Abstract
We report a case of an ethanol and massive gamma-butyrolactone (GBL) intoxication, the precursor of the recreational drug gamma-hydroxybutyric acid (GHB), resulting in life-threatening metabolic acidosis (pH 6.5) with a highly increased anion- and osmolal gap. Rapid analysis using gas chromatography revealed a GHB plasma concentration of 4400 mg/L, far above the upper limit concentration of 1000 mg/L found in adult fatalities attributed to GBL. Full recovery was established following supportive treatment including haemodialysis. This is the first report of a combined ethanol/GBL intoxication as a cause of high serum anion- and osmolal-gap metabolic acidosis.
Keywords
Case description
Emergency medical personnel were called by a patient’s friend around 23:00. The medical team found the patient unconscious and cyanotic; unresponsive to verbal command and painful stimuli, with absent cornea reflexes and dilated pupils not reacting to light. There were no external signs of violence, and medication blister packs were not found. Blood pressure was 110/50 mmHg, electrocardiography monitoring showed slow atrial fibrillation (28–40 bpm) and O2 saturation was low (80%). He had vomited and possibly aspirated gastric content into his lungs. Oxygen was administered, and the patient was intubated without any need for sedation or relaxation.
Upon arriving at the emergency department, laboratory analysis revealed a massive mixed acidosis (pH 6.53; pO2 196.4 mmHg, pCO2 74.4 mmHg; bicarbonate 6 mmol/L; BE – 35.2 mmol/L); a high lactate (4.03 mmol/L; N < 1 mmol/L) and a blood ethanol concentration of 279 mg/dL (2.79‰). Urine screens for the presence of tricyclic antidepressants, barbiturates, benzodiazepines, amphetamine, cocaine, methadone, opiates, methamphetamine and tetrahydrocannabinol were negative. There was no ketonuria or glycosuria. The patient’s lips and tongue were swollen, without necrosis of the mucosae, and he was profoundly hypothermic (31.5℃, bladder probe), even though he had been readily found inside his home and transported in a heated ambulance. He became progressively more hypotensive. Echocardiography showed normal left and right ventricular function and normal filling pressures, pleading against heart failure as an underlying mechanism of the shock and acidosis. Correction of acidosis was attempted by reversing the hypercapnia, administration of bicarbonate and by increasing body temperature using a forced-air warming device. Except for a modest reduction in lactate, this was without effect.
Chronology of clinical and biochemical data.
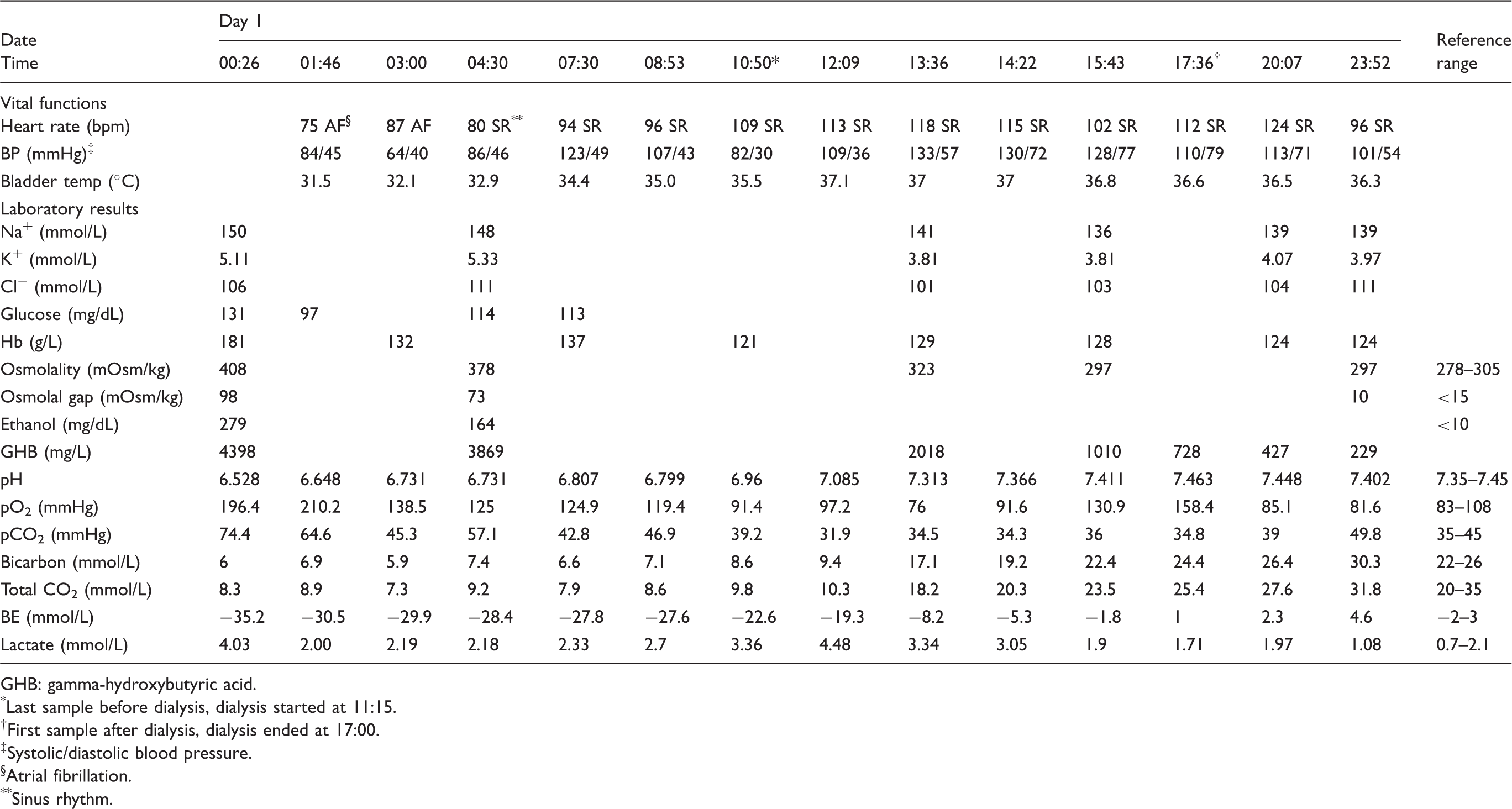
GHB: gamma-hydroxybutyric acid.
*Last sample before dialysis, dialysis started at 11:15.
†First sample after dialysis, dialysis ended at 17:00.
‡Systolic/diastolic blood pressure.
§Atrial fibrillation.
**Sinus rhythm.
Four hours after starting haemodialysis, the patient purposefully reacted to painful stimuli. The decision to stop dialysis was based on the normalization of the serum osmolality to 297 mOsm/kg (normal 278–300 mOsm/kg).
Three and a half days after admission, the patient could be weaned from the ventilator. The patient remained confused for the following two days and was discharged on day 7. To date, current cognitive functioning is normal.
Discussion
For quite a number of years, GHB and one of its precursors GBL have been known as substances of abuse, causing euphoria and enhancing sexual pleasure. GBL is usually taken in doses of 1–2 g. Higher doses result in ‘passing out’, a desired effect in some users, and current evidence shows that GHB is highly addictive. 2 Clinical features related to GHB/GBL intoxication include deep coma with sudden awakening, bradycardia, severe hypoventilation and hypothermia. Fatalities – usually caused by hypoxia – have been reported for 20 years, with the highest GHB level ever observed in (femoral) blood being 2937 mg/L in a fatal GHB overdose. 3
Here, we report a case of massive GBL intoxication, far above lethal doses, resulting in a life-threatening metabolic acidosis (pH 6.5) with a high anion- and osmolal gap. As far as we know, this is the first report of an ethanol/GBL intoxication as a cause of a high serum anion- and osmolal-gap metabolic acidosis.
Except for the deep hypothermia and the long-lasting acidosis, the clinical features of this case are not remarkable. After an overdose, reduced concentrations of consciousness are common, and unresponsive coma is reported in 18% of patients with GHB mono-intoxication. 4 The degree of hypothermia noted (31.5℃), however, was unexpected as the patient had not been exposed to a cold environment. Hypothermia <36℃ is reported fairly frequently in GHB/GBL intoxication ranging from 28% to 69%,5,6 but it is unclear how often significant hypothermia (<34℃) occurs. The profound hypothermia in this patient appears to be accentuated by the drug itself and may have contributed to the deep level of coma.
A striking feature reported here is the high anion gap (34 mmol/L), the high osmolal gap (98 mOsm/kg) and the metabolic acidosis with pH values <6.8 for 4 h despite attempts at correction. Detailed acid–base analyses with the traditional acid–base methodology of the first arterial blood gas in combination with the biochemistry panel 7 reveals a triple acid–base disorder. A respiratory acidosis, a high anion metabolic acidosis and a metabolic alkalosis (rise in anion gap of 24 is only compensated for by a decrease in bicarbonate of 19 mmol/L) were present at admission. The Stewart approach reveals a metabolic acidosis due to 37 mEq/L of unmeasured anions. The hypochloraemic metabolic alkalosis is confirmed by this method and shown to be caused by a rise in apparent strong ion difference due to a low chloride level, especially when corrected for the patients’ hypernatraemia. This was probably caused by vomiting, contraction alkalosis due to dehydration or both.
Although the patient was in a hypotensive shock, the rise in lactate was clearly insufficient to account for the degree of metabolic acidosis. Other causes of severe acidosis such as diabetic ketoacidosis (absence of ketones and glucose in urine, normoglycaemia), salicylate intoxication (spectrophotometric blood analysis) and bowel ischaemia (absence of clinical signs) were ruled out. From the onset, it was clear that the patient was ethanol intoxicated. Although it is possible that this contributed to the acidosis, the effect is generally considered to be minor. 8 In this patient, the concentration of hydroxybutyrate in the serum was below the limit of detection (3 mg/L). Likewise, the high osmolal gap is only partially explained by the ethanol intoxication. The 279 mg/dL ethanol accounts for 60.54 mOsm/kg which leaves one-third of the osmolal gap (37.46 mOsm/kg) unaccounted for. According to the amount of fluid remaining in the obtained container, the patient maximally can have ingested 70 mL of GBL corresponding to 78.7 g of GBL (measured purity: 98.3%, GBL specific gravity: 1.144 g/mL). GBL is converted completely to GHB within minutes in the body, and the intake here resulted in a measured GHB blood concentration of 4398 mg/L as determined by gas chromatography-mass spectrometry (GC-MS). 1 The unaccounted osmolal gap of 37.46 mOsm/kg theoretically corresponds to a GBL concentration of 3220 mg/L. When taking into account the analytical variability of the measurement of all analytes involved in the calculations (osmolality [CVa 0.7%], sodium [CVa 1.1%], urea [CVa 1.6%], glucose [CVa 1.8%], ethanol [CVa 4.1%] and GHB [CVa 8%]), this value is consistent with the GHB measured. GHB has a pKa of 4.7 resulting in a 99% ionization at physiological pH and a 63% ionization at a pH of 6.3 at admission. This high ionization grade is hypothesized to have contributed largely to the metabolic acidosis. GBL, measured later in the left over samples, could not be evidenced. Freezing and thawing of the samples probably hydrolyse the GBL as indicated by several authors.
As GHB/GBL is not a substance classically associated with a high anion-gap metabolic acidosis (HAGMA), the finding of the massive GHB concentration was a direct result of the method used in our lab, allowing the simultaneous and fast detection, without preanalytical cleanup, of several substances potentially causing HAGMA. Classical ethylene glycol methods would not measure GHB, and suitable GC-MS techniques for rapid detection of this illicit drug are not widely available. 9 Furthermore, with a molecular weight of 104.11, the theoretical contribution of a GHB blood level of 1040 mg/L, which is in the same range as the highest blood concentration described in cases of GHB fatalities, would only increase the serum osmolality by 10 mOsm/kg. So even in potentially lethal doses of GHB, the contribution to an increase in osmolality will be small. However, occasionally, as in this case, the double-gap acidosis may be an important clue in the diagnosis of this life-threatening intoxication. In view of the acidosis and the physical characteristics of the molecules involved, haemodialysis can be life saving.
Speculatively, we can hypothesize that the simultaneous ingestion of high amounts of alcohol interfered in the metabolism of GHB with a long-lasting acidosis and coma as result. Several authors, however, state that ethanol will act synergistically with GHB to induce coma. 10
In conclusion, we report a high anion-gap and osmolal-gap acidosis following the ingestion of a potentially lethal combination of ethanol/GBL and suggest that this intoxication be considered in the differential diagnosis of this acid–base disorder.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
LH.
Contributorship
LH, NVR, MH and SS were involved as medical doctors. HN and GD performed the laboratory analyses. LH, CLC, HN and WvdB wrote the first draft and prepared the manuscript for submission. All authors reviewed and approved the manuscript.
