Abstract
Background
Hyperemesis gravidarum, which affects 0.3–2.3% of pregnancies, is defined as excessive vomiting during pregnancy and usually starts in week 4 or 5 of gestation. Symptoms include weight loss, dehydration, ketonaemia, ketonuria, fasting acidosis, alkalosis due to hydrochloric acid loss and hypokalaemia and its exact cause is unknown. The present study was undertaken to investigate the relationship between prealbumin, ghrelin, nesfatin-1 and obestatin concentrations in pregnancies associated with hyperemesis gravidarum.
Methods
A total of 40 pregnant females with hyperemesis gravidarum and 38 pregnant females without hyperemesis gravidarum as controls were included in this study. Serum concentrations of prealbumin, ghrelin, obestatin and nesfatin-1 were measured.
Results
There were no significant differences in age, gestational week, gravidity and parity between the two groups. Body mass index was significantly lower in cases than in controls. Serum ghrelin and prealbumin concentrations were significantly lower in cases than in controls (P <0.05 and P < 0.001, respectively). There was no significant difference in serum concentrations of obestatin and nesfatin-1 between the two groups. There was no significant association between body mass index and serum ghrelin, nesfatin-1, obestatin or prealbumin concentrations in patients with hyperemesis gravidarum.
Conclusions
Decreased serum concentrations of ghrelin and prealbumin in patients with hyperemesis gravidarum are independent of body mass index. Based on our results, we believe that ghrelin may be considered to play a role in the aetiopathogenesis of hyperemesis gravidarum and that hyperemesis gravidarum may result in disruption of the relationship between nesfatin-1 and ghrelin. In addition, we believe that the measurement of serum prealbumin may be used for assessing nutritional status in pregnancy.
Introduction
Hyperemesis gravidarum (HG), which affects 0.3–2.3% of all pregnancies, is defined as excessive vomiting during pregnancy and usually starts at 4 to 5 weeks’ gestation. It may lead to severe symptoms, including weight loss, dehydration, ketonaemia, ketonuria, fasting acidosis, alkalosis due to hydrochloric acid loss and hypokalaemia.1–3 Although the exact cause is unknown, various metabolic and neuromuscular factors have been implicated in its pathogenesis. 2
Ghrelin is synthesized in the fundus of the stomach 4 and has been identified as an endogenous ligand for the growth hormone secretagogue receptor. It is believed to function as an orexigenic (appetite-stimulating) signal from the stomach when an increase in metabolic efficiency is necessary. 5
Nesfatin-1 is an 82-amino acid peptide, recently discovered in the brain which is derived from nucleobindin2. 6 It controls feeding behaviour, water intake and glucose homeostasis. 7
Obestatin, a 23 amino-acid amidated peptide, was initially shown to decrease appetite and weight gain thus opposing the effects of ghrelin. Obestatin was also found to bind to GPR39, although its effects on food intake and ability to activate GPR39 are still controversial. 8
Prealbumin (Transthyretin) is synthesized in the liver and functions as a transport protein. Serum prealbumin differs from albumin, in that it has a relatively short half-life of two days, does not accumulate in the body and does not undergo redistribution. Therefore, when studying a shorter timeframe, measurement of prealbumin is a better indicator to evaluate nutritional status than the measurement of serum albumin concentration, and any fluctuations in nutritional status can be noticed rapidly. 9
The eating process is related to homeostatic and hedonic regulatory mechanisms. Gut-derived hormones are important signals in appetite regulation, and the gut–brain connection plays a key role. 10 Numerous studies have measured serum ghrelin, nesftain-1 and obestatin in relation to eating habit, including conditions such as anorexia, cachexia and obesity.11–13 It has been suggested that the measurement of these hormones may be used as potential therapeutic agents for obesity and eating disorders.14,15
Although HG is closely associated with the loss of appetite, decreased weight gain and negative energy balance, few studies have investigated the possible roles for ghrelin, nesfatin-1 and obestatin in HG.16–19 In addition, there are conflicting results regarding serum ghrelin concentrations in patients with HG.16–19 We believe this to be the first study to investigate the measurement of serum concentrations of prealbumin, ghrelin, nesfatin-1 and obestatin in pregnant females with HG.
Materials and methods
Subjects
This was a case–control study, with a total of 40 pregnant females with HG (cases) and 38 pregnant females who were perfectly healthy and whose pregnancies were following a normal course (controls). All gestational ages were between 6 and 14 weeks, as determined by crown rump-length measurement in the first trimester of pregnancy in cases where there were more than three days’ difference between ultrasonographic measurement and the last menstrual period. The diagnosis of HG was based on the presenting symptoms: persistent nausea and vomiting (>4 times/day), ketonuria >800 mg/L in a random urine specimen and electrolyte imbalance that required intravenous replacement and the need for hospital admission. Those females who had dyspepsia, history of diabetes or gestational diabetes, history of gastric or intestinal surgery, abnormal thyroid function tests, urinary tract infection, previous multiple pregnancies, chronic systemic diseases, history of gastrointestinal surgery, neuro-psychiatric eating disorder and any psychiatric symptoms were excluded from this study. Body mass index (BMI) was calculated as the weight in kilograms divided by the square of height in meters (kg/m2). This study was approved by the Ethical Committee of Diskapi Yildirim Beyazit Education and Research Hospital and written informed consents of the participants were obtained. The study was performed in accordance with the principles enumerated in the Helsinki Declaration of 1975.
Measurement of ghrelin, obestatin and nesfatin-1
After an 8–12 h overnight fast, venipuncture was performed between 8:00 am and 9:00 am, and blood samples were collected into plain tubes for obtaining serum. Blood samples were centrifuged at 2.500 × g for 15 min at 4℃ within 30 min of collection, and serum samples were stored at −80℃ until analysis. Measurements of ghrelin (Phoenix Pharmaceuticals, Inc., USA) obestatin (Biovendor Research and Diagnostic Products) and nesfatin-1 (Phoenix Pharmaceuticals Inc., USA) were performed on an EPOCH analyser (BioTek Instruments, Inc, USA) using the commercially available enzyme-linked immunosorbent assay (ELISA) kits in accordance with the manufacturers’ instructions. The analytical ranges of the assays were: ghrelin, 0–100 ng/mL; obestatin, 0.412–100 ng/mL and nesfatin, 0.78–50 ng/mL.
Measurement of prealbumin
Prealbumin was measured on a Siemens Advia 1800 using the commercially available prealbumin kit (Siemens Healthcare Diagnostics Inc, USA). The analytical range of the prealbumin assay was 50–500 mg/L.
Statistical analysis
Data were presented as mean values with standard deviation (SD) or median with minimum and maximum values of ranges for continuous variable. Normality of distribution was determined for numerical variables using the Kolmogorov–Smirnov test. Data with a normal distribution were analysed using the independent samples test. The Mann–Whitney U test was used to analyse non-normally distributed data. Pearson analysis was used to examine the correlation between parameters. Analysis of covariance (ANCOVA) was used to analyse the differences in ghrelin, nesfatin-1, obestatin and prealbumin concentrations and the covariate factor (BMI).
Receiver-operating characteristics (ROC) curve analysis was performed to determine cut-off points in the prediction of HG. All statistical analyses were performed using the software SPSS 18.0 (SPSS Inc., Chicago, Il, USA) program. For all statistical tests, two-tailed P value <0.05 indicated the statistical significance of the results.
Results
Demographic data of the patients with hyperemesis gravidarum and the control groups.
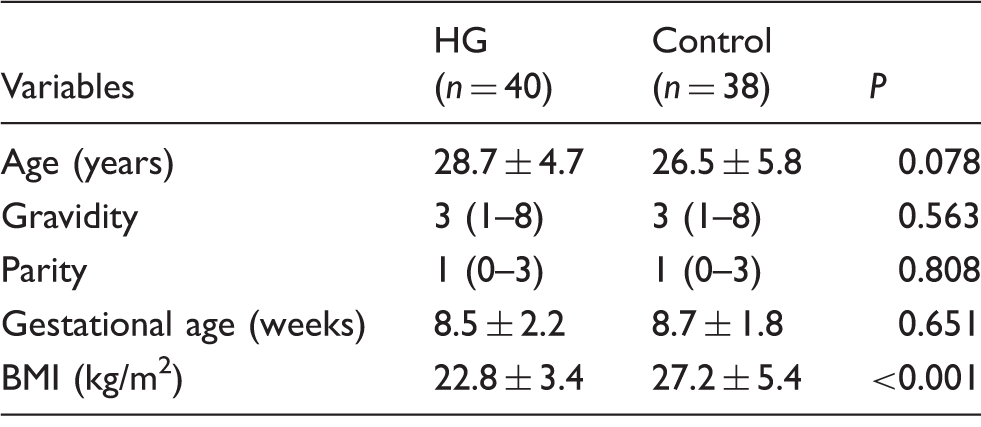
HG: hyperemesis gravidarum; BMI: body mass index.
Biochemical parameters of patients with hyperemesis gravidarum and control groups.

HG: hyperemesis gravidarum; AST: aspartate aminotransferase; ALT: alanine aminotransferase.
Concentrations of serum ghrelin, nesfatin-1, obestatin and prealbumin in females with and without hyperemesis gravidarum.
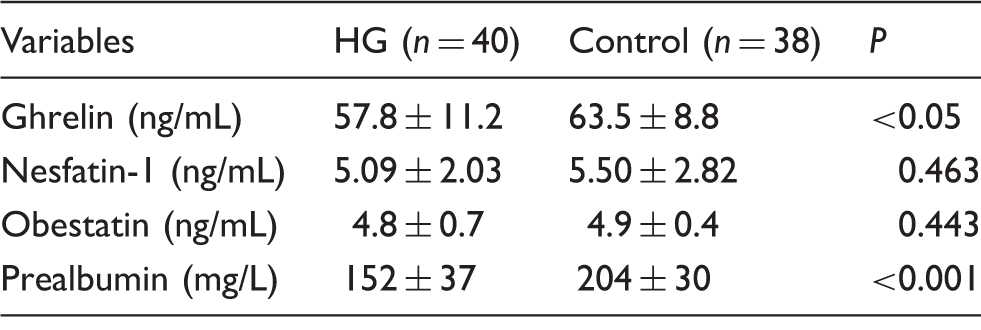
HG: hyperemesis gravidarum.
By ANCOVA, there was no significant association between BMI and ghrelin (F = 0.069, P = 0.793), nesfatin-1 (F = 1.67, P = 0.199), obestatin (F = 0.2, P = 0.656) or prealbumin (F = 0.116, P = 0.735) concentrations in those patients with HG.
Pearson correlations of nesfatin-1, ghrelin and obestatin with other parameters for control groups.
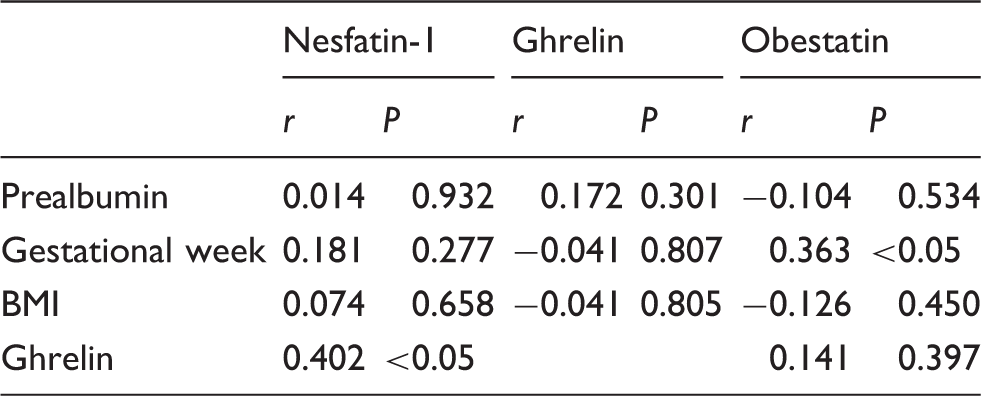
BMI: body mass index.
Effect of ghrelin, nesfatin-1 and obestatin concentrations in prediction of HG.
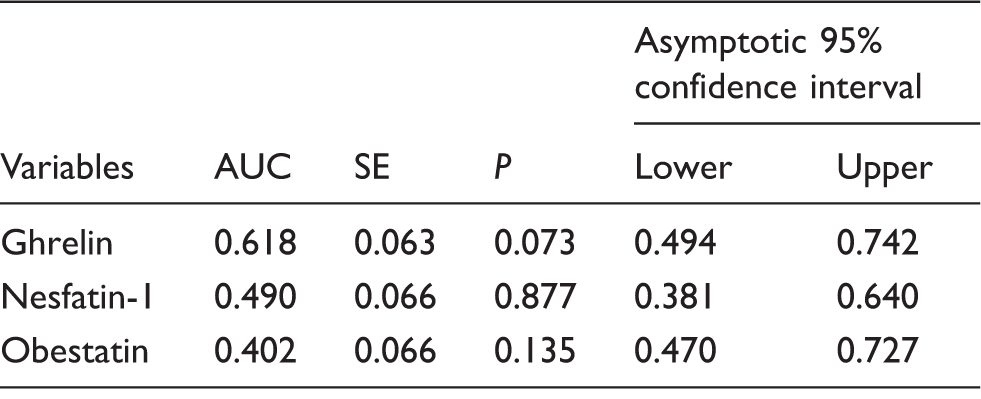
AUC: area under the curve.
Discussion
The aetiology of HG is complex and is based on various metabolic, neuromuscular and psychological factors. 2 In addition, malnutrition, anorexia nervosa, chronic infectious disease, cachexia and other debilitating eating disorders associated with chronic negative energy balance have many clinical features in common with HG.11–13 It has also been shown that a connection between the hormones involved in energy regulation and HG exists.2,17,20
Although the exact pathophysiological mechanisms that cause HG are unclear, it has been suggested that HG is associated with increased concentrations of endocrine hormones, including human chorionic gonadotropin and maternal serum oestrogen. An association with Helicobacter pylori seropositivity and elevated concentrations of serum amylase has also been proposed. Moreover, ethnicity and social factors also play an important role in the development of HG. 21
Ghrelin is a 28-amino-acid orexigenic hormone that stimulates growth hormone release and enhances feeding and weight gain to regulate energy homeostasis. The effects of Ghrelin on food intake and energy homeostasis are largely mediated by arcuate NPY/AgRP neurons. Ghrelin as a key modulator of energy metabolism during negative energy balance and starvation. 22 It is secreted by the stomach and duodenum and is involved in the short term regulation of appetite. 23 Ghrelin and its associated mRNAs are also present in human placenta. 24 Gastric ghrelin production is regulated by nutritional and hormonal factors. Inhibitory signals seem to include somatostatin, interleukin-1beta, growth hormone itself, high-fat diet and vagal tone. 5 The concentration of ghrelin in the circulation rises during fasting and just before a meal and decreases after food intake. 23
There are conflicting reports relating to concentrations of ghrelin in patients with HG. Gungor et al. 17 assessed how concentrations of acyl and des-acyl ghrelin changed in response to HG, and they observed no significant difference between those pregnant females with, and those without HG. 17 On the other hand, some studies have shown that concentrations of ghrelin are significantly increased in patients with HG compared with normal pregnant females.16,19 Unlike previous data, in the present study, we found that serum concentrations of ghrelin were significantly lower in patients with HG, compared with normal pregnant females. We believe that these findings may be interpreted in two ways. First, that low ghrelin concentrations might play a role in the aetiopathogenesis of HG, possibly through causing a decrease in both appetite and weight gain. This may explain the finding of a negative energy balance in pregnant females with HG. Secondly, it may be that in response to HG, ghrelin concentrations fall and this may be related to the duration of hyperemesis. However, longitudinal data are not available either in the literature or in the present study. HG pathogenesis has complicated and multifactorial causes, and further work is required including longitudinal studies of patients in all trimesters of pregnancy. Notwithstanding this, the present study nevertheless involved a larger cohort of pregnant females (n = 78) compared with the maximum number studied in previous reports (max. n = 56).16–19
We analysed the BMI as a covariate for variations in serum concentrations of ghrelin and prealbumin and following ANCOVA found no significant association, implying that the changes in ghrelin and prealbumin concentrations were independent of BMI.
Obestatin is mainly present in the stomach, but its action is focused on appetite inhibition in contrast to the action of ghrelin. 25 It has been shown that the plasma concentrations of obestatin are higher in non-pregnant females than during pregnancy. 26 However, in females who are pregnant, studies have shown that there is no difference in obestatin concentrations between those with HG and those without.17,18 These findings were confirmed in the present study. In addition, in the present study, we observed a positive correlation between obestatin concentration and gestational week in the control group. However, we have been unable to find any confirmation of this finding in the literature. In the present study, we only studied serum concentrations of obestatin in females in the first trimester of pregnancy. This needs to be extended to include females in all three trimesters.
Nesfatin-1, a recently discovered satiety hormone, is produced in hypothalamic nuclei, including the paraventricular, supraoptic, and arcuate nuclei, the lateral hypothalamic area and the nucleus tractus solitarius in the brainstem. 27 Intracerebroventricular injection of nesfatin-1 decreases food intake in a dose-dependent manner, whereas injection of an antibody neutralizing nesfatin-1 stimulates appetite. 28 In one study, Gungor et al. 17 evaluated nesfatin-1 concentrations, in females with HG. Their results showed that nesfatin-1 concentrations in those females with HG were found to be significantly higher than they were in the healthy pregnant group. 17 In contrast, our findings showed no significant difference in nesfatin-1 concentrations in those with HG and those without. However, our findings have also shown a positive correlation between serum concentrations of ghrelin and nesfatin-1 in controls, but not in the HG cases. Stengel et al. 29 demonstrated that NUCB2/nesfatin-1 immunosignals are mainly localized in mucosal endocrine X/A-like cells within a distinct subpopulation of vesicles different from those containing the orexigenic hormone ghrelin in the rat stomach. 29 Inhoff et al. 30 reported that peripheral injection of desacyl ghrelin at a dose preventing the orexigenic action of ghrelin activates NUCB2/nesfatin-1 immunoreactive neurons of the arcuate nucleus. It has been proposed that ghrelin and nesfatin-1 participate in the regulation of energy balance by means of the brain–stomach connection. 31 In light of this information, the relationship between the ghrelin and nesfatin-1 might be disrupted in the females suffering from HG.
We believe this to be the first study to investigate the value of serum prealbumin concentrations in females with HG. The measurement of prealbumin has been validated as a measure of nutritional status among various populations of patients with acute, chronic, and critical illness. It has also been widely used to predict outcomes such as length of hospital stay, development of infection and even mortality. 32 In the present study, we found that serum concentrations of prealbumin were significantly lower in those females with HG compared with controls. It may be proposed that serum prealbumin concentration could be used for assessing nutritional status during pregnancy.
We found a significant elevation of urea nitrogen, haemoglobin, haematocrit and platelets in those females with HG, findings which we believe to be attributable to dehydration.
In conclusion, our study has shown that decreased concentrations of serum ghrelin and prealbumin observed in females with HG are independent of BMI. Based on our results, we believe that ghrelin may be considered to play a role in HG aetiopathogenesis or be reduced in response to HG. We also believe that HG may disrupt the relationship between nesfatin-1 and ghrelin. Finally, measurement of serum prealbumin could be used to assess nutritional status during pregnancy. Further studies with larger numbers of individuals are required on this subject to ascertain whether these ideas are true.
Footnotes
Acknowledgements
The authors would like to thank the research assistants Hulya Ozcan for kind help.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the Ethical Committee of Diskapi Yildirim Beyazit Education and Research Hospital (number: 20.09.2013-10/08).
Guarantor
GO.
Contributorship
GO researched literature and conceived the study. GO gained ethical approval, developed protocol, made data analysis and wrote the first draft of the manuscript. ASOE, ND were involved in patient recruitment and data collection. FU, ZG were involved in literature search and data analysis. GE was involved in writing, critical reviews and literature search. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
