Abstract

Sawyer et al. 1 assessed the rate of cardiac troponin I (cTnI) outliers on the Abbott ARCHITECT analyzers. Importantly, they focused on the critical region where the difference between the results of duplicate measurements exceeded the allowable difference, with one of the two measurements above the 99th percentile cutoff (≥0.04 µg/L) and termed this a ‘critical outlier’. 1 Their findings are noteworthy in that the critical outliers appear to be false positives with the root cause unknown but unlikely to be related to an analyzer malfunction. Our laboratory, since November 2012, has used the Abbott ARCHITECT cTnI assay for routine clinical care. In addition to regular commercial quality control (QC) material, we monitor performance using a low cTnI concentration patient-pool QC material (approximately 0.03 µg/L).2,3 Recently, our laboratory switched to the 500-test reagent pack from the 100-test reagent pack and through monitoring with the low-pool QC material identified possible positive cTnI outliers at this critical region. After assurances from the manufacturer that the analyzer was functioning appropriately, we set up a series of experiments utilizing a pool of patient samples with ‘normal’ cTnI concentrations to explore the extent of positive outliers and to assess if it was related to a specific analyzer or cTnI reagent lot.
Within-run precision using ‘normal’ cTnI patient-pools to identify a specific lot and instrument outlier effect.
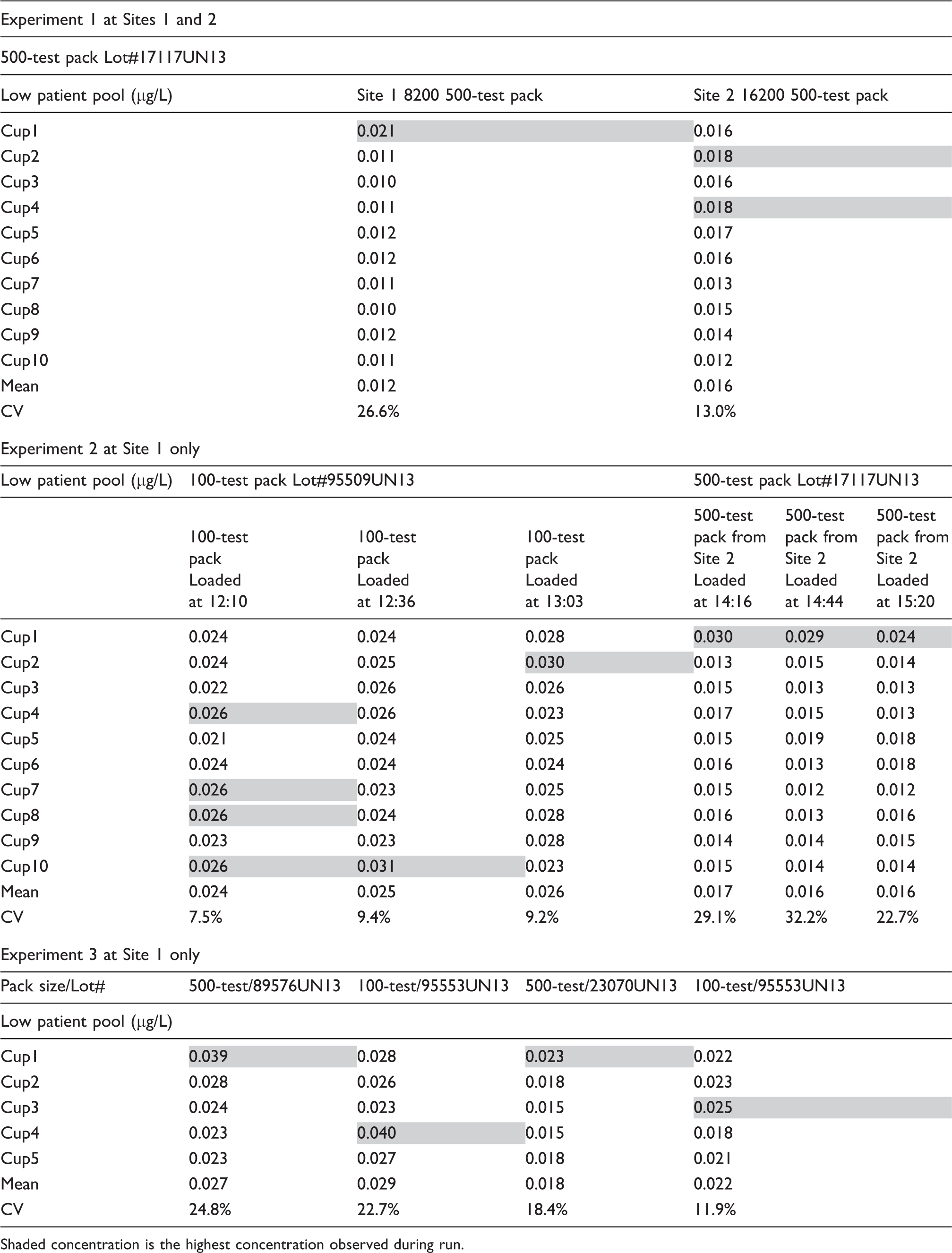
Shaded concentration is the highest concentration observed during run.
Sawyer et al. did not indicate whether a 500- or 100-test reagent lot was used during their study, nor were they able to identify if the critical outlier was with the first result after a period of inactivity. Our experiments suggest that contributing factors to variability include a period of inactivity on the analyzer (i.e. in running mode with no samples being processed) and the reagent pack size. During our investigation, at no time did we observe differences ≥0.03 µg/L with the normal pool that may have been misinterpreted clinically as a significant change. 4 We were able to uncover this positive first result outlier only with a normal cTnI patient-pool, thus reaffirming the importance of monitoring cTnI assays with appropriate QC material below the 99th percentile, regardless of using a high-sensitivity or sensitive assay.3,5 Finally, our findings indicate a renewed importance of performing within-run precision testing when evaluating cTnI reagent lots.
Footnotes
Acknowledgements
Abbott Diagnostics for technical support and evaluation/scout kits.
Declaration of conflicting interests
PK has received grant support and/or honorarium for work related to cardiac biomarkers from Abbott Laboratories, Beckman Coulter, Randox Laboratories, Roche Diagnostics.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
PK.
Contributorship
PK, LC, SL designed the experiments. LC and SL oversaw the analyses of samples. PK and AD analysed the results and drafted the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
