Abstract
Introduction
The biologic anti-tumour necrosis factor alpha (anti-TNFα) agents infliximab and adalimumab are monoclonal antibodies with binding specificity to TNFα, which are used for the treatment of Crohn’s disease. Clinical response is varied from complete with mucosal healing, to primary non-response, loss of response and adverse drug reactions. Measuring trough blood levels of infliximab and adalimumab may guide clinical management. The sample handling requirements for infliximab and adalimumab were previously unknown.
Aim
The aim of this study was to determine the in vitro stability of infliximab and adalimumab in samples stored for up to seven days at room temperature.
Methods
Samples were stored as clotted whole blood or serum at room temperature for up to seven days, before being frozen (−20℃) and analysed as a batch for either infliximab or adalimumab.
Results
No significant difference between the concentration of infliximab and adalimumab measured in samples stored as serum or whole blood for seven days at room temperature, as compared to baseline was found (t-test; infliximab: P = .35 [serum], P = .38 [whole blood]; adalimumab: P = .12 [serum], P = .49 [whole blood]).
Conclusion
The stability of infliximab and adalimumab at room temperature for seven days allows samples to be posted direct from clinics and research centres to the analysing laboratory.
Introduction
The biologic anti-tumour necrosis factor alpha (anti-TNFα) agents infliximab and adalimumab are monoclonal antibodies with binding specificity to TNFα and are used for the treatment of Crohn’s disease. These drugs are expensive (for standard dosage for one year, the costs of drug are £9000–£12,500)
1
and are administered intravenously (infliximab) or subcutaneously (adalimumab). In some patients, treatment with anti-TNFα agents is extremely effective, leading to mucosal healing and a complete clinical response. In other patients, the therapy is less effective due to one of several problems:
Primary non-response (a clinical response to these agents is never observed) Loss of response (although a good clinical response is initially observed, this is not maintained and the drug becomes ineffective) Adverse drug reactions
It has been suggested that measuring trough blood levels of infliximab and adalimumab may be an important tool in managing clinical response to these expensive agents,2,3 but the evidence is limited. It is established that a detectable anti-TNFα trough level (immediately predose) is associated with a higher rate of clinical and endoscopic remission.4,5
The PANTS (Personalised Anti-TNFα Therapy in Crohn’s disease, UK Clinical Research Network Study Portfolio: http://public.ukcrn.org.uk/search/StudyDetail.aspx?StudyID=14175) study aims to further understand the relationship between trough drug levels and clinical response. This multicentre study uses a central laboratory at the Blood Sciences Department at the Royal Devon and Exeter Hospital for analysis of drug levels and other analytes. There are 90 different UK recruitment sites for the PANTS study. In order to simplify sample handling at multiple centres, we recognized sending blood directly from the clinic to the central laboratory via the postal service would be practical, convenient and cost effective. This approach means that blood samples for anti-TNFα drug level analysis would be stored as whole blood at room temperature for a number of days before reaching the central laboratory for processing.
Aim
The aim of this work was to determine the in vitro stability of infliximab and adalimumab in samples stored for up to seven days at room temperature, in clotted whole blood and serum.
Methods
We undertook an experiment to determine the stability of infliximab and adalimumab under different storage conditions, to see if posting blood samples (precentrifugation) to the central laboratory from the clinic was a viable approach. We investigated the stability of both drugs in clotted whole blood and serum for up to seven days at room temperature by comparison with the concentrations measured at baseline. Blood samples were collected from patients undergoing infliximab (n = 7) and adalimumab (n = 7) therapy. For each patient, eight serum tubes were collected at a single time point.
To investigate the stability of samples stored as serum, one sample was centrifuged immediately upon receipt in the laboratory, and an aliquot (300 µL) of serum was taken for storage at −20℃ (baseline sample). The remainder of the primary tube was stored at room temperature. Every 24 h for seven days, an aliquot (300 µL) of serum was taken for storage at −20℃ until analysis.
The remaining seven samples were used to investigate the stability of samples stored as whole blood at room temperature, i.e. without immediate centrifugation: one tube was centrifuged every 24 h for seven days, and at each of these time points, an aliquot of serum (300 µL) was taken for storage at −20℃ until analysis.
All of the aliquots collected were stored frozen until they were defrosted and analysed in a single batch for infliximab or adalimumab drug levels. Adalimumab concentration was determined using K9657 TNFα blocker monitoring adalimumab drug level, and infliximab concentration was determined using K9655 TNFα blocker monitoring infliximab drug level (both kits from Immundiagnostik AG, Stubenwald-Allee 8 a, D 64625 Bensheim), on a DS2® ELISA processing system (DYNEX Technologies Ltd, Worthing, UK) platform. The principle of the infliximab assay is that free infliximab binds to the specific monoclonal anti-infliximab antibody coated on the plate upon incubation with serum. After washing, a peroxidase-labelled antibody is added, and there is a second incubation. After a final wash, the peroxidase substrate tetramethylbenzidine is added, and after a short incubation, acidic stop solution is added to terminate the reaction. The intensity of the yellow colour is directly proportional to the concentration of free infliximab in the sample. A dose-response curve of the absorbance (optical density at 450 nm) against concentration is generated, using the values obtained from standard solutions, and the concentrations of free infliximab are determined directly from this curve. The principal of the adalimumab assay is the same as the infliximab assay. The intra-assay % coefficient of variation of the infliximab assay ranges from 1.8% at 4.1 mg/L (n = 24) to 9.7% at 24.2 mg/L (n = 24). The intra-assay % coefficient of variation of the adalimumab assay ranges from 1.5% at 2.7 mg/L (n = 24) to 2.6% at 18.0 mg/L (n = 24).
Results
We found that there was no significant difference between the concentration of infliximab and adalimumab measured in samples stored as serum or whole blood for seven days at room temperature, as compared to baseline (t-test; infliximab: P = .35 [serum], P = .38 [whole blood]; adalimumab: P = .12 [serum], P = .49 [whole blood]) (Figure 1).
Mean change in concentration of infliximab (n = 7 for samples stored as serum and samples stored as whole blood) and adalimumab (n = 7 for samples stored as serum and n = 5 for samples stored as whole blood) for samples stored at room temperature for up to seven days. There was no significant change in the infliximab concentration for samples stored for seven days (P = .35 for samples stored as serum; P = .38 for samples stored as whole blood), nor in the adalimumab concentration for samples stored for seven days (P = .12 for samples stored as serum; P = .49 for samples stored as whole blood). Error bars show 95% CI. Note. No data were collected for adalimumab stored as serum for one day.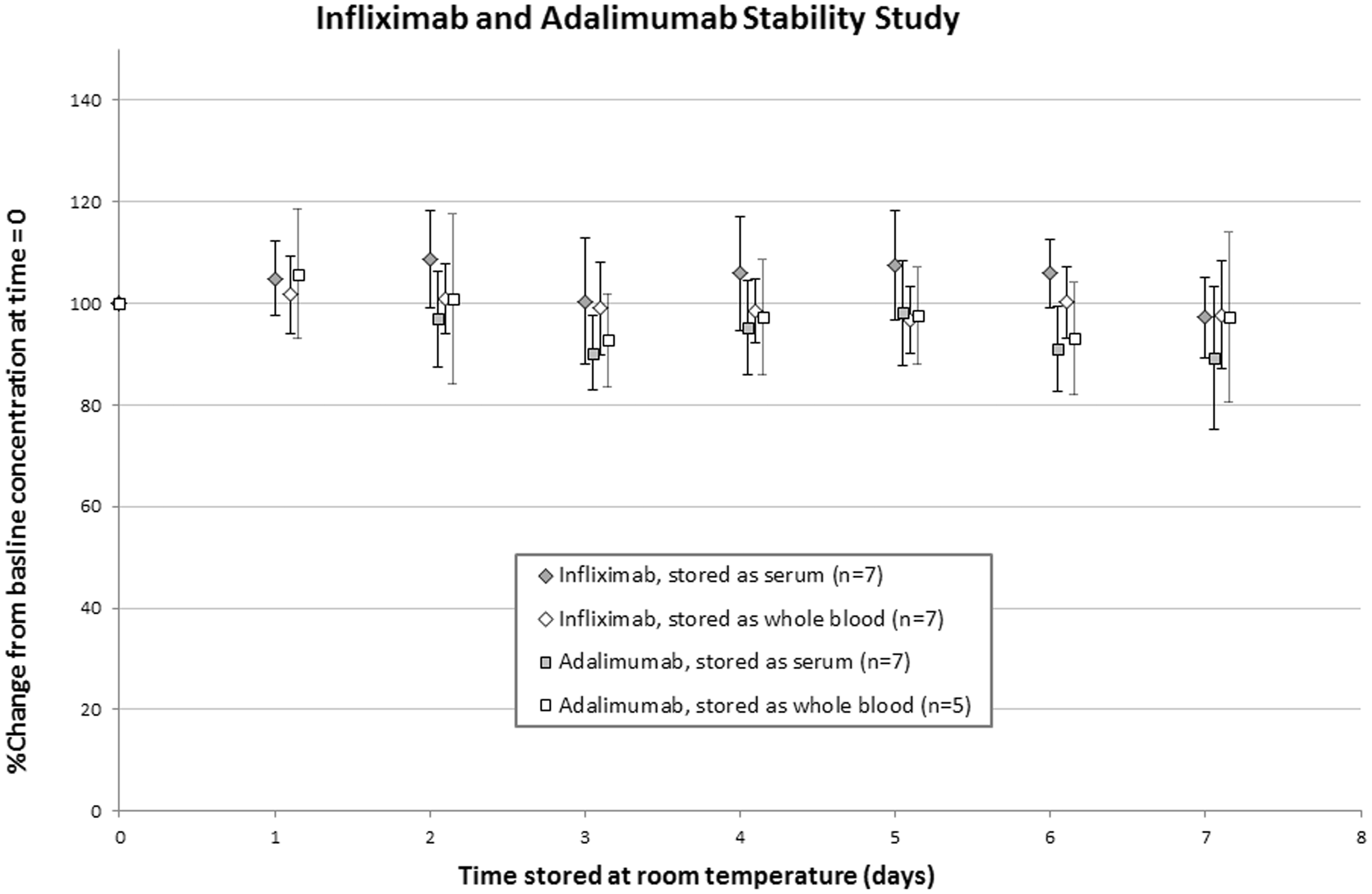
Conclusions
The stability of infliximab and adalimumab at room temperature for seven days on whole blood enables samples to be posted at room temperature direct from clinics and research centres to the analysing laboratory. This finding significantly increases the clinical utility of measuring infliximab and adalimumab, as samples do not need rapid or special processing at the point of collection.
Footnotes
Acknowledgements
We would like to thank Rebecca Crofts, John Vidler, Dave Marshall and Ian Clarke for sample preparation. Dr Tim McDonald is supported by an NIHR CSO Fellowship. BioHit Healthcare provided kits for this study.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
MP.
Contributorship
CB, TA, RB, MP, KS and TM designed the experiment. MP carried out the experiment. MP and TM carried out the data analysis. MP wrote the manuscript and prepared the figure. TM and TA edited the manuscript. All authors reviewed the manuscript and approved the final version of the manuscript.
