Abstract
Background
This study examined the new high-performance liquid chromatography analyser HLC-723GX (GX) and investigated its ability to both measure glycosylated haemoglobin (HbA1c) values and determine whether haemoglobin variants could cause interference with these measurements in the Japanese population.
Methods
For the basic GX examination, the within- and between-run precision, linearity of measurements, correlation of HbA1c values with current systems and the interference of chemically modified haemoglobin were determined. GX interference caused by the haemoglobin variant was examined by analysing 39 clinical laboratory samples that contained haemoglobin variants.
Results
Good within- and between-run precision were found, with the coefficients of variation at ≤1.0%. A wide range of HbA1c measurement values were confirmed, with the HbA1c values strongly correlated with the results of the currently used HLC-723G8 system. Chemically modified haemoglobins were prepared by adding glucose, sodium cyanate, acetaldehyde or acetylsalicylic acid to normal blood samples. None of these samples had any influence on the HbA1c values determined by GX. GX analysis showed haemoglobin variants that eluted after HbA0 and were similar to HbD, or HbS had HbA1c values that were close to those measured by boronate affinity chromatography and immunoassay. GX found lower HbA1c values in blood that contained HbE or haemoglobin variants, which elute before or at nearly the same time as HbA0.
Conclusions
GX is useful for the analysis of HbA1c samples that contain HbD, HbS, HbC and haemoglobin variants, even though the elution times are similar. However, a countermeasure is needed in order to avoid overlooking other haemoglobin variants in Japan.
Introduction
Glycosylated haemoglobin (HbA1c) is a blood glucose control marker used in the diagnosis of diabetes mellitus and has been recognized as a convenient control marker that helps prevent complications.1–3 A large number of assay systems and reagent kits are commercially available for HbA1c measurements, with most based on the principles of high-performance liquid chromatography (HPLC) or immunoassay. Among all of these approaches, cation-exchange HPLC is one of the primary analytical methods used in the world today. Chemically modified haemoglobins produced in vivo or haemoglobin variants arising from inherited globin chain abnormalities are known to interfere with these measurement results. For example, carbamylated haemoglobin, which is increased in the blood of patients during dialysis treatments, can cause interference. However, advances in column technology now ensure this interference will only be minimal. In addition, there is no interference in some of the commercially available analysers because the major haemoglobin variants such as HbD, HbS and HbC can be separated from normal haemoglobin components via HPLC separation in conjunction with having the actual HbA1c value available for the subsequent calculations.4,5 Although these major haemoglobin variants are detected throughout the world, the frequencies of each variant can differ in different populations. 6
In Japan, the type and frequency of the detected haemoglobin variants differs greatly from that found in non-Japanese populations. The overall detection frequency of the variants within the Japanese population is lower, with a rate of about 1 in 2500 people. At the present time, there are about 160 haemoglobin variants that have been discovered in the Japanese population. In almost all cases, there are no clinical symptoms associated with these variants.7,8 HbS and HbC are frequently detected in American and African blacks, while HbE is observed in Chinese and Southeast Asian populations. However, these variants are not highly distributed in Japanese populations. 9 In addition, since the distribution of haemoglobin variants differs in the Japanese population, there is a chance that Japan’s medical community may overlook the significance of haemoglobin variant interference when performing HPLC measurements of HbA1c.
The currently marketed HLC-723G8 HPLC analyser (G8; Tosoh Co., Tokyo, Japan) utilizes a standard mode for HbA1c measurements, with an analysis time of 1 min per sample. This device is also equipped with a variant mode that has a longer analysis time of 1.6 min per sample, and thus is capable of minimizing the interference from HbD, HbS and HbC due to the column separation. 10 Haemoglobin variants are infrequently detected in Japan, as the standard analysis mode model is the preferred setting since it allows for a faster determination time. When G8 is used in the variant mode, the haemoglobin variants with similar electrical properties to HbD, HbS and HbC elute after HbA0, while with the standard mode, they co-elute with HbA0. In these types of cases, even though the measured HbA1c value is falsely decreased from the actual value, the chromatograms do not usually show any abnormal peaks. 11 In addition, the haemoglobin variants detected in the Japanese population are often asymptomatic and therefore tend to be missed.
As a countermeasure, we have previously used a method that uses two steps to detect the haemoglobin variants. 11 In the first step, blood samples collected prior to meal intake that are suspected of containing haemoglobin variants are selected based on the correlation between the blood glucose values and the LA1C+ fraction values reported by the G8 for the same sample. This method is based on the principle that a high correlation exists between the blood glucose concentration and the LA1C+ fraction value, for which the main component is the labile HbA1c.
The LA1C+ fraction value in the specimen containing the inseparable haemoglobin variant will be lower than that which is expected for the LA1C+ value obtained from the blood glucose value. Therefore, the calculated value can be utilized to detect the indistinguishable haemoglobin variant. Subsequently, we then used high-resolution HPLC to check the samples for the presence of haemoglobin variants.
Recent development of the small and convenient new HPLC analyser HLC-723GX (GX; Tosoh Co.) for HbA1c determination has made it possible to measure HbA1c values using a separation mode that is similar to the variant mode of the G8 analyser. However, there have been no previous studies that have estimated the ability of this device to identify the haemoglobin variants in the Japanese population.
Therefore, the aim of this study was to examine this newly developed HPLC analyser and investigate its ability to both measure HbA1c values and determine whether haemoglobin variants could cause interference with these measurements in the Japanese population.
Materials and methods
Instruments and reagents
The GX uses a high-resolution separation mode that is similar to the variant mode of the G8 analyser. This device uses a cation-exchange column and three elution buffers of different salt concentrations. The analysis time is 2.2 min per sample, with each normal blood sample separated into six fractions: A1A; A1B; F; LA1C+; SA1C and A0. Labile HbA1c and carbamylated haemoglobin, both of which are known to interfere with measurements, were eluted in the LA1C+ fraction.
In contrast to the previous Tosoh systems, this new separation technique uses six fractions, with the net result of being able to achieve a higher level of specificity for the HbA1c analysis. Haemoglobin variants, which include HbD, HbS and HbC, are separated at longer elution times than that for HbA0 and are identified in the V0, V1 and V2 windows. By applying a correction calculation based on the volume of the separated haemoglobin variant fractions, HbA1c values with minimal interference from the haemoglobin variants can be obtained. The supplied column and reagents were exclusively used throughout the study, with all analyses performed in accordance with the procedures listed in the manufacturer’s instruction manual.
As a comparison system, we used the G8 in the standard, variant and beta-thalassaemia modes, 12 with the supplied exclusive columns and reagents used for all analyses. As the control measurement for HbA1c values in samples that contained the haemoglobin variants, we used the G8 instrument in the boronate affinity mode. 13 As an additional control measurement, we used the Tina-quant II HbA1c assay (Roche, Basel, Switzerland), 14 which is an immunoassay for HbA1c, in combination with the Modular Analytics automated biochemical analyser (Roche) in accordance with the manufacturer’s instruction manual. For comparison of the separability of the haemoglobin variants by GX, we used a high-resolution HPLC method. This method utilized the analytical column and elution buffer for the standard mode of the G8 device, as these gradient conditions can be used to separate the various haemoglobin variants. 11
Samples
The National Glycohemoglobin Standardization Program (NGSP) was used for HbA1c. Blood samples were handled in accordance with the ethical guidelines of our institution. The samples used for the fundamental examination contained two different concentrations of the quality control samples (haemoglobin A1c control level 1 and level 2; Tosoh). All patients’ blood was collected in a blood collection tube containing sodium fluoride (Terumo, Tokyo, Japan) and subsequently submitted to the clinical laboratory of our hospital for blood glucose and HbA1c determinations. To study the interference of the haemoglobin variants on the HbA1c determinations performed by the GX, blood samples that contained the haemoglobin variant were obtained from patients at our hospital and then stored at −80℃ until being analysed. Out of the 39 blood samples collected, two contained HbG-Taichung (alpha 74 Asp → His), one contained HbG-Szuhu (beta 80 Asn → Lys), four had HbE (beta 26 Glu→Lys) and four were unidentified but strongly suspected to be HbE. Although the remaining 28 cases indicated that the significant peak of the haemoglobin variant was beyond 20% in the high-resolution HPLC analysis, final diagnoses were not made for these samples due to the judgement of the doctor in charge. With regard to these cases, we defined any subject with an abnormal peak area beyond 20% as being positively suspected of having haemoglobin variants. This definition was based on the theory that the content of haemoglobin variants with a heterozygous point mutation in the alpha-globin chain is 25%. 7 The study blood samples were classified into four groups based on the characteristics of the haemoglobin variants detected during the routine high-resolution HPLC and G8 chromatograms.
Type A: Haemoglobin variant peak occurred at a shorter elution time than the HbA0 observed in the high-resolution HPLC.
Type B: Haemoglobin variant peak was similar to the elution time of the HbA0 observed in the high-resolution HPLC.
Type C: Haemoglobin variant peak was at a longer elution time than that observed for HbA0 in the high-resolution HPLC. These cases were determined to not have HbD, HbS or HbC based on their pedigree information and other clinical findings.
HbE: An abnormal peak was observed between HbA1c and HbA0 on the chromatograms by the standard mode of the G8. Although this abnormal peak was not observed by the high-resolution HPLC analysis, it was observed close to HbA2 in the beta-thalassaemia mode of the G8 device. All patients in these cases were from Southeast Asia.
Examination protocol
For the GX precision tests, the within-run precision was investigated by performing 30 consecutive assays using haemoglobin A1c control level 1 and level 2 quality control samples along with two different patient blood concentrations. The GX between-run precision was investigated by analysing the same samples over a 10-day period. All blood samples were divided into small quantities and stored at −80℃ until the examination. Linearity of measurement was investigated using blood samples from patients with low (2.3%) and high (20.5%) HbA1c concentrations. To prepare the haemolyzed specimens, the blood samples were diluted with Haemolysis and Wash Solution (Tosoh) at a dilution ratio of 200:1. In the next step, we investigated the dilution linearity of the HbA1c measurements by performing a 10-step dilution sequence using two haemolyzed samples, followed by GX analysis.
A total of 100 patient specimens that had been submitted for routine testing were used to investigate the correlation of the HbA1c values, with all values determined by the current G8 system. Specimens were analysed by the GX and compared to the results of the standard and variant modes of the G8. To investigate influences of chemically modified haemoglobins, namely the labile haemoglobin, carbamylated haemoglobin, acetaldehyde-modified haemoglobin and acetylated haemoglobin were generated by adding glucose, sodium cyanate, acetaldehyde or acetylsalicylic acid to a volunteer’s blood with 5.9% HbA1c. Each of the substances were prepared using a five-step process in which a series of concentrations were added to test tubes containing the volunteer’s blood, after which all of the samples were maintained at 37℃ for 1 h. The maximum final concentration of each added substance was 10 g/L for glucose, 250 mg/L for sodium cyanate, 250 mg/L for acetaldehyde and 500 mg/L for acetylsalicylic acid. All of these concentrations were set at levels higher than the highest concentration that may exist pathophysiologically. During the GX measurements, if the HbA1c values showed a change of more than ±0.2% with respect to the non-supplemented sample, measurement interference was considered to have occurred.
To investigate haemoglobin variant interference on the HbA1c determination at our hospital, we collected 39 blood samples that contained the haemoglobin variant, with all samples stored at −80℃ until use. These samples were analysed by the GX and by the standard and variant modes of the G8 instrument. All results were compared with measurements obtained by the G8 boronate affinity mode and the Tina-quant II HbA1c, which normally exhibit minimal interference due to haemoglobin variants. To study the separability of the haemoglobin variants, chromatograms of the GX were compared with the high-resolution HPLC and the beta-thalassaemia mode of the G8. This protocol was approved by the Human Studies Committee of Nihon University Itabashi Hospital (reference No. RK-1109-0286).
Statistical analysis
Changes in the values were analysed using a paired t-test. A value of P < 0.05 was considered significant.
Results
Precision, linearity and correlation
Precision data for the HbA1c (%) assay on the HLC-723GX.
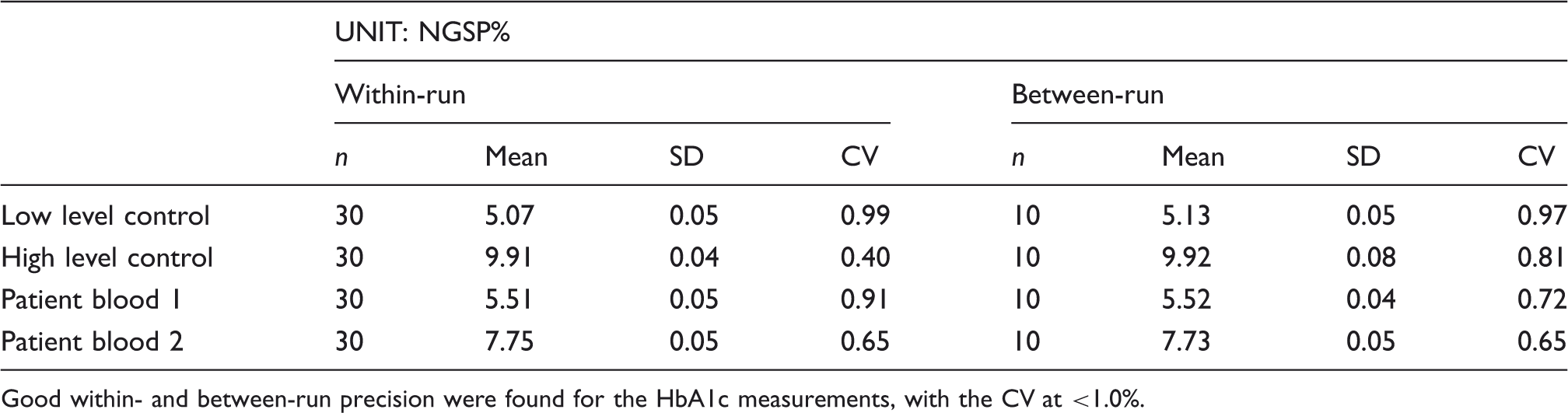
Good within- and between-run precision were found for the HbA1c measurements, with the CV at <1.0%.
Influence of chemically modified haemoglobin
Labile HbA1c caused no interference with the HbA1c measurements in the presence of glucose concentrations up to a maximum of 10 g/L. Similarly, there was no interference seen from carbamylated haemoglobin up to 250 mg/L of sodium cyanate content or acetaldehyde-modified haemoglobin up to 200 mg/L acetaldehyde content. Our results also confirmed that the LA1C+ fraction value increased in a linear manner from approximately 2% to 5%. In addition, increases in the additive concentration of acetylsalicylic acid were not accompanied by any change in the LA1C+ fraction values, with the HbA1c measurements only showing a slight upward trend. The increase in the HbA1c value was less than 0.2% with respect to the non-supplemented sample, even with a maximum additive concentration of 500 mg/L.
Influence of haemoglobin variants
Analytical data for HbA1c (%) in cases with some haemoglobin variants.

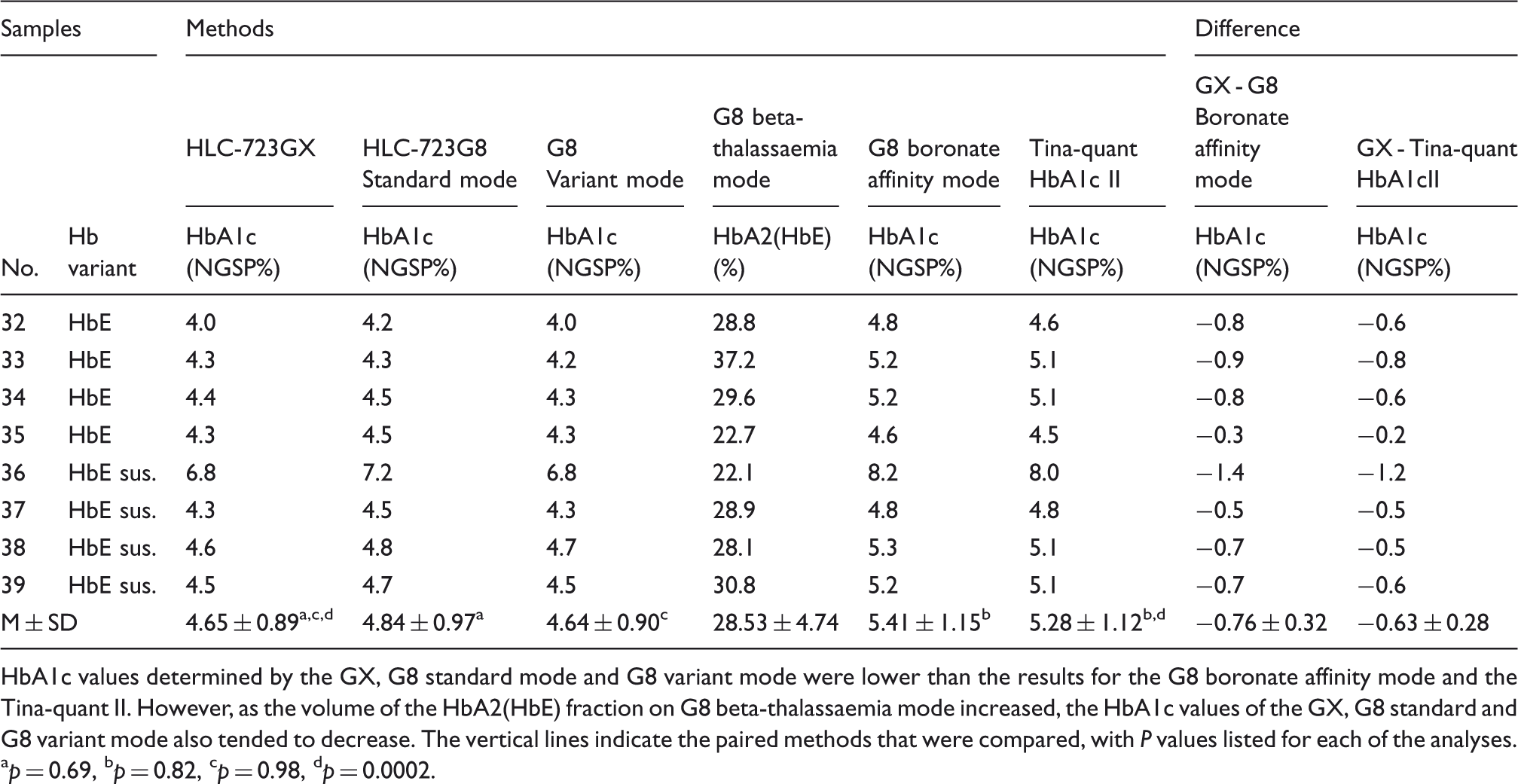
V0, V1 and V2 of GX are windows used for separating HbD, HbS and HbC. In the samples containing Hb variants which have shorter elution times than HbA0 (Type A) and elution times near to HbA0 (Type B), almost all HbA1c values determined by the GX were lower than those for the G8 boronate affinity mode or the Tina-quant II (used as the reference methods). The samples containing haemoglobin variants which eluted within the V0 or V1 window (Type C) had similar values to those for the reference methods. The vertical lines indicate the paired methods that were compared, with P values listed for each of the analyses. ap = 0.89, bp = 0.31, cp = 0.85, dp = 0.82, ep = 0.86
When the Type C cases containing the haemoglobin variant that eluted after the HbA0 were assayed using the GX, variant peaks were detected for two samples from case 23 and case 26 in the V0 window, which is the time where the HbD normally elutes. Seven haemoglobin variant cases, including HbG-Szuhu and HbG-Taichung, showed variant peaks within the V1 window, which is the point where HbS elutes. None of the samples used in this evaluation exhibited any variant peaks in the V2 window, which is the time where the HbC normally elutes. Measured GX values closely matched the results for the variant mode of the G8 and for the Tina-quant II (P = 0.85–0.86 > 0.05). The concentrations of HbA1c measured by the G8 boronate affinity mode for case 24 and case 30 were significantly higher than those for the Tina-quant II. All Type C values measured by the G8 standard mode showed significantly lower levels as compared to the Tina-quant II values.
Figure 1 shows the typical chromatograms in Type A, B and C, with the chromatograms arranged in order of the elution time of the haemoglobin variant. In the Type A cases, abnormal peaks due to haemoglobin variants or their glycosylated forms were observed by the GX and G8 chromatograms. These abnormal peaks interfered with the HbA1c fraction (Figure 1, shown in black). For example, the abnormal peak in case 1 interfered with the front portion of the HbA1c fraction, especially in the G8 variant mode analysis. In other cases, the abnormal peak was misidentified as being the HbA1c fraction (as was seen in the GX measurement of case 6). In the Type B cases, which have an elution time near to HbA0, no abnormal peaks were observed for either the chromatograms of the standard and variant modes of the G8, or for the GX. Since haemoglobin variants can be indistinguishable when only using chromatograms to determine their presence, a cautious approach is required to ensure that these haemoglobin variants will not be overlooked. Similar to the Type B cases, no abnormal peaks were observed for the Type C cases when using the standard mode of the G8. However, haemoglobin variants with longer elution times than HbA0 were separated and identified in the V0 or V1 window when using the variant mode of the G8 and the GX.
Chromatograms of blood samples with haemoglobin variant assayed by several HPLC methods. On the chromatograms of the G8 standard mode, abnormal peaks were observed only in the Type A samples. With the G8 variant mode and GX, abnormal peaks were observed with the Type A and Type C samples. For the Type B samples, unlike the results in the G8 standard mode, there were no abnormal peaks observed.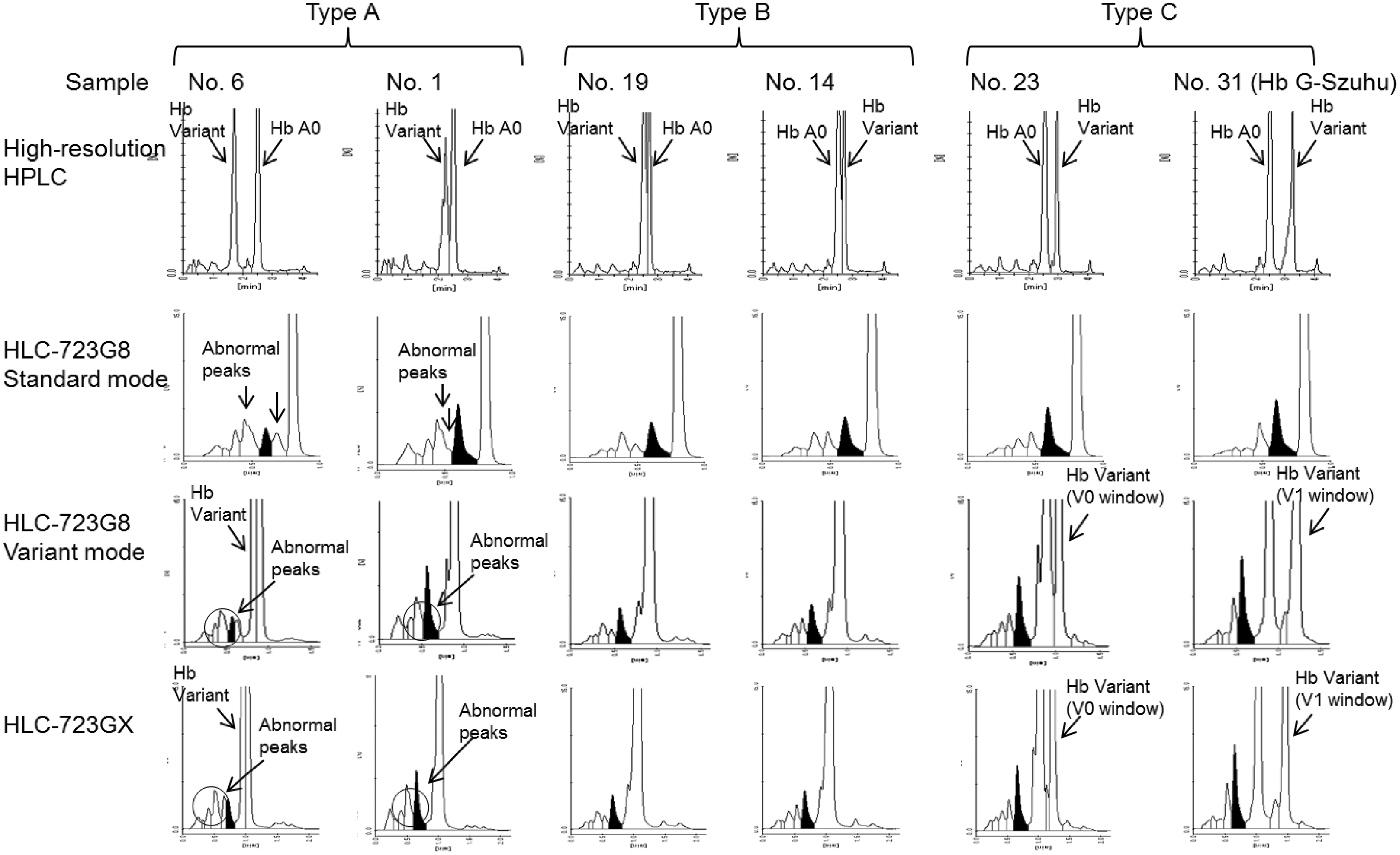
Table 3 shows the analytical data for the cases that contained the HbE and HbE-suspected haemoglobin. The HbA1c values determined by the GX, and the standard and variant modes of the G8 all matched (P = 0.69–0.98 > 0.05), with all measurements 0.2–1.4% lower than the results for the G8 boronate affinity mode and the Tina-quant II HbA1c. In these cases, the HbE elution time was similar to that for HbA2 in the G8 beta-thalassaemia mode, with a fraction volume of 22.1–37.2%. In addition, as the fraction volume of HbE increased, interference with the HbA1c values also tended to increase for the GX and the G8 standard and variant modes. The chromatograms for case 32 are seen in Figure 2. Moreover, as seen in all of the samples listed in Table 3, the same abnormal peak was also observed for case 32. This peak was identified between the HbA1c and HbA0 fractions by GX and by the standard and variant modes of the G8. HbE, however, was not separated by any of these analysis modes. When the HbA2 separation was examined by the G8 beta-thalassaemia mode, both HbE and HbE-suspected Hb eluted at the same time as the HbA2.
Chromatograms of the sample containing HbE (No. 32) assayed by several HPLC methods. While an abnormal change or peak was observed between the HbA1c and HbA0 fractions when using the G8 standard mode, the G8 variant mode and the GX, HbE was not separated by any of these analysis modes. When using the G8 beta-thalassaemia mode, results showed that HbE eluted at the same retention time as that for HbA2. Analytical data for HbA1c (%) in cases containing HbE or suspected to be HbE. HbA1c values determined by the GX, G8 standard mode and G8 variant mode were lower than the results for the G8 boronate affinity mode and the Tina-quant II. However, as the volume of the HbA2(HbE) fraction on G8 beta-thalassaemia mode increased, the HbA1c values of the GX, G8 standard and G8 variant mode also tended to decrease. The vertical lines indicate the paired methods that were compared, with P values listed for each of the analyses. ap = 0.69, bp = 0.82, cp = 0.98, dp = 0.0002.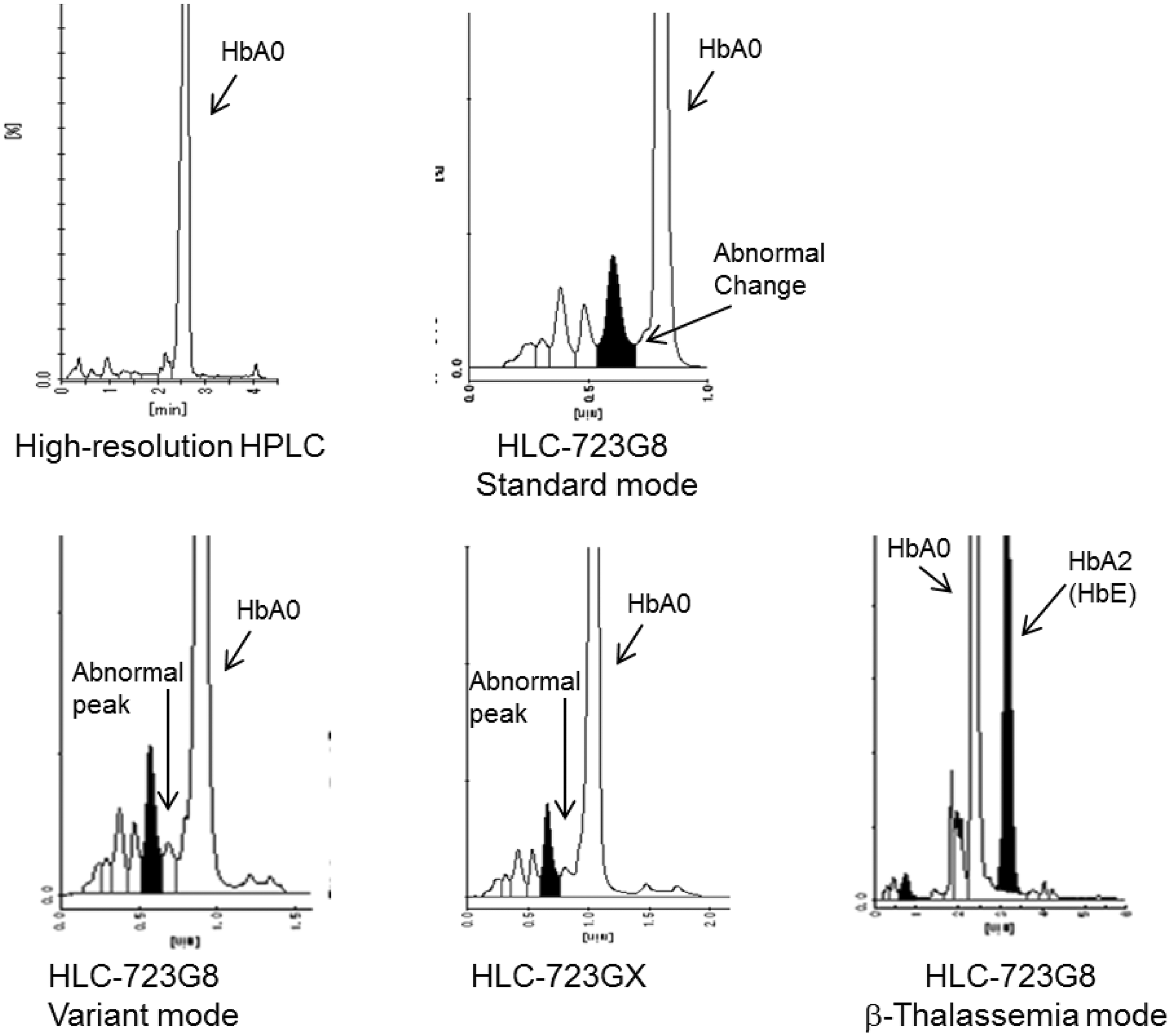
Discussion
We used the GX to determine the HbA1c values in blood that contained haemoglobin variants that were different from the major variants, such as HbD, HbS and HbC.
Prior to performing the study, we first examined the basic performance of the GX and determined there was good within- and between-run precisions for the HbA1c measurements (CVs ≤ 1.0%), sufficient linearity of the measurements, high correlation between the standard and variant modes of the G8 and no interference from chemically modified haemoglobins. On the basis of these results, the basic measurement performance of the GX was judged to be in no way inferior to the currently used systems.
In our present study, all blood samples were collected from Japanese and Southeast Asian populations, as samples from these subjects could contain undefined haemoglobin variants. Our results showed that all HbA1c concentrations measured for Type A by the GX, the G8 standard mode and the G8 variant mode were all lower than those determined for the controls. However, since the glycosylation materials may interfere with the HbA1c fraction, this could induce false high levels, such as was seen in our case 1. 15 As we can determine the existence of haemoglobin variants for Type A cases, we also need to carefully check the results of chromatograms for Type B cases. With the Type B haemoglobin variants, both the GX and G8 falsely reported finding decreased HbA1c values with no abnormal peaks in the chromatograms. Since these haemoglobin variants can be indistinguishable, a cautious approach is required in such cases. To avoid these problems, it is recommended that the relationship between the LA1C+ fraction and blood glucose be analysed in conjunction with high-resolution HPLC in order to determine which of the samples may contain Hb.
The HbA1c values of Type C measured by GX or by G8 were almost same as the values of the Tina-quant HbA1c II. In some of the Type C cases, however, values measured by the G8 boronate affinity mode were found to be slightly higher than those reported by the Tina-quant HbA1c II. This discrepancy may be related to the principle that the boronate affinity methodology is based on. 13
The widely used variant mode of the G8 analyser is able to perform measurements without any interference by HbD, HbS or HbC. 4 When we examined haemoglobin variants with Type C electrical properties, i.e. variants with elution times similar to HbD or HbS, the HbA1c values determined by GX closely matched the variant mode of the G8 analyser. Although we did not examine the HbC haemoglobin variant in this study, since the elution time has been shown to occur after that of HbS, this implies that GX could also be used to separate this variant. Moreover, the HbA1c value determined by the GX additionally appears to be closely matched to that determined by G8. Thus, this suggests that GX will be a useful HPLC system for HbA1c determination in countries where these haemoglobin variants are common.
Due to recent elevations in the number of immigrants from Southeast Asia, Japanese clinical laboratories are increasingly encountering samples that contain HbE. Unfortunately, when the standard and variant modes of the G8 analyser are used with only salt concentration gradients, HbE cannot be separated from HbA0. The only way the HbE peak can be confirmed is to use the beta-thalassaemia mode of the G8 analyser. 12
In cases where the HbE cannot be separated when using GX, G8 standard mode and the G8 variant mode, the concentrations of HbA1c may be lower than the values reported when using the G8 boronate affinity mode or the Tina-quant II HbA1c, as HbE co-elutes with HbA0. When we obtain such information during routine testing with the G8 in our hospital, primary physicians are informed that HbE is strongly suspected as being the haemoglobin variant.
In the current study, when GX and the variant mode of G8 were used to assay the samples, we found an abnormal peak between HbA1c and A0 in the chromatograms due to the glycosylated form of HbE. If this abnormal peak is checked in the G8 variant mode, an alarm will automatically occur and it will suggest the existence of HbE. Thus, even when the isolation of HbE is difficult, the presence of this function on the G8 device is very useful, thus adding this system to the GX would be of benefit. Moreover, the same abnormal peaks observed for the HbE trait can sometimes be seen due to problems with the elution buffer, the column or the analyser. Therefore, addition of a beta-thalassaemia analysis mode to the GX would also be expected to further enable the confirmation of HbE.
Conclusions
The GX is a useful HPLC system for determining HbA1c in populations where common haemoglobin variants such as HbD, HbS and HbC traits exist. In countries such as Japan, however, there are a wide variety of haemoglobin variants that exist and that could interfere with the HbA1c measurements. Therefore, we must take care not to overlook these variants in routine analyses performed in clinical laboratories. As a potential solution, we recommend that countermeasures similar to our approach that used correlation regression analysis and high-resolution HPLC be implemented. Furthermore, in order to definitively confirm the HbE trait, the beta-thalassaemia mode, which can separate HbE from HbA0, also needs to be included as a part of the routine analysis.
Footnotes
Acknowledgements
The authors wish to thank the Tosoh Corporation and Roche Diagnostics for providing the analysers and assistance with sample measurements performed in this study.
Declaration of conflicting interests
None declared.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
This protocol was approved by the Human Studies Committee of Nihon University Itabashi Hospital (reference No. RK-1109-0286).
Guarantor
TN.
Contributorship
TM and TN researched literature and conceived the study. TS, MN and AS were involved in protocol development, gaining ethical approval, patient recruitment and data analysis. TM wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
