Abstract
Background
Patients with metal-on-metal hip replacements require testing for cobalt and chromium. There may also be a need to test for titanium, which is used in the construction of the femoral stem in total hip replacements. It is not possible to use quadrupole inductively coupled plasma mass spectrometry due to interferences.
Methods
Titanium was measured using inductively coupled plasma optical emission spectroscopy using the emission line at 336.1 nm and Y (internal standard) at 371.0 nm. Internal quality control materials were prepared for blood and serum and concentrations assigned using a sector field-inductively coupled plasma mass spectrometer. A candidate whole blood certified reference material was also evaluated.
Results
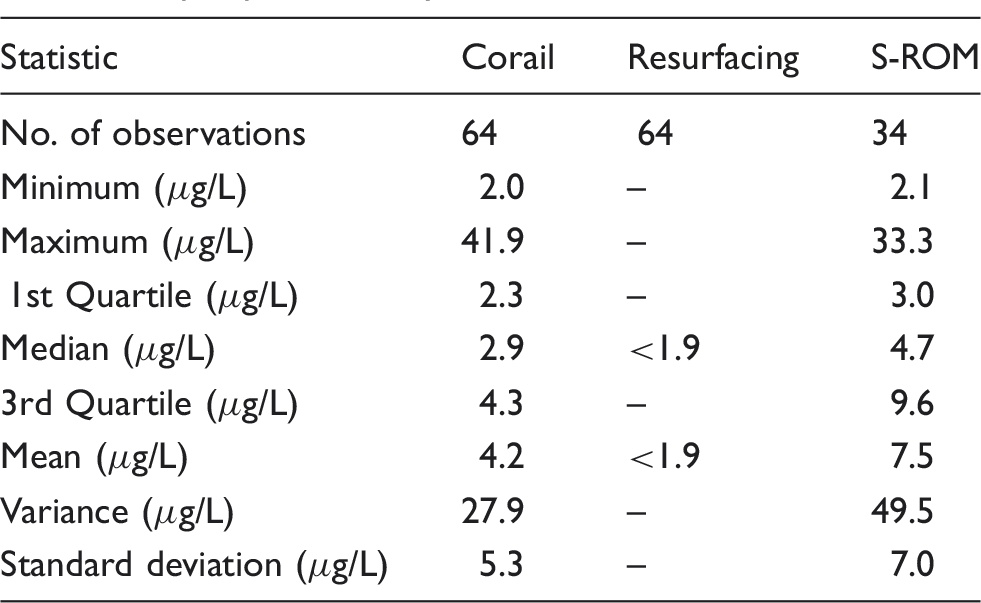
The method had detection and quantitation limits of 0.6 and 1.9 µg/L, respectively. The respective bias (%) and measurement uncertainty (U) (k = 2) were 3.3% and 2.0 µg/L (serum) and − 1.0% and 1.4 µg/L (whole blood). The respective repeatability and intermediate precision (%) were 5.1% and 10.9% (serum) and 2.4% and 8.6% (whole blood). The concentration of titanium was determined in patients’ samples, serum (median = 2.4 µg/L, n = 897) and whole blood (median = 2.4 µg/L, n = 189). Serum is recommended for monitoring titanium in patients, since the concentration is higher than in whole blood and the matrix less problematic. In hip fluid samples, the concentrations were much higher (mean 58.5 µg/L, median 5.1 µg/L, n = 83).
Conclusions
A method based on inductively coupled plasma optical emission spectroscopy was developed and validated for measuring titanium in clinical samples.
Keywords
Introduction
In terms of enhancing quality of life, the restoration of hip function using artificial hip replacements for patients suffering from a loss of motion, pain and discomfort is one of the most successful orthopaedic operations undertaken. 1 Common reasons for replacing the hip joint are damage caused by osteoarthritis, dysplasia, osteonecrosis and hip fractures, which commonly occur in older females as a complication of osteoporosis or a fall.1,2 The replacement of failing hip joints has, in general, had a positive effect on the quality of life.
There are two main approaches to hip-replacement surgery using metal-on-metal (MoM) systems: total hip replacements (THRs), where the ball of the femur is removed and replaced with a stemmed femoral ball component; and resurfacing arthroplasty, where the femur is capped or re-surfaced with a hollow metal head. 3 A variety of materials are used in the construction of joint-replacements, but the most common is a cobalt-chromium-molybdenum (Co-Cr-Mo) alloy (ASTM F75, approximate ratio 63:30:6). 3 Other materials and components of the implant used include titanium (Ti) stems and cup shells, along with plastic and ceramic parts. Although there are different implant systems currently in use, based on the materials used as the bearing surface, they fall into four main categories: metal-on-plastic (MoP); ceramic-on-ceramic; ceramic-on-plastic and MoM.3,4
All materials that are used in hip implants will wear with use and release wear particles. 2 This is true of MoP and MoM implants, and although MoM articulations release a greater number of particles than the number of particles released by MoP joints, the actual volumetric wear of MoM joints is usually lower. In general, joints using metal components release particulates in the nm range, whereas MoP-generated polyethylene particles are µm in diameter. 1 Corrosion, particularly of the stem, will also result in the release of metal ions, in particular Ti, 3 which is the main metal used for the manufacture of the stem used in THR surgery. The presence of metal and polyethylene wear particles in the area around the joint can lead to local inflammation and soft tissue damage.5,6 This can ultimately lead to failure of the hip replacement, destruction of the bone and muscle and a poor prognosis for the patient.
There are a variety of possible causes for failure of the prosthetic hip, but a common feature in all of them is the generation of metal wear particles and metal ions. 7 The generation of metal ions has been reported to reach very high concentrations: 50 times control for blood concentrations of Co and 100 times for Cr. 7 This is of increasing concern as MoM THRs were primarily developed for younger and more active patients many of whom may require a second implant, and in extreme cases these concentrations may well have toxicological endpoints such as deafness, loss of vision and cardiomyopathy, most likely due to high Co concentrations.8,9 The concentration of Ti in patients with MoM THRs has not been widely investigated clinically due to the paucity of reliable analytical methods available; however, information on the concentrations of Ti in these patients may be clinically useful, particularly those showing signs of tissue damage but with low concentrations of whole blood Co and Cr.
In 2010, the UK Medicines and Healthcare products Regulatory Agency (MHRA) issued a Medical Device Alert (MDA/2010/033) concerning all MoM hip replacements. 10 The problem identified related to a small number of implanted patients, who had developed progressive soft tissue reactions to the wear debris associated with MoM articulations. The action required by this document was that certain categories of patient with this type of hip replacement should be considered for the measurement of blood Cr and Co. In cases in which the result for either of the elements was greater than seven parts per billion (ppb) (7 µg L−1; 135 nmol L−1 Cr, 120 nmol L−1 Co), a second measurement should be performed three months after the first. In 2012, this was updated by further guidance, providing recommendations for follow-up of both symptomatic and asymptomatic patients. 11
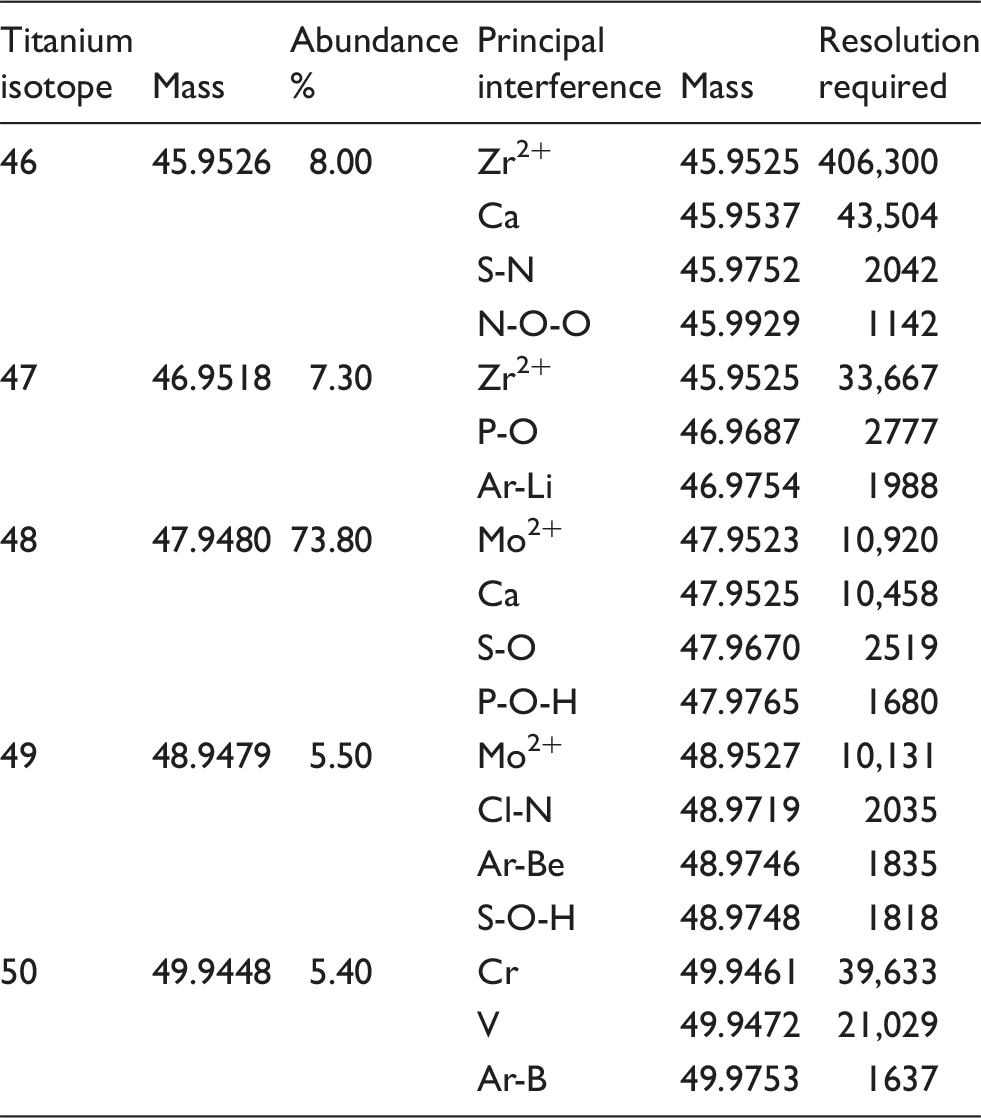
Possible isobaric and polyatomic interferences for the Ti isotopes.
A more practical and cost-effective solution would be the use of inductively coupled plasma optical emission spectroscopy (ICP-OES), a technique related to ICP-MS but which uses the emission lines from the analyte of interest rather than the ions generated in the plasma. The technique has been applied to the measurement of Ti in blood, urine and hair 17 and in a human spleen biopsy sample 18 from patients with hip replacements. It has the advantage of low limits of detection, long linear ranges and simultaneous multielemental capabilities. The analysis is not affected by any specific spectroscopic interference, and so a method was developed for the measurement of Ti in samples from patients with MoM THRs. We have used this method successfully for the routine measurement of Ti in hip-replacement patients for over four years. Here, we report on measured Ti concentrations for the serum, whole blood and hip aspirate samples from a large population of orthopaedic patients undergoing routine monitoring.
Materials and methods
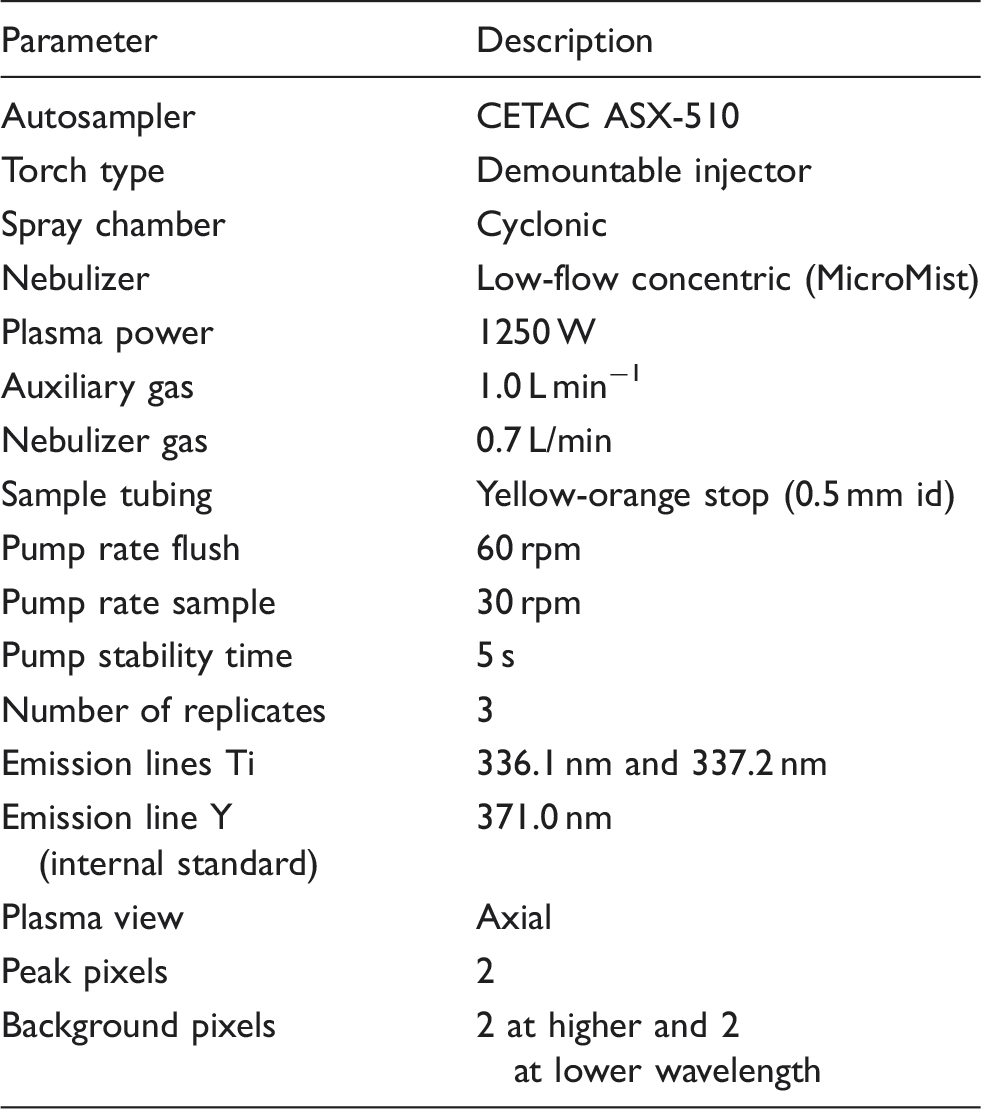
Instrumentation
Instrumental operating conditions for the iCAP 6300 MFC series ICP-OES instrument.
Reagents
Single element stock solutions (1000 mg/L) of Ti and Y traceable to NIST (Certipur, Merck) were used to prepare calibration standards. All solutions were prepared in dilute HNO3 (0.5% v/v) prepared using trace element free concentrated acid (Primarplus, Trace Element Grade, Fisher) and reverse osmosis deionized water > 18.2 MΩ cm resistivity (Millipore, Synergy System). Prior to the analysis of patient samples, a solution of Zn (2 mg/L) traceable to NIST (Certipur, Merck) was used to check the system performance, as recommended by the instrument manufacturer. The IQC materials were prepared from equine whole blood in EDTA (TCS Biosciences Ltd.) and bovine calf serum (Selbourne Biological Services). Commercially available serum and blood reference materials (Seronorm L2, Sero) and a candidate certified reference material (LGC8276, LGC) were also used.
Sample preparation
Whole blood samples were supplied in collection tubes containing EDTA to prevent clotting. Blood samples were sent for measurement of Ti in a variety of tube types including trace element Vacuette tubes (Greiner Bio-One, Stonehouse, UK), Monovette (Sarstedt, Leicester, UK) and Vacutainer (BD, Oxford, UK). Serum samples were supplied by the sender in secondary tubes after sample preparation in the hospital requesting the analysis. Hip-aspirate samples were collected in the clinic from the fluid accumulated around the hip prosthesis using a syringe, and the samples obtained were stored in white-topped Universal tubes (25 mL, Teklab, Durham, UK). All samples were refrigerated (4–8℃) until analysis, usually within five days of taking the sample.
All the different sample types were diluted 1:5 with a diluent made up of 0.5% v/v HNO3 acid containing the internal standard Y at a concentration of 20 µg/L. After dilution, the blood and hip-aspirate samples were centrifuged (Rotanta 460) at 3000 rpm for 10 min to reduce the potential for blockage of the nebulizer or torch injector by organic debris in the samples.
Calibrant and IQC preparation
Intermediate Ti standards with concentrations of 0, 0.5, 1.0, 2.0, 4.0, 8.0 and 16.0 µg/L were prepared in reverse osmosis water by dilution from an intermediate stock standard (10 mg/L). The calibration standards were made from these by diluting 1:5 with the diluent used for the samples, so that the internal standard concentration was the same in both standards and samples. The blood and serum IQC specimens were also diluted 1:5 with the same diluent. The instrument software generated a linear regression fit for the data using an unweighted linear equation and provided values for the slope, intercept and correlation coefficient.
Quality control material validation
In-house whole blood and serum IQC samples were prepared from equine whole blood and bovine calf serum, both spiked gravimetrically at 8.0 µg/L with Ti. Aliquots of these IQCs and a commercially available serum quality control material Seronorm L2 (Ref. 203105, Lot 0903107) with an indicative value for Ti of 12.9 µg/L were analysed by SF-ICP-MS to establish their values. The in-house prepared IQCs were used in the validation of the ICP-OES method and as IQC specimens during routine sample analysis, to confirm the accuracy of the calibration. A candidate whole blood certified reference material (LGC8276, LGC) with an indicative value of 10 ± 1 µg/L for Ti was also used as an additional IQC material.
The analytical protocol used to establish the values for Ti by SF-ICP-MS involved a standard addition calibration with standards of 0.1, 0.2, 0.5 and 1.0 µg/L spiked into Seronorm whole blood L2 reference material and diluted 1:50 with HNO3 (0.5% v/v) and Triton X-100 (0.005% v/v) containing 5 µg/L gallium as internal standard (m/z 71). The instrument was operated in high-resolution mode (R = 10,000) using a Micromist nebulizer (200 µL/min), a Scott-type spray chamber cooled to 5℃ and Ni cones and Ti was measured using the 47Ti isotope.
Method performance evaluation
The accuracy of the ICP-OES method was established by comparison to the values determined by SF-ICP-MS for the in-house prepared serum and blood IQC materials and the indicative value for the candidate certified reference material (CRM). The limits of detection and quantitation were determined by replicate (n = 8) measurement of a standard with a Ti concentration of 1.0 µg/L prepared in the same way as the calibration standards, so with an actual concentration of 0.2 µg/L. The within run precision was used to assess the repeatability by replicate analysis (n = 6) of the IQC materials and the candidate CRM. The precision over a longer time period was used to establish the intermediate precision by replicate (n = 2) analysis of an IQC material on 10 separate days. A Type A evaluation of the intermediate precision data was used to determine the uncertainty of the measurement with a coverage factor k = 2. The linearity of the calibration curve was evaluated by assessment of the correlation coefficient and the linear range for 10 separate experiments. Carry-over was investigated by running the blank preceded by a high concentration sample a number of times (n = 10).
Statistics
Statistical analyses were completed using Excel 2010 (Microsoft Office). Because some samples submitted for analysis were negative controls, i.e. they were samples from patients with hip resurfacings where no Ti is present in the device, it was decided that for statistical analysis samples with a concentration < limit of quantitation (LOQ) were included in the data-set as 0.5 × LOQ (0.75 µg/L). In total, 188 whole blood, 891 serum and 82 hip-aspirate samples were measured, but 102 whole blood, 530 serum and 74 hip-aspirate results were included in the statistical analysis. It is clear from the frequency distribution graphs that none of the data-sets are normally distributed, we have therefore also reported non-parametric statistical measures. For a subset of 16 patients, it was possible to directly compare the serum and whole blood Ti concentrations using regression analysis and analysis of variance to determine the significance of the linear relationship between the two (P = 0.001).
Results and discussion
Establishment of the IQC values
As a result of the limited availability of IQC materials for the measurement of Ti in samples from hip-replacement patients, it was necessary to prepare suitable specimens spiked with Ti at a clinically relevant concentration. These were used during the validation of the method to establish the analytical performance parameters, as well as for the routine measurement of patient samples. A useable number (n = 500) of specimens were prepared from equine whole blood and bovine serum by spiking gravimetrically with 8.0 µg/L Ti, using methods developed in-house and used for the preparation of external quality assurance (EQA) materials used for the UKNEQAS for Trace Elements EQA scheme (www.surreyeqas.org.uk). The use of human blood and serum was considered unethical because of the volume of endogenous material required to provide a suitable number of samples. Due to the low prevalence and heterogeneous nature of the hip-fluid samples, it was not possible to prepare an IQC specifically for this sample type.
To establish the concentration of Ti in the two IQC materials, the final Ti concentration in these specimens were determined by SF-ICP-MS in high-resolution mode (R = 10,000), which can provide the most accurate values, unaffected by the dominant polyatomic interferences found in ICP-MS (Table 1). At this resolution setting the N-, P- and S-based interferences are effectively removed from the 47Ti isotope, and there is still sufficient signal to measure this lower abundance isotope (7.3%). A summary of the interferences for each of the Ti isotopes and the resolution required to remove them are shown in Table 1. The isobaric interference on the most abundant 48Ti isotope (73.8%) from 48Ca (0.2%) makes it impossible to use this isotope for the analysis, because of the high concentration of circulating Ca (2.15–2.55 mmol/L) and the resolution (R = 10,457) required to remove this isobaric interference. The other four Ti isotopes (m/z: 46 (8.0%); 47 (7.3%); 49 (5.5%); 50 (5.4%)) that could potentially be used are also affected by a range of high abundance interferences including: polyatomics; doubly charged ions; and isobaric interferences. 14 SF-ICP-MS is sophisticated and expensive instrumentation and does have the power to resolve the polyatomic species on the Ti isotopes at m/z 47 and 49, but not the isobaric interferences from Ca on m/z 46 (R = 43,504) and 48 (R = 10,457), or Cr (R = 39,632) and V (R = 21,028) on m/z 50.
Using the high-resolution SF-ICP-MS method, serum spiked at 8.0 µg/L Ti was measured as 9.2 µg/L (9.1–9.3 µg/L, n = 9) and whole blood was measured as 8.6 µg/L (8.6–8.7 µg/L, n = 9). The determined values are raised compared with the spike due to the Ti present in the base material. Commercially available Seronorm serum L2 with an indicative value by a single laboratory SF-ICP-MS measurement of 12.9 µg/L for Ti was also analysed, and the measured concentration was found to be 11.4 µg/L (11.1–11.7 µg/L, n = 9). These in-house prepared IQC materials were used to validate the ICP-OES method and also to evaluate the performance of the assay during the routine measurement of patient samples.
Method performance characteristics
Method performance characteristics for the measurement of Ti in orthopaedic samples by ICP-OES at 336.1 and 337.2 nm.
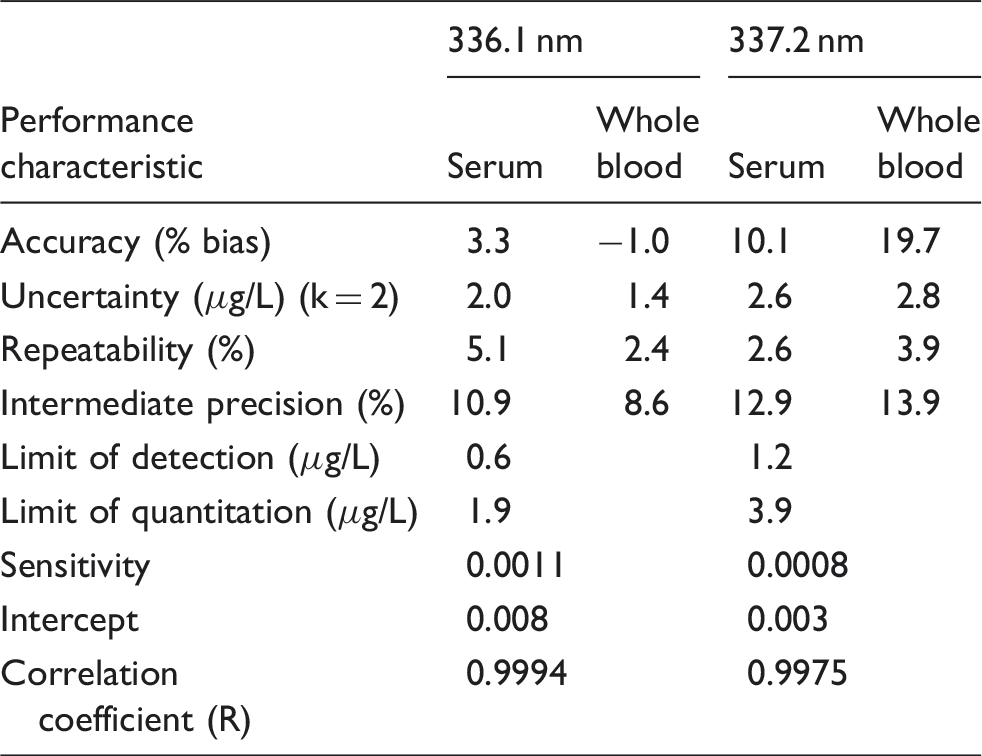
Note: Calibration curve data were for 11 analytical runs on different days with different operators. The values for bias are determined in comparison to the results for the analysis by SF-ICP-MS in high resolution mode.
The LOD for the primary emission line at 336.1 nm was determined by replicate measurement (n = 8) of a low concentration standard and then multiplying the standard deviation by 3 to give a value of 0.6 µg/L which is in reasonable agreement with others 12 using ICP-OES with an ultrasonic nebulizer for the measurement of Ti in human blood plasma, who achieved an LOD of 0.9 µg/L using the same emission line. The LOQ was determined by multiplying the standard deviation by 10, to give a value of 1.9 µg/L. For comparison, these values using SF-ICP-MS 15 were determined to be 0.011 and 0.066 µg/L in MR mode.
Patient sample analysis
Patient results (mean and 95% confidence limits) for Ti in serum, whole blood and hip-aspirates of patients with a range of MoM hip replacements.
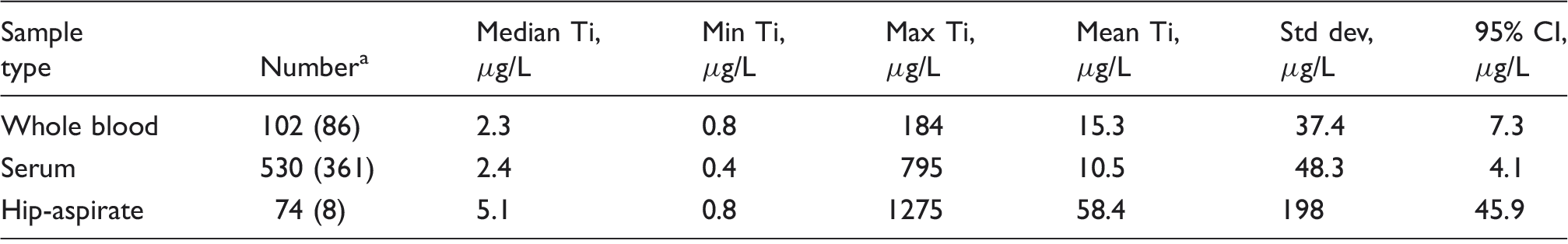
Number of samples included in statistics. The number of samples excluded from these statistics because Ti was not detected is given in parentheses. Samples with values < LOQ were included as 0.75 µg/L (half the LOQ). In total, 188 whole blood, 891 serum and 82 hip-aspirate samples were analysed.
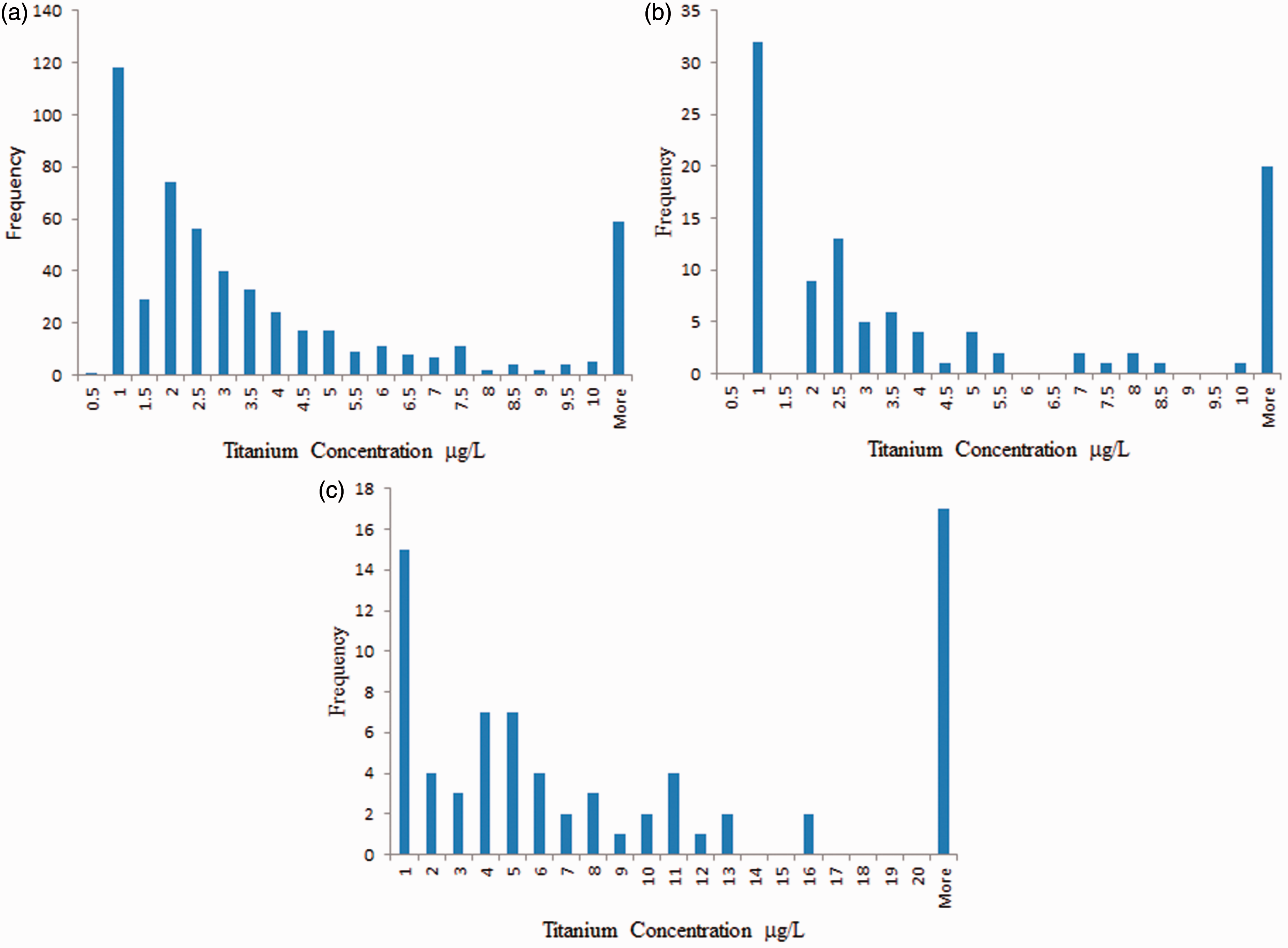
Frequency distributions for Ti in (a) serum (n = 530), (b) whole blood (n = 102) and (c) hip-aspirate samples (n = 74).
The appropriate sample type to assess hip functioning has not yet been established clinically, but in the UK, the MHRA requires analysis of Co and Cr in whole blood for the assessment of joint wear. This minimizes the possibility of sample contamination from Cr during preparation of the serum sample; however, this is not such an issue for Ti which is highly inert and not necessarily present in the device used for venopuncture, unlike Cr. Although we did not systematically investigate the potential for Ti contamination during our study, we found no evidence of Ti contamination, and therefore recommend using serum samples for the assessment of Ti by ICP-OES because on an individual patient basis the concentration is greater than whole blood and the serum matrix is less likely to block the torch injector, which was found to occur when aspirating large numbers of whole blood samples at the dilution level used.
Patient results for Ti in serum of patients with different MoM hip-replacement systems.
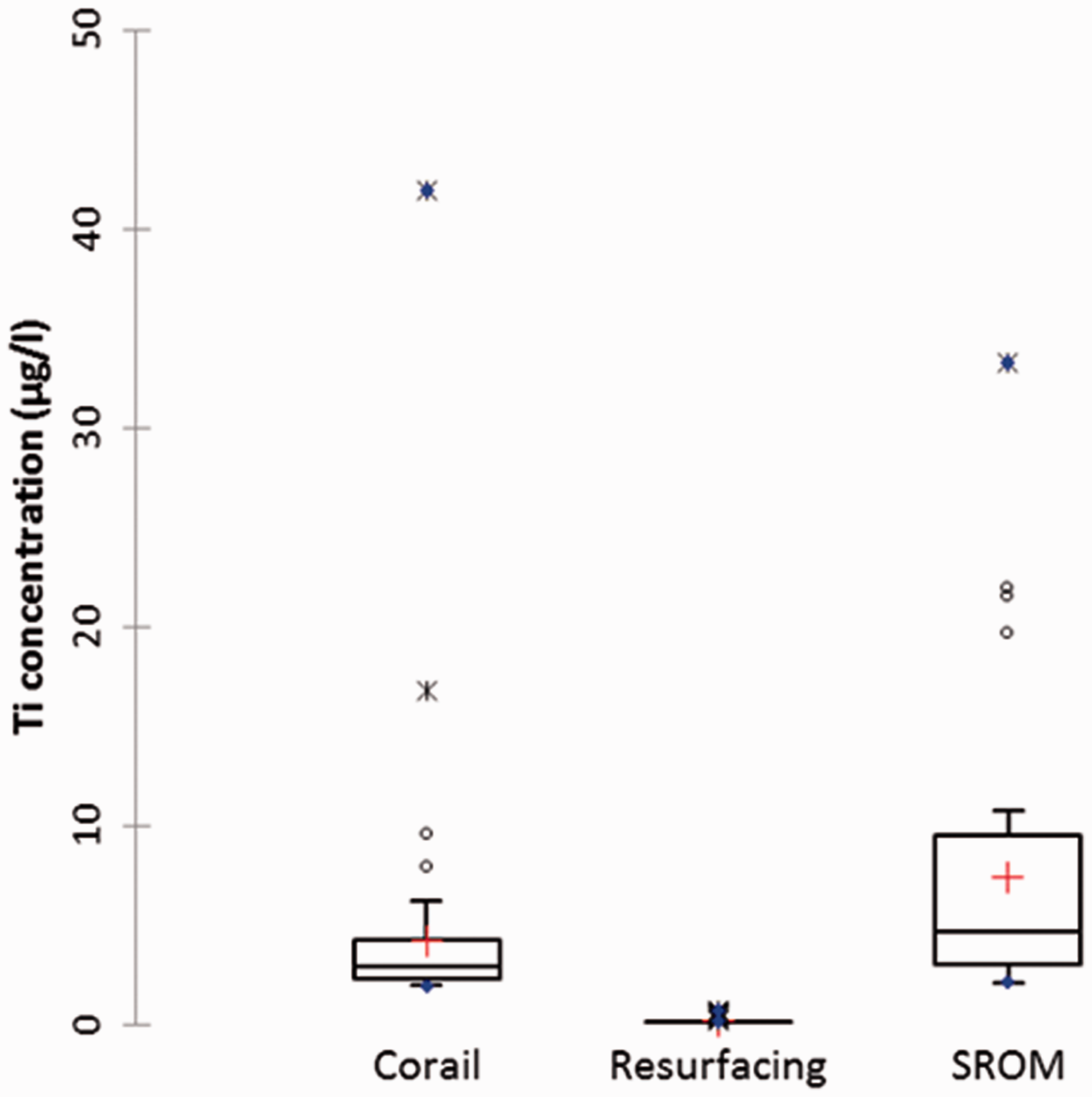
Patient serum Ti results for different MoM hip-replacement systems. The boxes show the interquartile range, the horizontal line in the box is the median value, red cross shows mean value, the other points were removed as outliers.
In conclusion, we have developed a simple and cost-effective method based on the use of ICP-OES, to measure Ti in samples from hip-replacement patients. The method provides a suitable detection limit for the investigation of patients with MoM hip-replacements which contain structures composed of Ti. Using this method, it is now possible to investigate the wear of these joints via the measurement of Ti. In future work, we hope to comment on the clinical significance, if any, of raised Ti in hip-replacement patients.
Footnotes
Acknowledgements
The authors thank Dominique Debellis, Thermo Scientific, for measurement of Ti in the IQC specimens by SF-ICP-MS.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The research was funded internally as part of the instrument validation. Thermo Scientific provided access to a SF-ICP-MS instrument free of charge.
Ethical approval
Not applicable.
Guarantor
CFH.
Contributorship
CFH, DL and AT conceived the study. CMcK and MR undertook initial protocol development. CH validated the method. AT, CFH and DL researched the literature. All authors contributed to the manuscript preparation, review and approved the final version.
