Abstract
Background
Various conjugated cholesterol metabolites are excreted in urine of the patients with metabolic abnormalities and hepatobiliary diseases. We aimed to examine the usefulness of precursor ion scan and neutral loss scan for the characterization of conjugated cholesterol metabolites in urine.
Methods
A mixture of authentic standards of conjugated cholesterol metabolites was used for investigating the performance of the present method. The urine of patients with Niemann–Pick diseases type C and 3β-hydroxysteroid dehydrogenase deficiency were analysed by precursor ion scan of m/z 97, 74, and 124.
Results
A precursor ion scan of m/z 97 was effective for identifying conjugates with ester sulphates on hydroxyl groups whereas ester sulphates on phenolic alcohols were signalled by a neutral loss scan of 80 Da. Monosaccharide-conjugated cholesterol metabolites were signalled by a precursor ion scan of m/z 113. Although precursor ion scan of m/z 74 and 124 was effective for finding glycine- and taurine-conjugated metabolites, high intensity of product ions (m/z 74 and 124) disturbed measurement of other multiply conjugated metabolites. The urine samples contained many conjugated cholesterol metabolites, and there were several disease-specific intense peaks. We found several unknown intense peaks with three known peaks in urine of the Niemann–Pick type C patient. In the patient with 3β-hydroxysteroid dehydrogenase deficiency, intense peaks that were tentatively identified as 5-cholenoic acid sulphates and their glycine and taurine conjugates were present.
Conclusion
The method should lead to the discovery of new urinary biomarkers for these disturbances of cholesterol catabolism and transport.
Keywords
Introduction
Cholesterol is converted to bile acids, vitamin D, and steroid hormones. Although cholesterol and its esters are lipid-soluble substances, their metabolites are much more hydrophilic because of the addition of hydroxyl, carbonyl, and/or carboxyl groups. The hydroxyl and carboxyl groups may be in turn modified by ‘conjugation’ with amino acids such as glycine or taurine, 1 sulfuric acid, 2 glucuronic acid, 3 N-acetylglucosamine (GlcNAc), 4 glucose, 5 and galactose. 6 These conjugations have profound effects on the fate of such molecules. Conjugates may be less tightly bound to albumin, and therefore enter the glomerular filtrate, increasing renal excretion. In addition, if conjugates are accumulated in the hepatocyte because of inadequate canalicular transport into bile, they may regurgitate into plasma and be eliminated in urine. Thus, the presence of conjugates of cholesterol metabolites in urine could indicate inborn defects of cholesterol metabolism or transport and serve as biomarkers for these genetic diseases. Inborn defects of cholesterol metabolism include 3β-hydroxysteroid-Δ5-C27-steroid dehydrogenase (3β-HSD) deficiency,7,8 Δ4-3-oxosteroid 5β-reductase deficiency,9,10 oxysterol 7α-hydroxylase deficiency, 11 and sterol 27-hydroxylase deficiency. 12 In patients of 3β-HSD deficiency, severe cholestasis, pruritus, jaundice, and cirrhosis appear as clinical symptoms. Giant cell change is shown as a pathological finding. 7 On the other hand, Niemann–Pick disease, type C (NPC), 13 is known as inborn defect of cholesterol transport, and patients are common in Europe, the United States, and Japan. In patients of NPC, intrahepatic cholestasis, splenohepatomegaly, and neurological and psychiatric symptoms appear, and accumulation of cholesterol, sloughing off of Purkinje cell, and swelling of axon appear as pathological features. 14
Liquid chromatography/electrospray ionization tandem mass spectrometry (LC/ESI-MS/MS) appears to be the best technique for analysis of urinary conjugated cholesterol metabolites.15–19 If we have preliminary information on the nature of disease-specific molecules, we should be able to analyse target molecules with high sensitivity by using selected reaction monitoring mode. However, there are many unknown biomarker candidates including molecules for which synthetic reference standards are unavailable. Taguchi et al. reported that precursor ion scan and neutral loss scan were effective for focused analysis of lipids possessing common partial structures, 20 and the technique is reviewed by Griffiths and Sjövall. 21
We have previously investigated the characteristics of fragmentation patterns obtained by low-energy collision-induced dissociation (CID) of various conjugated cholesterol metabolites. 22 According to conjugation types, characteristic product ions and/or neutral losses were observed in the product ion spectra. Thus, precursor ion scan and neutral loss scan have the potential to be effective for focused analysis of conjugated cholesterol metabolites.
In this study, we investigated the usefulness of precursor ion scan and neutral loss scan using characteristic fragment pattern of various conjugated cholesterol metabolites for focused analysis. We then applied the technique to the analysis of urine samples from a patient with NPC, a disease of cholesterol transport, and another with 3β-HSD deficiency, a genetic defect in bile acid biosynthesis, to validate the usefulness of the method.
Material and methods
Chemicals and materials
Cholic acid (CA), chenodeoxycholic acid (CDCA), deoxycholic acid (DCA), ursodeoxycholic acid (UDCA), and lithocholic acid (LCA) were purchased from Nakalai Tesque, Inc. (Kyoto, Japan). Glycine- and taurine-conjugated bile acids, 23 bile acid 3-sulphates, 24 bile acid 3-glucuronides, 25 bile acid 24-glucuronides, 26 bile acid 7β-GlcNAc conjugates, 27 bile acid 3-glucosides, 28 bile acid 24-galactosides, 29 3β-sulfooxy-7β-N-acetylglucosaminyl-5-cholen-24-oic acid (SNAG-Δ5-CA), and its glycine (SNAG-Δ5-CG) and taurine conjugates (SNAG-Δ5-CT) were synthesized in our laboratories using previously reported methods. 30 17β-Estradiol 3-sulphate was purchased from Sigma-Aldrich Corp. (St. Louis, MO, USA). 17β-Estradiol 3-glucuronide, 17β-estradiol 17-glucuronide, estrone 3-glucuronide, 17α-estradiol 3-sulphate, 17β-estradiol 17-sulphate, estriol 3-sulphate, estriol 17-sulphate, dehydroepiandrosterone sulphate, epiandrosterone sulphate, pregnenolone sulphate, pregnanolone sulphate, corticosterone 21-sulphate, cortisone 21-sulphate, dehydroepiandrosterone glucuronide, pregnenolone glucuronide, cortisol glucuronide, cholesterol sulphate, cholestanol sulphate, epicoprostanol sulphate, and cholesterol glucuronide were purchased from Steraloids, Inc. (Newport, RI, USA). 17β-Estradiol 3-sulphate 17β-glucuronide was purchased from Toronto Research Chemicals, Inc. (Toronto, Ontario, Canada).
All standard specimens were dissolved in water/ethanol (1:1, v/v) at a concentration of 100 mg/L as stock solutions. A mixture of all reference compounds was prepared, with a concentration of 2.0 µmol/L for each compound. Ultrapure water was prepared using a PURELAB ultra apparatus (Organo Co., Ltd, Tokyo, Japan). All other chemicals were analytical grade and solvents were HPLC or LC/MS grade.
Urine from an NPC patient was collected at Tottori University Hospital. The patient is a male 3-month-old infant. The patient was receiving oral UDCA when the urine sample was collected. Urine from a patient with 3β-HSD deficiency (a male 6-month-old infant) and a normal infant (a female 3-month-old infant) were donated by the Junshin Clinic Bile Acid Institute (Tokyo). A healthy adult is a 21-year-old man. All experiments were carried out according to the protocol approved by the ethical committee of the Graduate School of Medicine in Tohoku University.
Focused analysis conditions for conjugated cholesterol metabolites
Mass spectrometric analysis was performed using an API 5000 mass spectrometer (AB Sciex Pte. Ltd, Framingham, MA, USA) equipped with an ESI probe. It was coupled with a Nanospace SI-2 LC system (Shiseido Co., Ltd, Tokyo). Ion spray voltage, declustering potential, and turbospray gas probe were set at –4500, –80 V, and 700℃, respectively. Curtain gas, ion source gas1, ion source gas2, and collision gas were nitrogen and their flow was set to 25, 40, 60, and 6 units, respectively. A 100 µL of water and 100 µL of standard solution (2.0 µmol/L each) were mixed, and 50 µL of aliquots were injected into the LC/ESI-MS/MS system. Analytes were concentrated at the inlet edge of a trapping column (Shim-pack MAYI-C8, 4.6 mm i.d. × 10 mm, Shimadzu, Corp., Kyoto) using a mixture of 20 mmol/L ammonium acetate solution (pH 7.0) and methanol (9:1, v/v) as a mobile phase for trapping at a flow rate of 1 mL/min. The six-port valve position was switched 3 min after and the analytes were separated on a YMC-Pack Pro C18 (5 µm, 2.0 mm i.d.× 250 mm, YMC, Co., Ltd, Kyoto) as an analytical column. Compounds were eluted by a linear gradient elution mode at a flow rate of 0.2 mL/min with the following condition: Solvent A, 20 mmol/L ammonium acetate solution (pH 7.0) and methanol (19:1, v/v); solvent B, (1:19, v/v); 50% solvent B was linearly changed to 80% solvent B against A over 120 min and then the rate of solvent B was kept at 80% for 20 min. The rate of solvent B was then switched to 50% and kept for 40 min. Collision voltages were set at –50, –60, –50, –80, and –55 V on precursor ion scans of m/z 74, 97, 113, 124, and m/z 202, respectively. Neutral loss scan of 80, 162, and 221 Da were set using a collision voltage of –50, –50, and –50 V, respectively. In analysis of GlcNAc conjugates, neutral loss spectra were measured at a scan range from m/z 230 to 900, and in other cases, precursor ion spectra and neutral loss spectra were measured at a scan range from m/z 200 to 900 with a scan time of 2 s. Accuracy of retention time and m/z were calculated as coefficient of variation (CV%). Inter-day accuracy of retention time was within 1.6% and intra-day accuracy was within 0.2%. Both inter-day and intra-day accuracy of m/z were within 0.03%. Data were collected and processed using Analyst 1.4.1 data collection and integration software.
Results and discussion
Focused approach for analysis of conjugated cholesterol metabolites
First, we analysed a mixture of authentic conjugated cholesterol metabolites using the Q1-scan mode. Since we know retention times and m/z of all standard compounds, we can identify every peak by ion extraction (data not shown). Most authentic standards were found at the retention times from 8.55 to 126 min. However, C27 compounds (cholesterol 3-sulphate, cholestanol 3-sulphate, epicoprostanol 3-sulphate, and cholesterol 3-glucuronide) were not eluted even at 140 min under the present analytical conditions. For more rapid elution of those compounds, more lipophilic mobile phases containing dichloromethane or chloroform are likely to be required.
Figure 1(a) is a total ion current (TIC) chromatogram using a precursor ion scan of m/z 74 for extraction of conjugated cholesterol metabolites possessing a glycine moiety. We were able to find 17 metabolites in the chromatogram and detect all metabolites possessing a glycine moiety, which we used as reference compounds in this study. The detected precursor ions included doubly charged deprotonated molecules because they have two acidic groups (carboxylic acid and sulfuric acid) in their molecules. In the TIC chromatogram using precursor ion scan of m/z 74, some pairs, such as peaks 8 and 9, peaks 10 and 11, and peaks 15 and 16, were not separated from each other; however, individual compounds can be distinguished by their m/z values. The TIC chromatogram in Figure 1(b) is obtained using precursor ion scan of m/z 124. The peaks in this chromatogram were derived from all taurine-conjugated metabolites used as standards in this study. Although their chromatographic separations were incomplete, they were able to be distinguished by their m/z values as was the case with glycine-conjugated metabolites.
Total ion current chromatograms of a mixture of authentic conjugated cholesterol metabolites: (a) precursor ion scan of m/z 74, (b) precursor ion scan of m/z 124, (c) precursor ion scan of m/z 97, (d) neutral loss scan of 80 Da, (e) precursor ion scan of m/z 113, (f) neutral loss scan of 162 Da, (g) neutral loss scan of 221 Da, and (h) precursor ion scan of m/z 202. All tandem mass spectrometric and chromatographic conditions were as shown in ‘Material and methods’ section. The detected standard substances were numbered in each chromatograms. The peak list was shown in Table 1. e.g. The peak 2 in chromatogram (a) corresponds to the peak No. A2, GUDCA 3-sulphate, and the peak 15 in chromatogram (b) corresponds to the peak No. B15, TLCA.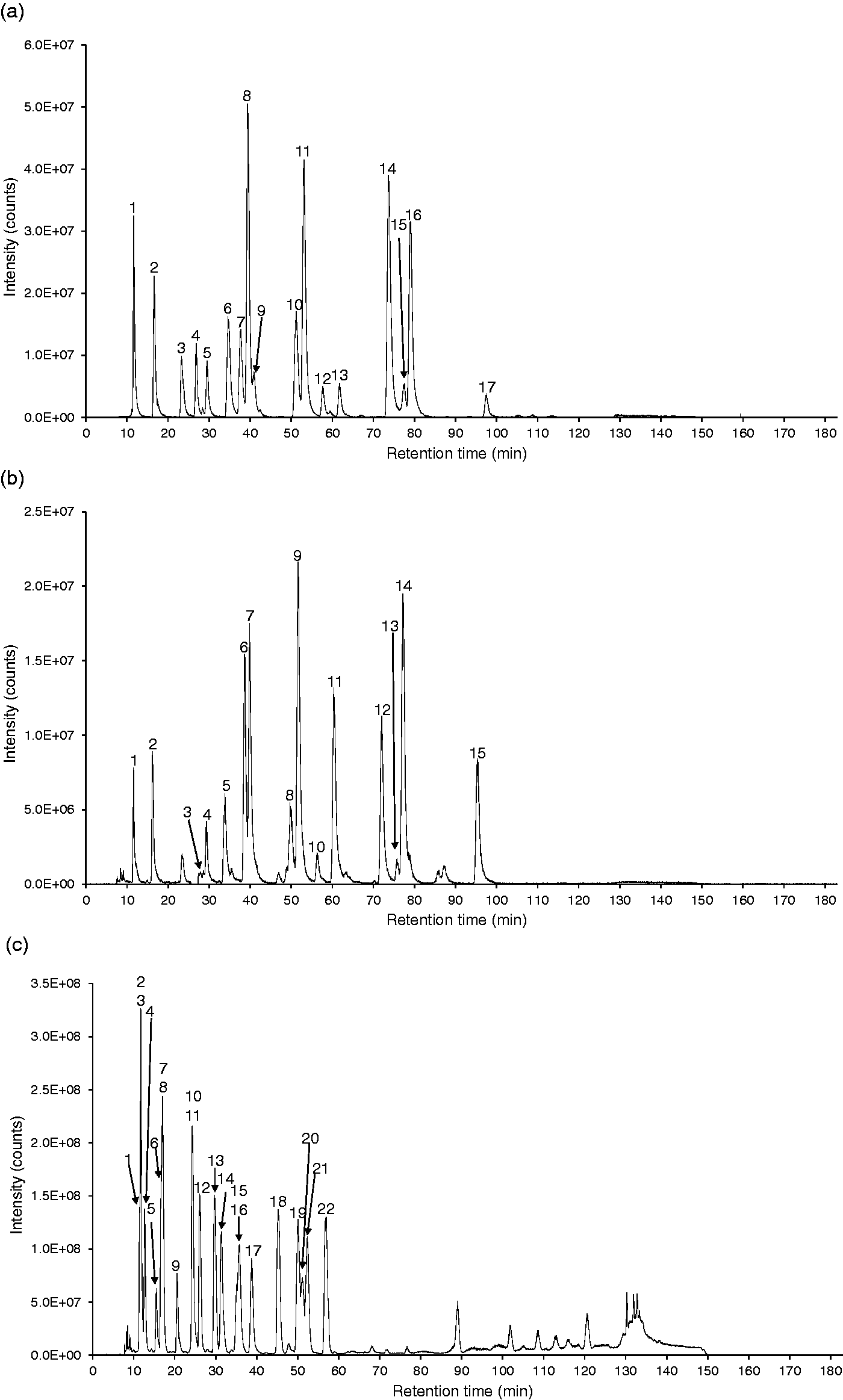
Next, we performed precursor ion scan of m/z 97 for extraction of sulphate conjugates; the TIC chromatogram is shown in Figure 1(c). Peaks in this chromatogram included sulphate conjugates with the sulphate moiety being esterified with the aliphatic, secondary hydroxyl groups such as a 3α-hydroxyl (or 7α-hydroxyl group) of bile acids and a 17β-hydroxyl group on estrogens. Sulphate groups on phenolic hydroxyl groups do not generate an m/z 97 fragment. Therefore, although estradiol 17-sulphate was clearly detected in the TIC chromatogram on precursor ion scan of m/z 97, estradiol 3-sulphate did not give a signal in the same chromatogram. Sulphate conjugates on the phenolic hydroxyl group were found in the TIC chromatogram on neutral loss scan of 80 Da as shown in Figure 1(d). Neutral loss of 80 Da was also able to extract amino acid-conjugated bile acid 3-sulphates in addition to estrogen C-3 sulphates. No non-amidated bile acid 3-sulphates were identified as precursors for the neutral loss of 80 Da, suggesting that somehow interaction between the side chain amino acid and the hydroxyl group at C-3 occurs. Thus, precursor ion scan of m/z 97 is effective for group-specific extraction of sulphate conjugates in which the sulphate group is linked by an ester bond to aliphatic alcohol substituents. On the other hand, neutral loss of 80 Da is effective for extraction of sulphate conjugates in which the sulphate is linked to a phenolic hydroxyl group although double conjugates with glycine or taurine can be found in the TIC chromatogram on neutral loss of 80 Da even in the case of sulphate conjugates linked to aliphatic alcohols. This fact is useful for identifying N-acyl amidates containing glycine or taurine.
Figure 1(e) shows a TIC chromatogram using the precursor ion scan of m/z 113. This product ion has been found in product ion spectra of monosaccharide-conjugated compounds other than GlcNAc conjugates, although acyl galactosides exhibit relatively lower intense peaks on the TIC chromatogram. 22 According to our previous paper, a precursor ion scan of m/z 175 and neutral loss scan of 176 Da aids in the identification of the mode of conjugation of glucuronides, in ester linkage with the carboxyl group, in ether linkage with an aliphatic hydroxyl group, or a phenolic hydroxyl group. Thus, this approach will work well for discrimination of conjugation types of glucuronides possessing the same molecular masses. 22
List of m/z of precursor ion and retention times of all standard substances detected in Figure 1.

CA: cholic acid; CDCA: chenodeoxycholic acid; DCA: deoxycholic acid; GCA: glycine-conjugated cholic acid; GCDCA: glycine-conjugated chenodeoxycholic acid; GDCA: glycine-conjugated deoxycholic acid; GLCA: glycine-conjugated lithocholic acid; GlcNAc: N-acetylglucosamine; GUDCA: glycine-conjugated ursodeoxycholic acid; LCA: lithocholic acid; SNAG-Δ5-CA: 3β-sulfoxy-7β-N-acetylglucosaminyl-5-cholen-24-oic acid; SNAG-Δ5-CG: glycine-conjugated 3β-sulfoxy-7β-N-acetylglucosaminyl-5-cholen-24-oic acid; SNAG-Δ5-CT: taurine-conjugated 3β-sulfooxy-7β-N-acetylglucosaminyl-5-cholen-24-oic acid; TCA: taurine-conjugated cholic acid; TCDCA: taurine-conjugated chenodeoxycholic acid; TDCA: taurine-conjugated deoxycholic acid; TLCA: taurine-conjugated lithocholic acid; TUDCA: taurine-conjugated ursodeoxycholic acid; UDCA: ursodeoxycholic acid.
Focused analysis of urine from NPC and 3β-HSD deficiency patients
Peak list of conjugated cholesterol metabolites detected in abundance in urine of a patient with NPC and a patient with 3β-HSD deficiency.
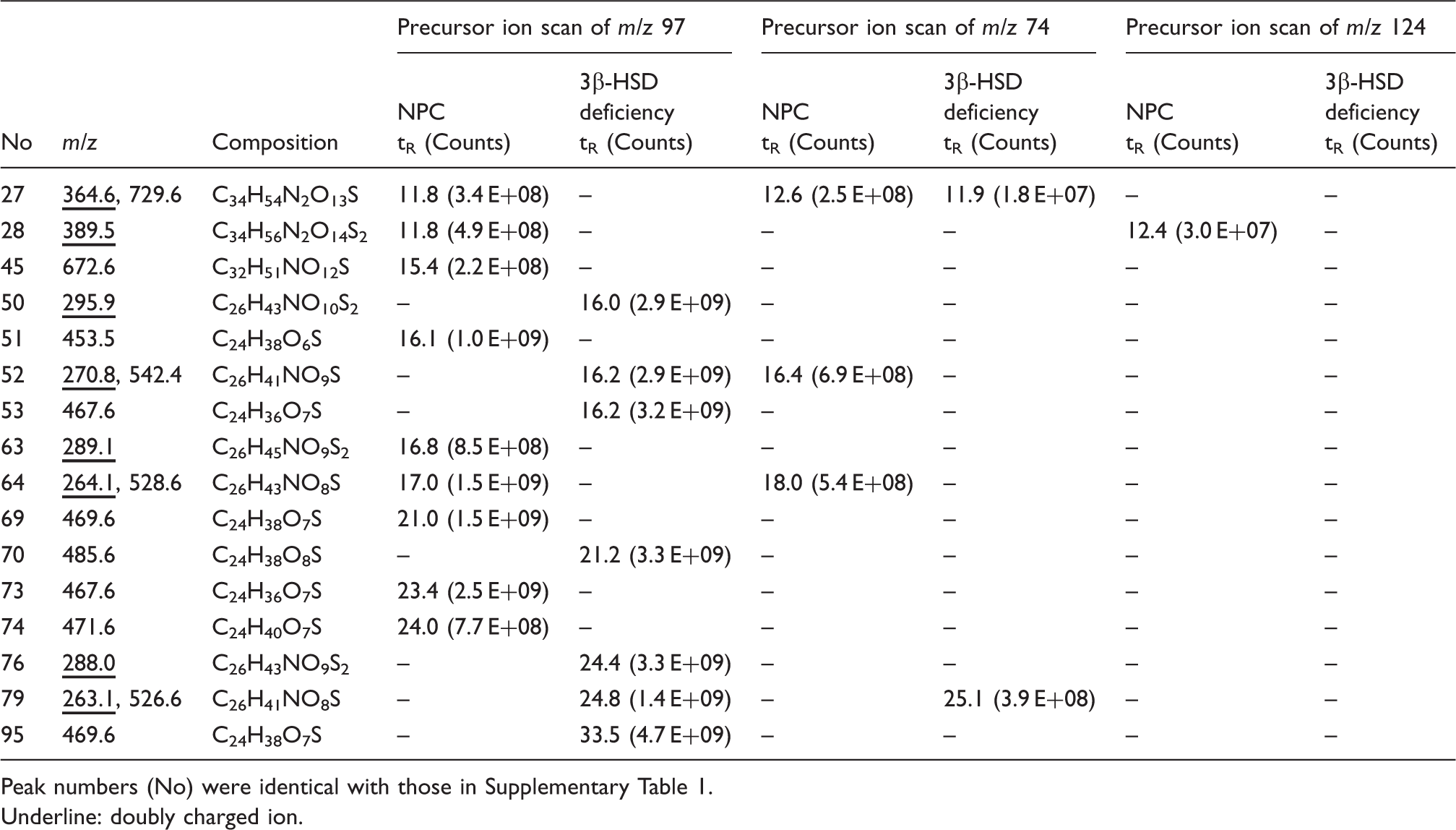
Peak numbers (No) were identical with those in Supplementary Table 1.
Underline: doubly charged ion.
Next, we performed precursor ion scans of m/z 74 and 124 for extracting glycine and taurine conjugates, respectively; the results also were put into the same tables. Taurine and sulphate double conjugates provided doubly charged deprotonated molecule [M-2H]2– without a singly charged one. Glycine-conjugated and sulphate double conjugates provided both signals with parallel intensity. This information also helped to distinguish sulphated glycine and taurine conjugates.
The urine sample obtained from an NPC patient provided many characteristic intense peaks although unconjugated UDCA 3-sulphate, glycine-, and taurine-conjugated UDCA 3-sulphates were found at the retention times of 24.0 min (No. 74), 17.0 min (No. 64), and 16.8 min (No. 63), respectively, presumably because the patient was ingesting UDCA as possible therapy. The peak (No. 51) at the retention time of 16.1 min, a precursor ion at m/z 453.5, has C24H38O6S as its elemental composition corresponding to a cholenoic acid sulphate conjugate possessing a hydroxyl group and one double bond, e.g. monohydroxy-5-cholenoic acid sulphate or possibly a sulphate of a hydroxyl, oxo-bile acid. The peak (No. 69) showing m/z 469.6 as a precursor ion at the retention time of 21.0 min corresponds to a dihydroxycholenoic acid sulphate or a hydroxy oxo bile acid sulphate, but the position and conformation of functional groups is not known. In addition, the peak (No. 73) with a retention time of 23.4 min, m/z 467.6, corresponds to a monohydroxy-monooxo-5-cholenoic acid sulphate. Alvelius et al. reported abnormal accumulation of SNAG-Δ5-CT, SNAG-Δ5-CG, and SNAG-Δ5-CA, in a patient with NPC. 31 We have synthesized these compounds as noted earlier. The retention time of SNAG-Δ5-CT was 11.8 min (No. 28, m/z 389.5), that of SNAG-Δ5-CG was 11.8 min (No. 27, m/z 364.5 and 729.6), and that of SNAG-Δ5-CA was 15.4 min (No. 45, m/z 672.6). Although their intensities were weaker than unknown three cholenoic acid sulphate conjugates, they were specific to our NPC patient.
In urine from 3β-HSD deficiency patient, the peaks at the retention times of 16.0 min (No. 50), 16.2 min (No. 52, 53), 21.2 min (No. 70), 24.4 min (No. 76), 24.8 min (No. 79), and 33.5 min (No. 95) were specific to our 3β-HSD deficiency patient. HSD catalyses the shift of the double bond at C-5 to C-4 and at the same time oxidizes the hydroxyl group at C-3. Therefore, a deficiency of this enzyme should result in the accumulation of 3β-hydroxy, Δ5 bile acids that in principle could be either C24 or C27, depending on whether peroxisomal oxidation of the C8 side chain occurs. Indeed, the occurrence of 3β,7α-dihydroxy-5-cholenoic acid and 3β,7α,12α-trihydroxy-5-cholenoic acid in urine has been reported, indicating that side chain oxidation proceeds.7,32 Thus, it seems likely that the peaks having the retention times of 16.0 min (No. 50, m/z 295.9), 16.2 min (No. 52, m/z 270.8 and 542.4), and 21.2 min (No. 70, m/z 485.6) are taurine conjugated, glycine conjugated, and unconjugated 3β,7α,12α-trihydroxy-5-cholenoic acid 3-sulphate, respectively. The peaks with retention times of 24.4 min (No. 76, m/z 288), 24.8 min (No. 79, m/z 263.1 and 526.1), and 33.5 min (No. 95, m/z 469.6) are perhaps taurine conjugated, glycine conjugated, and unconjugated 3β,7α-dihydroxy-5-cholenoic acid 3-sulphate, respectively. We did not detect the C27 homologues of these compounds.
Conclusion
In this study, we demonstrated the utility of focused metabolomics analysis for urinary conjugated cholesterol metabolites using precursor ion and neutral loss scans. Our work shows the usefulness of precursor ion scan and neutral loss scan analysis using characteristic fragmentations for conjugation moieties in extracting the desired peaks from very complicated matrices.
Using precursor ion scans analysis of urine from two patients with different diseases, NPC and 3β-HSD deficiency, we found some characteristic peaks specific for an NPC patient (No. 51, 69, and 73) and 3β-HSD deficiency (No. 50, 52, 70, 76, 79, and 95). The present method gives promise of identifying new biomarkers for early diagnosis of diseases characterized by urinary excretion of conjugated cholesterol and/or bile acid derivatives.
Footnotes
Declaration of conflicting interests
None declared.
Funding
This study was supported in part by KAKENHI (Grant-in Aid for Challenging Exploratory Research: 22659007).
Ethical approval
The ethical committee of the Graduate School of Medicine in Tohoku University approved this study (REC number: 2013-1-535).
Guarantor
NM.
Contributionship
MM collected data and wrote the manuscript. MS, JG, and HY contributed to the discussion. KO, MT, and HN collected urine sample. TI synthesized authentic standards. AFH contributed to the discussion and reviewed/edited the manuscript. NM designed the study, contributed to the discussion, and reviewed/edited the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
