Abstract
Background
Nephrotic syndrome is related to immune system dysfunction. Soluble human leukocyte antigen-G has been suggested to have an immunomodulatory role. Additionally, human leukocyte antigen-G expression may be influenced by the 14-base pair insertion/deletion polymorphism. However, this molecule has not been investigated in nephrotic syndrome.
Methods
Fifty-five children with nephrotic syndrome were enrolled: 24 primary nephrotic syndrome patients and 31 recurrent nephrotic syndrome patients. A group of 120 healthy subjects were included as reference controls. Additionally, 22 patients in nephrotic syndrome remission after treatments were also included. Both nephrotic syndrome patients and healthy subjects were genotyped for the 14-base pair insertion/deletion polymorphism. Plasma soluble human leukocyte antigen-G concentrations and serum immunoglobulin concentrations were determined.
Results
Nephrotic syndrome patients showed significantly higher levels of both soluble human leukocyte antigen-G and immunoglobulin E compared to normal controls. Nephrotic syndrome patients presented a higher frequency of the −14-base pair allele than did normal controls. Soluble human leukocyte antigen-G concentrations in remission patients were dramatically lower compared to in nephrotic syndrome patients. Moreover, soluble human leukocyte antigen-G and immunoglobulin E were moderately correlated in nephrotic syndrome patients.
Conclusions
The present study demonstrated that plasma soluble human leukocyte antigen-G concentrations were significantly elevated and that a relationship between serum total immunoglobulin E in nephrotic syndrome patients and the human leukocyte antigen-G −14-base pair allele may be a risk factor for nephrotic syndrome. These findings suggest that soluble human leukocyte antigen-G may be used as a monitoring marker for nephrotic syndrome patients' condition.
Introduction
Nephrotic syndrome (NS) is a common chronic glomerular disease in children characterized by heavy proteinuria, hypoalbuminemia, generalized edema and hyperlipidemia. 1 The most common form of NS in children is minimal change NS, 2 although the aetiology of NS remains poorly understood. Previous studies have suggested that NS is an immune-mediated disorder of T-lymphocyte function and promotes the production of permeability factor,3,4 which alters the glomerular filtration barrier, leading to massive proteinuria. 5 However, numerous studies have linked NS with atopic disorders (asthma, allergic rhinitis and atopic eczema) with elevated serum immunoglobulin E (IgE) concentrations. 6 Previous studies have proposed that atopy exerts a role in the pathogenesis of NS and that proteinuria may be triggered by various allergens.7,8 Therefore, NS is considered to be an immunoregulatory-related disorder.
Human leukocyte antigen-G (HLA-G), a non-classical HLA class Ib molecule, differs from other classical class I HLA (-A, -B and -C) molecules by its restricted tissue distribution and limited polymorphism in the coding region. Four membrane-bound (HLA-G1, -G2, -G3 and -G4) and three soluble isoforms (HLA-G5, -G6 and -G7) are encoded by alternative splicing of the same primary transcript. 9 In addition, soluble HLA-G (sHLA-G) can be shed from the membrane-bound isoforms through proteolytic cleavage of metalloprotease. 10 The immunoregulatory properties of HLA-G molecule are mainly related to the inhibition of the function of various immune cells, such as T cells, NK cells and antigen-presenting cells. The inhibitory functions are mediated by the interaction between HLA-G and four inhibitory receptors expressed on immune cells, including: immunoglobulin-like transcripts (ILT)-2/CD85j, ILT-4/CD85d, killer immunoglobulin-like receptor (KIR)2DL4/CD158d and BY55/CD160. In addition to its inhibitory role in immune cells, HLA-G can induce the generation of regulatory T cells, which play a comprehensive immunemodulatory role. 11
Since it was first detected in placental trophoblast cells of the maternal–fetal interface and reported to maintain a tolerogenic status during pregnancy, the HLA-G antigen has been found to be widely expressed in a variety of pathological conditions, such as transplantations, tumors and viral infections. 12 Additionally, many in vitro studies have described the expression and role of HLA-G in immune-mediated disorders. A study investigated by Tahan and Patiroglu 13 found higher levels of sHLA-G in atopic asthmatic patients compared to in non-asthmatic patients and controls. 13 Catamo et al. 14 found that the 5′ upstream regulatory region and 3′ untranslated region (UTR) polymorphisms of HLA-G gene were risk factors for rheumatoid arthritis (RA). However, Kim et al. 15 failed to find an association between two single-nucleotide polymorphisms of the HLA-G gene and RA. 15 However, no studies have examined the potential associations between sHLA-G and NS.
Moreover, a 14-bp insertion/deletion polymorphism (rs16375) in the 3′-UTR of the HLA-G gene affects mRNA stability and protein expression levels. 16 Particularly, the del allele in the mRNA is correlated with a higher protein expression level of HLA-G.16,17 We and other authors have reported that this polymorphism is associated with numerous disorders, including human cytomegalovirus infection, RA, systemic lupus erythematosus and juvenile idiopathic arthritis.18–20
Considering currently known characteristics about NS and the immunoregulatory role of sHLA-G, the aim of the present study was to evaluate the prevalence of the 14-bp insertion/deletion polymorphism and plasma concentrations of sHLA-G in a cohort of children with NS and normal controls, as well as to explore the potential relationship between sHLA-G and total IgE in NS patients.
Methods
Subjects
The comparison of clinical characteristics between NS patients and normal controls.
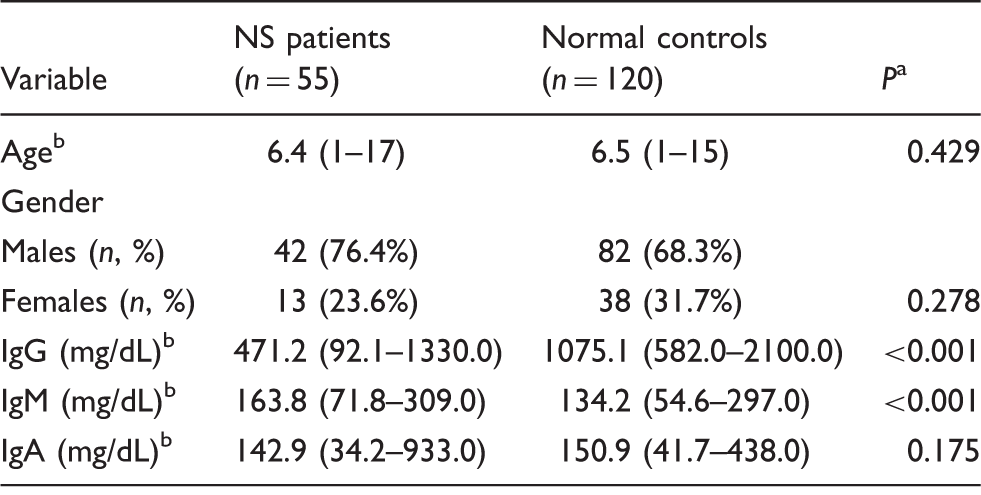
NS: nephrotic syndrome; IgG: immunoglobulin G; IgM: immunoglobulin M; IgA: immunoglobulin A.
The results of the Chi-square (χ2) test and Mann-Whitney U test.
Data are presented as the mean (range).
Patients with edema, severe proteinuria (≥3 + protein on urine dipstick for >3 consecutive days) and serum albumin < 25 g/L were defined as relapse. Remission was defined as total subsidence of edema and normal protein excretion (trace or negative protein on urine dipstick for ≥3 consecutive days or < 4 mg/m2 per hour). 22
According to the suggestions of International Study of Kidney Disease in Children, the initial treatment for children with NS is daily oral prednisone 2 mg/kg per day (maximum of 60 mg/day) for four weeks, followed by a decreases of 1.5 mg/kg per day for the next four weeks if responsive, and then an additional decrease over eight weeks. The treatment for relapses changes the regimen of daily oral prednisolone to 60 mg/m2 per day (maximum 60 mg/day) until three consecutive days of trace or negative urine protein on the dipstick, followed by maintenance alternate-day prednisolone at a dose of 1.5 mg/kg, and gradual decreasing doses by approximately 0.1–0.5 mg/kg for at least seven weeks.22,23
Nephrotic patients with infectious diseases, gross hematuria, allergic disorders, persistent hypertension, congenital kidney dysfunction, low complement levels and those who were undergoing any other specific immunotherapy within the previous four weeks were excluded from the study. Renal biopsies were performed in all 55 patients, all of whom showed minimal change NS.
Blood samples were taken from NS patients during the nephrotic phase (before starting the steroid treatment) and remission phase (four weeks after steroid treatment), as well as from normal controls. The separated serum and plasma were stored at −80 ℃ until analysis.
The study was approved by the ethics committee of the Second Affiliated Hospital of Wenzhou Medical University. The parents or legitimate guardians of all subjects provided written informed consent before beginning the study.
Genotype of 14-bp insertion/deletion polymorphism
The HLA-G 14-bp insertion/deletion polymorphism was detected as described previously. 24 Briefly, genomic DNA extracted from peripheral whole blood leukocytes was amplified by polymerase chain reaction (PCR) using specific primers to detect the HLA-G 14-bp insertion/deletion polymorphism. PCR products were then resolved by 10% non-denaturing polyacrylamide gel electrophoresis and visualized by ethidium bromide staining. PCR products showed bands of 288 bp, 274 bp or both. Partial PCR products were directly sequenced.
sHLA-G enzyme-linked immunosorbent assay
sHLA-G molecules were determined using a specific recognizing shed HLA-G1 and HLA-G5 enzyme-linked immunosorbent assay (ELISA) kit for sHLA-G (sHLA-G kit; Exbio, Prague, Czech Republic), which employs the monoclonal antibody MEM-G/9. Each sample final concentration was determined at an optical density of 450 nm (Spectra Max 250, Molecular Devices, Sunnyvale, CA, USA) according to the standard curves (range: 0–125 U/mL) in three independent measurements. When the concentration exceeded 125 U/mL, samples were diluted to calculate the sHLA-G value. The detection limits were 1 U/mL. The procedures were performed according to the manufacturer's instructions.
Measurement of serum immunoglobulin
Serum samples were stored at −80℃ before the immunoglobulin assay. Serum total IgE concentrations were determined using an enzyme immune assay method with the ImmunoCAP System (Pharmacia, Uppsala, Sweden). Serum total IgG, IgM and IgA concentrations were determined using standard enzyme immunoassay techniques (Beckman Coulter Inc., CA, USA).
Statistical analysis
Statistical analysis was performed using SPSS 17.0 software (SPSS, Inc., Chicago, IL, USA). The frequency difference of the 14-bp insertion/deletion polymorphism between NS patients and normal controls as well as the goodness of fit for genotype frequencies were analysed using the chi-squared (χ2) test according to Hardy–Weinberg equilibrium. The odds ratio (OR) represents the relative risk. The non-parametric Mann-Whitney U test or Chi-square (χ2) test was used to compare quantitative variables between the different study groups. The correlation between plasma sHLA-G concentrations and serum IgE concentrations in nephrotic patients was performed using Spearman's coefficient of correlation (rs). A value of P < 0.05 was considered to be statistically significant.
Results
Serum total IgG, IgA and IgM concentrations
Serum total IgG concentrations were dramatically decreased in NS patients (median, 471.2 mg/dL; range, 92.1–1330.0 mg/dL) compared with normal controls (median, 1075.1 mg/dL; range, 582.0–2100.0 mg/dL; P < 0.001; Table 1). Serum total IgA concentrations in NS patients were also lower than in normal controls, but the difference was not significant difference. In contrast, serum IgM concentrations were increased in NS patients (median, 163.8 mg/dL; range, 71.8–309.0 mg/dL) compared with in normal controls (median, 134.2 mg/dL; range, 54.6–297.0 mg/dL; P < 0.001; Table 1).
HLA-G 14-bp insertion/deletion polymorphism
Genotypic and allelic frequencies of 14-bp insertion/deletion polymorphism in NS patients and normal controls.
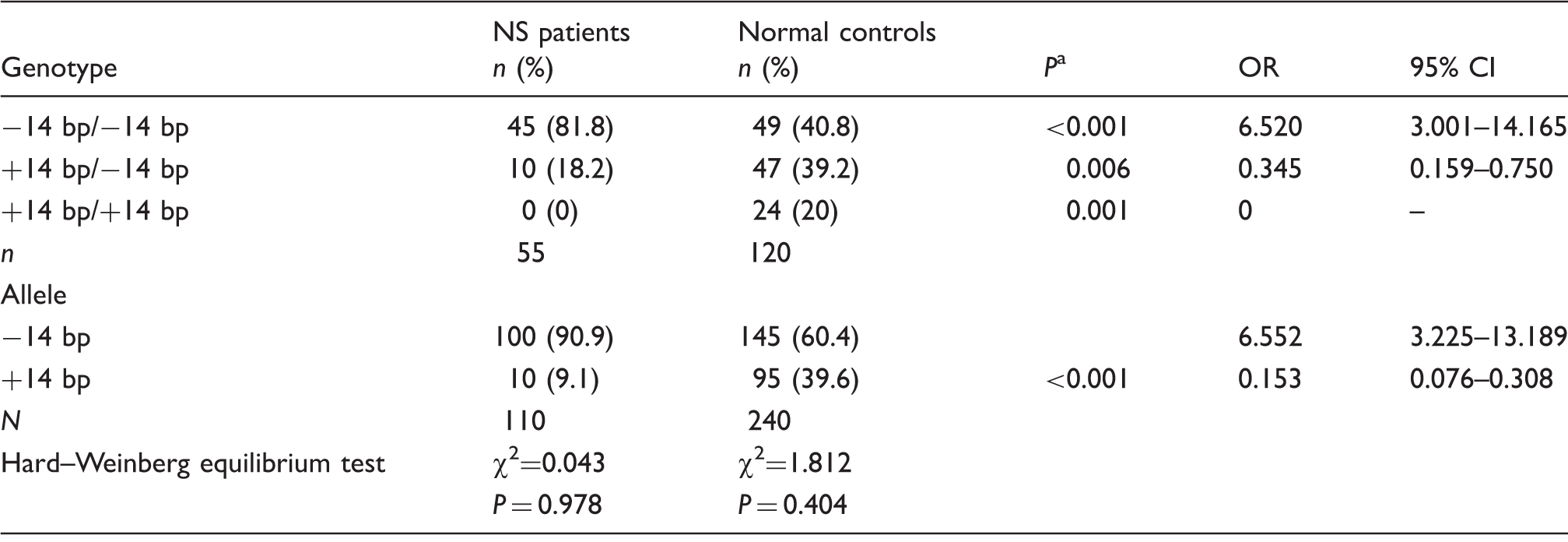
NS: nephrotic syndrome; OR: odd ratio; 95% CI: 95% confidence interval.
The results of the Chi-square (χ2) test.
Plasma sHLA-G concentrations in NS patients
The data showed that plasma sHLA-G concentrations in total NS patients (median, 95.20 U/mL; range, 25.00–187.56 U/mL) were significantly higher than in normal controls (median, 29.66 U/mL; range, 0.97–143.69 U/mL; P < 0.001; Figure 1). In addition, normal controls have lower sHLA-G concentrations compared with both PNS (n = 24; median, 93.20 U/mL; range, 25.64–187.56 U/mL) and RNS patients (n = 31; median, 96.75 U/mL; range, 25.00–169.84 U/mL) (P < 0.001 for both; Figure 1). Our data also showed that there were significant differences in sHLA-G concentrations in NS patients with remission (n = 22; median, 38.84 U/mL; range, 8.58–82.10 U/mL) and in NS patients who did not reach remission (n = 8; median, 93.38 U/mL; range, 44.39–156.60 U/mL; P = 0.001) or normal controls (median, 29.66 U/mL; range, 0.97–143.69 U/mL; P = 0.014; Figure 1).
Distribution and comparison of plasma sHLA-G concentrations in NS patients, remission patients, non-remission patients and normal controls. Values are shown as medians (horizontal lines in boxes), quartiles (25th and 75th percentiles, boxes) and P values between groups. Comparison was performed with Mann–Whitney U test.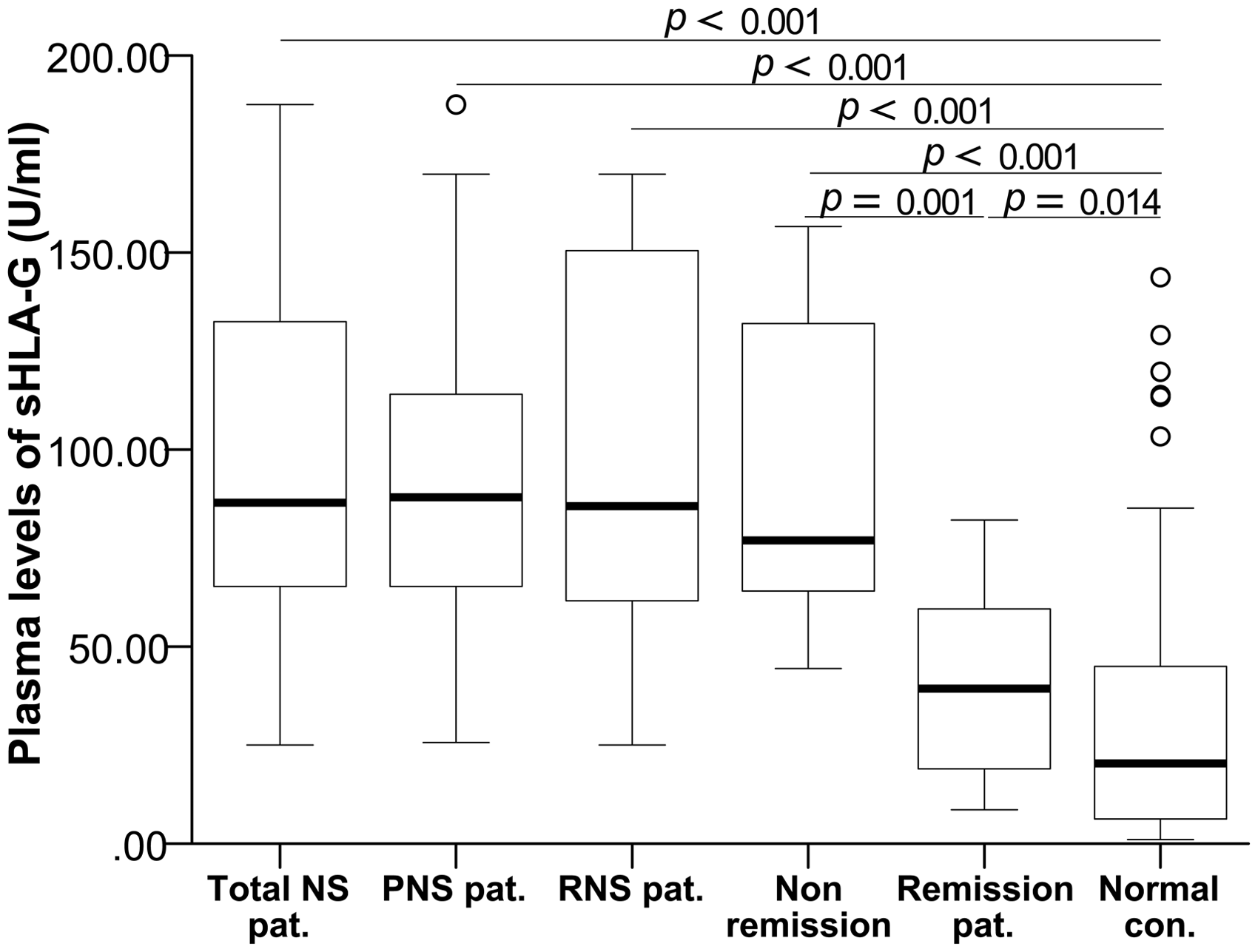
Serum total IgE concentrations in NS patients
Serum total IgE concentrations were significantly higher in total NS patients (median, 711.57 IU/mL; range, 53.96–3000.00 IU/mL) than in normal controls (median, 37.92 IU/mL; range, 1.84–141.30 IU/mL; P < 0.001; Figure 2). Serum total IgE concentrations in both PNS (median, 770.50 IU/mL; range, 60.95–3000.00 IU/mL) and RNS patients (median, 665.94 IU/mL; range, 53.96–3000.00 IU/mL) were clearly higher than in normal controls (P < 0.001 for both; Figure 2).
Distribution and comparison of serum total IgE concentrations in NS patients and normal controls. Values are shown as medians (horizontal lines in boxes), quartiles (25th and 75th percentiles, boxes) and P values between groups. Comparison was performed with Mann–Whitney U test.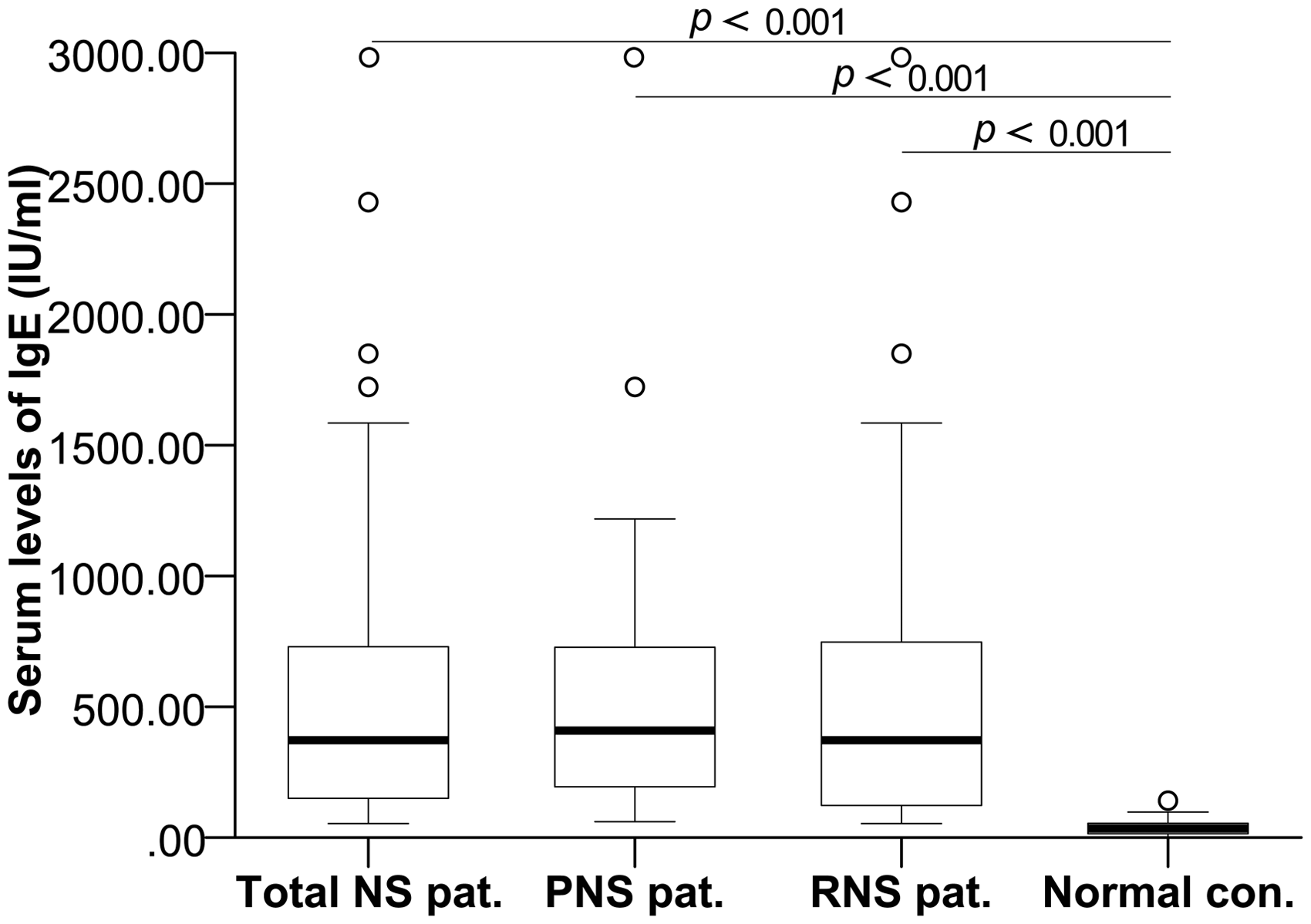
Relationship between sHLA-G and IgE
We observed a significant and moderate relationship between plasma sHLA-G concentrations and serum total IgE concentrations in total NS patients (r = 0.532 and P < 0.001; Figure 3). A significant relationship between plasma sHLA-G concentrations and serum total IgE concentrations in normal controls was not observed (data not shown). No significant relationship was observed between plasma sHLA-G concentrations and serum total concentrations of IgG, IgA and IgM (data not shown).
Correlation between plasma sHLA-G concentrations and serum total IgE concentrations in NS patients. r is a spearman correlation coefficient.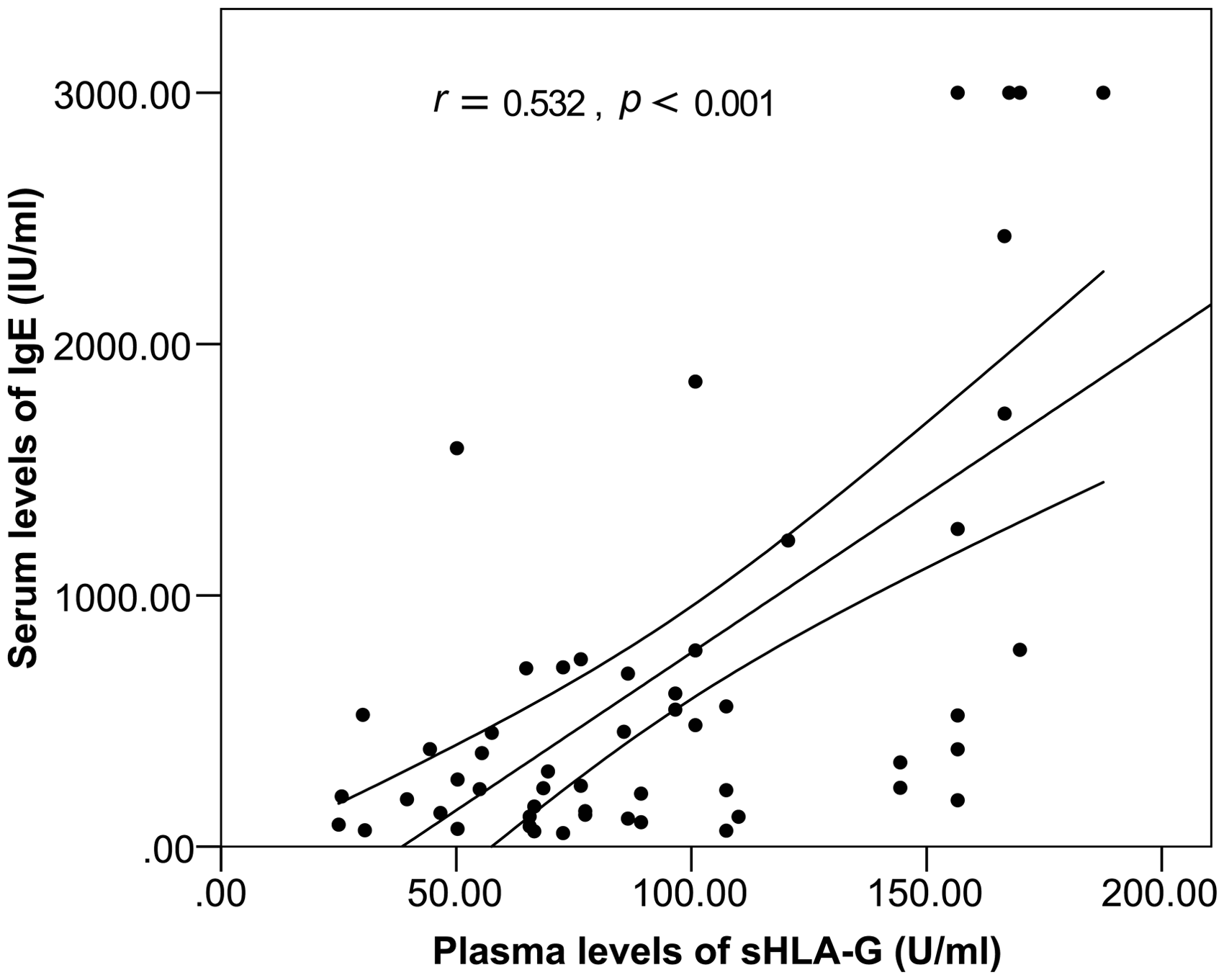
Discussion
The pathogenic mechanism of NS is currently unclear. In recent decades, numerous studies have indicated that the immune system is strongly associated with NS.1,3 It has been shown that the primary abnormality in NS results from T-cell dysfunction.3,4 However, many other findings have demonstrated an association between NS and the elevation of various cytokines,25–29 as well as abnormal concentrations of serum immunoglobulin,28,30 suggesting involvement of a dysfunction of the humoral immune system. However, the causal relationship between humoral immunity and T-lymphocyte dysfunction is not clearly understood.
In recent years, studies have reported that increased serum IgE concentrations in patients with NS were closely associated with NS and allergic disorders.6–8,28,31 The synthesis of IgE is mainly mediated by two cytokines, interleukin (IL)-4 and IL-13, released by T-helper 2 (Th2) cells. 32 Increased concentrations of IL-4 and IL-13 have been reported in children with NS.26–28,33,34 Moreover, some studies have described that NS shows an imbalance between Th1 and Th2 cytokines,35,36 with the polarity toward a greater Th2 response.35,37,38
In this regard, HLA-G may exert its immunoregulatory properties on NS. Similarly to its membrane-bound counterparts, the soluble HLA-G molecule may inhibit the activity of CTL and NK cells and induce the apoptosis of cytotoxic CD8+T cells and CD8+ NK cells.9,39 Moreover, sHLA-G can inhibit alloproliferation of CD4+T cell by sealing cell cycle progression and induce regulatory T cells.11,40 In addition, sHLA-G has the capability to modulate the production of Th2-type cytokines, which alters the Th1/Th2 balance. 41
However, no studies have documented the involvement of sHLA-G in NS to date. In the present study, we assessed the plasma sHLA-G concentrations in children with NS, as well as explored the potential relationship between sHLA-G and total IgE. First, an abnormity of serum immunoglobulin concentrations was observed in NS patients compared to in normal controls, which was consistent with the results of previous studies,28–31 indicating the presence of an aberrant humoral immune system. More importantly, NS patients showed significantly elevated concentrations of sHLA-G compared to normal controls, as well as both the PNS and RNS patient subgroups. In addition, a significant and moderate relationship between sHLA-G and total IgE was observed in NS patients. Our data suggested that NS patients had increased sHLA-G concentrations compared with normal controls, indicating that sHLA-G plays a role in regulating immune-mediated disorders, probably not in a direct way. A previous study supported that sHLA-G could induce the production of IL-4 from PBMCs co-cultured with HLA-G-expressing cells, while decreasing the concentrations of IFN-γ and TNF-α,42,43 which implied that sHLA-G contributes to the disturbed cytokines production. In addition, high concentrations of HLA-G may induce a Th2-type response and has been examined in vitro. 44 Therefore, we speculated that the positive correlation between sHLA-G and total IgE in NS patients may be mediated through Th2-type cytokines, such as IL-4 and IL-13. Furthermore, the concentrations of sHLA-G in remission phase were dramatically reduced compared with NS attack phase. Thus, sHLAG may be used as a monitoring marker for NS patients' condition.
It was previously shown that the HLA-G 14-bp insertion/deletion, a gene polymorphism in exon 8 of the gene, may play a pivotal role in HLA-G mRNA stability and protein expression mainly through post-transcriptional regulatory mechanisms. 16 In fact, the presence of the −14-bp allele modulated higher levels of HLA-G mRNA and increased HLA-G concentrations.16,17 A previous study conducted by Rizzo et al. 45 revealed a relationship between the HLA-G 14-bp polymorphism and the clinical outcome in methotrexate-treated RA patients. In contrast, Stamp et al. 46 did not observe a difference between the HLA-G 14-bp polymorphism and the response to methotrexate. The finding that the RA patients with the 14-bp deletion allele were positively correlated with improvement in disease status has also been reported. 47 Another study revealed that in SLE patients, the −14bp/−14bp genotype presented higher disease activity compared with +14bp/−14bp. 20 In addition, Veit et al. 19 reported that the HLA-G −14bp allele was a potential susceptibility factor for juvenile idiopathic arthritis patients, mainly in females, but observed no difference in the 14-bp polymorphism in RA patients when compared with control subjects. 19
In the present study, we also assessed the prevalence of the 14-bp insertion/deletion polymorphism in the HLA-G gene in NS patients, there have been no studies examining the association between this polymorphism and NS. Our data suggested a significantly higher prevalence in the HLA-G 14-bp allele distribution in NS patients compared with normal controls, revealing that the −14-bp allele was a risk factor for the disease, particularly in −14-bp/−14-bp homozygotes. Further investigation should focus on whether an association exists between this polymorphism and the steroid treatment response, as well as the activity of NS.
In conclusion, our study confirmed that plasma sHLA-G concentrations dramatically increased accompanied by a moderate correlation with elevated serum total IgE concentrations in NS patients. The genotypic and allelic frequencies of the 14-bp polymorphism in the HLA-G gene in NS patients and normal controls were significantly different. More importantly, the HLA-G −14-bp allele may be a risk factor for NS, and the sHLA-G molecule may be related to NS prognosis. Our findings support that the immune system participates in the aetiopathogenesis of NS, likely involving the sHLA-G molecule. A longitudinal study in specific subgroups of children with NS would be of real value to our understanding of the pathogenesis of NS in children.
Footnotes
Acknowledgements
We would like to thank Dr. Zhifa Shen for his assistance and guidance in this research.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by grants from the National Science Foundation of China (no. 81071365) and the Zhejiang Provincial Natural Science Foundation (LY13H190006).
Ethical approval
The study was approved by the ethics committee of the Second Affiliated Hospital of Wenzhou Medical University (REC number: L-2015-14).
Guarantor
XZ.
Contributorship
ML, YL, QH, JL and HW collected samples. QY, MC and HW performed laboratory detection. YL, WH and XZ designed the study, and analysed the data. YL wrote the first draft of the manuscript. All authors reviewed the manuscript and approved the final version of the manuscript.
