Abstract
Introduction
Associations of insulin-like growth factor-II (IGF-II) and insulin-like growth factor binding protein-2 (IGFBP-2) with cardiovascular risk have been inadequately studied. We hypothesized that IGF-II and IGFBP-2 associate with longitudinal trends in lipid profiles in type 2 diabetes patients.
Subjects and methods
Four hundred and eighty nine subjects with type 2 diabetes (age 27–87 years) from the Salford Diabetes Cohort were studied. Longitudinal clinical information was extracted for an eight-year period (2002–2009) from an integrated electronic dataset of primary care and hospital data.
Results
There were 294 male subjects and mean age was 62.9 years. At baseline, IGF-II concentration was 602 ng/mL. HDL cholesterol at baseline was associated with log-IGF-II concentration in a model adjusted for age, gender, baseline body-mass index (BMI), estimated glomerular filtration rate (eGFR) and lipid-lowering therapy. IGFBP-1 and IGFBP-2 were associated with high HDL-cholesterol. A higher circulating IGF-II concentration at baseline was also associated with longitudinal increase in HDL-cholesterol in mixed-effects regression analyses independent of IGF-I, IGFBP-1, IGFBP-2, IGFBP-3, age, gender, eGFR, BMI and lipid-lowering therapy. Log-transformed baseline concentrations of IGFBP-1 and IGFBP-2 were also associated with longitudinal elevation in HDL-cholesterol. No association was observed for IGF-II or IGFBP-2 with longitudinal LDL cholesterol trends.
Conclusion
Our analyses based on ‘real world’ data demonstrate that higher baseline IGF-II and IGFBP-2 predict increased HDL concentration over time, implicating IGF-II in modulation of circulating HDL-cholesterol concentrations.
Keywords
Introduction
The insulin-like growth factors (IGF-I and IGF-II) are critical to growth and metabolism. 1 The biological role of IGF-II in embryonic growth and early development is well established, but the role of this peptide in both health and disease within adults has not been adequately clarified. 2
There is evidence to suggest a role for IGF-II in adiposity and lipid metabolism. Low IGF-II has been associated with subsequent weight gain in individuals with and without type 2 diabetes,2,3 though there is some evidence that suggests the contrary. 4 We have previously demonstrated in individuals with type 2 diabetes, significant positive correlations of serum IGF-II concentration with total cholesterol and HDL cholesterol. 5 Furthermore, another study primarily involving older subjects not on lipid lowering therapy demonstrated that IGF-II and its principal carrier protein IGF binding protein (IGFBP)-3 were positively correlated with serum HDL cholesterol and total cholesterol concentration. 6 The latter finding is not surprising as the biological activities of IGF-II are governed by its interactions with IGFBPs, including IGFBP-1, IGFBP-2 and IGFBP-3.
The hypothesis of this study was that baseline concentrations of IGF-II and IGFBP-2, which has preferential IGF-II affinity, were biomarkers for longitudinal associations in total cholesterol, HDL cholesterol and triglycerides. We analysed ‘real-world’ data in a Caucasian population with type 2 diabetes. IGF-I, IGFBP-1 and IGFBP-3 were also studied for longitudinal associations with these lipid measures.
Subjects and methods
Sample population
A total of 489 patients with type 2 diabetes were recruited from the city of Salford in North-West England. The city has approximately 9100 subjects with type 2 diabetes out of a total population of around 242,000. The Salford Longitudinal Diabetes Cohort is representative of this background diabetes population and was recruited in 2002–2003 from patients under routine annual review in the diabetes outpatient clinics in the Salford Royal NHS Foundation Trust. The patients were community dwelling and there were no specific exclusion criteria. Approval was granted for this study by the local research ethics committee. Patients donated a blood sample for the measurements of IGFs and their binding proteins, which were collected only once (at baseline) and were non-fasting.
Clinical data
Longitudinal clinical and pharmacological data were obtained from a comprehensive linked electronic database of primary and secondary care information related to the study patients. Measurements of serum total cholesterol, serum HDL cholesterol, serum LDL cholesterol, serum triglycerides and glycated haemoglobin (HbA1c) for the years 2002–2009 were requested as a part of routine medical care by the patients’ healthcare providers, and results of these investigations were collected for each of the years between 2002 and 2009. As all the study patients were recruited from the Salford area, the majority of the blood tests were performed at the clinical laboratories of the Salford Royal Hospital.
For this study, clinical and biochemical measurements and prescriptions of lipid lowering therapy for the year 2002 were considered as being baseline data. For the longitudinal analysis, subjects were considered to have been on 3-hydroxy 3-methylglutaryl coenzyme A inhibitors (‘statins’) or fibrates if they were prescribed these drugs for any duration between 2002 and 2009. Duration of diabetes was defined at baseline (2002) as the number of years since the first diagnosis of diabetes at that time.
Measurement of IGF proteins and cholesterol concentration
IGF-I was measured using the Immulite 1000 immunoassay (Siemens UK, Camberley). The analytical sensitivity of this assay was 20 ng/mL and intra- and inter-assay coefficients of variation (%CV) were <4.5% and <8.4%, respectively. IGF-II was measured using an enzyme-linked immunosorbent assay (ELISA) that was developed using antibodies previously used in other studies. 7 This assay had an analytical sensitivity of <10 ng/mL, with intra-assay and inter-assay, sensitivities were <6% and <10%, respectively. A locally developed ELISA was used to measure IGFBP-1 and this assay detects all phosphoforms of this protein as reported in a previous study. 8 This assay had intra-assay and inter-assay %CV of <9% and <15%, respectively. The lower limit of detection of IGFBP-1 assay was 0.1 ng/mL. IGFBP-2 measurements were performed using a commercial ELISA kit manufactured by RayBiotech Inc (Norcross, GA, USA) with a manufacturer reported sensitivity of < 20 pg/mL. This assay had an intra-assay %CV of <10% and an inter-assay %CV less than 12%. The Immulite 1000 immunoassay was used to measure plasma IGFBP-3 and had an analytical sensitivity of 0.1 µg/mL. The within-assay %CV for this assay was <6% and the between-assay %CV was <10%.
Measurement of total cholesterol, HDL cholesterol and triglycerides were performed using the Roche Integra 700 (Roche Diagnostics, Rotkreuz, Switzerland) platform from 2001 to 2004, and using the Roche Modular system (Roche Diagnostics, Rotkreuz, Switzerland) thereafter. The measuring range of HDL cholesterol was 0.08–3.10 mmol/L with a lower limit of detection of 0.08 mmol/L. The measuring range of triglycerides was 0.05–11.3 mmol/L with a lower limit of detection of 0.05 mmol/L.
LDL cholesterol values extracted from the healthcare records were estimated measures using the Friedewald equation. 9 Estimated glomerular filtration rate (eGFR) was calculated from serum creatinine data using the gender specific Modification of Diet in Renal Disease equation. 10
Statistical analysis
Statistical analysis was performed using the software package Stata 10SE (College Station, TX). Linear mixed-effect regression analysis was used to study longitudinal trends in serum total cholesterol and HDL cholesterol concentrations as outcomes of baseline serum IGF-II concentration. Circulating concentrations of IGF proteins and serum triglyceride were log-transformed in all analyses to adjust for skewness.
Results
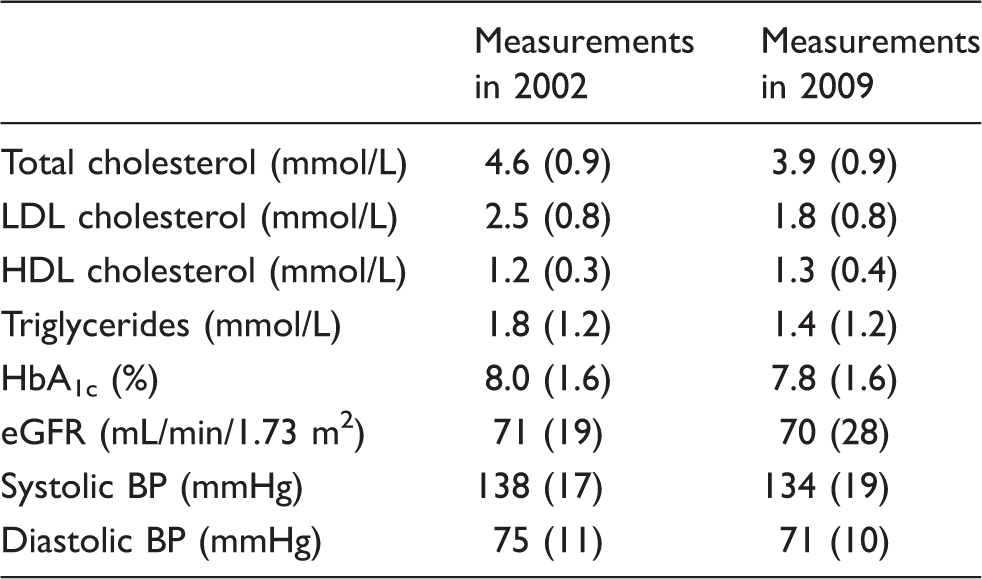
Characteristics of study population in 2002 and 2009. Values expressed as mean (SD) except serum triglycerides which are expressed as median (IQR). HbA1c was measured at both time points using a diabetes control and complications trial (DCCT) aligned assay.
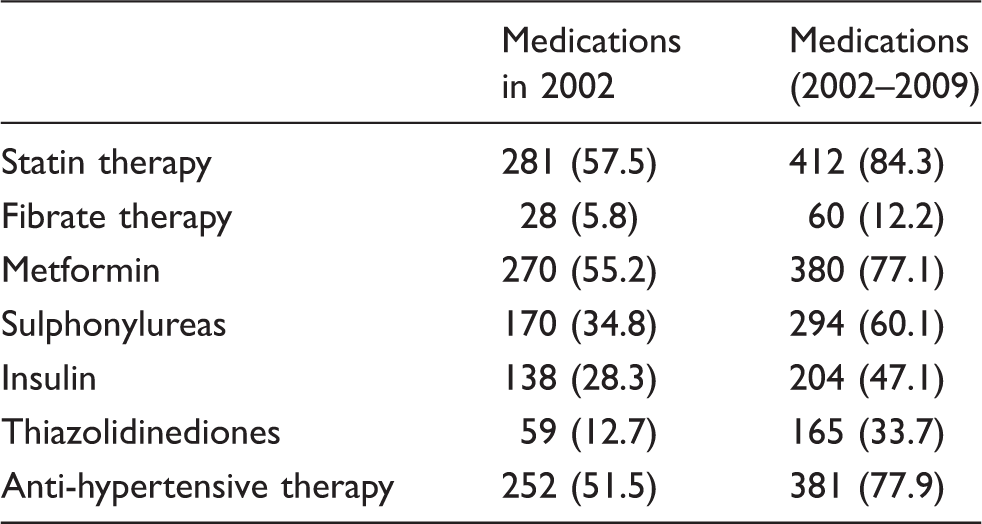
Prescription of selected medications in the study population at study commencement (2002) and the overall study duration (2002–2009) expressed as number of subjects (percentage of study population in parentheses; n = 489).
Vascular outcomes in the study population (expressed in numbers, with percentage of total study population in parentheses; n = 489).

Mean serum IGF-II concentration in the study sample population was 602 ng/mL (95% CI 585–621). Of the study population, 281 (57.5)% were on 3-hydroxy-3-methylglutaryl CoA (HMG CoA) reductase inhibitor (‘statin’) therapy at study commencement in 2002, while the numbers increased to 412 (84.3% of the study population) when statin prescription anytime over the study period (2002–2009) was considered. Each IGF protein was analysed for associations with baseline and longitudinal trends in the concentrations of HDL cholesterol, total cholesterol, LDL cholesterol and triglycerides. Results reported for all analyses are with respect to log-transformed values of IGF proteins and triglycerides.
Associations of IGF proteins with HDL cholesterol at baseline
HDL cholesterol at baseline was studied as an outcome of IGF-II concentration in a regression model that also included age, gender, eGFR, body-mass index (BMI) and diabetes exposure at study commencement. High log-IGF-II was positively associated with HDL cholesterol, and associations were unchanged on adjustment for fibrate or statin prescription (β = 0.21, 95% CI 0.09–0.33, P = 0.001). The association was also sustained on addition of IGF-I, IGFBP-1, IGFBP-2 and IGFBP-3 to the model (β = 0.25, 95% CI 0.05–0.46, P = 0.017). As expected, females had a higher HDL cholesterol at baseline as compared to males (β = 0.15, 95% CI 0.06–0.24, P = 0.001).
Association of IGF proteins with longitudinal HDL cholesterol trends over time
The serum HDL trends of the study population in 2002 and 2009 are illustrated in a kernel density plot (Figure 1).
Kernel density estimates of HDL concentrations for the study population at study commencement (2002) and completion (2009).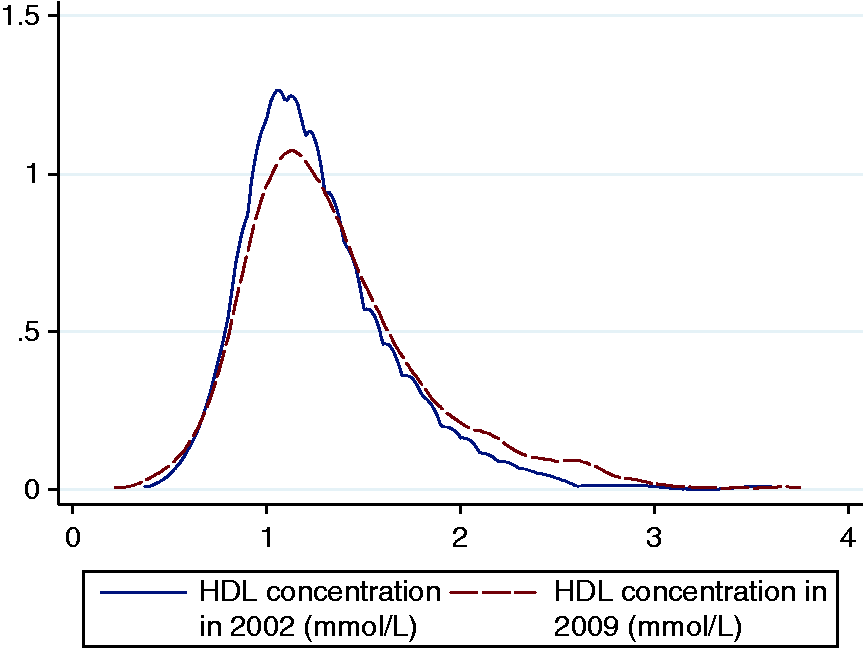
Importantly, we found that higher log-transformed IGF-II concentrations at baseline were predictive of longitudinal increases in serum HDL cholesterol over the period 2002–2009 in a model adjusted for age, gender, baseline eGFR, baseline BMI, diabetes duration, factored time interaction and lipid lowering therapy (β = 0.18, 95% CI 0.72–0.30, P = 0.001).
When all five studied IGF proteins were considered together in the age, gender, diabetes duration and lipid-lowering therapy adjusted model as covariates and factored for time-interaction, IGF-II remained a significant predictor of HDL cholesterol (β = 0.24, 95% CI 0.08–0.42, P = 0.005). Baseline concentrations of IGFBP-1 (β = 0.09, 95% CI 0.04–0.14, P = 0.001) and IGFBP-2 (β = 0.12, 95% CI 0.05–0.19, P < 0.001) also retained significance as predictors of longitudinal increases in HDL cholesterol. However, IGF-I (P = 0.64) and IGFBP-3 (P = 0.34) were no longer significantly associated with HDL cholesterol trends.
Figure 2 describes the mean annual measurements of serum HDL cholesterol per quartile of IGF-II (panel A) and IGFBP-2 (panel B). Importantly the highest quartile of baseline IGF-II was associated with a significantly greater increase in HDL-cholesterol than other quartiles.
Graphical representation of trends in HDL cholesterol in relation to quartiles of baseline IGF-II and IGFBP-2 in the study population for each year from 2002 to 2009. Q1 to Q4 represent quartiles of serum IGF-II (Panel A) and plasma IGFBP-2 (Panel B).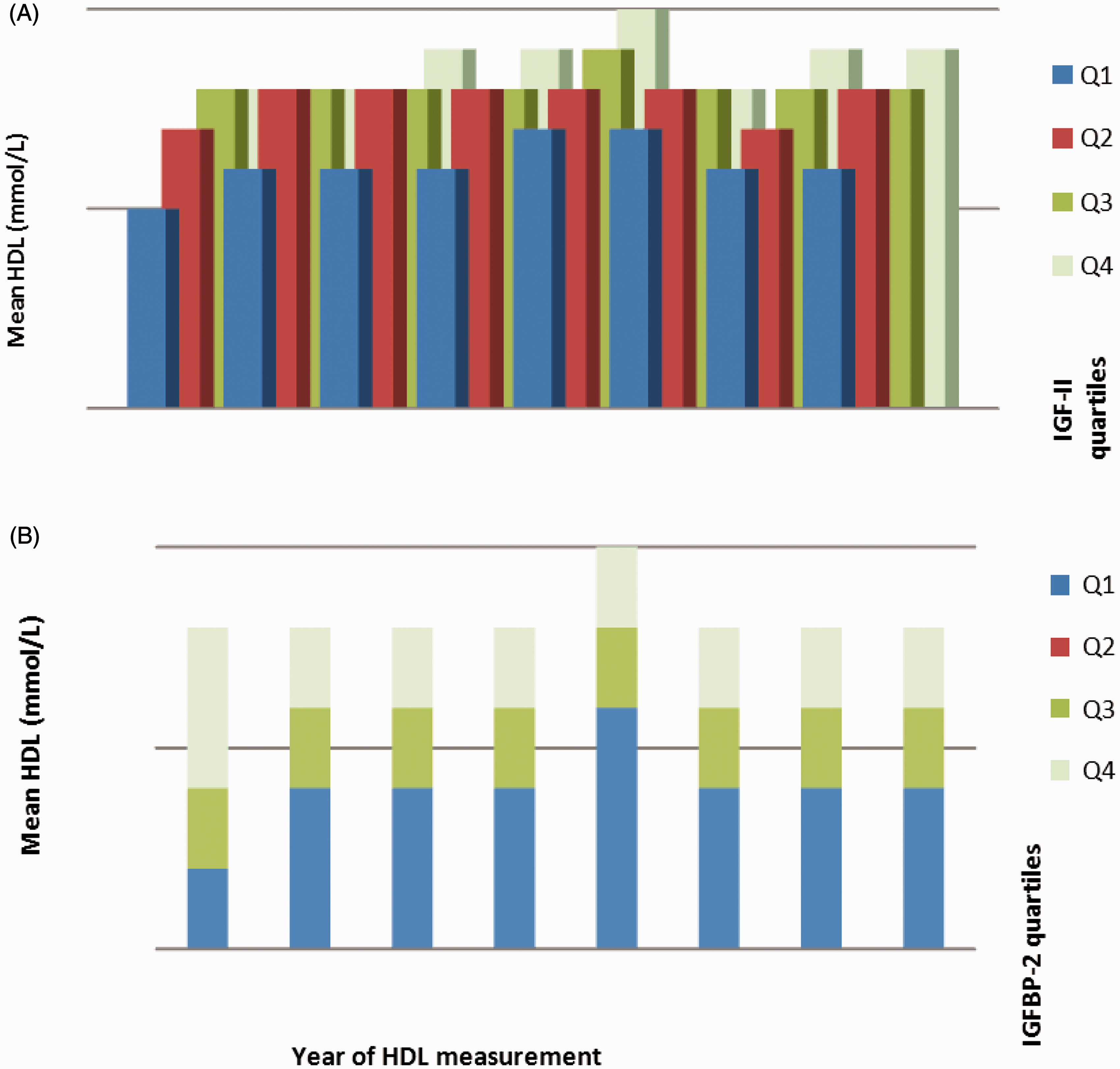
Associations of IGF proteins with baseline and longitudinal trends in total cholesterol
Higher serum IGF-II was also a significant predictor of serum total cholesterol at baseline in a model that also included age, gender, diabetes duration, baseline eGFR, baseline BMI and lipid lowering therapy at baseline (β = 0.30, 95% CI 0.02–0.58, P = 0.04). When all five measured IGF proteins (log transformed) were considered together with age, gender, diabetes duration, baseline eGFR, baseline BMI, time interaction and adjusted for the prescription of statins and fibrates, IGF-II was no longer associated with baseline total cholesterol concentrations (P = 0.19).
When IGF-II at baseline was studied as a predictor of longitudinal trends in total cholesterol between 2002 and 2009 in a model adjusted for age, gender, diabetes duration, baseline eGFR, baseline BMI, time duration and lipid lowering therapy, baseline IGF-II was associated with a longitudinal increase in total cholesterol over the study period (β = 0.39, 95% CI 0.18–0.60, P < 0.001). This association was lost on the addition of the other measured IGF proteins to the study model. Summary measurements of total cholesterol in the study population for the year 2002–2009 are described as annual box-plots (Figure 3).
Summary measurements of total cholesterol in the study population for the year 2002 to 2009 as annual box plots.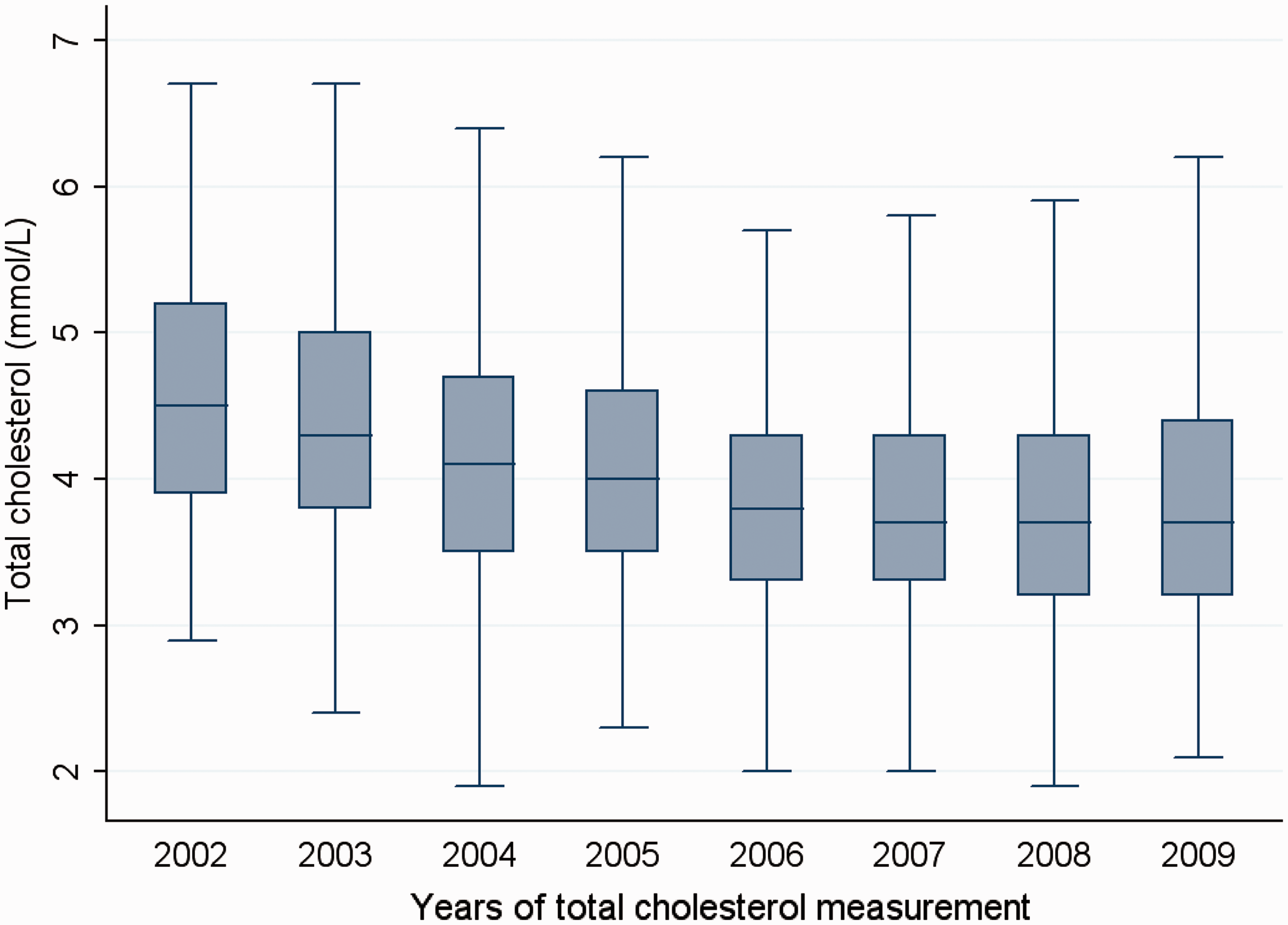
Associations of IGF proteins with LDL cholesterol at baseline and over time
No associations were seen for IGF-I, IGF-II, IGFBP-1, IGFBP-2 or IGFBP-3 with baseline or longitudinal trends in LDL cholesterol either. The models were adjusted for age, gender, diabetes duration, baseline eGFR, baseline BMI and lipid lowering therapy, and additionally for time interaction in the longitudinal analysis.
Associations of IGF proteins with serum triglycerides at baseline
High IGF-I (β = −0.29, 95% CI −0.46 to −0.13, P = 0.001), IGFBP-1 (β = −0.13, 95% CI −0.22 to −0.04, P = 0.003) and IGFBP-2 (β = −0.19, 95% CI −0.31 to −0.08, P = 0.001) were each associated with low baseline triglyceride concentration in models adjusted for age, gender, diabetes duration, eGFR, BMI and lipid lowering therapy. There was no association observed for IGF-II (P = 0.50) or IGFBP-3 (P = 0.54).
Associations of IGF proteins for longitudinal triglyceride trends
Log-transformed values of each of the IGF proteins were studied separately as a predictor of longitudinal log-triglyceride trends in models adjusted for age, gender, diabetes duration, baseline BMI, baseline eGFR, baseline HbA1c, time interaction and lipid lowering therapy. Higher IGF-II (β = 0.13, 95% CI 0.007–0.02, P < 0.001) and IGFBP-3 (β = 0.07, 95% CI 0.04–0.09, P < 0.001) were each associated with increased long-term triglyceride concentrations over time. IGF-I, IGFBP-1 and IGFBP-2 were not associated with long-term serum triglyceride trends.
Discussion
This study demonstrates novel temporal associations of higher baseline circulating IGF-II with longitudinal increases in HDL cholesterol over an eight-year period in a Caucasian population with type 2 diabetes. In other words, a higher IGF-II concentration at baseline is a marker of longitudinal elevation in HDL cholesterol. The findings of previous cross-sectional studies also support our results.5,6
The previously reported cross-sectional association of IGF-II with HDL cholesterol concentration was also replicated. Similar associations were demonstrated for IGFBP-1 (which acutely regulates IGF bioavailability) and IGFBP-2 (which has a preferential affinity for IGF-II) with baseline and longitudinal HDL cholesterol trends. Our findings may have relevance from a public health perspective, as numerous studies have previously demonstrated that increased HDL cholesterol is associated with a reduction in cardiovascular risk.
HDL cholesterol has been recognized for many years as having a potent and inverse association with cardiovascular disease,11–13 though the relation between higher HDL cholesterol and lower incidence of ischaemic heart disease may not be causal. 14 Dietary patterns, exercise, changes in body-weight and fibrate therapy may influence serum HDL concentration.15–17
The IGF-II peptide circulates in concentrations much higher than IGF-I in humans and like IGF-I has a structural homology with insulin. 18 Furthermore, IGF-II can interact with the insulin receptor which exists in two isoforms. IGF-II binds with isoform A of the insulin receptor (IR-A) with high affinity and this interaction is important in embryonic developments and relevant in neoplasia.19,20 The interaction of IGF-II with isoform B of the insulin receptor (IR-B) occurs with lower affinity. 21 The isoform B of the insulin receptor is the ‘classical’ insulin receptor through which insulin mediates the majority of its metabolic actions. All these factors suggest an important role for IGF-II in adult metabolism.
There is little clarity on the mechanisms behind IGF associations with lipid profiles. Animal studies in ageing rats have suggested that IGF-II administration is associated with favourable antioxidant and hepatoprotective effects. 22 IGF-II coencapsulation with rodent pancreatic islet cells has also been demonstrated to improve beta cell survival. 23 It is possible to speculate that IGF-II and IGFBP-2 have favourable effects on HDL cholesterol mediated through an increase in insulin sensitivity.
It is of significance that both insulin and IGF-II negatively modulate neuropeptide-Y (NPY) production in the paraventricular nucleus of the hypothalamus and thus are involved in regulation of appetite. 24 There is accepted to be an inverse relation between HDL cholesterol and body weight.25,26 As IGF-II interacts with the insulin receptors with considerable affinity, it is possible that IGF-II may also negatively regulate ApoA1 secretion in a manner similar to the action of insulin. Thus, there may be direct or indirect effects of IGF-II on HDL cholesterol metabolism.
IGFBP-1 is the principal acute determinant of IGF bioavailability and its concentration is primarily regulated by insulin. IGFBP-1 was associated with a long-term increase in HDL cholesterol concentrations in this study. Low IGFBP-1 has been associated with increased cardiovascular risk and has been associated with low HDL cholesterol in an earlier study. 27 IGFBP-1 is a recognized marker of insulin sensitivity,5,28,29 and the correlation of HDL cholesterol with insulin sensitivity 30 may explain the association of IGFBP-1 with HDL cholesterol.
IGFBP-2 has a preferential affinity to IGF-II, and low concentrations of this protein are a marker for the metabolic syndrome. A previous cross-sectional study has correlated IGFBP-2 concentration with low concentrations of serum triglycerides and LDL cholesterol. 31 In this study, higher baseline IGFBP-2 concentration was associated with high HDL concentrations at baseline and longitudinally as well as low baseline triglyceride concentration.
Our study has also identified associations of high IGF-II and IGFBP-3 with higher longitudinal serum triglyceride concentration trends. This is interesting because previous studies have reported inverse associations of IGFBP-3 with triglyceride concentration. 32 As IGFBP-3 generally serves to restrict IGF-I and IGF-II within the circulation, it is possible that the overall bioavailability of the IGF system is maintained if the circulating concentrations of IGF-II as well as IGFBP-3 are increased together.
However, in this study, there are important limitations in the analysis of results related to triglyceride associations with the IGF system, which may also be relevant to the other lipid measures analysed. Serum triglyceride concentrations are highly influenced by the nature of diet, exercise, glycaemic control and alcohol consumption. In this study, we did not have access to information regarding diet, exercise and the extent of alcohol consumption in the study population. While all patients had type 2 diabetes, they would have had varying degrees of insulin resistance, which is also correlated with triglyceride concentration. Results regarding longitudinal triglyceride trends may therefore not be reliable in some cases. We also did not have adequate information regarding the smoking status, which would have been an important predictor of cardiovascular risk in our study population.
We have determined that inter-individual variations in IGF-II concentrations could influence longitudinal trends in HDL concentration in a Caucasian population with type 2 diabetes, as could concentrations of IGFBP-1 and IGFBP-2. The primary importance of our findings at present would be in restating the importance of the IGF system in the determination of cardiovascular risk and attesting to a potentially vital biological role for IGF-II and IGFBP-2 in diabetes complications that merits clarification in future studies. Cardiovascular disease is a major cause of mortality in type 2 diabetes, and biomarkers that predict beneficial cardiovascular trends or improved macrovascular outcomes are likely to be of public health importance. The relation between IGF-II and established cardiovascular risk factors in this and other studies offers clues as to the factors that determine cardiovascular risk profile beyond the standard markers.
In summary, we report circulating IGF-II as a potential marker of longitudinal elevation of HDL cholesterol in a type 2 diabetes population. Future cardiovascular risk stratification strategies could include measurements of circulating IGF-II and these binding proteins, and research into these proteins may offer novel strategies for cardiovascular risk reduction in type 2 diabetes.
Footnotes
Acknowledgements
None.
Declaration of conflicting interests
No author has any competing interests.
Funding
The work was funded by the North-West Research and Development Fund.
Ethical approval
Ethical approval was granted by the Salford R&D Committee.
Guarantor
AHH.
Contributorship
RPN led on writing the paper and performed the data analysis; BF contributed to the statistical analyses; RLO, KWS and JEH carried out laboratory assays and contributed to the writing of the paper; AW, IL, RD, JMG and WERO are authorities in the fields of IGFs, metabolic risk and genetics and provided regular guidance and input to the paper; AHH co-ordinated the research team and is the corresponding author; JMG is an authority in the IGF field and is the overall lead for the project.
