Abstract
Background
Immunometric assays have recently shown higher specificity in the detection of human insulin than radioimmunoassays with almost no cross-reaction with proinsulin or C peptide. The introduction of the new insulin analogues on the market, however, has raised the need to define their cross-reactivity in these assays. Several studies have been published in this regard with different results.
Methods
The analogues studied were insulins lispro, aspart, glargine, detemir, and glulisine. Insulin concentrations were measured in Immulite® 2000 and Advia Centaur® XP (Siemens Healthcare Diagnostics), and Elecsys® Modular Analytics E170 (Roche). All samples were processed 15 times in the same analytical run following a random sequence. Those samples which showed statistically and clinically significant changes in insulin concentration were reprocessed using increasing concentrations of analogue, and this was done twice, using two different serum pools, one with a low concentration of insulin and one with a high concentration of insulin.
Results
In the Elecsys® E170 analyser, glargine showed statistical changes (comparison of mean concentrations with
Conclusions
In the Elecsys® E170 assay, relevant cross-reactivity was only detected with insulin glargine, whereas in the other analysers all analogues except glulisine showed significant interference.
Keywords
Introduction
Diabetes mellitus (especially type 2 diabetes) has become one of the most serious health problems of our time with a prevalence of approximately 14% in our country. 1
Chronic hyperglycaemia, even in the absence of symptoms, causes lesions in multiple tissues, making diabetes a leading cause of blindness, amputations and end-stage renal disease in developed societies. Additionally, diabetes poses a major risk for cardiovascular disease, both by itself and through its association with other risk factors such as hypertension and dyslipidaemia. 2
Insulin is used for intensive treatment in type 1 diabetes and in insulin-requiring type 2 diabetes when oral agents do not achieve adequate glycaemic control. Strategies combining basal and bolus insulin treatments are often applied to achieve more physiological insulin profiles and to maintain long-term glycaemic control, 3 using insulin analogues synthesized by recombinant DNA technology. 4
Currently available insulin analogues include fast-acting preparations: insulin aspart, insulin lispro, and insulin glulisine. Long-acting analogues include insulin glargine and insulin detemir.5–7 The fast-acting analogues are more rapid in onset and with a shorter duration than human insulin. The long-acting ones present a fairly constant action without peaks, similar to the physiological interprandial secretion of insulin in non-diabetic subjects. 4
Serum insulin quantification assays are required to aid in the evaluation of adequate insulin therapy compliance, or suspected overdose of insulin. 4 Furthermore, these tests are used in the differential diagnosis of hypoglycaemia in non-diabetic patients and may be useful in determining the pathogenesis of the different types of diabetes, as well as in evaluating the treatment of patients with insulin resistance.5,8
Initially, radioimmunoassay techniques were used for insulin measurement although they presented some limitations, such as cross-reactions with endogenous insulin precursors (proinsulin and C peptide) and interference with insulin antibodies. Also, mass spectrometry-based methods for insulin analogue assessment have been described, although their use is not widespread. 9
Immunometric assays have recently shown higher specificity than radioimmunoassays in the detection of human insulin. However, the introduction of the new insulin analogues on the market has raised the need to define their cross-reactivity in these assays. 5 Although specific immunoassays have been developed for pharmacokinetic studies of insulin analogues, these are used primarily for preclinical studies and research and are not commonly used in clinical laboratories.3,10
Studies have been conducted to evaluate the interference produced by some of these analogues (especially lispro, aspart, and glargine) in a selection of commercially available automated insulin immunoassays with different results. Some studies have shown a high cross-reactivity with all analogues, but others have shown almost no interference.3,5,10–15
The variability of results observed in the different studies demonstrates the importance of evaluating the available commercial assays, to achieve a greater understanding of what is being measured and the possible interferences and, thus, provide diagnostic results with greater sensitivity and specificity.
The purpose of this study was to determine the extent of interference caused by the new analogues (lispro, aspart, detemir, glargine, and glulisine) in three different automated analysers.
Methods
An analytical study was performed to examine the possible interference of insulin analogues in insulin quantification assays. The analogues used were five different commercial preparations: lispro (Humalog® and Eli Lilly and Company), aspart (Novo-Log™ and Novo Nordisk Pharmaceuticals), detemir (Levemir® and Novo Nordisk Pharmaceuticals), glargine (Lantus® and Aventis Pharmaceuticals), and glulisine (Apidra® and Aventis Pharmaceuticals), each with a concentration of 100 IU/mL. These were diluted using 7% fetal bovine serum (FBS) to concentrations of 10,000 mIU/L each, 10 times higher than the maximum concentration studied.
A serum pool was prepared using samples from patients attended in our laboratory who were not treated with any of the analogues under study and excluding those samples which presented anti-insulin antibodies. The interferent (insulin analogue) solutions prepared beforehand were then diluted 10-fold using this pool, thus obtaining our working solutions. Similarly, a control sample was prepared diluting the FBS to 1/10 using the serum pool.
Fifteen aliquots of each sample were processed in the same analytical run following a random sequence, according to the indications of the interference assessment procedure of the Spanish Society of Clinical Biochemistry and Molecular Pathology (SEQC) 16 for the study of the interference of endogenous samples, which in turn is based on Clinical and Laboratory Standards Institute criteria. 17 This procedure was carried out on each of the analysers under study: Elecsys® Modular Analytics E170, Roche Diagnostics, (assay range: 0.2–728 mU/L), and Immulite® 2000 XPi Immunoassay System and Advia Centaur® XP Immunoasssay System, Siemens Healthcare Diagnostics (both with assay ranges: 0.5–300 mU/L), following the manufacturers’ recommendations in all cases. All assays are standardized against WHO NIBSC 1st IRP 66/304 reference preparation.
The procedure was designed to detect interference at high concentrations, considering that if a certain substance does not interfere at high concentrations, it will not interfere at low concentrations either. In cases where statistically significant interference was observed, clinical significance was tested for comparing the results to the reference change values (RCV). The RCV can be defined as the critical difference that must be exceeded between two sequential results for a significant (or true) change to occur and incorporates the total variation associated with both results. 18 RCV is demonstrated by using the equation provided in the Statistical analysis section.
For those analogues which presented clinical significance, the interference was quantified, to establish the possible relationship between the degree of interference and concentrations of analogue and insulin. For this, the solutions prepared in the previous phase were used: the control sample and the samples with high concentrations of analogue. These were serially diluted to obtain samples with the same insulin concentration, but different concentrations of analogue: 0, 100, 300, 500, and 700 mIU/L, respectively. In the case of Siemens Healthcare Diagnostics instruments, the last two dilutions could not be used, for they exceeded the upper testing limit; the 300 mIU/L dilution was replaced with 250 mIU/L and two dilutions of 50 and 200 mIU/L were added, hence, resulting in samples of 0, 50, 100, 200, and 250 mIU/L of analogue. All samples were processed five times each.
To determine whether the insulin concentration of a sample has an effect on the interference, the same process was carried out using varying concentrations of analogue as before, on a pool of serum samples with a constant concentration of insulin, higher than the one used previously.
Statistical analysis
Dixon’s test was used to check for outliers. The mean and standard deviation were calculated, and the normality of the distribution and homogeneity of variance were tested for. The means of the control sample and problem sample were then compared, evaluating the results for statistical significance. For normally distributed variables the Student’s
In cases in which the interference detected was statistically significant, the clinical significance was tested for. To do this, the results were compared with the RCVs used in our laboratory, calculated from the data of within-subject biological variation
19
and the analytical imprecision obtained from our laboratory, using the formula
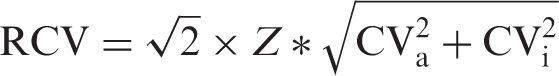
To calculate the interference dependent on the concentration of analogue, the data were plotted in graphs and in those cases with a linear distribution the correlation was studied using the Passing–Bablok regression model. In the cases in which the distributions were not linear, the data were adjusted using logarithmic, exponential, multiplicative, or reciprocal models.
For the study of interference dependent on the base concentration of insulin, a multiple linear regression model was used including the following variables: insulin concentration, concentration of the analogue, and the product of both or insulin-analogue interaction term.
Statistical analysis was performed using R v.2.13.0 (2011-04-13) Copyright© 2011. The R Foundation for Statistical Computing.
Results
Mean insulin values (mIU/L) and standard deviation of the control sample and the different analogues under study.
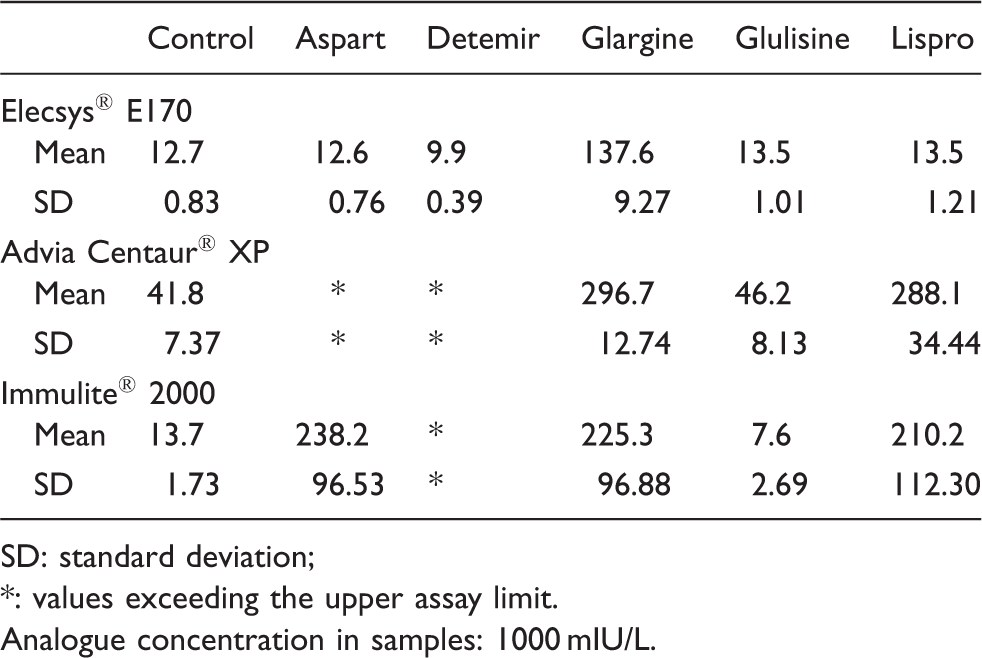
SD: standard deviation;
: values exceeding the upper assay limit.
Analogue concentration in samples: 1000 mIU/L.
All results followed a normal distribution except insulins detemir and glargine in the Elecsys® E170 analyser. All the analogues studied showed statistically significant interference except insulin aspart in the case of Elecsys® E170 and insulin glulisine in the Advia Centaur® XP analyser.
In Elecsys® E170, the difference of means obtained for glargine was 125.0 mIU/L, (95% CI: 119.8–130.1), its percentage difference with respect to the control sample was 986.2%, substantially higher than the RCV (59.8%). The other analogues presented percentage differences lower than the RCV and were therefore not clinically significant.
In Advia Centaur® glargine and lispro presented percentage differences with respect to the control sample of 610.3% and 589.7%, respectively, higher than the RCV (61.4%), thus, being clinically significant. The mean results obtained for aspart and detemir were higher than the analyser’s upper assay limit, resulting in percentage differences higher than 618.1%.
Regarding the Immulite® 2000 analyser, the percentage differences with respect to the control sample were: aspart 1636.1%, glargine 1541.7%, and lispro 1431.8%. All were markedly higher than the RCV (103.5%). Glulisine, however, showed no clinical significance (44.7%). The results obtained for detemir were higher than the analyser’s upper assay limit, resulting in percentage differences higher than 2086.1%.
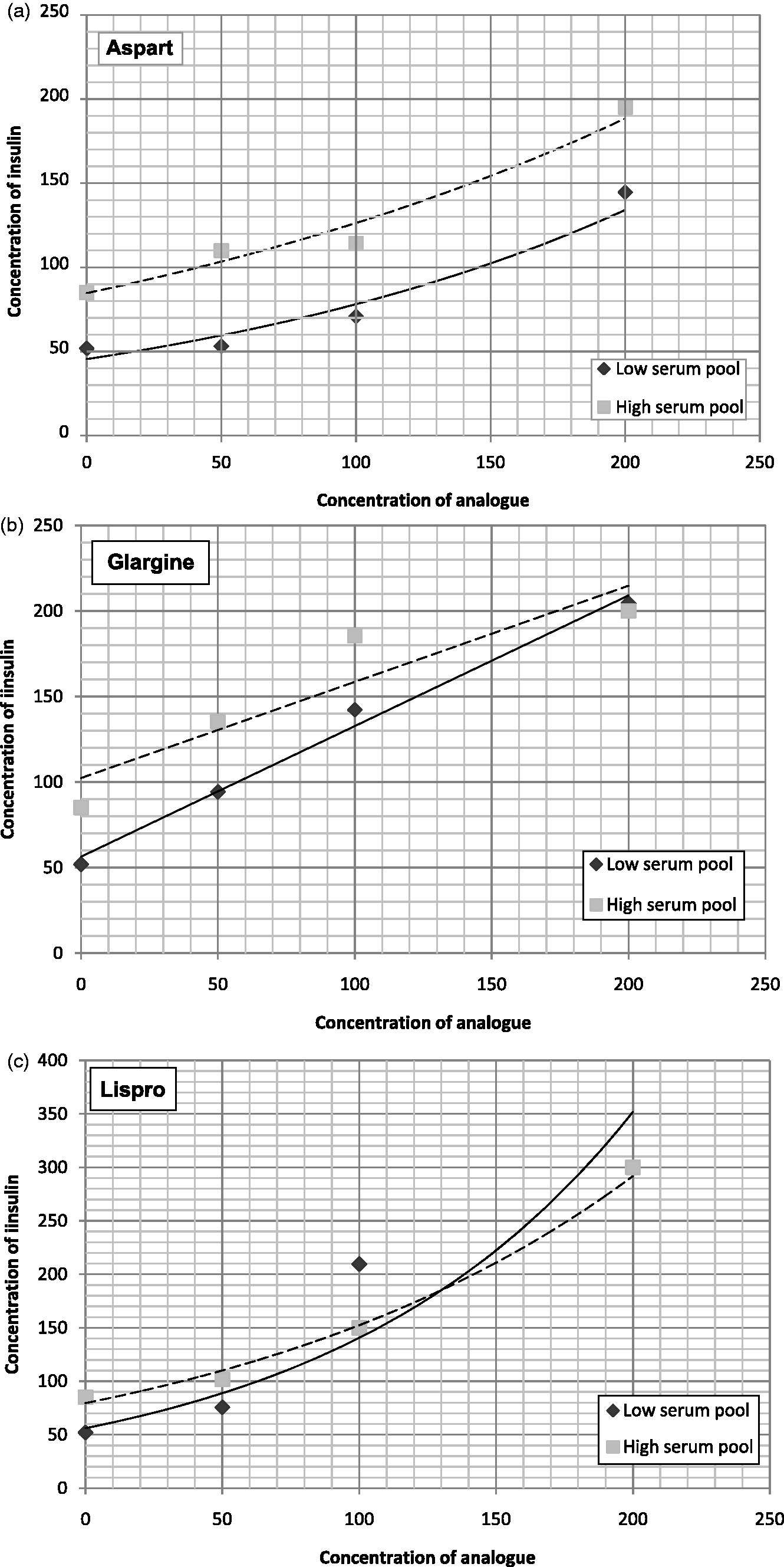
Figures 1 and 2 show the results of the study of interference depending on the concentration of analogue, of those which had presented statistically and clinically significant interference in the first phase. For the Advia Centaur® XP analyser, the analogues presented interference but this could not be quantified as two or more results exceeded the upper limit of the assay, making it impossible to plot the graphs. The same problem occurred with detemir in the Immulite® 2000 analyser.
Results of the study of interference depending on the concentration of analogue in the Elecsys E-170 analyser. Results of the study of interference depending on the concentration of analogue in the Immulite® 2000 analyser. (a) Aspart, (b) Glargine and (c) Lispro.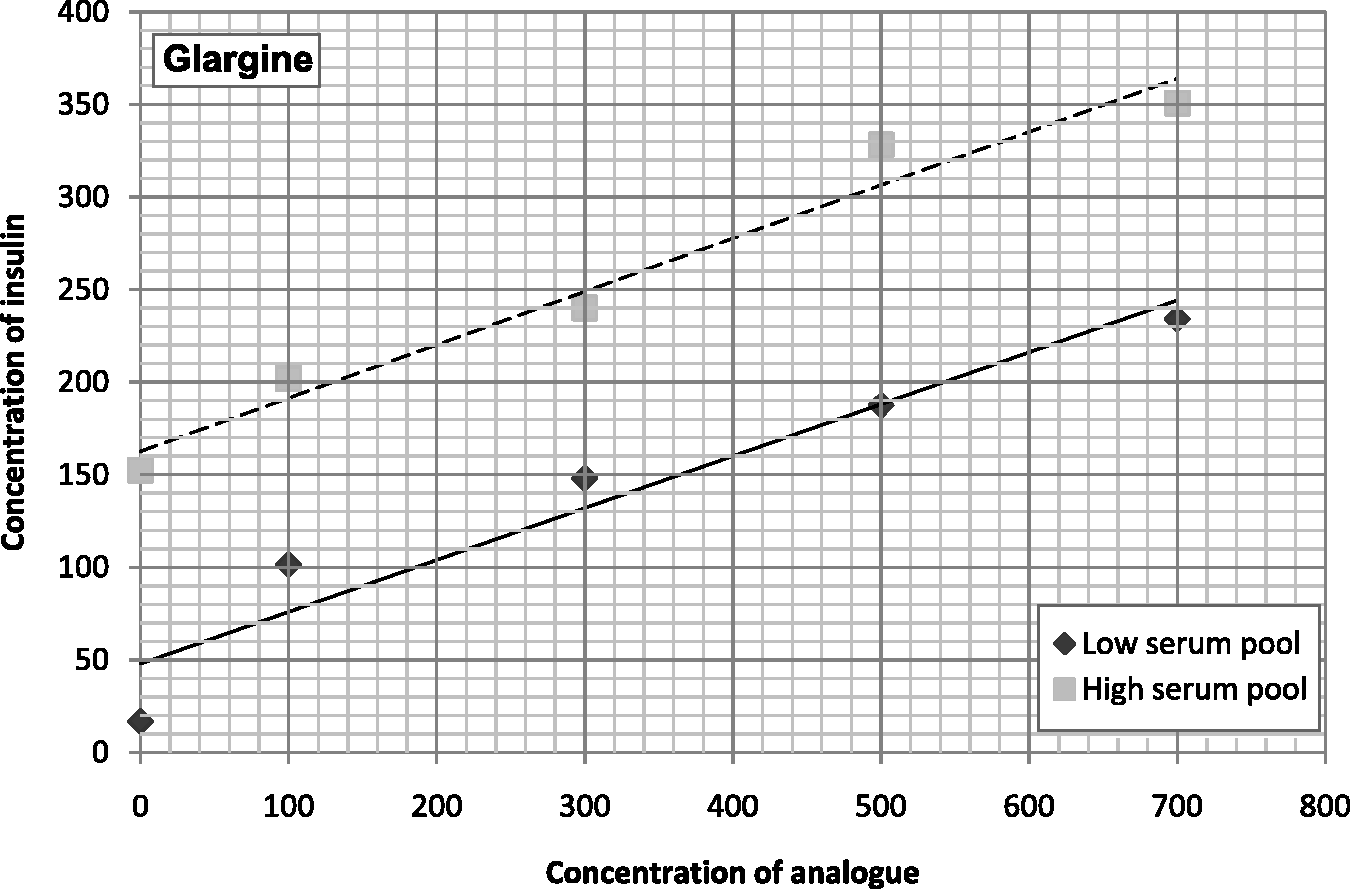
The results of the study of interference dependent on the base concentration of insulin by multivariate analysis showed that higher concentrations of insulin did not increase interference significantly, or the increase was small and not clinically significant.
Discussion
Cross-reactivity is a function of the binding of the antibodies employed in the assay used to determine the concentration of an analyte. Human insulin has antigenic determinants differing from the antigenic sites of insulin analogues. However, depending on the assay, human insulin may moderately decrease or increase cross-reactivity with insulin analogues. The simultaneous presence of the primary ligand (human insulin) and of the cross-reactant may have miscellaneous effects depending on the assay and on the analogue assessed. 20
The aim of this study was to determine the interference caused by the new analogues (lispro, aspart, detemir, glargine, and glulisine) in three different automated analysers.
The study showed significant differences regarding insulin glargine for Elecsys® E170 and with insulin aspart, detemir, lispro, and glargine in the case of Advia Centaur® XP and Immulite® 2000.
Interference was observed to increase linearly in the case of glargine in both Elecsys® E170 and Immulite® 2000, or exponentially in the cases of aspart and lispro in Immulite® 2000, with increasing concentrations of analogue. However, an increase in the concentration of serum insulin did not increase the cross-reactivity.
We observed that glulisine lacked clinically significant interference. All analogues possess modifications at the carboxy-terminal end of the B chain of insulin (aa. 28–29) but glulisine also has a modification at the amino-terminal end of that chain (aa. 3 asparagine is replaced by lysine) so this modification could be the cause of its lack of reactivity in the different assays.
We found that in Advia Centaur®, the concentrations measured were higher than in the other two analysers. The lack of standardization of insulin assays is well known and is currently being studied. The source of discrepancies in results among commercial methods of insulin immunoassays is likely multifactorial and not explainable by a single analytical performance characteristic. 21
Regarding the Elecsys® E170 analyser, our results agree with those previously published10,12,22,23 with respect to aspart, detemir, lispro, and glulisine. However, in our study, glargine presents clinically significant interference in this analyser, unlike Owen and Roberts’s. 10 This discrepancy could be explained by Agin et al.’s 3 report which shows that glargine in human serum undergoes a biotransformation that produces two metabolites M1 and M2, and the first (M1) is recognized by the Elecsys® E170 assay. Owen and Roberts perform their study in FBS and not in human serum, which probably does not possess the same activity as the human carboxypeptidase and therefore our results (performed in human serum) match Agin et al’s.3,10 Recently, Heurtault et al. 20 have conducted an extensive study with similar results.
Our results in the Advia Centaur® XP or Immulite® 2000 are also in agreement with previous studies,10,12 showing significant differences with respect to aspart, lispro, and glargine. Regarding the other two insulin analogues, detemir showed cross-reactivity in both analysers, whereas the reactivity presented by glulisine was not clinically significant.15,20
Our results are similar to others regarding analogue concentration, which appears to be an important parameter to take into account in the choice of the insulin assay to be used.10,20,24 We found no significant relationship between cross-reactivity and base concentrations of serum insulin. None of the studies revised have data to this respect.
There seems to be a large variability in insulin analogue cross-reactivities with different commercial assays. On the whole, it appears that Siemens’ Advia Centaur® XP analysers exhibit greater cross-reactivity with the analogues aspart, glargine, and lispro, whilst in their Immulite® 2000 analyser and Roche’s Elecsys® E170 analyser cross-reactivity is low.3,5,10–15 However, although we did find the Elecsys® E170 to be more precise and show lower cross-reactivity, this was not our case for the Immulite® 2000 analyser. Our results showed the greatest cross-reactivity with this analyser, and it also proved to be the least precise of the three.
Although specific immunoassays have been developed for research studies of certain insulin analogues, the combination of some of the various commercial immunoassays, taking interference into account, can be used to estimate the concentration of insulin analogues in the presence of human insulin. Knowledge of the cross-reactivity of the different insulin analogues is important not only for the correct interpretation of the results of patients treated with these analogues but also for detecting factitious hypoglycaemia in non-diabetic patients. Unfortunately, many times the manufacturers do not provide adequate information about the cross-reactivity that may occur with their assays. That is why studies like this should be carried out in clinical laboratories and should be performed following standardized protocols that will allow extrapolation of the results to other laboratories.
In summary, we found that only insulin glargine presented relevant cross-reactivity in the Elecsys® E170 assay, whilst in the other analysers all analogues except glulisine presented significant interference.
Footnotes
Acknowledgements
We are grateful to Abraham Ledesma, María Tapia and Rocío Díaz for their help in the completion of this study.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not required.
Guarantor
AD.
Contributorship
AD and PO researched literature and conceived the study. MR was involved in protocol development and data analysis. AD wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
