molecular weight squamous cell carcinoma antigen in these three cases. The spike recovery tests suggested the possible presence of an autoantibody against squamous cell carcinoma antigen. Moreover, the absorption test revealed that large squamous cell carcinoma antigen complexes were formed by the association of squamous cell carcinoma antigen with IgG in two cases and with both IgG and IgA in one case.
Conclusions
This study describes the existence of large molecular weight squamous cell carcinoma antigen that has complexed with immunoglobulin in the serum samples. The reason for the deviations between the two immunoassays might be due to differences of their reactivities against the squamous cell carcinoma antigen immune complexes with their autoantibody. To our knowledge, this is the first report to describe the coupling of squamous cell carcinoma antigen with IgA.
Introduction
Squamous cell carcinoma antigen (SCCA), known as a member of the serine protease inhibitor family, was originally isolated from human cervical squamous carcinoma tissues. 1 SCCA consists of two isoforms, SCCA1 and SCCA2, which share a 92% homology in terms of their amino acid sequences; the molecular weights of SCCA1 and SCCA2 are both about 45 kDa.2,3
Serum SCCA concentrations are elevated in patients suffering from various squamous cell carcinomas including those of the cervix, lung, and head and neck. In clinical laboratory medicine, the serum SCCA concentration is used for diagnosis, assessments of the efficacy of treatment, and predictions of the recurrence of these malignancies.4–8 SCCA is detected in normal squamous epithelial tissues, 9 and elevated SCCA concentrations are also found in patients suffering from benign skin diseases such as atopic dermatitis and psoriasis.10–12 SCCA was originally measured using a radio immunoassay method; at present, however, it is commonly measured using a non-radio immunoassay method, enabling its use as a routine clinical laboratory test. Nevertheless, measurements based on immunoassay techniques, including SCCA measurements, generally have some problems that remain to be solved. One of these problems is interference from serum proteins and antibodies, such as heterophilic antibodies, which can lead to falsely high or low results.13–19 Another problem is the difference in reactivities against antigen among different kits produced by various manufacturers, since each immunoassay uses different specific antibodies.
We have experienced three cases in which the SCCA concentrations differed considerably when measured using two different SCCA kits during a validation of SCCA measurements. Therefore, in the present study, we investigated the mechanisms responsible for the deviations in the SCCA concentrations in these three cases.
Material and methods
Measurement of SCCA concentrations
The serum samples used in this study were residual samples of those obtained after the completion of routine laboratory analyses. The residual serum samples were stored at −20℃ until analysis. The SCCA concentrations were measured using the following two methods: ARCHITECT SCC (Abbott Japan Co. Ltd, Tokyo, Japan), which is based on a chemiluminescent microparticle immunoassay (CLIA method) and ST AIA-PACK SCC (Tosoh Corporation, Tokyo, Japan), which is based on a fluorescent enzyme immunoassay (FEIA method).
ARCHITECT SCC is a two-step sandwich immunoassay; 25 µL of a serum sample and 50 µL of anti-SCCA mouse monoclonal antibody coated with paramagnetic microparticles in 2-morpholinoethanesulfonic acid buffer were incubated for 18 min at 37℃. After washing step with a phosphate-buffered saline solution, 50 µL of acridinium-labelled anti-SCCA mouse monoclonal antibody in 2-morpholinoethanesulfonic acid buffer was added and incubated for 4 min. Following a second washing step, 100 µL of a solution containing 1.32% (wt/vol) hydrogen peroxide was added to the reaction mixture and the background intensity was measured. Then, 300 µL of a solution containing 0.35 N sodium hydroxide was added and the chemiluminescent intensity (approximately 400–500 nm) was measured to calculate the SCCA concentration.
ST AIA-PACK SCC is a one-step sandwich immunoassay; lyophilized anti-SCCA mouse monoclonal antibody coated with magnetic beads and anti-SCCA mouse monoclonal antibody conjugated to bovine alkaline phosphatase were reconstituted with 100 µL of diluent buffer containing TritonX-100 in a plastic test cup. Then, 25 µL of a serum sample was added to the test cup and incubated at 37℃ for 10 min. After washing with a solution containing Tween 20, the enzyme-labelled antibody which bound to the beads was reacted with 4-methylumbelliferyl phosphate (fluorogenic substrate). The fluorescence intensity of fluorescent substance (4-methylumbelliferone) was measured at an excitation wavelength of 363 nm and at an emission wavelength of 447 nm to calculate the SCCA concentration.
Polyethylene glycol precipitation
Polyethylene glycol (PEG) precipitation was performed as described previously. 20 Briefly, the serum samples were mixed with an equal volume of 25% (wt/vol) PEG6000 solution (Wako Pure Chemical Industries Ltd, Osaka, Japan) or distilled water and centrifuged at 3500 r/min for 30 min. The supernatants were then collected, and the SCCA concentrations were measured using the CLIA method. The results were expressed as the ratio of the SCCA concentration after treatment with PEG6000 solution to that after treatment with distilled water.
Size exclusion chromatography
The serum samples were applied to a 7.8 mm × 30 cm TSKgel G3000SWXL column (Tosoh Corporation) and eluted with 0.1 M phosphate buffer containing 0.1 M NaCl and 0.1% NaN3, pH 7.0, at a flow rate of 0.5 mL/min. The eluted fractions were collected every minute from 11 min to 30 min. A serum sample that exhibited approximately the same SCCA concentration when measured using both immunoassays (the CLIA method, 61.5 ng/mL; the FEIA method, 57.9 ng/mL) was used as a control.
Spike recovery test
A ST AIA-PACK SCC CALIBRATOR SET (Tosoh Corporation) was used as a recombinant SCCA. A calibrator not containing SCCA was added to the serum samples as a reference. The recombinant SCCA (83.3 ng/mL) and serum samples were mixed at a ratio of 1:9. After the addition of the SCCA calibrator, serum samples were incubated at room temperature for 2.5 h. The recovery rate was represented as the rate of the measured concentration to the expected concentration. The spike test for SCCA in the size exclusion chromatography (SEC) fraction was performed in the same manner. Briefly, recombinant SCCA (49.8 ng/mL) was added to each fraction at a ratio of 1:9 and the fractions were incubated overnight at room temperature. The SCCA concentrations were then determined using the FEIA method.
Absorption test of immunoglobulin
Anti-human immunoglobulin antibody was utilized for the absorption of immunoglobulin binding SCCA; 10 µL of serum samples and 100 µL of goat anti-IgG (H + L chain) (Human) antibody (Medical & Biological Laboratories Co. Ltd, Nagoya, Japan), goat anti-IgM (µ chain) (Human) antibody, goat anti-IgA (α chain) (Human) antibody, or saline were mixed and incubated overnight at 4℃. After centrifugation, the SCCA concentrations were measured using the CLIA method. The recovery rates were represented as the ratio of the SCCA concentration after incubation with each antibody to that after incubation with saline.
Results
Validation of SCCA measurements using the CLIA method and the FEIA method
First, we measured the recombinant SCCA concentrations in a dilution series of concentrations using both the CLIA method and the FEIA method. The results showed that the recombinant SCCA concentrations measured using the CLIA method were higher than those measured using the FEIA method (y = 0.618 x + 0.357) (Figure 1(a)), while the dilution linearity indicated a good performance for the quantification of SCCA for both methods.
Correlation between SCCA concentrations determined using the CLIA method and using the FEIA method and the time courses of the SCCA concentrations. The SCCA concentrations of recombinant SCCA (a) and of 123 serum samples (b) were determined using the CLIA method and the FEIA method. The x-axis shows the SCCA concentrations measured using the CLIA method, and the y-axis shows the SCCA concentrations measured using the FEIA method. (a) Primary regression equation: y = 0.618 + 0.357; correlation coefficient, r = 0.999. (b) Primary regression equation: y = 0.673x − 0.456; correlation coefficient, r = 0.899. The SCCA concentrations of case1, 2, and 3 are represented as cross, closed triangle, and closed square, respectively. Those of other samples are represented closed circle. The time courses of SCCA concentrations in case1, 2, and 3 are shown in (c), (d), and (e), respectively. The SCCA concentrations determined using the CLIA method (closed square) and the FEIA method (open circle). The x-axis shows days or months after the initiation of the treatment.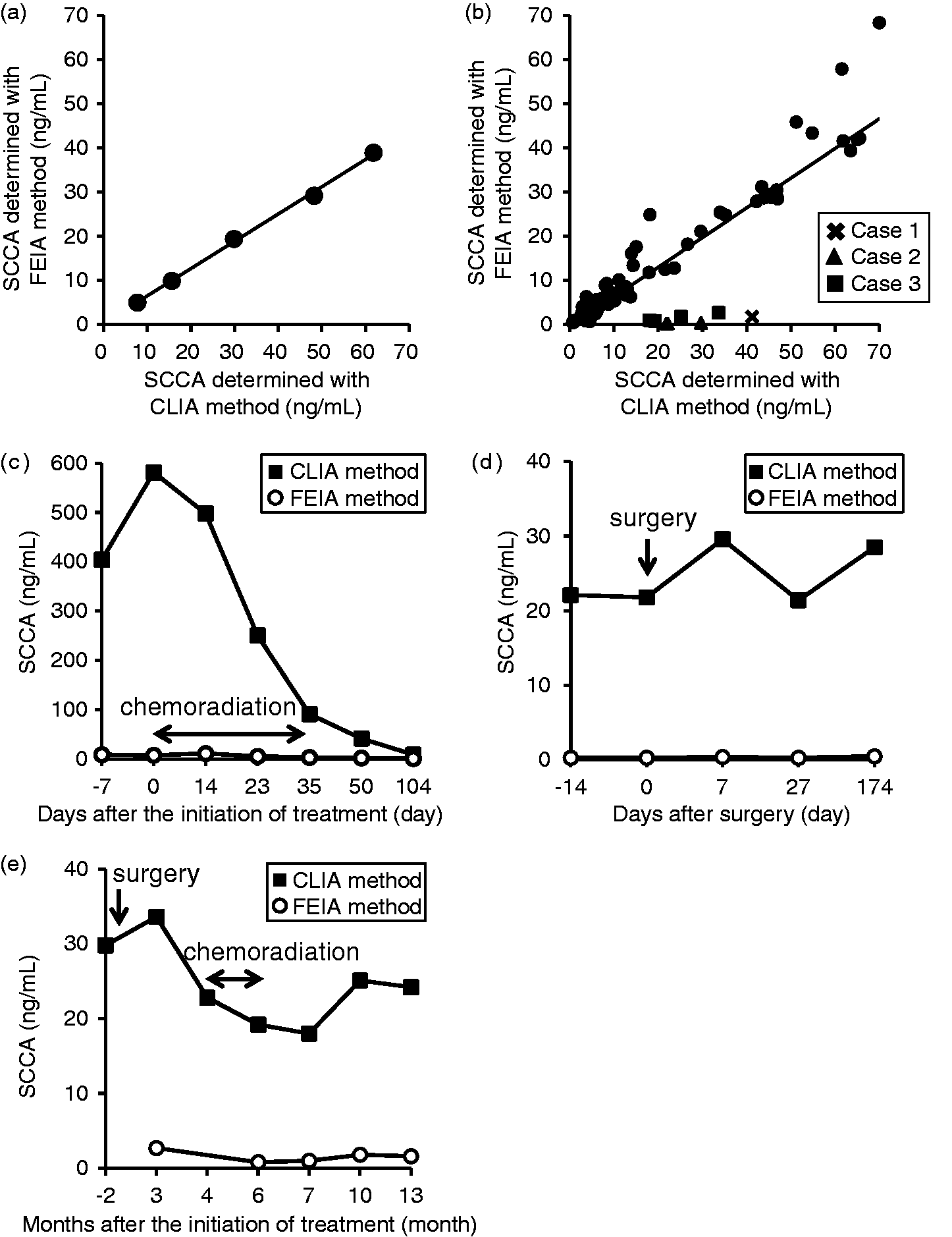
Correlation between SCCA concentrations determined using the CLIA method and those determined using the FEIA method in human serum samples
We next compared the SCCA concentrations determined using the CLIA method with those determined using the FEIA method for 123 serum samples. Although the SCCA concentrations of the serum samples were somewhat different between those measured using the CLIA method and those measured using the FEIA method (y = 0.673 x − 0.456), the SCCA concentrations in eight samples from three patients were extremely different; in these eight samples, the SCCA concentrations determined using the CLIA method were much higher than those determined using the FEIA method (Figure 1(b)), even when compared with the correlation for recombinant SCCA or the correlation observed in other serum samples. Case 1 represents a subject with relapsed esophageal cancer (pretreatment; the CLIA method 581.5 ng/mL, the FEIA method 7.3 ng/mL). Although SCCA concentrations measured with each method were quite different for all over the course, SCCA concentrations determined with both methods gradually decreased with chemoradiation (at 104 days after the initiation of the treatment; the CLIA method 8.5 ng/mL, the FEIA method 0.6 ng/mL) (Figure 1(c)). Case 2 represents a subject diagnosed with a thymoma. Preoperative SCCA concentration determined using the CLIA method was 22.1 ng/mL, while that determined using the FEIA method was 0.3 ng/mL. Although there were no evidence of the recurrence of the tumour after the curative surgical resection, the SCCA concentrations determined with both methods remained unchanged (the range of SCCA concentrations; the CLIA method 21.4–29.6 ng/mL, the FEIA method 0.3–0.5 ng/mL) (Figure 1(d)). Case 3 represents a subject who suffered from mandibular gingival cancer, hypopharyngeal cancer, and esophageal cancer simultaneously and SCCA concentrations determined using the CLIA method were much higher than those determined using the FEIA method and remained still high without any recurrence after the operative resection (Figure 1(e)).
Low recovery rates after PEG precipitation in deviated samples
Recovery rate after PEG precipitation in serum samples from three cases and a control.
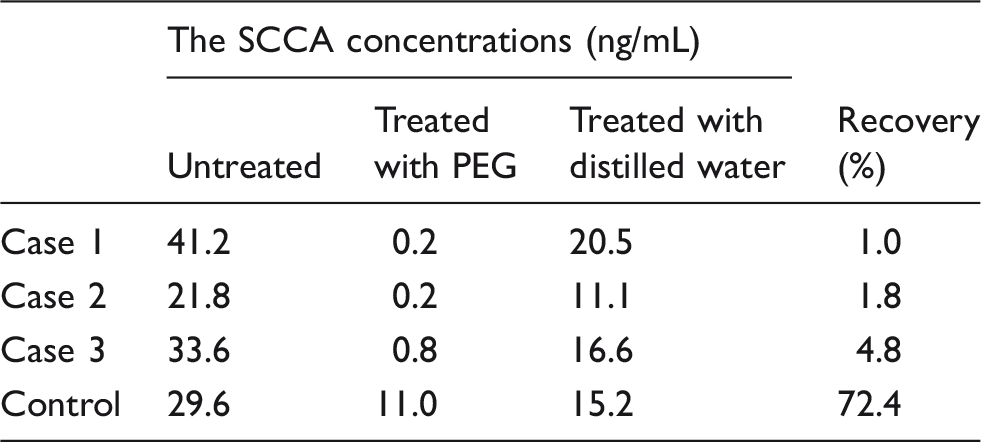
PEG: polyethylene glycol.
Evaluation of molecular size of SCCA using SEC
We next examined the molecular weight of SCCA using SEC. The recombinant SCCA and the control sample were eluted in the fraction obtained at around 21 min, the peak size of which was slightly smaller than an ovalbumin peak (approximately 47 kDa) (Figure 2(a) and (b)). In cases 1 and 3, the SCCA was eluted not only at the same molecular weight position as the control sample and the recombinant SCCA but also in a fraction corresponding to a size larger than the IgG peak (approximately 150 kDa) (Figure 2(c) and (e)). In case 2, the peak for SCCA was observed in a fraction corresponding to a size larger than the IgG peak (Figure 2(d)). These results suggested the presence of large molecular weight SCCA in these samples, indicating that the SCCA might have formed an immune complex with immunoglobulin, which can be detected with the CLIA method but not with the FEIA method.
Size exclusion chromatography showing the molecular size distribution of SCCA. The SCCA concentrations were determined using the CLIA method (closed square) and the FEIA method (open circle). SEC was conducted using recombinant SCCA (a) and serum for which similar SCCA concentrations had been obtained using the CLIA method and the FEIA method (b). SEC results for serum samples from cases 1, 2, and 3 are shown in (c), (d), and (e), respectively. The SCCA concentrations of the serum samples before SEC are shown above the figures. The arrows indicate the elution times of the molecular size markers, IgG (approximately 150 kDa) and ovalbumin (approximately 47 kDa).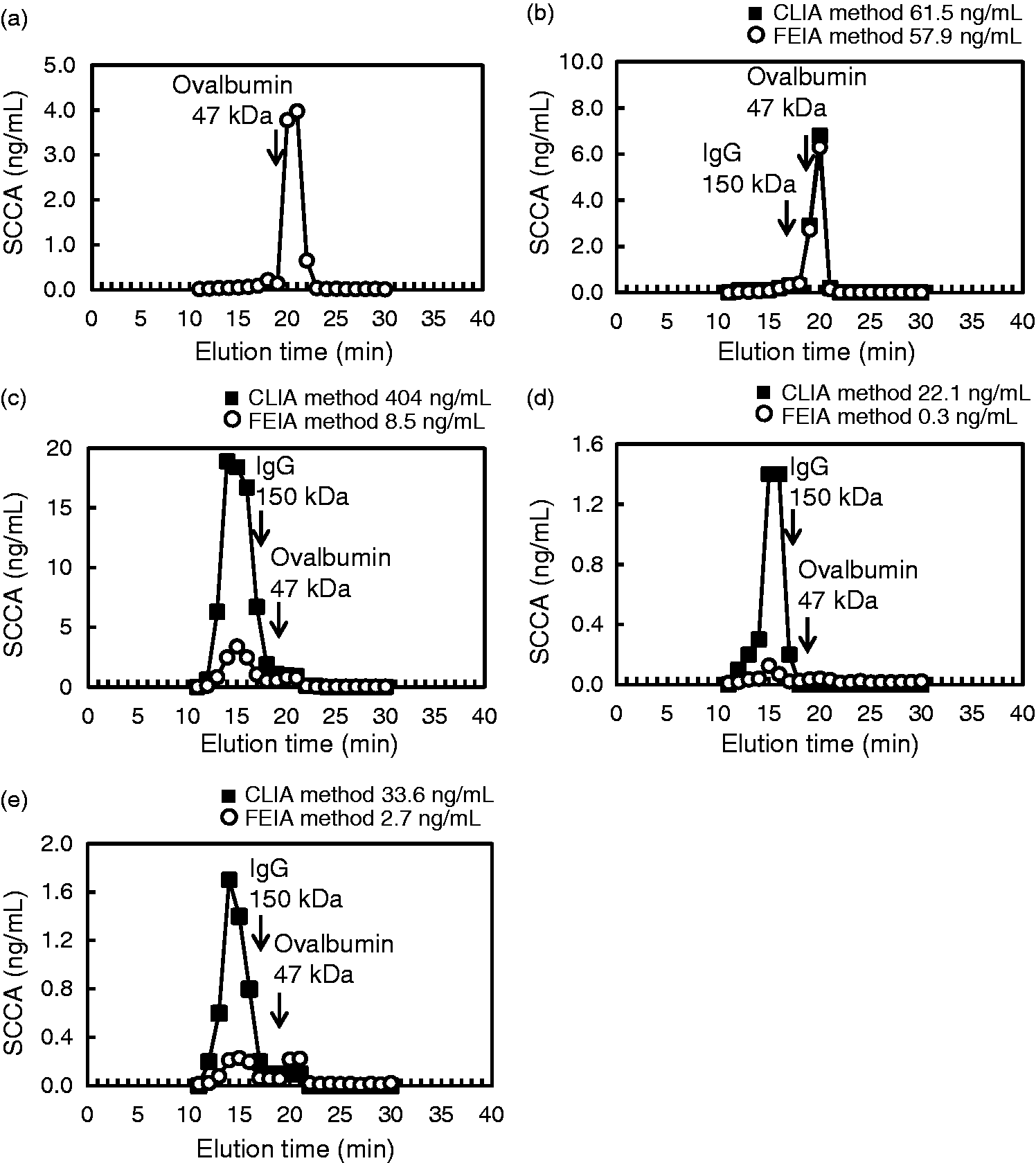
Detection of anti-SCCA antibody in patients’ serum samples using spike recovery tests
To investigate the possible presence of autoantibody against SCCA, we performed spike recovery tests. We used two samples as controls (control 1 and control 2) which exhibited similar SCCA concentrations determined using both the CLIA method and the FEIA method. The SCCA concentrations of control 1 and control 2 measured using the FEIA method were matched with cases 1 and 3 (control 1) and case 2 (control 2), respectively.
Recovery rate after spiked recombinant SCCA.
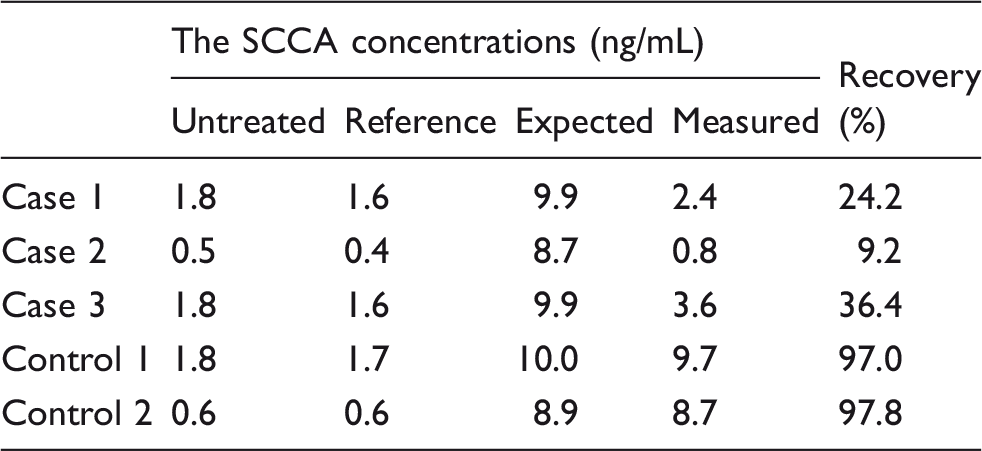
Spike tests using SEC-separated fractions
We next examined which SEC-separated fractions affected the added recombinant SCCA. The added recombinant SCCA was well recovered in the control sample (Figure 3(a)), suggesting that any fraction did not affect recombinant SCCA. Unlike the control sample, the concentrations of the added recombinant SCCA determined using the FEIA method were lower in the fractions corresponding to the molecular size of IgG (approximately 150 kDa) in cases 1 and 2 (Figure 3(b) and (c)). The added recombinant SCCA concentrations were not obviously reduced in any fraction in case 3 (Figure 3(d)). This result suggested that excess immunoglobulin bound to SCCA might exist, at least in cases 1 and 2.
Spike tests using SEC-separated fractions. Recombinant SCCA was added to each SEC-separated fraction from the control (a), case 1 (b), case 2 (c), and case 3 (d). The SCCA concentrations of each fraction before the addition were analyzed using the CLIA method (closed square) and the FEIA method (open circle). The SCCA concentrations of each fraction after the addition of recombinant SCCA were measured using the FEIA method (open triangle). The arrows indicate the elution times of the molecular size markers, IgG (approximately 150 kDa) and ovalbumin (approximately 47 kDa).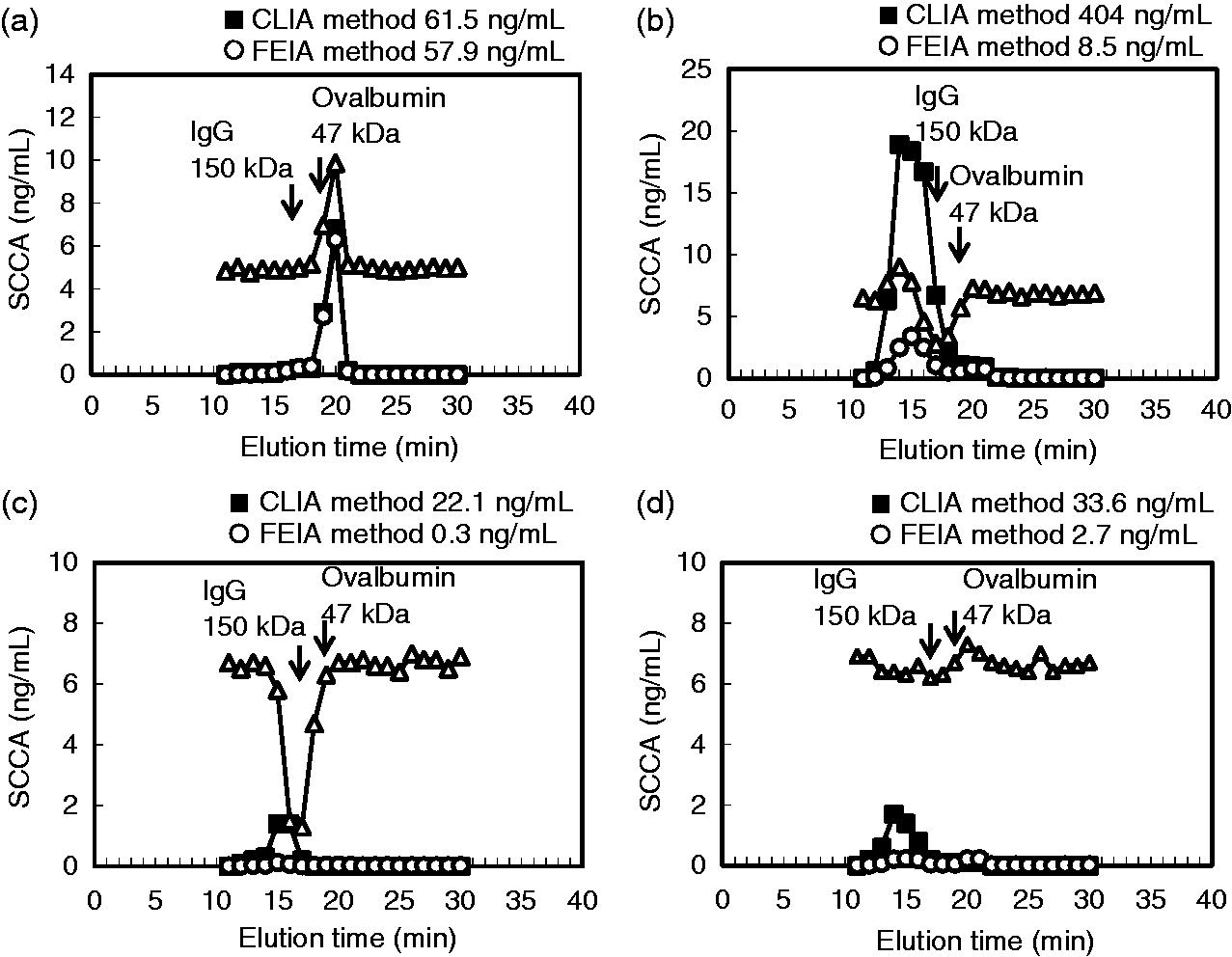
Absorption test for immunoglobulin using anti-human immunoglobulin antibody
Effects of SCCA concentrations on the absorption of anti-human immunoglobulin antibody.
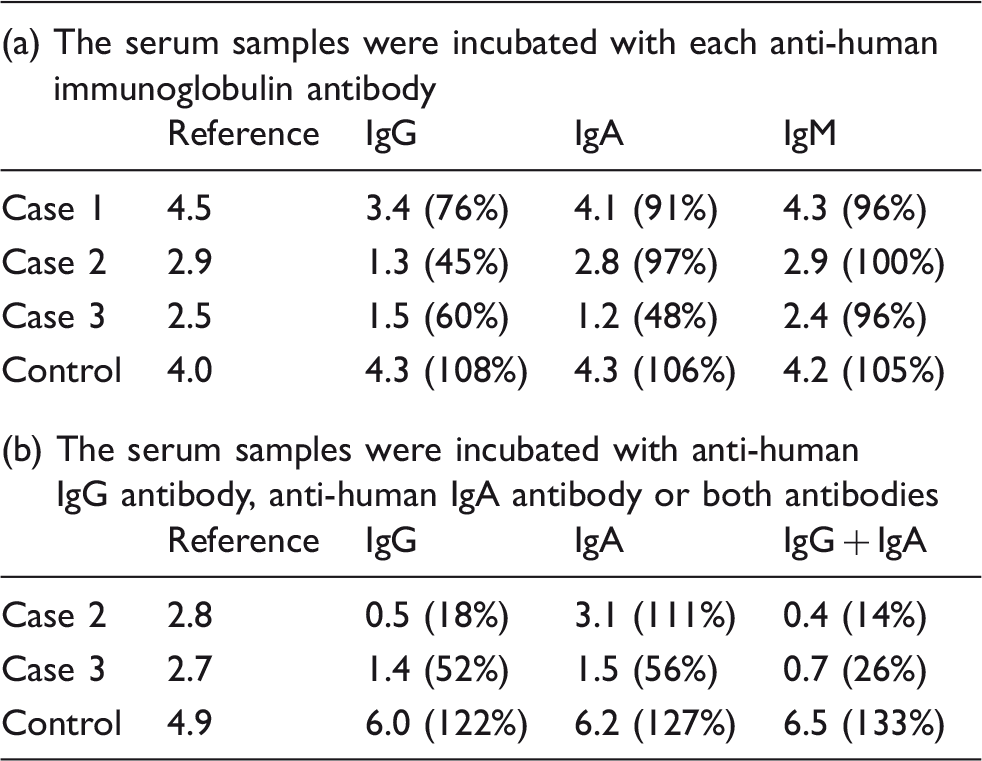
Note: Each datum shows the SCCA concentrations (ng/mL). Recovery rate is shown in brackets.
To determine the involvement of IgG and IgA with SCCA in case 3, we further performed an absorption test using a combination of anti-human IgG antibody and anti-human IgA antibody. As shown in Table 3b, case 2 exhibited the same absorption level after incubation with only anti-human IgG antibody and after incubation with both antibodies, while the recovery rate in case 3 was decreased to a greater degree when the serum was incubated with both antibodies than when it was incubated with each antibody alone. These results suggest that a large molecular SCCA was formed by association with IgG in cases 1 and 2, while association with both IgG and IgA occurred in case 3.
Discussion
In this report, we experienced three deviated cases when we examined the correlation between the SCCA concentrations determined using the CLIA method and that determined using the FEIA method (Figure 1(b)), and the SCCA concentrations determined using the CLIA method in these cases remained unexplainable high after treatment (Figure 1(c) to (e)). We demonstrated that the deviations could be explained by the existence of large molecular weight SCCA bound to immunoglobulin (Tables 1 to 3, Figures 2 and 3). Moreover, in one case, the result from the absorption test suggested the possible existence of SCCA bound to IgA (Table 3), which has not yet been reported, to our knowledge.
Regarding the performance of the assays for the measurement of SCCA, both the CLIA method and the FEIA method were deemed to possess a sufficient capacity necessary for clinical laboratory testing, while a correlation test showed that the values determined using the CLIA method were higher than those determined using the FEIA method (Figure 1(a)), which might reflect a possible difference in setting standard SCCA value between the two methods. As shown in Figure 1(b), however, the SCCA concentrations of the deviated cases departed even from the correlation slope of the diluted recombinant SCCAs between the two methods. Since the serum concentrations of thyroid-stimulating hormone (TSH) were reportedly deviated between different immunoassays in a patient with an immune complex consisting of TSH and its autoantibody, 21 we suspected that the reason for these deviations observed in these cases might also be caused by the existence of an immune complex of SCCA with its autoantibody. To demonstrate the presence of SCCA bound to its autoantibody, we first performed a PEG precipitation assay and an SEC assay and found that large molecular SCCAs might exist in the three deviated cases (Table 1, Figure 2). From these results, we hypothesized that the deviation observed in the three cases might be due to the difference in reactivity to the immune complex form of SCCA between the two methods. Actually, the results of the SEC assay suggested that immune complexes of SCCA in the three deviated cases were only detected using the CLIA method, and not with the FEIA method. One possible explanation for the different performances in detecting the immune complexes of SCCA is that the epitope of SCCA, which is recognized by the anti-SCCA antibody used in the FEIA method, might be masked by autoimmunoglobulin.
We also demonstrated the presence of immunoglobulins that had reacted with SCCA using the spike recovery test (Table 2), suggesting that the subtype of immunoglobulin complexed with SCCA was IgG in cases 1 and 2 and both IgG and IgA in case 3 (Table 3). Interestingly, the spike recovery test showed different results when recombinant SCCA was added to the serum and when it was added to the SEC-separated fraction in case 3; thus, the patient’s immunoglobulin, probably consisting of IgG and IgA, could bind to SCCA in the serum, but not in the SEC-separated fraction. We think that the reason for this difference was the different conditions underlying the reaction in the serum and elution buffer. Moreover, the recovery rate of the spike recovery test was slightly higher in case 3 than for the other two cases. Therefore, in case 3, the binding ability of immunoglobulin to SCCA was weaker than in the other two cases.
Until now, the existence of immune complexes of tumour markers with their autoantibodies has been reported for some tumour markers, including prostate-specific antigen and α-fetoproteins.22–25 An important clinical problem regarding tumour markers is their low diagnostic specificity for target tumours. However, some reports have suggested the possibility that the combination of an immune complex form and a free form may provide more useful information in terms of diagnosis.23–25 Regarding SCCA, the presence of SCCA bound to IgM in serum from patients with hepatocellular carcinoma has been recently reported, 26 and some studies have suggested the possibility that SCCA bound to IgM might be a useful marker for hepatocellular carcinoma and chronic liver disease.27–29 Since the total SCCA concentration is not higher in subjects with hepatocellular carcinoma, the immune complex form of SCCA might be a novel biomarker for some cancers other than typical squamous cell carcinoma.
The results of the present study suggested the existence of an immune complex of SCCA with IgG autoantibody in three cases and with IgA autoantibody in one case. Although a previous study reported that IgG autoantibodies in serum from patients with psoriasis react with SCCA in psoriatic stratum corneum, 30 this is the first report of IgA autoantibody binding to SCCA in the patient’s serum.
In summary, we found three cases in which the SCCA concentrations measured using the CLIA method deviated from those measured using the FEIA method. The results from the present study indicate that large molecular weight SCCA complexed with IgG (two cases) and IgG plus IgA (one case) existed in the serum samples from these three deviated cases. Therefore, the reason for the deviation observed between the two immunoassays might be due to differences in the reactivities to immune complexes of SCCA with its autoantibody.
Footnotes
Acknowledgements
We would like to thank Mr. Kazuhiro Shimazu (Tosoh Corporation) for technical assistance with the SEC assay.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Nissui Pharmaceutical Corporation Ltd (Tokyo, Japan) and by the JSPS KAKENHI (Grant Number JP16H00608).
Ethical approval
This study was approved by the Institutional Research Ethics Committee of the Faculty of Medicine, The University of Tokyo (Number 3333-95).
Guarantor
YY.
Contributorship
EM collected samples, performed the experimental work and analyzed the data. EM and MK wrote the first draft of the manuscript. AT and HS participated in several experiments. YY conceived the study, coordinated the study design and helped to draft the manuscript. All the authors reviewed and edited the manuscript and approved the final version of the manuscript.
