Abstract
Background
Anyplex II HPV HR (Anyplex_HR; Seegene, Seoul, Korea) is a new multiplex real-time polymerase chain reaction assay for screening cervical cancer, and it is designed to detect 14 high-risk human papillomaviruses along with all the genotype information in a single tube. The aim of this study was to evaluate the performance of the Anyplex_HR in comparison to that of the Cobas 4800 HPV (Cobas_4800; Roche Molecular Diagnostics, Pleasanton, CA, USA) and the Hybrid capture 2 (HC2; Qiagen GmbH, Hilden, Germany).
Methods
The performance of the Anyplex_HR for high-risk human papillomavirus genotype detection was prospectively evaluated against that of the HC2 and the Cobas_4800 at the National Cancer Center using 400 cervical samples. All discrepant samples were confirmed by polymerase chain reaction with type-specific primers followed by sequencing.
Results
The overall agreement and kappa value of the Anyplex_HR with the Cobas_4800 were 98.0% and 0.96, respectively. The level of agreement between the two assays and the corresponding kappa values for human papillomavirus16, human papillomavirus18 and other high-risk human papillomaviruses were 99.5%, 99.8% and 98.8%, and 0.98, 0.96 and 0.97, respectively. The agreement and kappa value of the HC2 with the Cobas_4800 were 95.3% and 0.91. The human papillomavirus positivity of the Anyplex_HR and the Cobas_4800 in low-grade squamous intraepithelial lesion/high-grade squamous intraepithelial lesion samples demonstrated 100% concordance. Both the Anyplex_HR and the Cobas_4800 showed excellent results in the precision test.
Conclusions
The Anyplex_HR is comparable with the Cobas_4800 and the HC2 for human papillomavirus DNA testing, and it may prove more useful for follow-up testing and patient management by providing genotyping information additional to human papillomavirus16 and human papillomavirus18.
Introduction
Cervical cancer is the second most commonly diagnosed cancer and the third leading cause of cancer death among females in less developed countries, with 527,600 new cases and 265,700 deaths estimated to occur annually. 1 Human papillomavirus (HPV) has been identified as the leading cause of cervical cancer in women. 2 The consolidated causal link between high-risk HPVs (hrHPVs) and cervical cancer has led to the introduction of HPV DNA testing in screening programmes along with cytology testing.
In the United States, hrHPV testing is recommended for women presenting with atypical squamous cells of unknown significance (ASCUS) cytology, and co-testing for cytology and HPV is recommended for screening women aged ≥30 years. 3 HPV nucleic acid testing has recently emerged as the most likely candidate to replace cytology for primary screening.4,5 HPV testing with HPV16 and/or HPV18 genotyping could provide improved detection of women at high risk for cervical cancer, relative to that achieved with cytology testing. 6 There is clear scientific evidence that screening based on validated tests for the DNA of oncogenic HPV as a primary test and applying an appropriate protocol are more effective than screening based on cytology in preventing invasive cancers of the uterine cervix. 7 In April 2014, the FDA approved the use of an HPV test (Cobas) for primary cervical cancer screening in women above 25 years-of-age, without the need for a concomitant Papanicolaou (Pap) test. 8
Anyplex II HPV HR (Anyplex_HR; Seegene, Seoul, Korea) is a recently developed multiplex, real-time polymerase chain reaction (PCR) assay designed to detect and simultaneously distinguish 14 unique hrHPV genotypes in a single tube. The Anyplex_HR has unique features for specific amplification (HPV-specific dual priming oligonucleotides [DPO]) 9 and cyclic-catcher melting temperature analysis (cyclic-CMTA) that facilitate the analysis of multiple analytes in a single tube to overcome the multiplexing limitations of conventional real-time PCR. 10
We believe this to be the first study to compare the analytical performance of the Anyplex_HR, the Roche HPV 4800 test (Cobas_4800; Roche Molecular Diagnostics, Pleasanton, CA, USA) and the Hybrid capture 2 (HC2; Qiagen GmbH, Hilden, Germany).
Materials and methods
Clinician-collected cervical specimens in liquid medium (Cobas PCR cell collection media, Roche Molecular Diagnostics) were prospectively collected from 400 women who visited the department of gynecology and oncology for the treatment of cervical cancer or for follow-up at the National Cancer Center in Korea between November 2014 and February 2015. The specimens were used for routine clinical testing for HPV DNA (Cobas_4800). The performance of the Anyplex_HR and the HC2 was evaluated in comparison to that of the Cobas_4800 using 400 archived routine cervical samples (200 positive samples and 200 negative samples detected by the Cobas_4800). The cytology results of these 400 samples represented 290 samples negative for intraepithelial lesions or malignancies (NILMs), 40 samples with ASCUS, 22 with low-grade squamous intraepithelial lesions (LSILs), 42 with high-grade squamous intraepithelial lesions (HSILs) and six with atypical glandular cell samples. The average age of the participants was 52 years (range: 23–81 years). The Cobas_4800 test was carried out using the Cobas 4800 system (Roche Molecular Diagnostics), comprising the Cobas x 480 instrument and the Cobas z 480 analyzer, which are fully automated instruments for nucleic acid preparation and real-time PCR, respectively. Nucleic acid extraction using 400 µL of collection media per subject yielded 150 µL of nucleic acid eluted sample, which was used for Cobas x 480 real-time PCR (Roche Molecular Diagnostics) to detect HPV DNA. All assay procedures were carried out following the manufacturers' instructions. The residual cell suspensions were stored at 4–8 ℃ for up to two weeks until analyzed by the Anyplex_HR and the HC2 HPV DNA test. For the Anyplex_HR, HPV DNA was extracted using an automated extraction instrument (NIMBUS, Hamilton, Reno, NV, USA). The Anyplex_HR assay was carried out using a CFX96 real-time thermocycler (Bio-Rad, Hercules, CA, USA) according to the manufacturer's instructions. Briefly, each PCR reaction was performed in 20 µL of reaction mixture containing 5 μL of extracted DNA and PCR master mix. The thermal cycler conditions consisted of initial incubation at 50 ℃ for 4 min for the activation of the uracil DNA glycosylase system, denaturation at 95 ℃ for 15 min, 50 cycles of denaturation (30 s at 95 ℃), annealing (1 min at 60 ℃) and elongation (30 s at 72 ℃).
The Cobas_4800 test and the Anyplex_HR are able to detect 14 HR HPV genotypes (HPV types 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68). Both the Cobas_4800 and Anyplex_HR real-time PCR assays also detect the aforementioned internal-control target gene (human β-globin gene) to ensure the quality of prepared nucleic acids.
For the HC2 assay, 4 mL of the originally collected specimens in Roche transport media were processed with the Sample Conversion Kit (Digene-Qiagen, Valencia, CA, USA) according to manufacturer's instructions. After conversion, a 75-µL aliquot of the sample was used for the HC2 test, according to the manufacturer's protocol. After denaturation of the nucleic acids, the single-stranded HPV DNA from the specimens was hybridized with mixed RNA probes specific for 13 h HPV genotypes (HPV types 16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59 and 68).
Type-specific PCR and sequencing
All discrepant samples were confirmed by PCR with type-specific primers followed by sequencing. Type-specific PCR was a nested PCR targeting E6/E7 oncogene and performed as described previously. 11 Briefly, in this assay, consensus primers for the first-round amplification of a broad spectrum of mucosal HPV genotypes, including all high-risk HPV genotypes, were combined with type-specific primers for nested PCR amplifications. The PCR conditions used with E6 consensus primers were 15 min at 95 ℃, 40 cycles of 94 ℃ for 1 min, 40 ℃ for 1 min and 72 ℃ for 2 min. Second-round PCR with type-specific primers was performed under the following conditions: 15 min at 95 ℃, 35 cycles of 94 ℃ for 30 s, 55 ℃ for 30 s and 72 ℃ for 45 s, according to Sotlar et al. 11 with minor modifications. All PCRs were performed using HotStarTaq Plus Master Mix Kit (Qiagen GmbH) with 15 pmol of each primer. Five microliters of the PCR products served as a template for the nested PCRs; 10 μL of the amplification products were analysed by electrophoresis on 2% agarose gels.
PCR sequencing was performed using the PGMY primers, 12 and the respective sequences of the HPV DNA regions corresponding to the two primer sets were read using the Applied Biosystems 3730xl DNA analyzer (Life Technologies Co., Carlsbad, CA, USA).
Precision
The precision of the assays was assessed by comparing the positive control results from 15 replicates using clinical specimens with low (quantitation cycle [Cq] ∼37), intermediate (Cq 32.2) and high (Cq 26.7) concentrations of HPV DNA, in triplicate per run a day on five different days. The relative standard deviation (SD) of the Cq values was calculated as a measure of stability of the Cobas_4800. For the Anyplex_ HR, only the qualitative results are presented.
Statistical analyses
Statistical analyses for the reproducibility study were performed as follows. All statistical analyses were performed using Analyse-it for Microsoft Excel ver. 2.20 (Analyse-it Software, Leeds, UK). The total SD and the total coefficient of variation (CV) were calculated. The data were summarized as the percentage of positive and negative results and the associated exact 95% confidence intervals (CIs) for agreement estimates. Kappa coefficients (κ) with 95% CIs were calculated to estimate the agreement between the results of the HPV DNA tests.
Results
Agreement
Overall concordance among assay results
Overall concordance among assay results (n = 400).
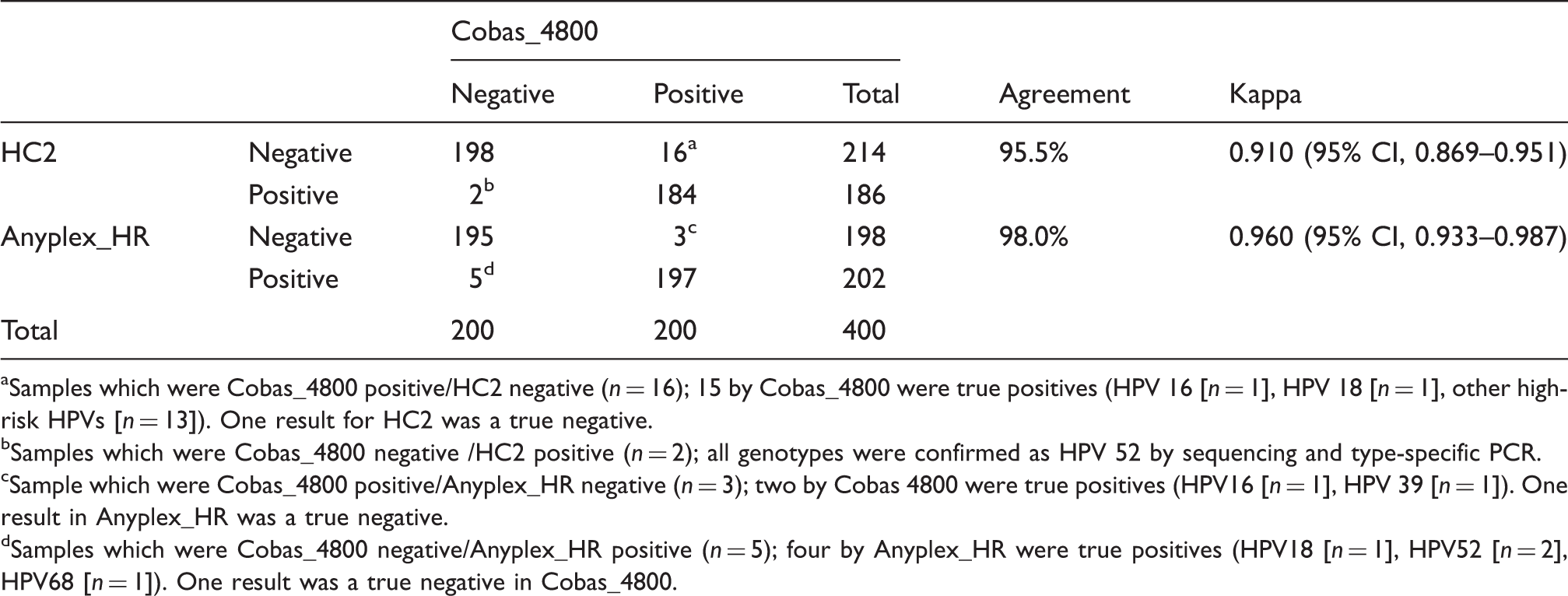
Samples which were Cobas_4800 positive/HC2 negative (n = 16); 15 by Cobas_4800 were true positives (HPV 16 [n = 1], HPV 18 [n = 1], other high-risk HPVs [n = 13]). One result for HC2 was a true negative.
Samples which were Cobas_4800 negative /HC2 positive (n = 2); all genotypes were confirmed as HPV 52 by sequencing and type-specific PCR.
Sample which were Cobas_4800 positive/Anyplex_HR negative (n = 3); two by Cobas 4800 were true positives (HPV16 [n = 1], HPV 39 [n = 1]). One result in Anyplex_HR was a true negative.
Samples which were Cobas_4800 negative/Anyplex_HR positive (n = 5); four by Anyplex_HR were true positives (HPV18 [n = 1], HPV52 [n = 2], HPV68 [n = 1]). One result was a true negative in Cobas_4800.
Concordance between Anyplex_HR Detection and Cobas_4800 for genotypes 16 and 18 and other hrHPVs
Concordance between Anyplex II HPV HR Detection (Anyplex_HR) and the Cobas 4800 HPV test (Cobas_4800) using clinical samples for 14 high-risk human papillomaviruses (HPVs).
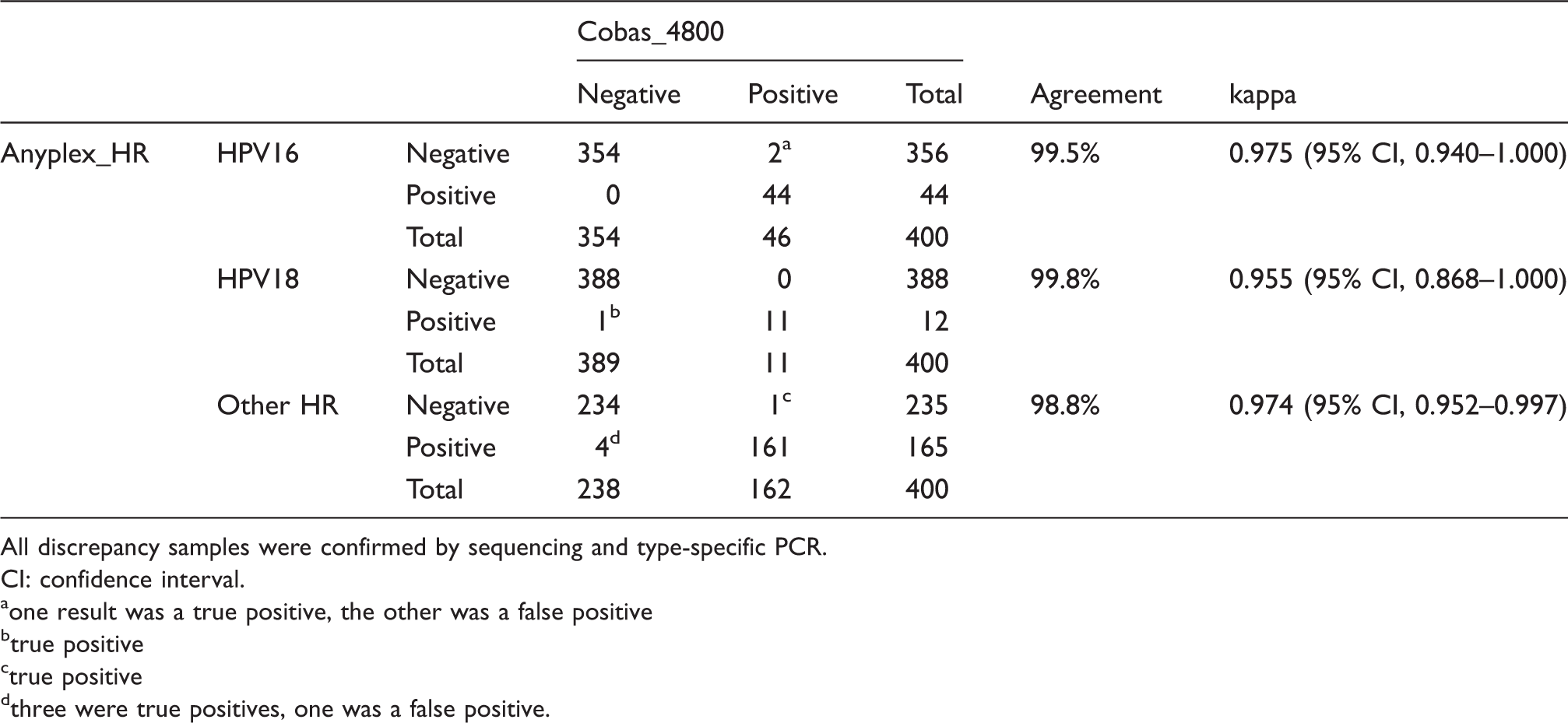
All discrepancy samples were confirmed by sequencing and type-specific PCR.
CI: confidence interval.
one result was a true positive, the other was a false positive
true positive
true positive
three were true positives, one was a false positive.
The agreement and kappa values for genotype 18 and other hrHPVs were 99.8%, 0.955 (95% CI, 0.868–1.000) and 98.8%, 0.974 (95% CI, 0.952–0.997), respectively. The Cobas_4800 showed one false negative result for a HPV 18 sample that was positive by the Anyplex_HR. For the other hrHPV genotypes, five samples showed discrepancy between the Cobas_4800 and the Anyplex_HR. Among them, four samples showed Cobas_4800 negative/Anyplex_HR positive results, of which three were confirmed as true positives (HPV genotype 52 [n = 2], 68 [n = 1]) and one was a false positive by the Anyplex_HR. One sample with a Cobas_4800 positive/Anyplex_HR negative result was found to be a true positive. All individual discrepant results are shown in supplementary Table 4.
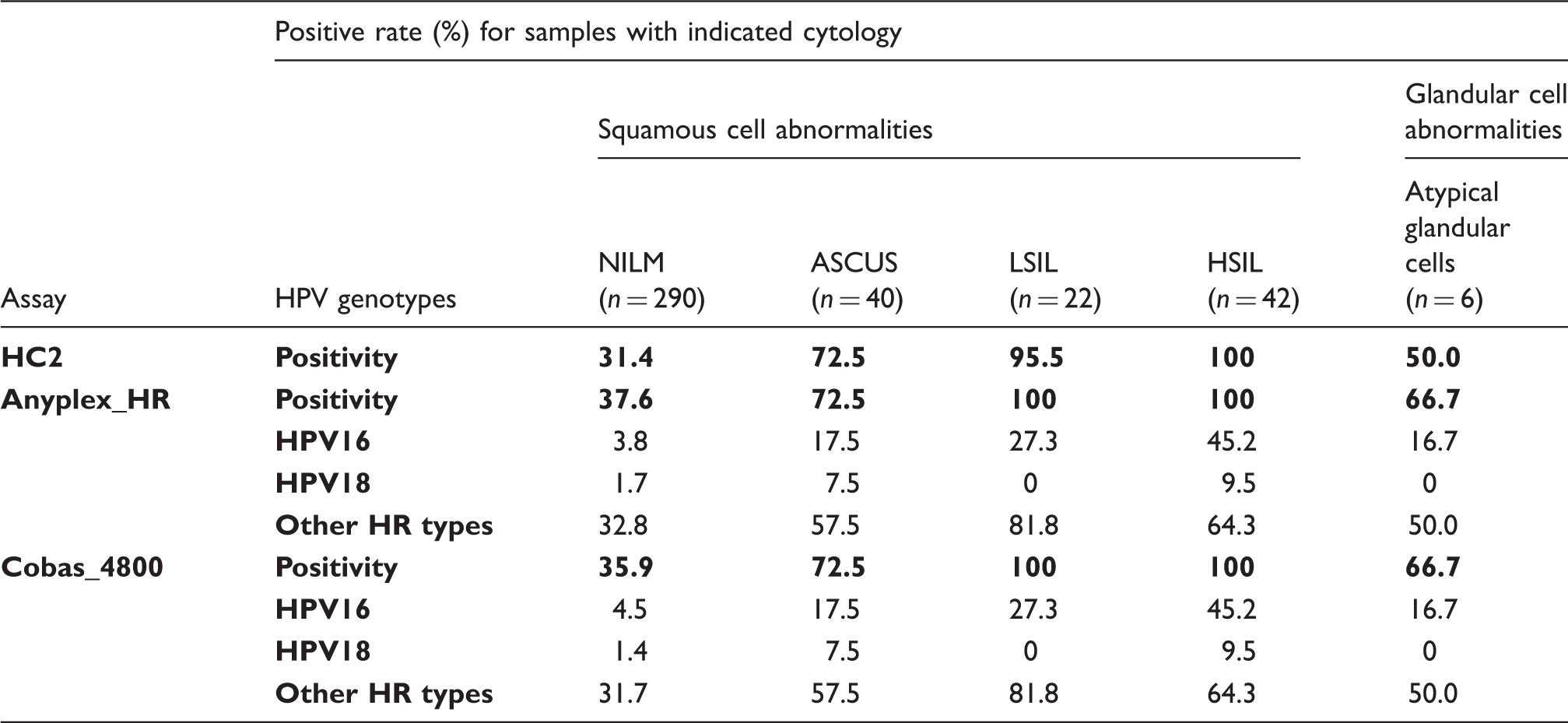
Concordance between Anyplex_HR Detection and Cobas_4800 according to cytology results
Results of cervical cytology related to human papillomavirus (HPV) DNA detection with Anyplex II HPV HR Detection (Anyplex_HR) and the Cobas 4800 HPV test (Cobas_4800).
Precision
The precision tests showed that there was 100% detection of three different levels (low, medium and high concentration) of pooled clinical specimens using the Anyplex_HR and the Cobas_4800 (Supplementary Table 1). Analysis of variance of the Cq values from the Cobas_4800 yielded total CVs ranging from 1.8% to 8.9% for the pooled clinical samples (repeatability ranging from 1.6% to 8.9%) (Supplementary Table 2).
Discussion
In this study, we compared the performance of two real-time PCR assays (the Anyplex_HR and the Cobas_4800) and the HC2. The overall agreement (98.0%) between the Anyplex_HR and the Cobas_4800 was very good. In addition, the performance of the Anyplex_HR was more similar to that of the Cobas_4800 than to that of the HC2. The agreement of genotype 16 and 18 detection was very high (99.5% and 99.8%, respectively) between the Anyplex_HR and the Cobas_4800. Most of the discrepant results were obtained for positive samples with Cq values greater than 35 of the result on the Cobas_4800. The discrepant cytology results between the Anyplex_HR and the Cobas_4800 were, in all cases, in samples with NILM. There was no difference in the HPV detection rate between the Anyplex_HR and the Cobas_4800 in the cytology results of the LSIL and HSIL groups, showing that both techniques had a sensitivity of 100%. In our study, the detection rate of HPV in samples with negative cytology was relatively high (37.6% for the Anyplex_HR and 35.9% for the Cobas_4800) because most patients are treated immediately after diagnosis.
For other hrHPV genotypes, five samples showed discrepancy between the results yielded from the Anyplex_HR and the Cobas_4800. Most discrepant samples (n = 3), which were negative by the Cobas_4800 and positive by the Anyplex_HR, were confirmed as true positives (HPV genotype 52 [n = 2], 68 [n = 1]). The Anyplex_HR could detect more alternative hrHPV genotypes than the HC2 was able to.
In the present study, the co-infection detection rates of the Anyplex_HR and the Cobas_4800 were 10% (20/200) and 8.5% (17/200), respectively. This result is similar to that of a previous study that reported multiple HPV infections in approximately 15.2% (21/138) of samples in Korea using the Seegene multiplex kit. 13 Several studies have reported that multiple infections are significantly associated with a risk of cervical neoplasia.14,15
The Anyplex_HR has unique features for HPV-specific DPO 9 and cyclic-CMTA that facilitates the analysis of multiple analytes in a single tube. 10 These techniques have already been validated for the detection of respiratory viruses. 10 Thus, Anyplex_HR can differentiate all the 14 HR HPV genotypes. In the Cobas_4800, one reaction tube with four different reporter dyes was used for each HPV DNA test, and the results of the Cobas_4800 were reported as either positive or negative for HPV types 16 and 18, with a pooled result for the remaining 12 HR genotypes. Thus, the Cobas_4800 could not distinguish between the 12 other HR HPV genotypes besides HPV 16 and 18. Both real-time PCR assays also detected the internal-control target gene (human β-globin gene) to ensure the quality of the prepared nucleic acids.
Single genotyping is important to predict the impact of vaccination on genotype distribution.16,17 A reduction in the prevalence of HPV16/18 from 19.1% to 6.5% was observed in sexually active young women after the introduction of HPV immunization. 18 The use of HPV genotyping tests is recommended in order to predict the potential severity of a precancerous state with abnormal cytology, even if the patient is a young woman. 19
Overall the agreement between the Cobas_4800 and the HC2 was relatively good, with kappa values of 0.763–0.832.20,21 The Cobas_4800 has also been shown to demonstrate a lower rate of cross-reactivity to low-risk HPV genotypes, and has the advantage over the HC2 test of simultaneously providing HPV16 and HPV18 identification. 22 The limitations of the HC2 assay were that it could not detect other HR HPV types such as HPV66, nor could it distinguish individual genotypes or identify infection with multiple genotypes.
Performance of real-time PCR assays in cervical cancer screening settings has shown consistently high absolute clinical sensitivity for both CIN2 + and CIN3+, as well as clinical sensitivity and specificity comparable to those of the HC2 in previous studies.20,23,24 Recently, it was reported that the Cobas_4800 could represent a more accurate methodology for primary cervical cancer screening than that of liquid-based cytology.23–26
One of the limitations of the present study was that the test performance of the Anyplex_HR was not evaluated in a primary screening setting. Cervical cancer screening is performed using cervical cytology, a HPV test or a combination of the two. 27 HPV DNA testing has been proven as an accurate methodology for primary cervical cancer screening, either in co-testing in conjunction with cytology, 28 or as a stand-alone test. 19 The overall performance of cytology for the detection of CIN2 + cervical lesions was poor (sensitivity of ∼53%),4,29 and relative to that of the conventional Pap test, liquid-based cervical cytology is not better for the detection of high-grade cervical intraepithelial neoplasia.27,30 HPV testing with separate HPV16 and HPV18 detection provided an alternative, more sensitive and efficient strategy for cervical cancer screening than methods based solely on cytology.31,32 HPV-based screening provides 60–70% greater protection against invasive cervical carcinomas than that provided by cytology. 6 In addition, stored samples were not tested at the same time by the Anyplex_HR and the Cobas_4800 methods. Another limitation was that low numbers of samples of abnormal cervical cytology were included in this study.
In conclusion, the Anyplex_HR was comparable to the Cobas_4800 HPV and the HC2, and thus, it could be used for HPV DNA testing. This test would also be useful for follow-up testing and patient management by providing genotyping information beyond HPV16 and HPV18 positivity. However, the clinical performance of the Anyplex_HR should be analysed in a population of women with abnormal cervical cytology who have been referred for colposcopy.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Seegene (Seoul, Korea) (no. 1441360).
Ethical approval
This study has been approved by the Institutional Review Board of the National Cancer Center, Seoul (IRB No. NCC2014-0183).
Guarantor
SHH
Contributorship
Study conception and design: SHH and DHL.
Acquisition, analysis, or interpretation of data: NRH, JJ, MCL, JYK and CWY.
Drafting of manuscript: SHH.
Statistical analysis and interpretation: JJ.
Administrative, technical, or material support: DHL and SYP.
All authors reviewed and edited the manuscript and approved the final version of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
