Abstract
Background
Traditionally, testing for syphilis has consisted of initial screening with a non-treponemal test, then retesting reactive specimens with a treponemal test. Recent availability of a chemiluminescent microparticle immunoassay for detecting antibodies against Treponema pallidum has led several laboratories in China to adopt chemiluminescent microparticle immunoassay for screening of syphilis, with subsequent testing of reactive serum samples with non-treponemal tests. We evaluated the utility of chemiluminescent microparticle immunoassay for routine screening of syphilis.
Methods
Antibodies against Treponema pallidum were screened in 20,550 serum samples using chemiluminescent microparticle immunoassay. Chemiluminescent microparticle immunoassay-positive samples were reflexively tested with rapid plasma reagin tests and Treponema pallidum particle agglutination assays. Dot-immunoblot assays were used to confirm results of chemiluminescent microparticle immunoassay-positive and Treponema pallidum particle agglutination-negative serum samples.
Results
Overall, 267 samples (1.3%) were chemiluminescent microparticle immunoassay-positive, and 185 (69.3%) of those chemiluminescent microparticle immunoassay-positive serum samples were also Treponema pallidum particle agglutination-positive. Samples’ signal to cut-off ratio for chemiluminescent microparticle immunoassay correlated with diagnostic reliability, as greater samples’ signal to cut-off ratio corresponded with greater concordance between chemiluminescent microparticle immunoassay and Treponema pallidum particle agglutination results. Dot-immunoblot testing of 82 chemiluminescent microparticle immunoassay-positive and Treponema pallidum particle agglutination-negative serum samples showed that 16 samples (19.5%) were Dot-immunoblot-positive, 28 (34.2%) were indeterminate and 38 (46.3%) were negative.
Conclusions
Because there is a certain percentage of false-positive results using chemiluminescent microparticle immunoassay for routine screening of syphilis, further analysis by Treponema pallidum particle agglutination is recommended to confirm diagnostic results. While in screening populations discrepancies between chemiluminescent microparticle immunoassay and Treponema pallidum particle agglutination results are quite prevalent, confirmation by immunoblot assay may be useful.
Introduction
Syphilis is a sexually transmitted disease (STD) caused by the spirochete Treponema pallidum (TP). Nearly eliminated from China 50 years ago, 1 syphilis has now returned to the country. 2 According to China’s national STD surveillance system and sentinel site network, primary and secondary syphilis alone affected 5.7 out of 100,000 people in 2005. The rate of congenital syphilis has increased recently, from 0.01 per 100,000 live births in 1991 to 19.68 per 100,000 live births in 2005, with an average yearly increase in incidence of 71.9%. 3
Since the natural course of infection is characterized by periods of latency and the disease can mimic a variety of conditions in its clinical presentation, 4 laboratory examination is of particular importance in syphilis diagnosis. Moreover, TP can be cultured only in vivo, 5 and direct microscopy is possible only when lesions are present, so serological testing is currently the most widely used laboratory technique to diagnose syphilis and monitor its course after treatment.
Serological tests are divided into two types of assays: non-treponemal tests and treponemal tests. All non-treponemal tests, such as venereal disease research laboratory test and rapid plasma reagin (RPR) test, measure anticardiolipin antibodies produced during active infection. Treponemal tests, such as enzyme-linked immunosorbent assay (ELISA), TP particle agglutination assay (TPPA), fluorescent treponemal antibody-absorption test and Western blot detect antibodies directed against specific TP antigens. Traditional testing for syphilis has consisted of initial screening with a non-treponemal test, followed by retesting reactive specimens with a more specific treponemal test. 6 With recent availability of automated treponemal enzyme and chemiluminescent microparticle immunoassays (EIA/CMIA), some high-volume clinical laboratories in China have adopted treponemal EIA/CMIA for screening followed by testing of reactive serum samples with a non-treponemal test.7,8
EIA/CMIA may be a more sensitive (95–99%) and specific (98–99%) screening technique,9,10 and thus can shorten patients’ seronegative window following TP infection. 11 We have previously found that CMIA has higher sensitivity than ELISA and TPPA (96.7 vs. 93.4% and 91.0%). 12 However, we found in clinical practice that a significant number of CMIA-positive samples were confirmed negative with TPPA.
The purpose of the present study was to investigate the consistency between CMIA and TPPA in a routine screening population and the characterization of patients with discordant syphilis serology (CMIA-positive and TPPA-negative).
Materials and methods
Population and samples
The study population included 20,550 participants (10,575 males and 9975 females) recruited to the having attended Peking University First Hospital for various reasons from June to December 2014. Participants’ mean age was 48 years (ranged from 10 to 97 years). All participants gave informed and written consent before participating in this study. All protocols were approved by local Scientific Ethics Committee.
Blood samples were collected from the antecubital vein. After clotting, blood was centrifuged at 1200 g for 10 min to obtain serum for CMIA, RPR and TPPA tests. Serum samples were stored at −20℃ for retesting by dot-immunoblot (dot-IBT).
Laboratory procedures
Treponemal CMIA was performed as the initial screen for syphilis using an Architect syphilis TP assay (Abbott Laboratories, Abbott Park, IL, USA), which is a two-step immunoassay for qualitative detection of TP antibodies in human serum or plasma. The assay was performed manufacturer’s instructions.
No further testing was done on specimens that were non-reactive. CMIA-reactive specimens were subsequently tested with RPR (Kehua Inc., Shanghai, China) and TPPA (Fujirebio Inc., Japan). Samples that were CMIA-positive and TPPA-negative were further analysed with Euroimmun dot-IBT on a EUROBlotMaster and EUROLineScan (Euroimmun Medizinische Labordiagostika AG, Germany). TPPA, RPR and dot-IBT were performed following the manufacturers’ instructions.
Statistical analysis
Statistical calculations were performed using MedCalc version 6 (Medcalc Software, Mariakerke, Belgium). Descriptive statistics were used to analyse the data.
Results
Serum reactivity for CMIA, TPPA, and RPR
The screening results of the 20,550 serum samples and their subsequent testing are shown (Figure 1). For CMIA, 267 samples (1.3%) were positive. The samples’ signal to cut-off ratios (S/CO) ranged from 1.02 to 45.97. After duplicate tests, the 267 CMIA-positive samples underwent reflex testing with TPPA. TPPA results were positive in 185 samples (69.3%) and negative in 82 samples (30.7%). When the 267 CMIA-positive samples were also tested by RPR, 54 samples (20.2%) were reactive and 213 samples (79.8%) were non-reactive.
Sequence of clinical diagnostic syphilis testing based on serology results from various assays.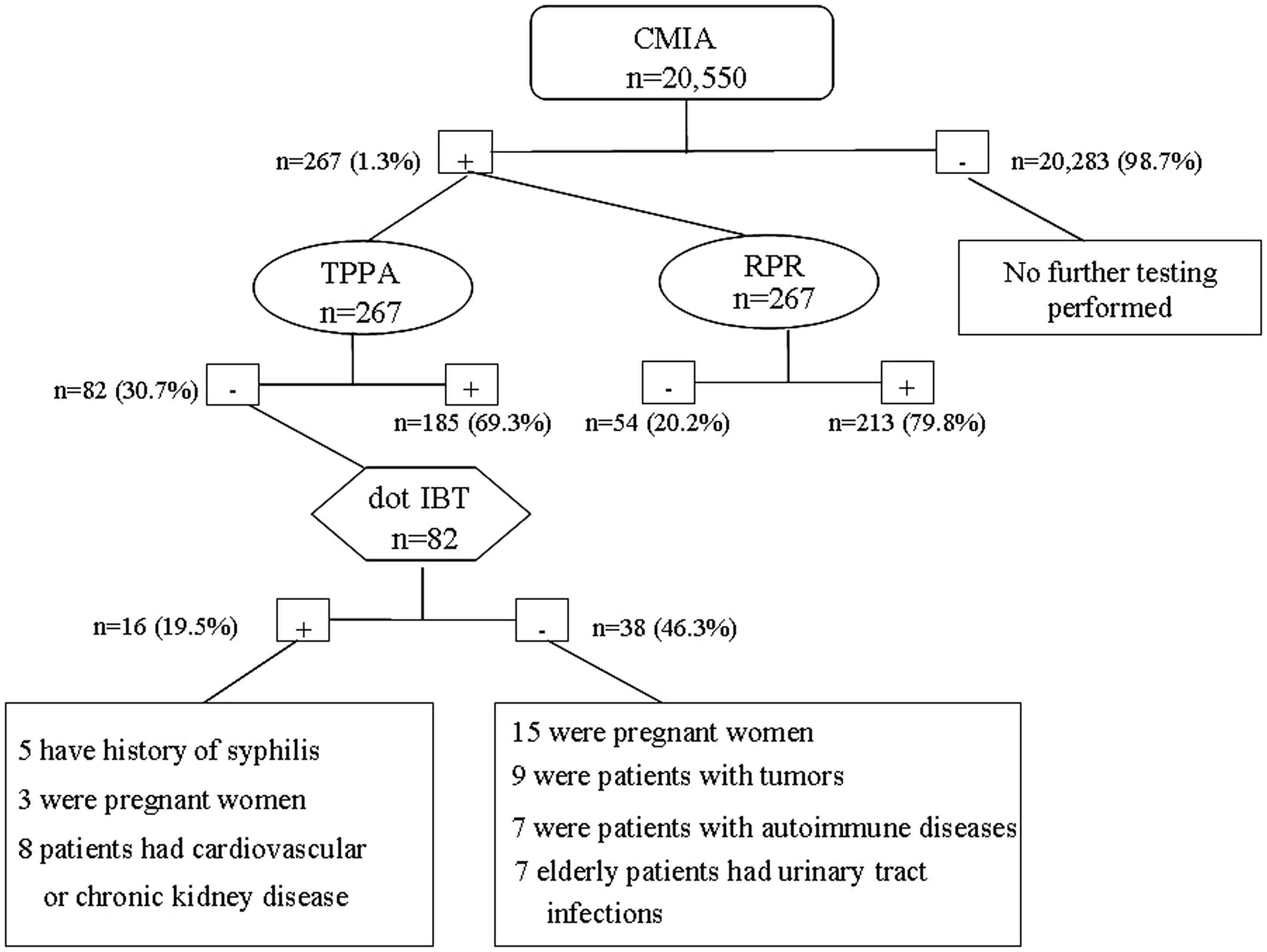
Correlation between CMIA and TPPA results
To determine if there was correlation between CMIA and TPPA results, we analysed samples by their S/CO for CMIA. When the CMIA S/CO was >10, all 117 samples were TPPA-positive (Figure 2). RPR was reactive in 42 of the 117 samples (35.9%). When the CMIA S/CO was 6–9.99, 31/34 samples (91.2%) were TPPA-positive. RPR was reactive in 8 of the 34 samples (23.5%). When the CMIA S/CO was 3–5.99, 23/28 samples (60.5%) were TPPA-positive. RPR was reactive in 4 of the 28 samples (14.3%). When the CMIA S/CO was 1–2.99, 14/76 samples (18.4%) were TPPA-positive. RPR was non-reactive in all 76 samples.
Rate of positive Treponema pallidum particle agglutination assays (TPPA) when serum samples are divided into groups according to the signal to cut-off ratio (S/CO) for chemiluminescent microparticle immunoassays (CMIA).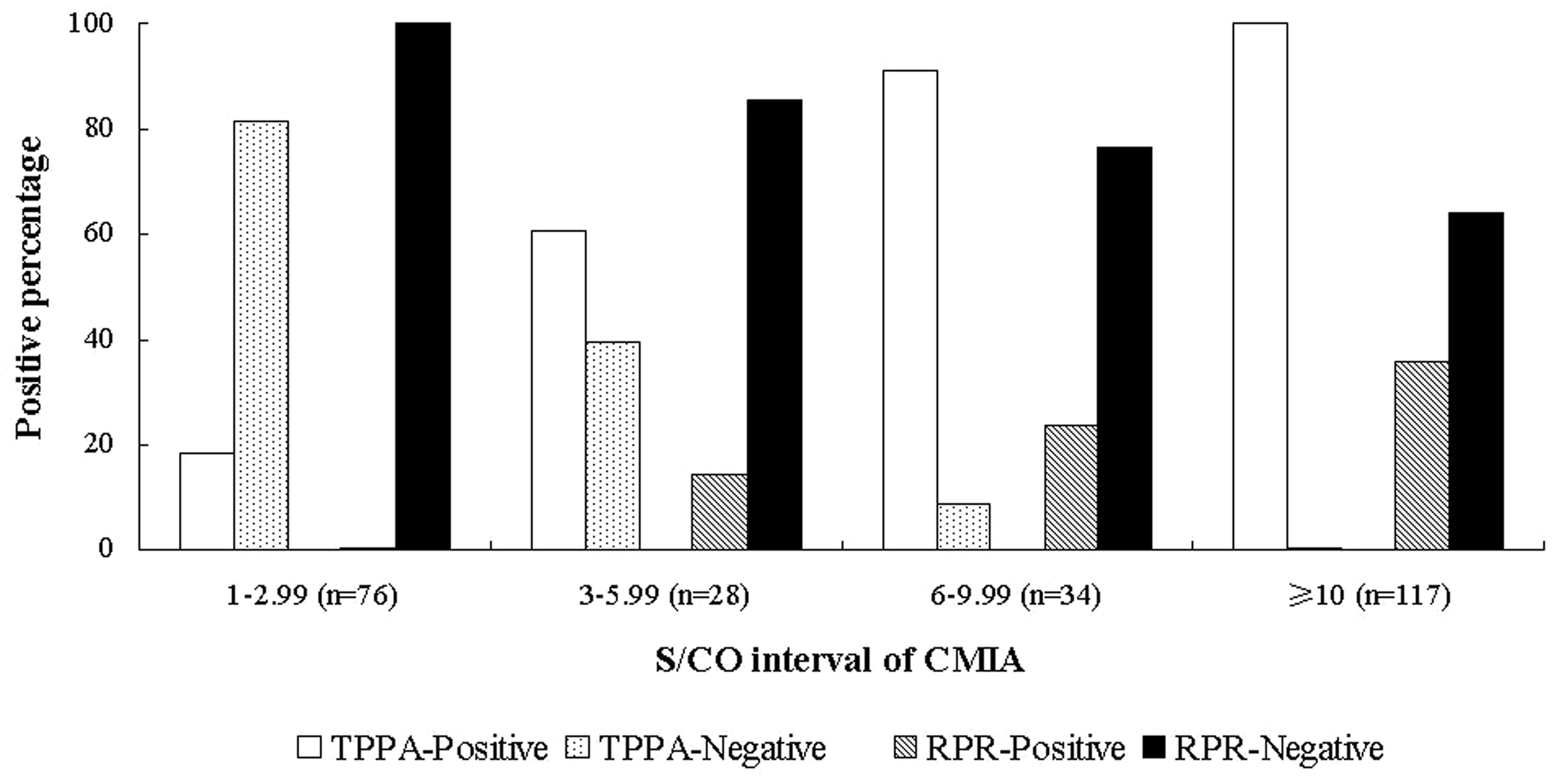
Characterization of samples with CMIA-positive and TPPA-negative results
There were 82 (30.7%) samples that were CMIA-positive but TPPA-negative, and 2 of these samples were RPR-positive (Figure 2). To further classify these samples, we performed dot-IBT on the 82 discordant serum samples. Dot-IBT results showed that 16 samples (19.5%) were positive, 28 (34.2%) were indeterminate and 38 (46.3%) were negative. CMIA S/CO values for these samples ranged from 1.12 to 9.87. Clinical data showed that of the 16 dot-IBT-positive samples, 5 patients had a history of syphilis, 3 were pregnant women, and 8 had cardiovascular or chronic kidney disease. In addition, of the 38 dot-IBT-negative samples – or CMIA false-positives – 15 were pregnant women, 9 were patients with tumours, 7 were patients with autoimmune diseases, and 7 were elderly patients with urinary tract infections.
Discussion
Diagnosis of syphilis is based on clinical evaluation, detection of the causative organism TP and confirmation of disease by sero-diagnosis. Most infected individuals have no symptoms or have transient lesions, and therefore a serological test is necessary for screening. Serological tests are divided into non-treponemal and treponemal tests and neither alone is sufficient for diagnosis.
Non-treponemal tests, such as RPR, can be used to monitor responses to treatment or to indicate new infections. However, these methods detect antibodies to cardiolipin and are not specific for treponemal infection. False-positive non-treponemal tests occur in 1–2% of the U.S. population and have been associated with multiple other conditions, including pregnancy, human immunodeficiency virus infection, intravenous drug use, tuberculosis, rickettsial infection, non-syphilis spirochetal infection, bacterial endocarditis and disorders of immunoglobulin production.13,14 In addition, non-treponemal test results can be falsely negative in longstanding latent infections. 6
Treponemal tests – such as ELISA, TPPA and dot-IBT, which are based on TP-derived antigens – allow detection of specific treponemal antibodies. These tests have higher sensitivity and specificity than non-treponemal tests and are used as confirmatory tests for syphilis after a reactive non-treponemal screening. However, because treponemal antibodies may last a lifetime after infection, they cannot distinguish between current and past infections and also cannot be used to evaluate therapeutic effects. Moreover, these methods are time-consuming and are not suitable for large-scale testing and automatic handling.
The CMIA assay used here offers the advantages of automation, higher testing throughput, and objective interpretation. In this study, we analysed detection results from routine clinical samples and evaluated the performance of CMIA for screening TP antibodies. The results showed that among 20,550 serum samples, 267 samples (1.3%) were CMIA-positive and 185 samples (69.3%) were TPPA-positive.
Further, we found that the quality of CMIA signal can help predict diagnostic accuracy of the test. When the S/CO for CMIA results was 1–2.99 and 3–5.99, 18.4% and 60.5% of samples were TPPA-positive, respectively. So, if CMIA screening results are 1–5.99 S/CO, samples should be tested reflexively using TPPA as a confirmatory treponemal test. When results of CMIA were >6 S/CO, over 90% of samples were TPPA-positive, indicating that for those samples CMIA results can be directly reported as positive without confirmation. But for pregnant women, patients with tumours or autoimmune disease, and elderly individuals >60 years old, additional detection methods beyond CMIA are recommended for confirmation because of false-positives. RPR tests should be reported only to assist clinical distinction between past or current infections. Therefore, individual clinical situations must be taken into account when considering diagnostic tests for syphilis.
A suitable confirmatory test should have, at least, sensitivity and specificity equivalent to those of screening assays. Manavi et al. 15 have suggested that, in the absence of a specific treponemal IgM EIA, a TPPA test should be performed whenever there is clinical suspicion of primary infection, as TPPA is more sensitive in primary infections. In this study, 82 (30.7%) serum samples with CMIA-positive results were TPPA-negative. Sixteen of the TPPA-negative serum samples (19.5%) were positive by dot-IBT. It would be reasonable to classify those TPPA-negative, CMIA-positive and dot-IBT-positive serum samples as true treponemal antibody seropositives, which would therefore regard TPPA results as false-negatives.
A previous study by Maple et al. 16 that examined Abbott’s Murex ICE syphilis test found that 11.6% of reactive serum samples were TPPA-negative, and, after confirmation by line immunoblot assay and fluorescent treponemal antibody test, 69% of those TPPA-negative but ICE-positive serum samples were true treponemal antibody seropositives. However, the differences between that study and ours can be attributed to different populations – Maple et al. 16 studied samples sent to a laboratory for confirmatory serology, while our samples came from a syphilis screening population.
Although there are no clinical data to identify stage of infection, we found that 13/16 serum samples (81.3%) were positive by CMIA, with S/CO values of 1.15–3.06. Low-level antibodies to certain syphilis antigens (TpN47, TpN17, TpN15, and TmpA) could suggest that these patients may be in primary stages of infection. In view of our findings, CMIA-positive and TPPA-negative serum samples may need further confirmatory tests, such as dot-IBT, for accurate diagnosis.
Overall, because there is a certain percentage of false-positive results using CMIA for routine screening of syphilis, further analysis by TPPA is recommended for accurate diagnosis. While screening populations have prevalent discrepancies between treponemal CMIA and TPPA results, this seems to be a function of very low levels of syphilis-specific antibodies. Therefore, confirming diagnostic results with additional assays, such as dot-IBT, may remain useful.
Footnotes
Acknowledgements
We are grateful to Dr. HaiXia Li (Department of Clinical Laboratory, Peking University First Hospital, China) for providing language help.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
The study protocol was approved by the Ethical Medical Board of Peking University First Hospital (2014[691]).
Guarantor
ZY Li.
Contributorship
Zhiyan Li is responsible for design of the work, analysis and interpretation of data and drafting the manuscript. Zhenru Feng is responsible for interpretation of data and editing the manuscript. Cunling Yan and Ping Liu are responsible for specimen collection and detection.
