Abstract
Recent studies have found that cardiac stem cells (CSCs) are present in the adult heart. CSCs play an important role in maintaining the balance of the number of myocardial cells. The purpose of this study was to examine characteristics of human CSCs and their correlation with clinical characteristics of patients. We collected heart auricles of 105 patients (age range, 1–78 years; mean, 55.6 ± 17.0 years) undergoing cardiac surgery to obtain CSCs. We assayed the percentage of c-kit positive (c-kit+) CSCs with flow cytometry. Plasma Nɛ-(carboxymethyl)lysine (CML) concentrations were measured by enzyme-linked immunosorbent assay. The percentage of c-kit+ CSCs was 4.96 ± 3.12% (0.98–17.17%), and this was significantly negatively correlated with age, the presence of diabetes mellitus (DM) and coronary heart disease (CHD) (r values were −0.797 [P < 0.01], −0.500 [P < 0.01] and −0.250 [P = 0.011], respectively). The percentage of c-kit+ CSCs was significantly negatively correlated with CML concentrations (r = −0.859, P < 0.01). The percentage of c-kit+ CSCs decreases with ageing and is further decreased in patients with DM and/or CHD. Furthermore, plasma CML concentrations may have potential as an indicator of the number of c-kit+ CSCs.
Introduction
Ischaemic heart disease is a major cause of death in developed countries. Currently, the main clinical treatment for ischaemic heart disease is palliative and aimed at preserving the function of remaining cardiac myocytes. Cell transplantation offers the potential to effectively reverse cardiac damage and restore cardiac function. 1 Recent studies have demonstrated the existence of cardiac stem cell (CSC)-like populations in adult hearts that can be self-renewing, clonogenic and multipotent, giving rise to cardiac myocytes, smooth muscle and endothelial cells in vivo and in vitro. This finding has undermined the notion of the adult heart as a terminally differentiated organ without self-renewal potential. 2 Resident CSCs have been found in the adult mammalian heart, including the mouse, rat, and human,2–8 and this has led to a new field of research for heart diseases and myocardial repair. The origin and significance of these cells has remained obscure for lack of a proper biological context. A previous study reported that c-kit+ cells have the properties of CSCs. 2
Animal studies have demonstrated low efficiency of donor cells isolated from older individuals.1,9 Human mesenchymal stem cells (hMSCs) derived from old patients are less robust than those from young donors. 1 Indeed, multiple effects of age on stem cell function have been confirmed, including effects on stem cell number, 10 proliferation potential, 11 senescence and apoptosis, 12 replicative capacity 13 and in vitro differentiation potential. 14 In animal experiments, ex vivo growth is less robust in stem cells from older donors compared with that in young donors, and old cells do not confer the same benefits as young cells after implantation into an experimental infarct.15,16 Age shows a progressive decrease in functionally competent human CSCs. 17 Therefore, in this study, we first studied characteristics of human CSCs and then investigated their relationship with clinical characteristics of patients.
Materials and methods
Study population
This study was performed in 105 consecutive patients undergoing cardiac surgery from November 2011 to November 2012, and it is in accordance with the approval of the Southeast University Medical Ethics Committee (Approval No: 2012ZD11KY28.0). Patient’s sex and age, smoking status, blood pressure, disease type and medication use were recorded. Fasting total cholesterol (TC), high-density lipoprotein (HDL) cholesterol, low-density lipoprotein (LDL) cholesterol, high sensitive C reactive protein (hsCRP) and renal function (estimated glomerular filtration rate [eGFR], formula: eGFR = 186 × [serum creatinine in mg/dl]−1.154 × [age]−0.203 × 0.742 [if female]) were measured in an accredited laboratory.
Glycoxidation product Nɛ-(Carboxymethyl)lysine (CML, a major advanced glycation endproduct [AGE]) concentration in patients’ plasma was determined using a CML enzyme-linked immunosorbent assay (ELISA) kit (Westang Bio-tech, Shanghai, China, Catalog No: F00438) according to the manufacturer’s instructions. A 5-mL blood sample was collected from each patient between 6:00 and 7:00 a.m. after an overnight fast (12–14 h) into EDTA (1 mg/mL). The sample was centrifuged at 3000 (rotations per minute) rpm for 15 min at room temperature within 90 min of collection, and the supernatant was stored at −80℃ for the CML assay. All ELISA assays were performed in duplicate.
Isolation and culture of CSCs
Human CSCs were isolated from adult human heart tissue (generally taken from the cardiac auricle) that was removed during cardiosurgery, e.g. valve replacement surgery. These surgical samples (approximately 0.4–2 g) were minced and seeded onto the surface of uncoated Petri dishes for culture of 2–3 weeks. The cell culture medium was DMEM/F-12 (Hyclone, Logan, USA) containing 10% fetal calf serum (Hyclone, Logan, USA), 2 mmol/L
Statistical analysis
Data analysis was performed using SPSS version 18.0 (SPSS Inc., Chicago, IL, USA) and CurveExpert 1.3 (Daniel G. Hyams, Crossgate Street Starkville, MS, USA). Data are shown as mean ± SD (standard deviation) or number and percentage for categorical variables, unless stated otherwise. A P value of <0.05 (two-tailed) was considered statistically significant. A logistic model was used to calculate plasma CML concentrations. Pearson’s correlation was used to test the association of two parameters. To describe the linear association of the percentage of c-kit+ CSCs and a set of exploratory variables, a multiple linear regression model was developed by the stepwise method.
Results
Clinical characteristics of the study population
Baseline characteristics of the study population (n = 105).
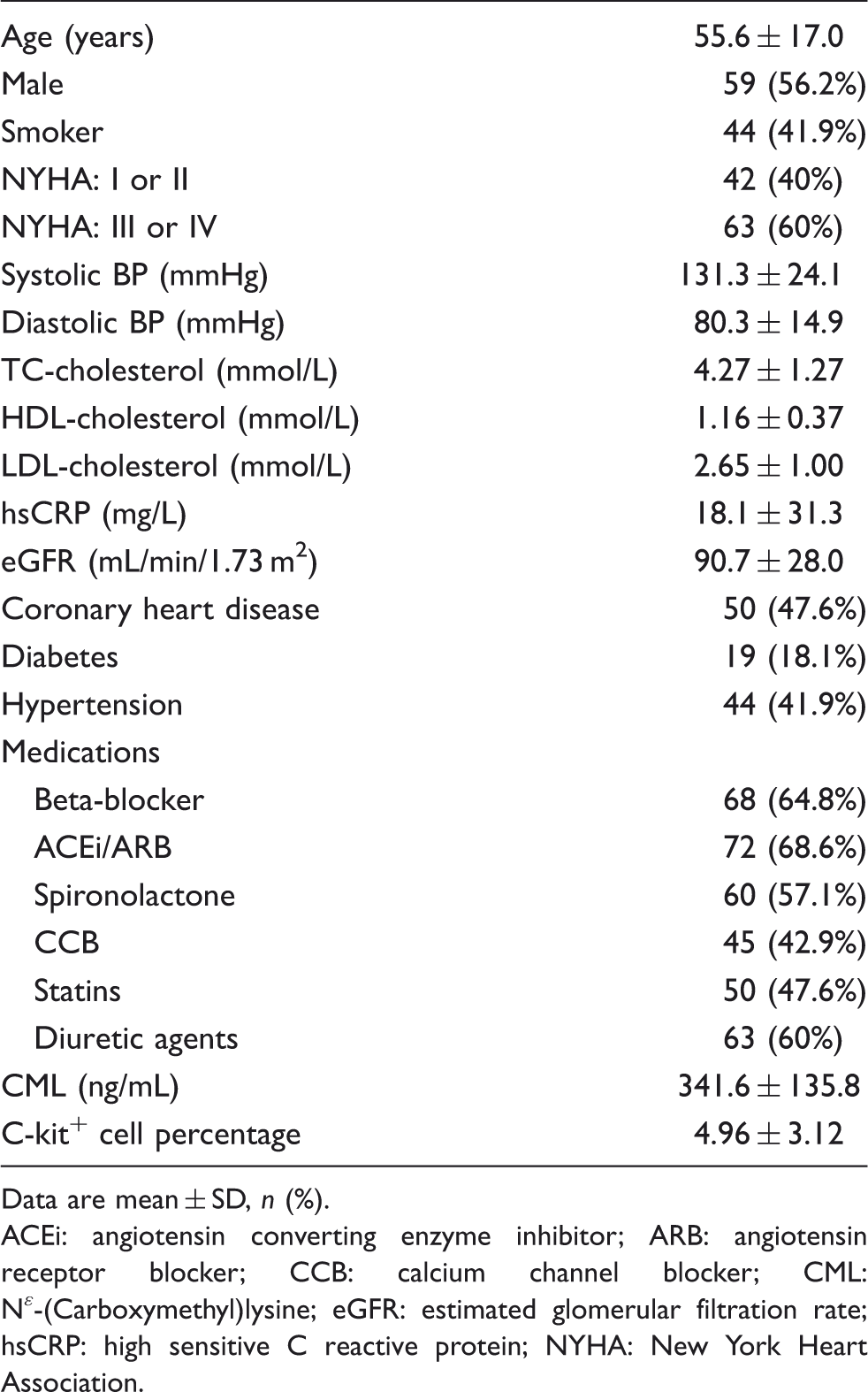
Data are mean ± SD, n (%).
ACEi: angiotensin converting enzyme inhibitor; ARB: angiotensin receptor blocker; CCB: calcium channel blocker; CML: Nɛ-(Carboxymethyl)lysine; eGFR: estimated glomerular filtration rate; hsCRP: high sensitive C reactive protein; NYHA: New York Heart Association.
Percentage of C-kit+ CSCs and its relationship with clinical indicators
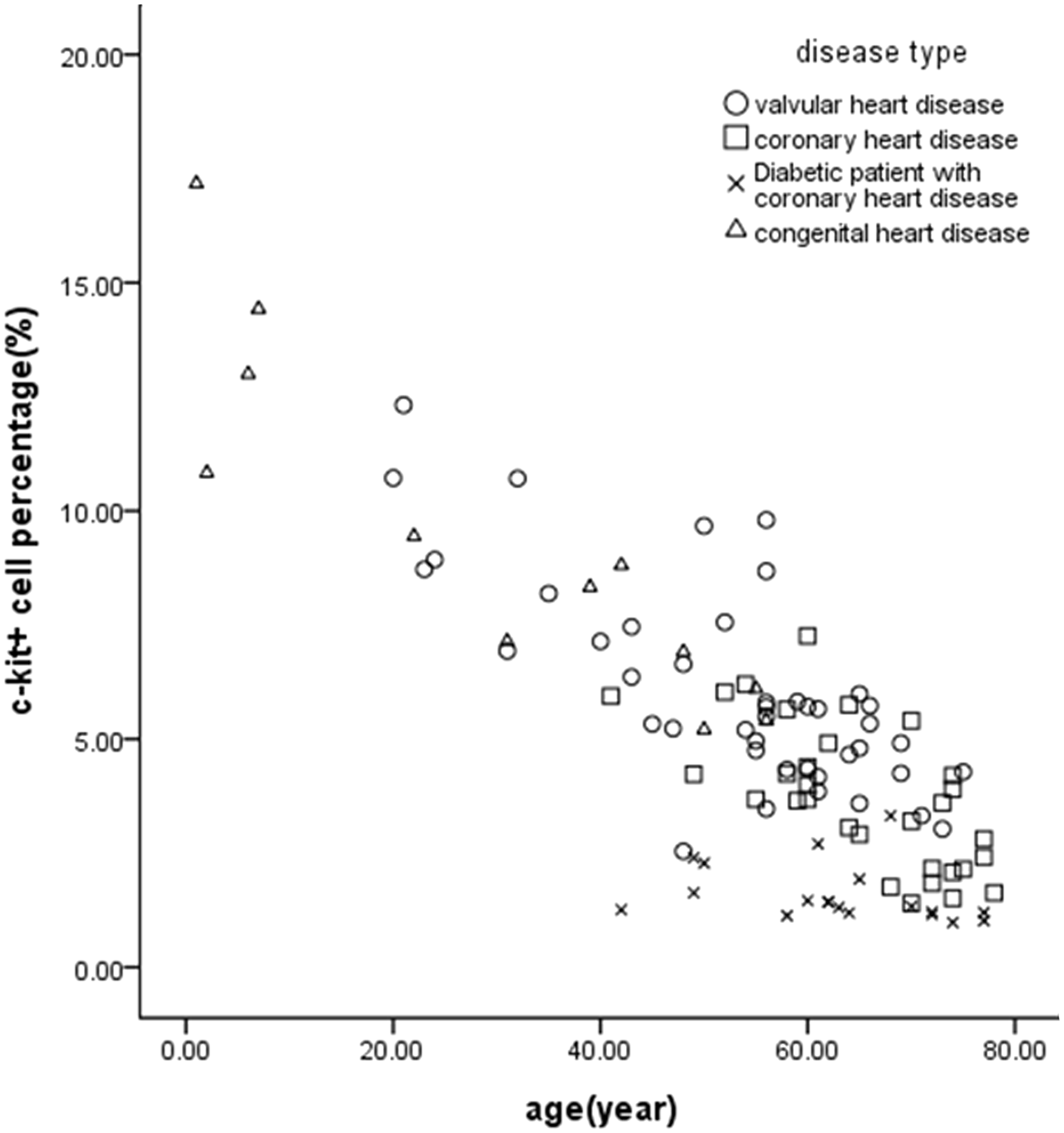
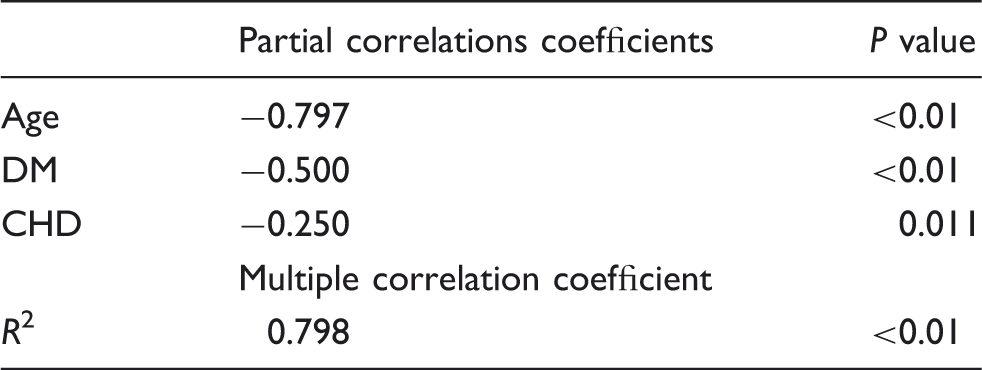
C-kit+ CSCs comprised 4.96 ± 3.12% (0.98–17.17%) of the entire cell population, and after one sorting the c-kit positivity could exceed 85% (Figure 1). Multiple linear regression analysis showed that the percentage of c-kit+ CSCs was significantly negatively correlated with age, DM and CHD (r values were −0.797 [P < 0.01], −0.500 [P < 0.01] and −0.250 [P = 0.011], respectively), which could explain 79.8% of the variation of the percentage of c-kit+ CSCs (P < 0.01) (Table 2 and Figure 2). Univariate linear analysis showed a significant correlation between eGFR and the percentage of c-kit+ CSCs (r = 0.383, P < 0.01). However, after adjustment for age, this relationship disappeared (r = 0.070, P = 0.642). No correlations were found between the percentage of c-kit+ CSCs and other clinical indicators, such as blood pressure, NYHA functional class, TC, HDL, LDL and hsCRP concentration. The use of 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors (statins), beta-adrenergic antagonists, calcium-channel blockers, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers and diuretic agents had no effect on the percentage of c-kit+ CSCs.
Representative tracings of the flow cytometry. Representative flow cytometric analyses of CPCs for expression of the cell surface markers c-kit: (a) 8.12%, before sorting; (b) 86.1%, after one sorting. Effect of age and type of disease on the percentage of c-kit+ cells. Multiple linear regression analysis showed that four diseases all had a significant effect on the percentage of c-kit+. However, after adjustment for age, the percentage of c-kit+ cells was significantly negatively correlated only with age (r = −0.797, P < 0.001), diabetes (r = −0.500, P < 0.001) and coronary heart disease (r = −0.250, P = 0.011), accounting for 79.8% of the variability in the percentage of c-kit+ cells (P < 0.001). Variables related to the percentage of C-kit+ cell in the study group (n = 105).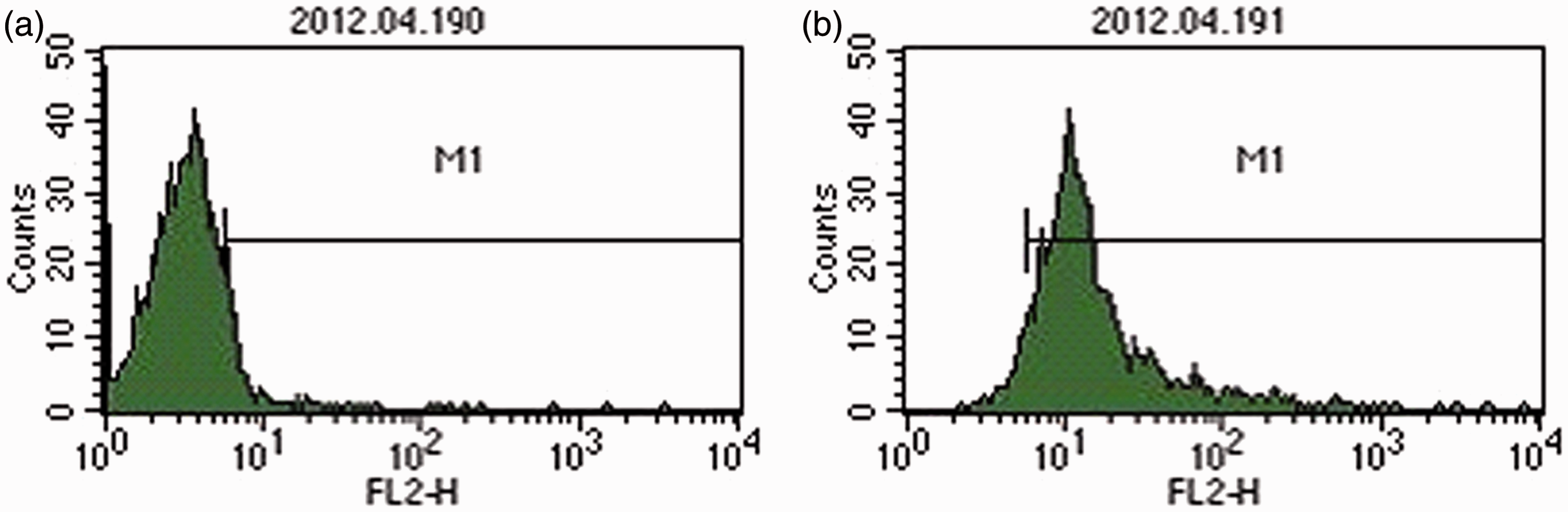
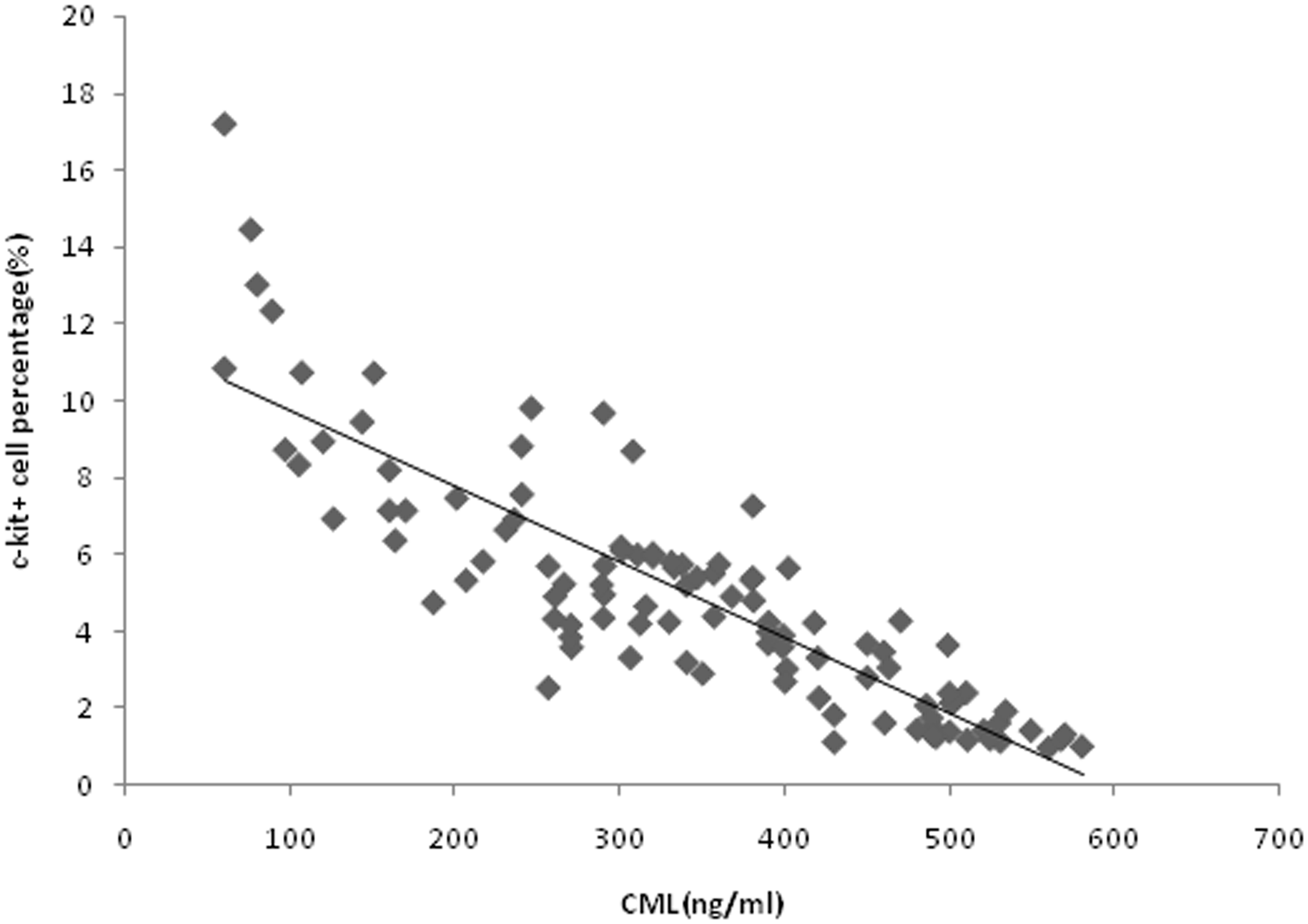
The mean (range) CML concentration was 341.6 ± 135.8 ng/mL (60.1–580.3 ng/mL). CML concentrations were significantly positively correlated with age, DM and CHD (r values were 0.769 [P < 0.01], 0.553 [P < 0.01] and 0.544 [P < 0.01], respectively), which could explain 86.1% variation of CML concentrations (P < 0.01) (Table 3 and Figure 3). Similarly, univariate linear analysis showed a significant correlation between eGFR and CML concentrations (r = −0.458, P < 0.01), but after adjustment for age, this relationship disappeared (r = 0.099, P = 0.507). Linear regression analysis showed that the percentage of c-kit+ CSCs was significantly negatively correlated with CML concentrations (r = −0.859, P < 0.01) (Figure 4).
Effect of age and type of disease on CML concentrations. Multiple linear regression analysis showed that four diseases all had a significant effect on CML concentrations. However, after adjustment for age, CML concentrations were significantly positively correlated only with age (r = 0.769, P < 0.001), diabetes (r = 0.553, P < 0.001) and coronary heart disease (r = 0.544, P < 0.001), accounting for 86.1% of the variability in CML concentrations (P < 0.001). Correlation between CML concentrations and the percentage of c-kit+ cell. Linear regression analysis indicated that the CML concentration had a significant negative correlation with the c-kit+ cell percentage (r = − 0.859, P < 0.001). Variables related to CML concentrations in the study group (n = 105).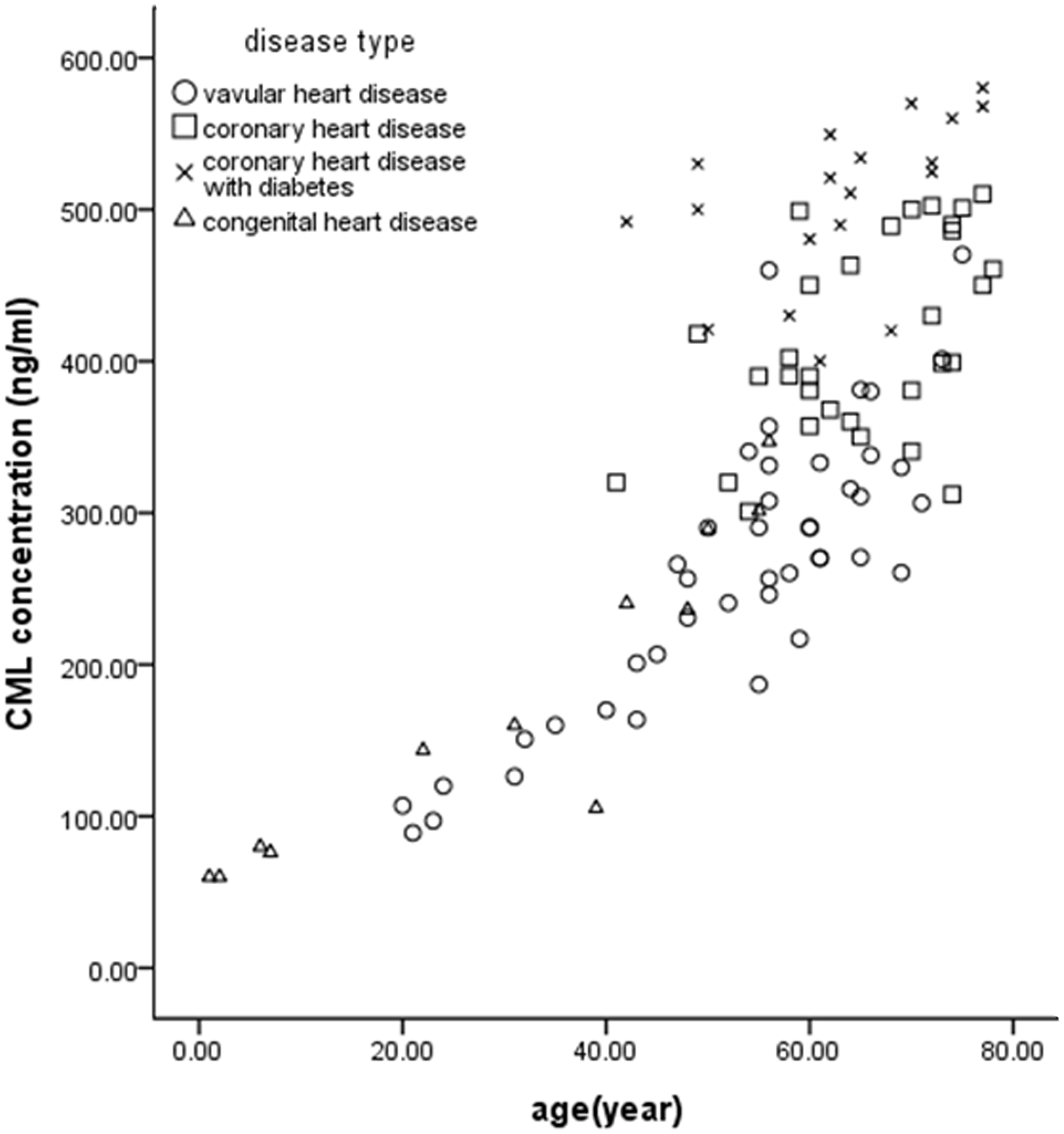

Discussion
The main finding of this study was that the percentage of c-kit+ CSCs in patients decreases with age. Furthermore, the presence of DM and CHD resulted in a further decrease in the percentage of c-kit+ CSCs. In addition, the percentage of c-kit+ CSCs was negatively correlated with plasma CML concentrations.
A growing number of studies have shown that the adult heart possesses a pool of clonogenic CSCs, which can differentiate into cardiac myocytes, smooth muscle cells and endothelial cell lineages in vitro and in vivo.2,18,19 In addition, studies have described the continuous formation of new cardiac myocytes in the adult mammalian normal and diseased heart.20,21 These findings have led to the idea that normal and pathological cardiac homeostasis involve death and renewal of cardiac myocytes.20,21 C-kit+ CSCs are self-renewing clonogenic, and multipotent, and give rise to three different cardiogenic cell lineages.2,18 In this study we also directly demonstrated that the c-kit+ cell from hearts can give rise to myocytes in vitro. However, the stem cell potential of c-kit+ cells in the heart remains controversial. Several studies have shown that c-kit+ cells might not give rise to cardiomyocytes, but act as endothelial progenitor cells.22–24 Even so, many studies in animals and humans have demonstrated that transplantation of c-kit+ CSCs was effective in improving left ventricular systolic function and reducing infarct size.25–27 Therefore, much further work on c-kit+ CSCs is needed.
CSCs reside in niches, including supporting nurse cells, junctional and adhesion proteins, that are located deep in the heart tissue for protection from damaging stimuli. These niches constitute a microenvironment in which stem cells divide, differentiate and die.28,29 The direct physical interactions between stem cells and their non-stem cell neighbours in the niche are critical in keeping stem cells in this specialized compartment and in maintaining stem cell characteristics.30–32 There are many factors related to cell senescence, which can limit the survival and regenerative potential of stem cells.33,34 These factors include telomere attrition and/or dysfunction, which increase cells’ sensitivity to apoptotic stimuli, 35 and micro-environmental changes, which restrict stem cell function and engraftment potential. 36 Furthermore, cell growth and clonogenic potential are both diminished in hMSCs from old donors. 1 Rota reported that DM decreased the number of c-kit+ CSCs, 37 which was consistent with the findings of our study.
CML is an advanced AGE. 10 AGEs are constituted by a heterogeneous group of molecules formed by non-enzymatic reactions of reducing sugars, ascorbate and other carbohydrates with amino acids, nucleic acids and lipids. 11 AGEs can lead to vascular damage by forming intra- and inter-molecular cross-links with matrix proteins in the vascular wall, causing increasing vessel rigidity, trapping lipoproteins within the arterial wall and disrupting the clearance of the vessel. 12 AGEs interact with the endothelial receptor of AGEs (RAGE), which induces procoagulant activity, impairment of endothelium-dependent relaxation, increased vascular permeability, migration of macrophages and T-lymphocytes into the intima and oxidative stress.13,14 One of the best characterized AGEs is CML. 10
Our study found that plasma CML concentrations increased with age and were further increased in patients with DM after adjustment for age. A previous study reported enhanced accumulation of CML in patients with DM 15 and with ageing, 38 and enhanced accumulation of CML appeared in heart tissue of eight patients with DM. 39 Our study also found that plasma CML concentrations in patients with CHD increased further after being adjusted for age. CML concentration was not an independent variable, but an intermediate rather than a confounding variable. CML has also been detected in atherosclerotic lesions as an endogenous biomarker for local oxidative stress. 16 Previous studies by our group have demonstrated that cardiac CML concentrations show the same changes as plasma CML concentrations. 40
Cardiac AGE accumulation is associated with a significant decrease in myocardial collagen solubility, an index of increased formation of collagen cross-link. 41 AGE formation associated with DM results in reduced extracellular matrix degradation and angiogenesis. 42 Therefore, long-term accumulation of CML gradually changes the microenvironment of CSCs.41,42 These changes can reduce the oxygen required by the CSCs, and CML itself may have an adverse effect on CSCs, proliferation of neural stem cells is decreased in AGE culture medium. 17 CHD can result in a reduction in the blood supply of the microenvironment in which stem cells reside and may reduce the number of CSCs. All of these changes lead to a gradual reduction in the content of c-kit+ cells and then pathological changes in the myocardium occur.
Rota 37 demonstrated that reactive oxygen species (ROS) played an important role in decreasing the number of c-kit+ CSCs, but the present study speculated that AGEs (especially CML) may play an important role. The generation of intracellular AGEs can disturb redox homeostasis by modifying protein and enzyme structure and function. 43 While oxidative stress can augment the formation of AGEs through glycoxidation, AGEs can also lead to enhanced formation of free radicals. 44 Studies indicate that ROS generation with subsequent increased oxidant stress is a potent factor initiating signal transduction and altered gene expression as a result of the AGE–RAGE interaction. 45 Therefore, although the focus of the two studies differed, the results are consistent. ROS and AGEs (including CML) can promote each other and function together.
Ageing and DM affect the structure of the heart, such as the loss of cardiac myocytes, cardiac myocyte hypertrophy and collagen deposition in the extracellular matrix, resulting in myocardial interstitial fibrosis and a reduction in cardiac function.46–51 Senescence of CSCs has been observed in aged and diseased hearts, which may lead to a decrease in regenerative capacity, 21 and hMSCs derived from old patients are less robust than those from young donors. 1 In young hearts, 1% of cardiac myocytes are renewed annually, but this rate decreases to 0.45% in ageing hearts. 20 Ageing cardiac myocytes do not show a lower rate of cell death compared with young cardiac myocytes, 52 but the formation of new cardiac myocytes decreases. Low cardiac myocyte renewal in ageing and DM leads to pathological changes in the heart. All of these data are indirect proof of a gradual decline in the number of CSCs with proliferation and differentiation potential. Currently, it is thought that CSCs may be a natural mixture of stromal, mesenchymal and progenitor cells. 53 However, the number of cardiac c-kit+ CSCs might be more important than their origin because the compensation efficiency depends on the number of c-kit+ CSCs rather than its source.
Conclusion
Our study shows that the percentage of c-kit+ CSCs decreases with ageing, DM and CHD. In addition, plasma CML concentrations may be a promising tool for assessment of CSCs. We speculate that pathological changes in aged and diseased hearts, especially CHD and DM, are based on the gradual loss of c-kit+ CSCs in the heart, and the remaining CSCs cannot effectively supplement the gradual loss of myocardial cells. Further investigations to elucidate the exact role of CML in pathological changes in the heart are required.
Footnotes
Acknowledgments
The authors are grateful to Ya-fei Zhang (Public Health Institute, Southeast University) for support with statistical analysis.
Declaration of Conflicting Interests
None.
Funding
This work was supported by grants from National Natural Science Found (No. 6590000029) and Chinese Medical Association (No. 09010140169).
Ethical approval
It is in accordance with the approval of the Southeast University Medical Ethics Committee (Approval No: 2012ZD11KY28.0).
Guarantor
GM.
Contributorship
SH researched literature, assessed the patient results database, completed the entire experiment, analysed the data and wrote the first draft of the manuscript, assisted by HX. GY assisted with preparing the results database and data analysis. WH and ZL provided the myocardial tissue. GM conceived the study, supervised SH and assisted with data analysis and manuscript drafting. All the authors reviewed and edited the manuscript and approved the final version of the manuscript.
