Abstract
Background
Rheumatoid factor causes positive interference in multiple immunoassays. Recently, negative interference has also been found in immunoassays in the presence of rheumatoid factor. The chemiluminescent microparticle immunoassay is widely used to determine serum alpha-fetoprotein. However, it is not clear whether the presence of rheumatoid factor in the serum causes interference in the chemiluminescent microparticle immunoassay of alpha-fetoprotein.
Methods
Serum alpha-fetoprotein was determined using the ARCHITECT alpha-fetoprotein assay. The estimation of alpha-fetoprotein recovery was carried out in samples prepared by diluting high-concentration alpha-fetoprotein serum with rheumatoid factor-positive or rheumatoid factor-negative serum. Paramagnetic microparticles coated with hepatitis B surface antigen–anti-HBs complexes were used to remove rheumatoid factor from the serum.
Results
The average recovery of alpha-fetoprotein was 88.4% and 93.8% in the rheumatoid factor-positive and rheumatoid factor-negative serum samples, respectively. The recovery of alpha-fetoprotein was significantly lower in the rheumatoid factor-positive serum samples than in the rheumatoid factor-negative serum samples. In two of five rheumatoid factor-positive samples, a large difference was found (9.8%) between the average alpha-fetoprotein recoveries in the serially diluted and initial recoveries. Fourteen rheumatoid factor-positive serum samples were pretreated with hepatitis B surface antigen-anti-HBs complex-coated paramagnetic microparticles. The alpha-fetoprotein concentrations measured in the pretreated samples increased significantly.
Conclusions
It was concluded that the alpha-fetoprotein chemiluminescent microparticle immunoassay is susceptible to interference by rheumatoid factor, leading to significantly lower results. Eliminating the incidence of negative interference from rheumatoid factor should be an important goal for immunoassay providers. In the meantime, laboratorians must remain alert to the negative interference by rheumatoid factor, and in some cases, pretreat rheumatoid factor-positive samples with blocking or absorbing reagents.
Introduction
Alpha-fetoprotein (AFP), a single polypeptide chain glycoprotein, was first found in fetal serum by Bergstrand and Czar. 1 Clinical research has shown that abnormal concentrations of maternal serum AFP during the second trimester of pregnancy are associated with poor pregnancy outcomes, such as Down syndrome. 2 Serum AFP concentrations have also been found to be elevated in patients with multiple malignancies, such as primary hepatocellular carcinoma (HCC). 3 In the case of HCC, a significantly higher incidence of tumour recurrence and poor tumour-free survival after treatment is noted in patients with high pretreatment concentrations of AFP. The use of AFP concentrations in the management of patients with HCC has also been well documented. 4 For patients in clinical remission following treatment, AFP concentrations generally decrease. Post-treatment AFP values that fail to return to normal strongly suggest the presence of residual tumour. 5 Therefore, it is very important to accurately measure serum AFP concentrations in patients with HCC and associated diseases.
The chemiluminescent microparticle immunoassay (CMIA) is widely used for the quantitative assessment of AFP in human serum or plasma. Investigations by the National Center for Clinical Laboratories showed that most clinical laboratories used the CMIA to measure serum or plasma AFP from 2010 to 2014 (www.clinet.com.cn). It is noteworthy that quite a few laboratories used the ARCHITECT AFP assay kits (Abbott Ireland, Sligo, Ireland) to measure AFP. Its instruction manual shows that multiple substances, such as heterophilic antibodies and human antimouse antibodies, could be potential sources of interference in the ARCHITECT AFP assay.
Rheumatoid factor (RF) is defined as an autoantibody against the Fc region of immunoglobulin G (IgG). It is frequently present in 60–80% of patients with rheumatoid arthritis (RA) and Sjögren's syndrome. 6 It is also found in individuals infected with the Epstein-Barr virus or parvovirus, and in 5–10% of healthy persons, notably, the elderly. The phenomenon of positive RF interference in some immunoassays has been known for quite some time. 7 As RF has the ability to bind to the Fc region of IgG via its own Fab region, it can facilitate the detection and capture of antibody, leading to falsely elevated or false positive results. 8 However, recent studies have found that RF caused both positive and negative interference in the serum hepatitis B surface antigen (HBsAg) enzyme-linked immunosorbent assay (ELISA) and in the plasma B-type natriuretic peptide (BNP) CMIA.9–11 Remarkably, there are few reports of negative interference by RF in the literature. Moreover, it is still unclear whether the negative interference caused by RF is an exceptional phenomenon produced by the HBsAg ELISA and the BNP CMIA or a denominator of immunoassays. Therefore, it was deemed reasonable to investigate whether RF causes interference, particularly negative interference, in serum AFP CMIA in this study.
Materials
Blood samples were collected in the Union Hospital Affiliated to Tongji Medical College, Huazhong University of Science and Technology, in 2014. The serum was isolated by centrifugation for 10 min at 1900 × g and stored at −20℃. Sixty RF-positive (26.0 KIU/L ≤ RF ≤ 1500.6 KIU/L) serum samples from patients with RA, 60 RF-negative (RF < 20 KIU/L) serum samples from healthy volunteers and 11 serum samples from patients with liver carcinoma with AFP concentrations ranging from 141.1 to 14652.3
Methods and results
RF concentrations measured by the BNII RF and the Immage 800 RF assays
Serum RF concentrations were determined using the BNII RF assay, and repeated measurement of serum RF concentrations was performed using the Immage 800 RF assay. A total of 60 RF-positive serum samples identified by these RF assays were brought into this study. A one-sample Kolmogorov-Smirvnov test showed that the RF concentrations in RF-positive serum samples were not distributed normally using the BNII RF assay and the Immage 800 RF assay (Z = 2.7,
AFP recovery in the RF-positive serum samples
Sixty serum samples with known AFP concentrations were diluted 1:9 with (a) RF-negative serum (control group) and (b) 60 with RF-positive serum (experimental group). Serum AFP was measured in the diluted serum samples using the ARCHITECT AFP assay in these two groups and AFP recoveries were calculated as described by Eriksson et al.
12
The average AFP recovery in the control group was 93.8% (range 82.6–110.4%); the corresponding values in the experimental group were lower at 88.4% (range 79.2–99.2%). A paired t-test showed that the recovery of the experimental group was significantly lower than in the control group (t = 4.5; Correlation between AFP recovery and RF levels.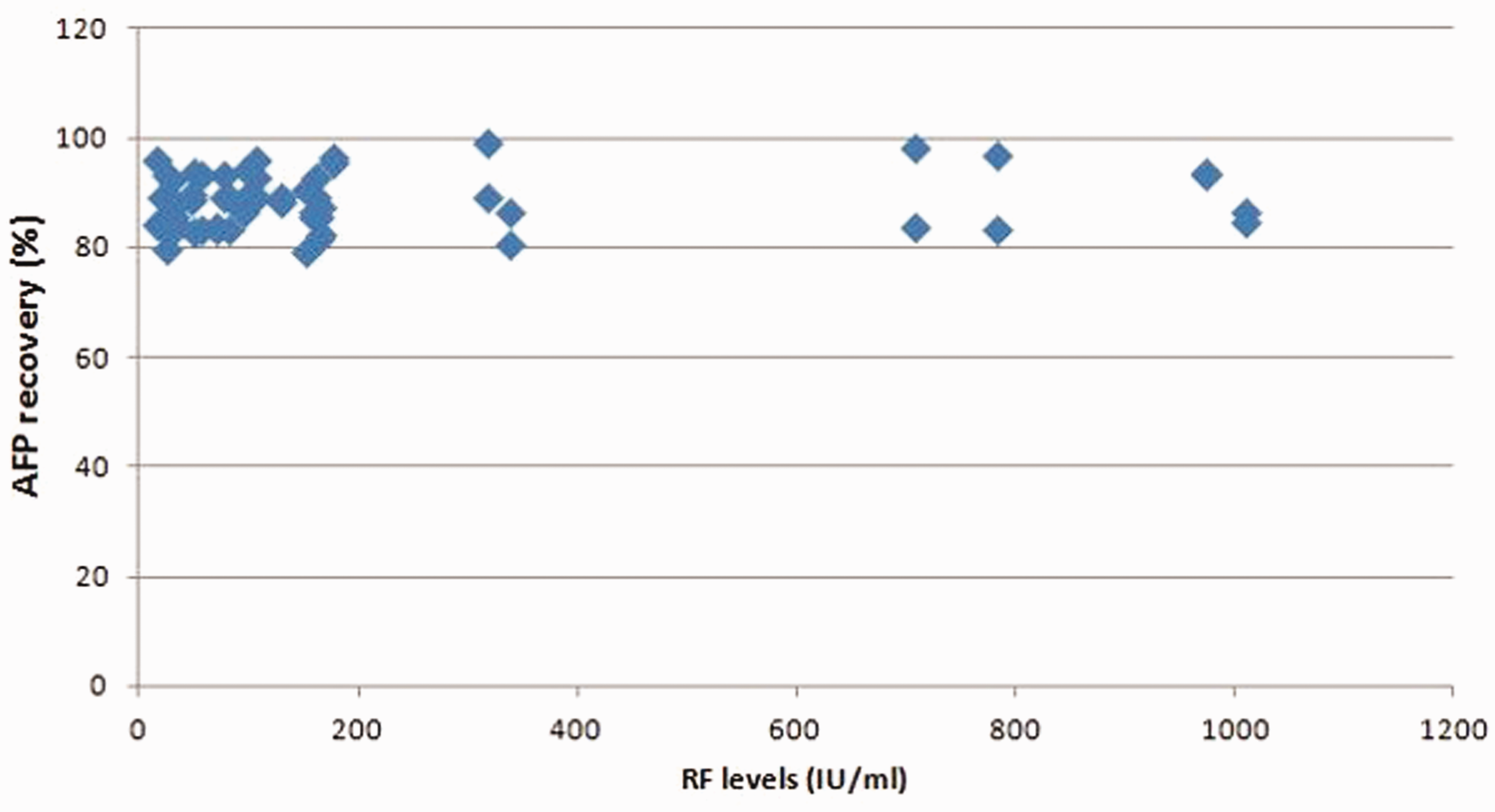
The effect of serial doubling dilutions on AFP recovery in RF-positive serum samples
AFP recovery (%) measured in diluted serum samples.
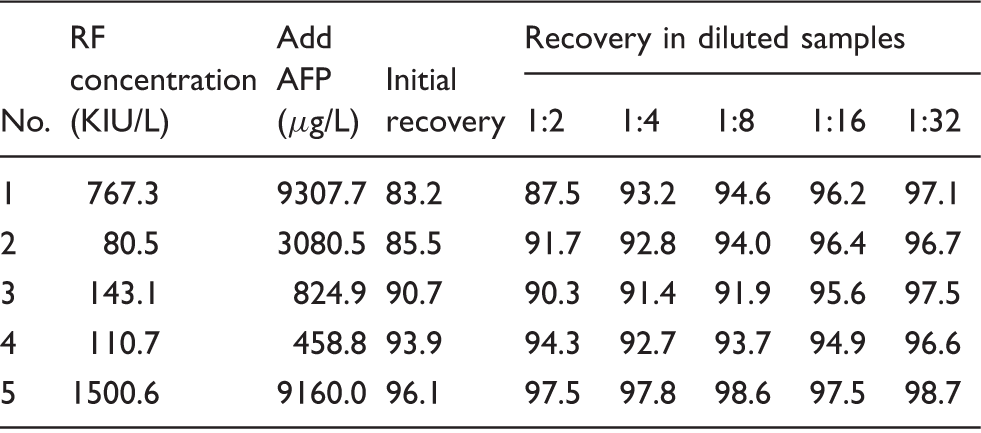
AFP: alpha-fetoprotein; RF: rheumatoid factor.
Estimation of AFP concentrations before and after the removal of RF
The HBsAg-anti-HBs complexes-coated paramagnetic microparticles were prepared before being added to RF-positive serum samples to remove RF and to RF-negative serum samples as control. These coated paramagnetic particles were prepared as follow: (i) 9 mL of paramagnetic microparticles coated with anti-HBs were incubated with 9 mL of sera with HBsAg concentrations of 200.0 KIU/L at 4℃ overnight on a rotary shaker; (ii) the mixtures were then centrifuged at 2230 × g for 8 min, and the pellets were washed three times with 5 mL of a phosphate buffer solution (pH 7.5); and (iii) the pellets were then dissolved in 1.5 mL of the phosphate buffer solution (pH 7.5).
AFP concentrations (
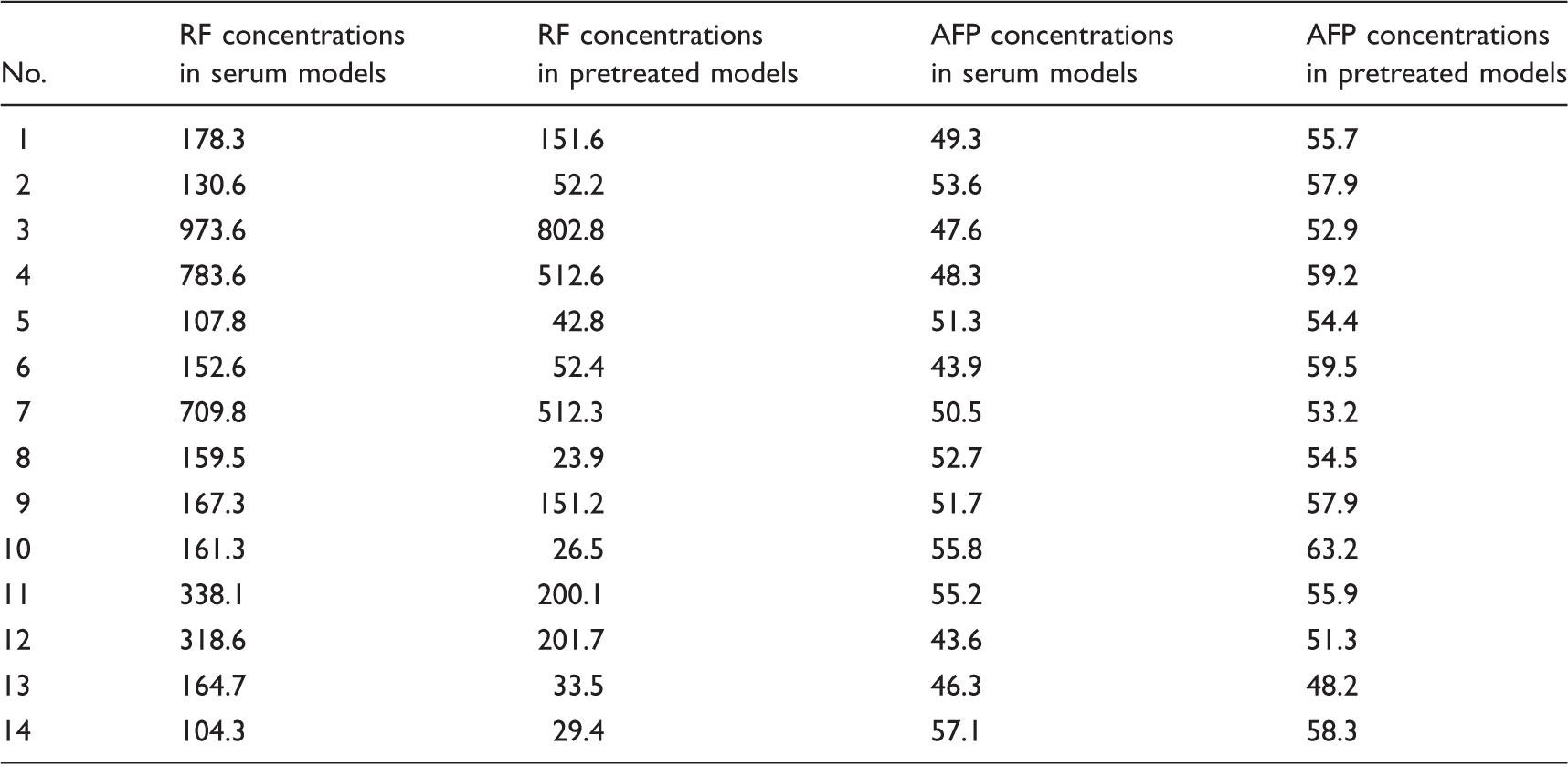
AFP: alpha-fetoprotein; RF: rheumatoid factor.
Discussion
Specific antibody immunoassays vary according to their antigens, platforms and performance characteristics, so false positivity is unlikely to be exhibited by more than one assay when multiple assays are used on a single specimen. 13 In order to exclude false-positive results, serum RF concentrations were quantified using the BNII RF and the Immage 800 RF assays, and 60 RF-positive serum samples identified by these two RF assays were brought into this study. The BNII RF assay uses an immunocomplex consisting of human IgG and anti-human IgG from animals as immobilized antigen to bind RF, whereas the Immage 800 RF assay uses human IgG to substitute the immunocomplex. Although an intercept of 24.1 was found suggesting methods’ bias, an correlation coefficient of 0.96 implied that good correlation was present in the RF concentrations measured by the BNII RF and the Immage 800 RF assays. These data give support to the integrity of RF-measurement.
RF, a known heterophilic endogenous antibody, can interfere with the immunoassays causing inaccurate measurement of BNP, thyroid stimulating hormone or troponin I, among others.9,13–15 In this study, the average AFP recovery was 93.8% in the RF-negative serum samples and 88.4% in the RF-positive serum samples. Compared to the RF-negative serum samples, AFP recovery decreased significantly in the RF-positive serum samples. Furthermore, 2 of the 60 RF-positive serum samples had AFP recoveries of <80.0%. These results indicate that potential endogenous interfering substances were present in the RF-positive serum samples. Serial dilution is a rapid and effective method to demonstrate the presence of endogenous interfering substances in serum samples.9–11,16–18 In this study, five RF-positive serum samples were performed serial doubling dilutions, and AFP recovery was estimated. In two samples, a large difference was found (9.8%) between the average AFP recoveries in the serially diluted and initial recoveries, and the maximum difference was 13.9% suggestive of non-linearity and lack of parallelism. These results also provided evidence that there were endogenous interfering substances in the RF-positive serum samples.
Blockage or removal of RF from the serum is one of the most effective methods identifying whether RF can falsely reduce results.9–11,16–18 Paramagnetic microparticles coated with HBsAg-anti-HBs complexes were used to remove RF from the serum in view of following reasons: (i) soluble antigen-antibody complexes can be precipitated with RF 19 ; (ii) anti-HBs used to label paramagnetic microparticles include the IgG and IgM subgroups; (iii) polystyrene particles coated with an immunocomplex in the BNII RF assay were used to bind RF in the serum; (iv) it is convenient to achieve pellets using a magnetic field or low speed centrifugal. Although the RF was not removed completely from the RF-positive serum using the paramagnetic microparticles, the measured AFP concentrations increased significantly, whereas there was no significant difference in AFP concentrations before and after the removal of RF in the RF-negative serum. These data highly support our hypothesis that RF causes negative interference in the AFP CMIA.
As RF reduced AFP measurements in the AFP CMIA, we investigated whether AFP recovery was associated with serum RF concentrations. When the RF concentrations were in the range of 26.0 to 1500.6 KIU/L, AFP recovery did not correlate with the serum RF concentration. As multiple serum samples were added to the RF-positive serum to estimate AFP recovery, we considered the influence of AFP variability on this result. To exclude any possible bias introduced by AFP heterogeneity, 30 of 60 serum samples in the experiment group were prepared by diluting a serum sample with an AFP concentration of 473.6
In this study, AFP recovery in RF-positive serum samples was not only significantly lower than that of RF-negative serum samples but also the AFP recovery in 2 of 60 (3.3%) serum samples was also significantly lower, which was considered to be inaccurate. Therefore, we can conclude that AFP CMIA assays are susceptible to interference by RF, which leads to significantly lower results. Eliminating and reducing the incidence of negative interference from RF should be an important goal for immunoassay providers. In the meantime, laboratorians must remain alert to the negative interference of RF, and in some cases, pretreat RF-positive samples with blocking or absorbing reagents.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval (including reference number)
This study was approved by the Ethics Committee of the Union Hospital Affiliated with Tongji Medical College, Huazhong University of Science and Technology (reference number: 2014-381). All of the patients signed an informed consent form.
Guarantor
YL.
Contributorship
HW and XB contributed equally to this work. Conceived and designed the experiments: YL. Performed the experiments: HW, XB and LX. Analysed the data: HW, XB and LX. Contributed reagents/materials/analysis tools: YL. Wrote the manuscript: YL.
