Abstract
Background
To date, the effects of exclusive breastfeeding duration and timing of solid food introduction on allergy prevention are unclear. The aim of this study was to determine the effect of variable feeding practices on intestinal inflammation in infants using faecal eosinophil cationic protein as a surrogate marker and to assess whether faecal eosinophil cationic protein is associated with serum immunoglobulin E.
Methods
Subjects (n = 206) were enrolled from the
Results
Faecal eosinophil cationic protein was significantly higher in exclusively breastfed infants compared with formula-fed infants and infants who were not exclusively breastfed at 6 months of age (P < 0.05). At 12 months, infants who were introduced to solid foods at 5–6 months had the lowest faecal eosinophil cationic protein compared with those who were introduced at earlier and later periods. There was no significant association between faecal eosinophil cationic protein and serum immunoglobulin E.
Conclusion
We found that breastfeeding exclusively for >6 months did not reduce serum immunoglobulin E, but rather increased intestinal inflammation. Faecal eosinophil cationic protein was not associated with total serum immunoglobulin E and specific immunoglobulin E and might not be a useful indictor of immunoglobulin E sensitization in infancy.
Keywords
Introduction
Because of the immaturity of the infant mucosal immune system, exposure to food antigens early in life could greatly influence the development of infant gut immunity. 1 Whether these food antigens would eventually lead to immunoglobulin E (IgE)-mediated sensitization or oral tolerance in young infants is uncertain, and seems to depend on a variety of factors. 2 Breast milk is the most important food to infants. Despite the fact that protection against infectious diseases is observed among breastfed children, 3 it is uncertain if breastfeeding for longer periods is an effective strategy for preventing allergies.4–7 In addition, the timing of solid food introduction for allergy prevention is also a matter of debate.4,8–10 Thus, investigations of infant feeding practices and their association with later atopic diseases are of interest to physicians.
As allergic diseases are not easy to diagnose in young infants, some well-established allergic biomarkers, such as total IgE and allergen-specific IgE, have been used for many years to promptly identify infants with atopic sensitization.11–14 Infants with atopic sensitization have a higher risk of developing allergic diseases in early childhood.15–17
As interactions between feeding practices and gut immunity are complicated and difficult to evaluate, using non-invasive parameters to elucidate intestinal inflammation would be helpful in clinical work. Eosinophil cationic protein (ECP) is an excellent marker for eosinophil activation in various allergic and gastrointestinal diseases. 18 Elevated ECP in serum and/or the presence of ECP in respiratory secretions have been shown to be of value in predicting subsequent wheezing and even asthma in infants.19–21 Additionally, ECP as a faecal marker of intestinal inflammation can help in the diagnosis and monitoring of food hypersensitivity reactions.18,19,22,23 Currently, data regarding faecal ECP levels in healthy infants is scarce, 24 particularly in relation to food practices. 25
In the present study, our first aim was to use faecal ECP as a marker for intestinal inflammation to monitor the intestinal inflammatory reactions between various feeding practices. Our second aim was to investigate the associations between risk factors, feeding practices and IgE sensitization from birth to 12 months of age. Our third aim was to assess if faecal ECP was correlated to serum IgE and if it could be used as an indicator of atopy in clinical work.
Methods
Study subjects
All participants in this study were enrolled from the
A total of 376 infants (10 twins) were enrolled soon after birth in the birth cohort study from March 2012 to October 2013. To avoid potential confounding factors, 39 neonates (7 twins) who had a gestational age of <36 weeks, any perinatal insults or major congenital anomalies were excluded. Eighty participants who dropped out during the one-year follow-up period and 51 participants who did not provide stool samples were also excluded. Finally, our analytical sample included 206 infants (three sets of twins).
Questionnaires
The study participants returned for follow-up clinic visit regularly and were checked by a neonatologist at 2, 4, 6 and 12 months. The parents of the enrolled participants answered the standardized questionnaires under the guidance of well-trained research assistants at all follow-up clinic visits at 2, 4, 6 and 12 months. Detailed information about one or both parents having a physician-confirmed history of asthma, rhinoconjunctivitis, atopic eczema, urticarial and/or food allergy, and other questions regarding the infants' demographic data, general health information, current feeding practices, timing to introduce solid foods and clinical atopic symptoms were also collected.
Determinations of serum allergen-specific and total immunoglobulin E (IgE)
Blood samples (3–5 mL) were collected from each infant at 12 months of age. The allergen-specific IgE was determined by ImmunoCAP™ Phadiatops infant (Phadia, Uppsala, Sweden), and total IgE was measured by ImmunoCAP (Thermo Fisher Scientific, Uppsala, Sweden). Phadiatop infant is an in vitro test for graded determination of immunoglobulin E (IgE) antibodies specific to food and inhalant allergens (i.e. house dust mite, cat, dog, birch, timothy, ragweed, wall pellitory, egg white, cow's milk, peanut and shrimp) that are relevant in the development of atopy in young children.14,26 Infants were characterized as IgE-sensitized if their ImmunoCAP™ Phadiatops levels were ≥ 0.35 PAU/L.14,27
Definitions used in this study
Stool sample collection and processing
The infants' stools were collected in plastic containers within two days before the study participants returned to the clinic for follow-up at 6 and 12 months of age. The methods of stool sample collection and processing have been described in our previous study. 28 Briefly, our research assistants weighed each stool sample and added an extraction buffer containing citrate after stool samples collection. The stool samples were then mixed homogenously and centrifuged for 15 min at 3000 rpm and 4 ℃. The supernatant was collected and frozen at −80 ℃ until it was used.
Determining the levels of faecal ECP by enzyme-linked immunosorbent assays
After thawing the infant stool sample supernatants, faecal ECP concentrations (ng/mL) were determined using an eosinophil cationic protein ELISA kit (MyoBioSource, San Diego, CA, USA), according to the manufacturer's instructions. To derive ECP levels in the infant stool (g), the measured faecal ECP units were adjusted and expressed as nanogram per gram of stool (ng/g).
Statistical analysis
The infants' demographic data were collected by questionnaires and analysed. Numerical variables were summarized by mean ± standard deviation (SD) or frequencies and percentages. Associations between categorical variables were studied by means of the chi-square test. Analysis of variance (ANOVA) test was used to compare continuous variables with normal distribution. The differences between continuous variables with non-normal distribution were estimated with the Kruskal-Wallis test. P-values < 0.05 were considered significant. Correlation coefficients between faecal ECP and serum IgE levels were determined using the Spearman rank correlation test. Statistical analysis was performed using IBM SPSS statistics version 20 (Armonk, NY, USA).
Results
Clinical characteristics
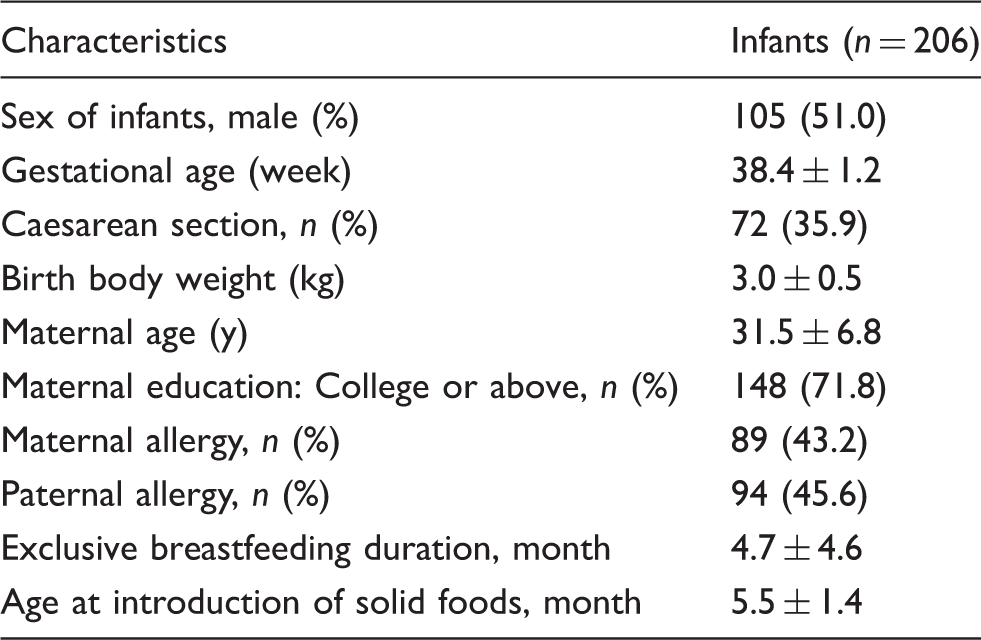
Demographic characteristics of infants (n = 206).
The relationship between feeding practices, faecal ECP, and serum IgE levels
Faecal eosinophil cationic protein and total serum immunoglobulin E levels in relation to feeding practices.
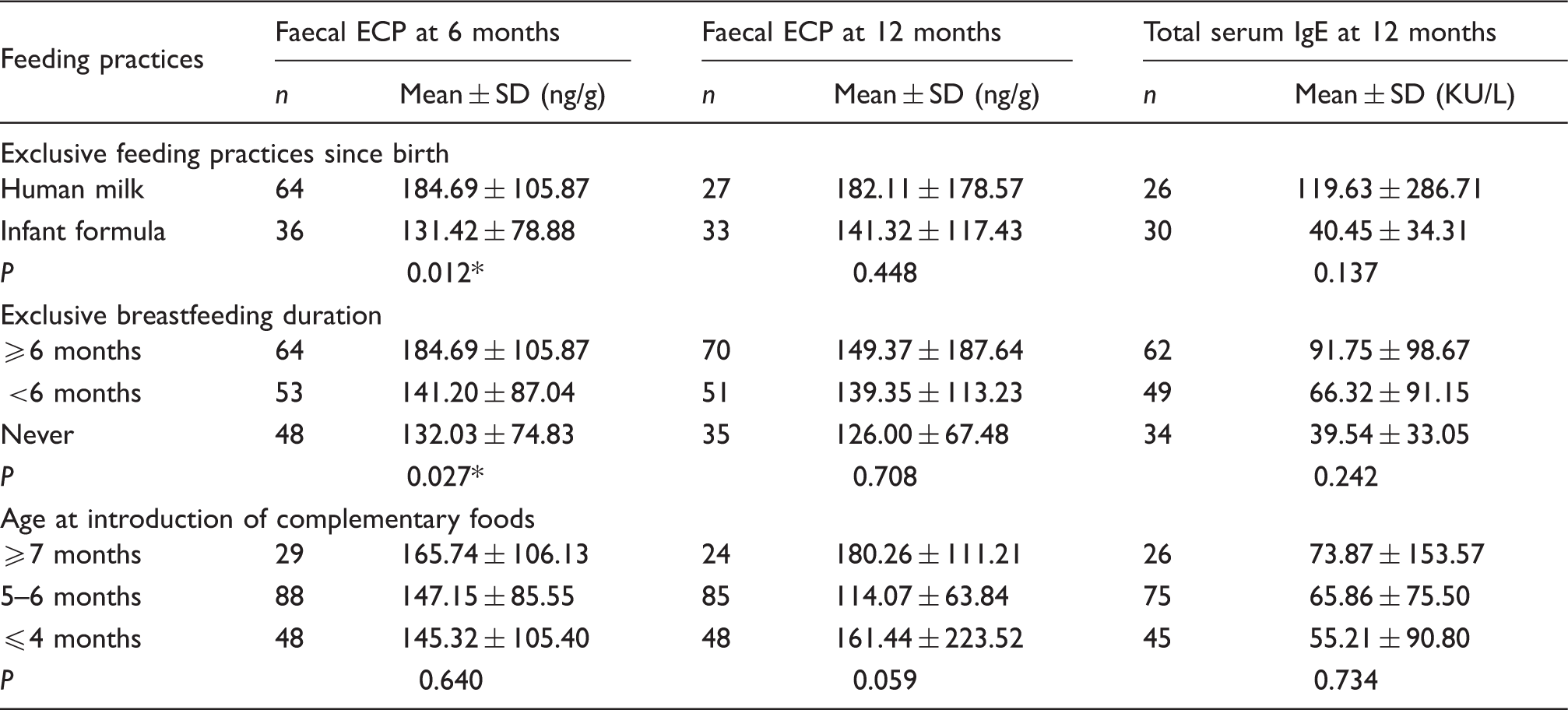
Statistically significant (P < 0.05).
ECP: eosinophil cationic protein; IgE: immunoglobulin E.
For timing to introduce solid foods, the lowest faecal ECP was found in infants who were introduced to solid foods at 5–6 months compared with infants who were introduced to solid foods earlier or later by 12 months of age (P = 0.059). The faecal ECP at 6 months of age and serum total IgE levels at 12 months of age were not affected by the time of solid food introduction (P > 0.05; Table 2).
The characteristics of infants who were IgE-sensitized at 12 months
Description of the characteristics of infants who were or were not IgE-sensitized at 12 months
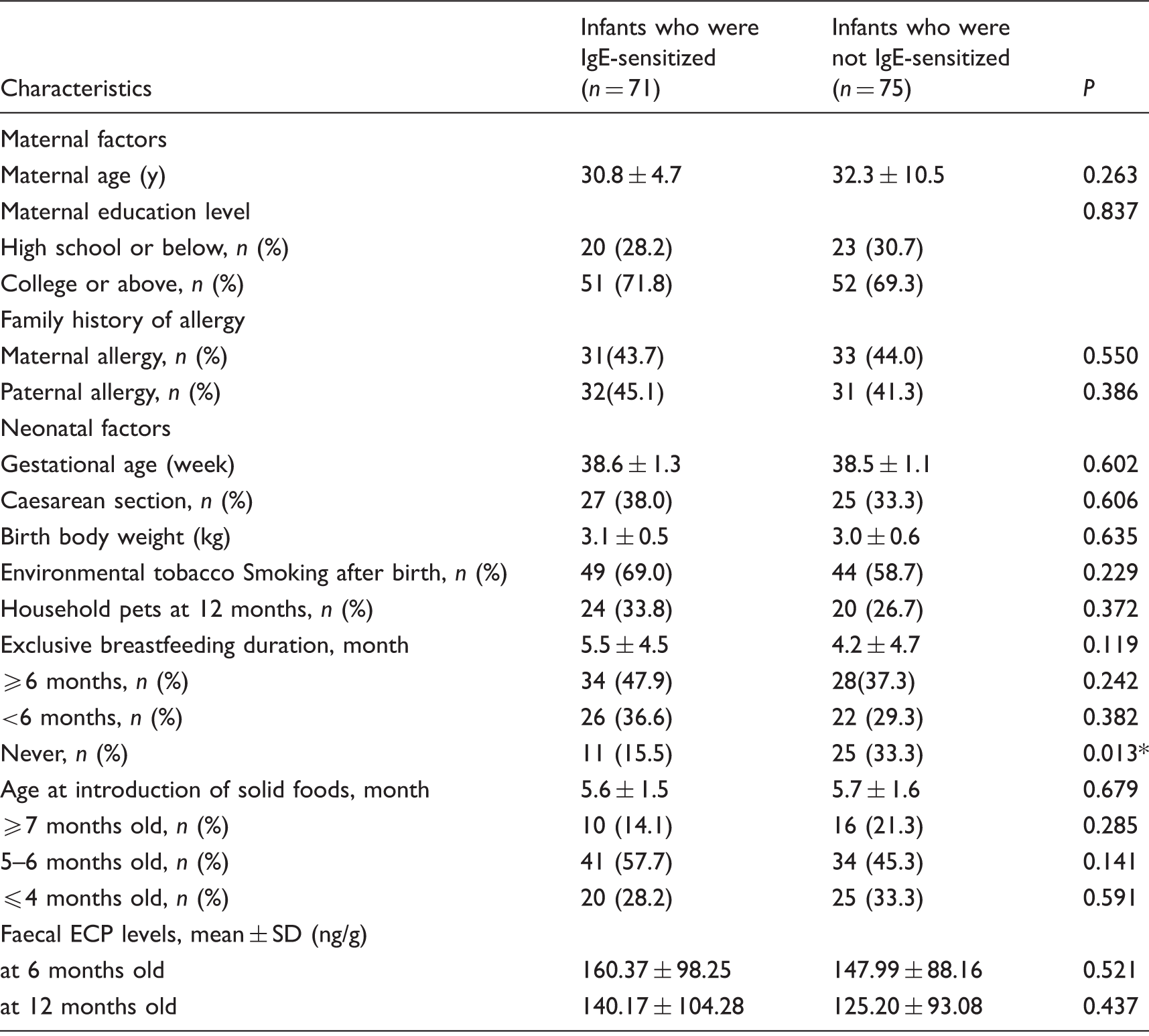
Note: Infants who were IgE-sensitized were those infants who had ImmunoCAP™ Phadiatops levels of ≥ 0.35 PAU/L.
Infants who were not IgE-sensitized were those infants who had ImmunoCAP™ Phadiatops levels of < 0.35 PAU/L. *Statistically significant (P<0.05).
Correlation between faecal eosinophil cationic protein and total serum Immunoglobulin E levels
In assessing the relationship between faecal ECP and total serum IgE levels, only a weak, positive correlation between faecal ECP and serum IgE levels at age 12 months was observed (r=0.124, P = 0.061). There was no statistically significant relationship between faecal ECP levels at 6 months and total serum IgE levels at 12 months (r=−0.08, P = 0.258; Figure 1).
Scatterplot for serum total IgE and faecal ECP levels at 6 and 12-months.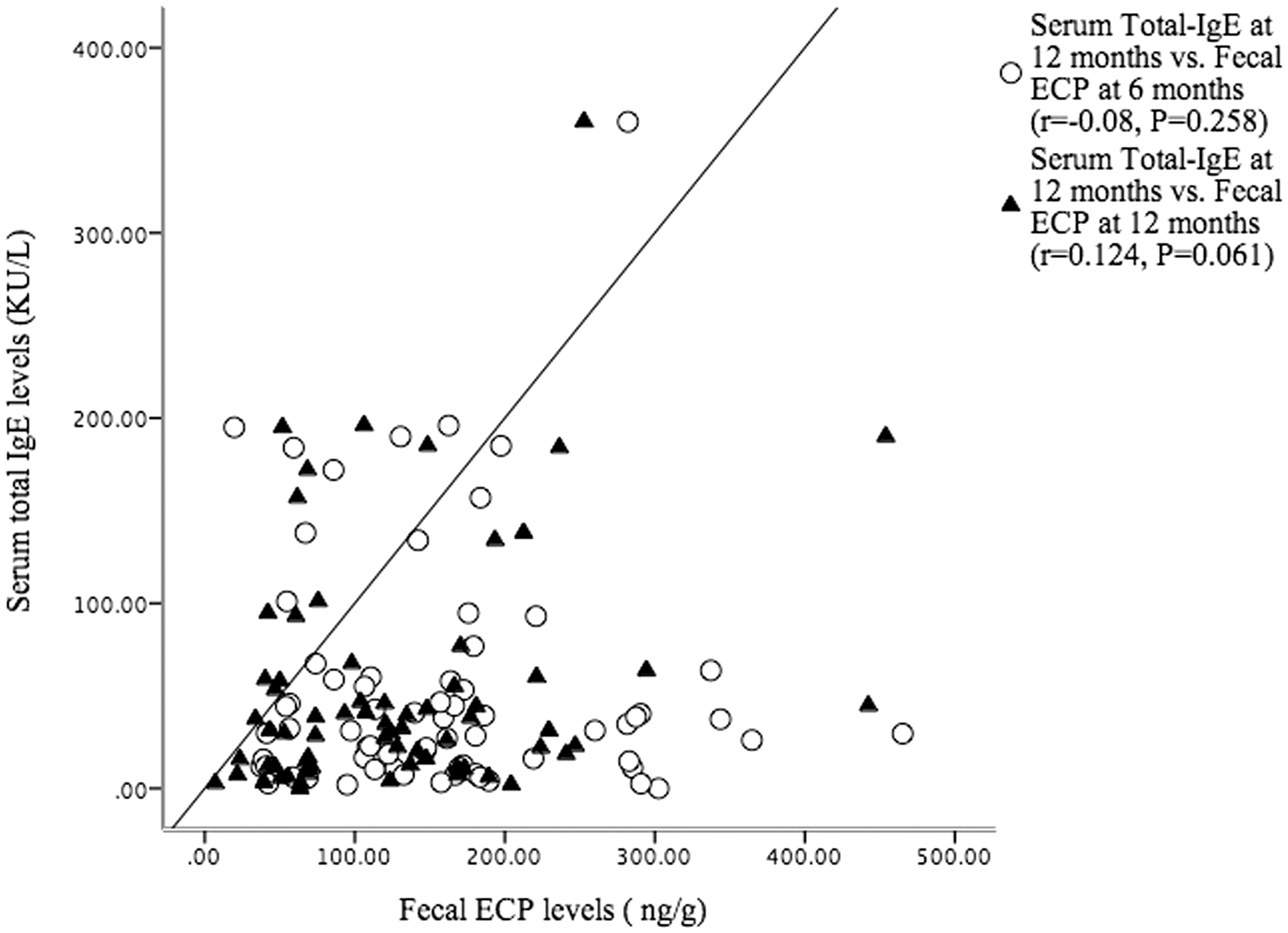
Discussion
This study reported infants' feeding practices in Taiwan. To the best of our knowledge, this is the largest study that used faecal ECP to monitor intestinal inflammation in infants, while also investigating the relationship between faecal ECP, total serum IgE, and allergen-specific IgE.
Breastfeeding is the preferred method of infant nutrition. In our study, only 36 infants (17.4%) were never breastfed, and 41.7% of lactating mothers chose to continue exclusive breastfeeding, while solid foods had been introducing to their children after four months of age. In general, the feeding choices, exclusive breastfeeding duration and the timing of solid food introduction were not different among our study participants, regardless of one or both parents having an allergic disease.
Many studies have attempted to assess the role of breastfeeding in the development of allergic disease, but the findings are inconsistent.4–7,29 Although expert bodies recommend that infants should be exclusively breastfed for 3–4 months to decrease the risk of allergies,4,30 it is uncertain that prolonged breastfeeding also has protective effects, because there was a low number of infants who were exclusively breastfed for longer than six months in those study populations. 4 In immunological food intolerance, mast cells, eosinophils, mucosal lymphocytes and epithelial cells are involved and related to each other in the development of mucosal inflammation. 31 Faecal ECP is an intestinal inflammatory marker that could be a promising non-invasive tool in monitoring intestinal inflammation in infants with food allergies.19,23 Our results show that faecal ECP levels were significantly higher in exclusively breastfed infants compared with formula-fed infants and infants who were never exclusively breastfed by the age of 6 months (P < 0.05) (Table 3). Perhaps, an explanation for increasing mucosal inflammation in prolonged exclusively breastfed infants (≥6 months) was imperfect oral tolerance against all the antigenic food proteins in the breast milk. Nevertheless, there was no evidence showing that a longer duration of exclusive breastfeeding increased IgE sensitization at the age of 12 months (Table 3). Exclusive breastfeeding seemed not the major determining factor that influenced IgE sensitization in the first year of life.
Currently, the optimal age for the introduction of solid foods varies greatly.4,8–10 Several studies investigated whether early introduction of solid foods, before four months, or delayed introduction, after 6 months, would promote or prevent allergies.4,9,29,32 In the present study, infants who were introduced to solid foods at 5–6 months had the lowest faecal ECP levels by 12 months of age, compared with infants who were introduced to solid foods earlier or later than 5–6 months. This indicates that exposure to solid food antigens during this period was not inducing an overt intestinal inflammatory response. In our study, there was no evidence showing that early or late introduction of solid foods influenced IgE sensitization in the first year of life (Table 3).
Silva et al. 24 reported that the mean and median faecal ECP levels were 1.93 μg/g and 1.20 μg/g, respectively, in 51 healthy Caucasian children (mean age 2.89 y). Those faecal ECP values are significantly higher than ours (median 124.24 ng/g, mean 145.20 ng/g). The difference could be due to the different methods of processing the stool samples, experimental conditions and differences in race and age. Currently, there is no standard technique for quantification, and the faecal ECP findings vary greatly and do not follow a normal distribution 18 ; therefore, comparing the results of the few paediatric studies published to date is difficult.23,24
Both ECP and IgE are important mediators of allergic inflammation, and they are used to monitor allergic diseases.11,20,21 ECP is an intestinal inflammatory marker for studying gastrointestinal disorders that are predominantly eosinophilic.18,23,24,33 Our results showed only a weak correlation between faecal ECP and serum IgE levels at 12 months (r=0.124, P = 0.061) (Figure 1). Also, there was no difference in faecal ECP levels at 6 and 12 months between infants who were or were not IgE-sensitized (Table 3). These findings suggest that faecal ECP might not be a useful indictor of IgE sensitization in infancy.
The strength of this study was its design. Detailed information regarding feeding practices and demographic characteristics of infants were obtained on a regular basis by questionnaires, and both blood and stool samples were collected prospectively. The timings of feeding practices matched the timings of sample collection. A major limitation of the study is that the number of participants was not large enough to make any definitive conclusions. Another limitation is that we did not investigate the effect of total breastfeeding duration. Additionally, the timing of specific food introduction in relation to faecal ECP levels was not evaluated.
In summary, we found that exclusive breastfeeding for longer than six months did not seem to reduce serum IgE levels, but instead showed a tendency to increase intestinal inflammation. Additionally, infants starting solids food during the fifth and sixth month showed the mildest intestinal inflammation by 12 months of age. As faecal ECP was not significantly associated with total serum IgE and allergen-specific IgE levels, we suggest that faecal ECP could be considered to be a specific intestinal inflammatory marker, but not an indicator of atopy in infancy.
Footnotes
Acknowledgements
All authors thank the study subjects and their parents for their participation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was funded by grants from Chang Gung Memorial Hospital, Keelung (CMRPG 2E0131, 2E0132, 2B0042).
Ethical approval
This study was approved by the Ethics Committee of Chang Gung Memory Hospital (101-4361C).
Guarantor
Huang JL
Contributorship
All authors were involved in the study design, participates recruitment, and written consent. Hua MC, Chen CC and Huang JL were involved in the laboratory work, statistical analysis and interpretation of its results. Hua MC wrote the first draft of manuscript, and Huang JL edited it. All authors reviewed the manuscript and approved the final version of the manuscript.
