Abstract
Background
Serum thyroglobulin (Tg) is useful for monitoring patients with differentiated thyroid cancer (DTC) but is limited by interference from anti-Tg antibodies (TgAb). We determined Tg assay discordance between a radioimmunoassay (RIA) and one of two immunometric assays (IMA) in DTC patients over a 9-year period to gauge assay performance against evidence of recurrent/progressive DTC.
Methods
Patients with DTC monitored for >1 year attending local clinics between September 2000 and January 2010 were included. All samples were analysed for Tg using both RIA and IMA. TgAb were measured on all Tg requests made after May 2006. Bias plots comparing RIA against IMA were established to calculate a 2-SD outlier limit. Clinical records were viewed to compare discordant Tg results against clinical evidence of recurrent/progressive DTC.
Results
Discordant Tg results were observed in 53/433 patients (12.2%). Four were discordant owing to a higher IMA result, one of which demonstrated recurrence. The remaining 49 patients demonstrated a disproportionately higher RIA result, of which four had recurrent/persistent disease. Twelve patients with a higher RIA result but no evidence of recurrence underwent thyrogen stimulation testing, which was negative in all 12. In many cases, assay discordance appeared more sensitive at indicating interference than direct measurement of TgAb.
Conclusions
Interference was evident with both Tg assays, such that neither could be solely relied upon to provide the correct result in the presence of TgAb. The concomitant measurement of Tg by RIA and IMA methods should be considered as an alternative to monitoring TgAb status.
Introduction
Thyroglobulin (Tg) is a large (660 kDa) dimeric protein expressed by follicular thyroid cells on which thyroid hormones are synthesized. Serum Tg measurement is useful clinically for the postoperative monitoring of patients diagnosed with differentiated thyroid cancer (DTC),1,2 with detectable and/or increasing Tg concentration indicating possible recurrent or persisting disease. Unfortunately, immunoassays for Tg can be subject to either negative or positive interference, primarily due to the presence of anti-thyroglobulin antibodies (TgAb) or heterophilic/human anti-murine antibodies (HAMA).3–8 Given that TgAb can be found in up to 25% of patients with thyroid cancer, 9 the potential for reporting aberrant Tg results is high and can give rise to under-treatment of active disease or over-investigation of individuals free of cancer. The inclusion of blocking agents such as animal protein within assay buffer has proven useful in limiting the potential impact of HAMA on Tg analysis. 10 However, the interference posed by TgAb has been a more difficult problem to overcome.
Current guidelines recommend the simultaneous measurement of TgAb alongside serum Tg to minimize the risk of misinterpretation in TgAb positive serum samples.1,2 While useful, recent evidence indicates that many TgAb assays currently in routine use frequently fail to detect circulating TgAb, even when assays are applied at their lowest operating analytical sensitivity, below manufacturer recommended cut-offs.11,12 Indeed, the classification of samples as TgAb-positive or TgAb-negative is dependent on the assay used, with some samples yielding differences in TgAb results of up to 100-fold across different assays.7,11
The poor analytical performance of TgAb assays has led to alternative strategies for testing for Tg assay interference. Tg recovery tests have been previously used for such purposes, but are now widely regarded as insensitive for detecting Tg-assay interference.9,13 Another alternative is to monitor discordance in Tg results when samples are analysed by different assay methods. Current Tg assays are based on either immunometric (IMA) or competitive radioimmunoassay (RIA) formats. TgAb are known to cause negative interference in IMA Tg assays,7,9,11 the most commonly used format in clinical practice. Conversely, while RIA methods are believed by some to be more robust, they may suffer from positive interference from TgAb and do not offer equivalent sensitivity as current IMA methods.7,9,14 New methods of measuring Tg such as tandem mass spectrometry offer an interesting means of combating existing problems of assay interference 15 ; however, such methods have yet to be widely clinically implemented.
Few studies have sought to correlate assay-specific Tg results against clinical evidence of recurrent DTC. Moreover, the high reported incidence of TgAb in patients with DTC (10–25%),9,16 combined with the current poor sensitivity in TgAb assays means that erroneous Tg results may pose a more significant problem in clinical practice than previously suspected.
In this study, we determined the incidence of discordant RIA/IMA Tg results in a group of DTC patients monitored long-term using both an automated IMA against a manual RIA method. We sought to compare the assay results against clinical evidence of recurrent DTC.
Methods
Study design and subjects
The study was performed at the Royal Infirmary of Edinburgh (RIE), UK. The study was a retrospective audit of patients attending local DTC clinics, with all tests performed (including dual Tg measurement) as part of the local standard of care. Results from all Tg and TgAb requests received at the Department of Clinical Biochemistry, RIE, between 1 April 2000 and 31 January 2010 were retrieved from the hospital laboratory computer system, iLAB (iSOFT, Banbury, UK), alongside results of standard thyroid function tests (thyrotropin (TSH), free thyroxine (FT4) and total tri-iodothyronine (TT3)). Requests on patients who had been made for reasons other than DTC were excluded from the study, as were patients with less than one year of follow-up post-diagnosis. Each request for Tg analysis received by the department was analysed by both RIA and IMA methods, with concurrent TgAb analysis also conducted on all samples taken after 31 May 2006. Tg results were then sorted on a Microsoft Excel© Spreadsheet and classified into either concordant or discordant groups as defined below. Clinical records of patients demonstrating discordant Tg results were then reviewed on a case-by-case basis on the hospital patient administration system (TrakCare™), examining Tg results against clinical evidence of recurrent or progressive disease.
Assays
All samples for Tg analysis were analysed by both RIA and IMA methods. The in-house RIA assay was performed at the Regional Endocrine Laboratory, University Hospitals of Birmingham, as described previously. 17 The assay utilizes a polyclonal rabbit anti-Tg antibody, calibrated against CRM 457. The lower reporting limit of the RIA assay was 5.0 µg/L.
For the IMA analysis, samples were analysed by one of two methods. For all samples taken prior to 31 May 2006, Tg analysis was performed by immunoradiometric assay (IRMA) using a test kit manufactured by Sanofi Diagnostics Pasteur (supplied by BioRad Laboratories, Harts, UK), described previously. 18 The lower reporting limit of the IRMA assay was 1.0 µg/L. IRMA analyses were performed at the Freeman Hospital, Newcastle upon Tyne, UK. All samples taken after 31 May 2006 were analysed at the RIE laboratory by immunochemiluminetric assay (ICMA) using an Immulite 2000 analytical system (Siemens, Camberley, UK) according to the manufacturer’s protocol. The reported inter-assay CVs were 5.6% and 7.2% at a concentration of 10 and 279 µg/L Tg, respectively. The laboratory lower reporting limit of the ICMA assay was 2.0 µg/L.
TgAb were analysed at RIE on a Siemens Immulite 2000 using a chemiluminescent immunometric assay. The reported inter-assay precision of the TgAb assay was 4.6% and 5.7%, at a TgAb concentration of 63 IU/L and 1644 IU/L, respectively. The lower reporting limit of the TgAb assay was 30 IU/L, derived from in-house precision data (not shown).
Standard laboratory thyroid function tests (TSH, FT4 and TT3) were analysed using an automated Abbott Architect analyser (Abbott Laboratories, Maidenhead, UK).
Assignation of discordant results
All samples that generated detectable Tg results by both RIA and IMA methods were gathered into a Microsoft Excel© spreadsheet and Bland–Altman plots generated to evaluate assay bias difference (ICMA + 43% bias versus RIA; IRMA: +26% bias versus RIA; Figure 1). All results were gathered irrespective of TgAb status. The bias difference was not observed to be concentration dependent (Figure 1). The 95% (2 SD) confidence interval was determined for the ratio of each IMA against RIA and subsequently used to classify Tg results as either concordant or discordant.
Bland–Altman plots showing comparative Tg results obtained from the RIA method against the ICMA (a) and IRMA (b) methods. Results that were undetectable by either (or both) methods were not plotted. (a) The ICMA assay had an overall positive bias of +43% against the RIA method (excluded values n = 1198). (b) The IRMA assay had an overall positive bias of +26% (excluded values n = 1100).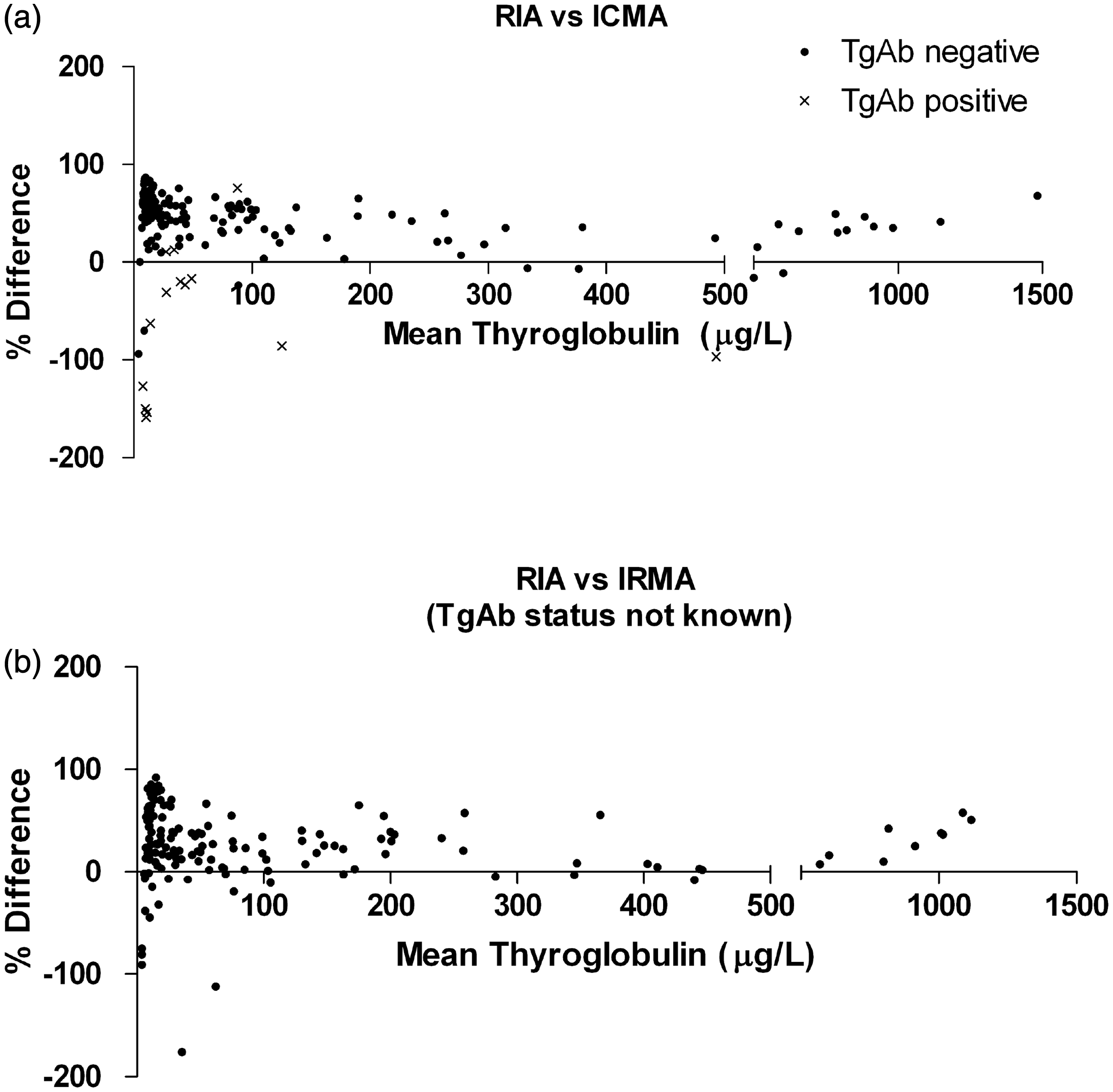
Statistical and graphical analyses
All data/results were compiled and sorted onto Microsoft Excel© spreadsheets. Graphs were plotted using GraphPad Prism Software (version 5.0). Patients monitored by both the IMA methods (IRMA/ICMA) were plotted as a continuum on each graph.
Results
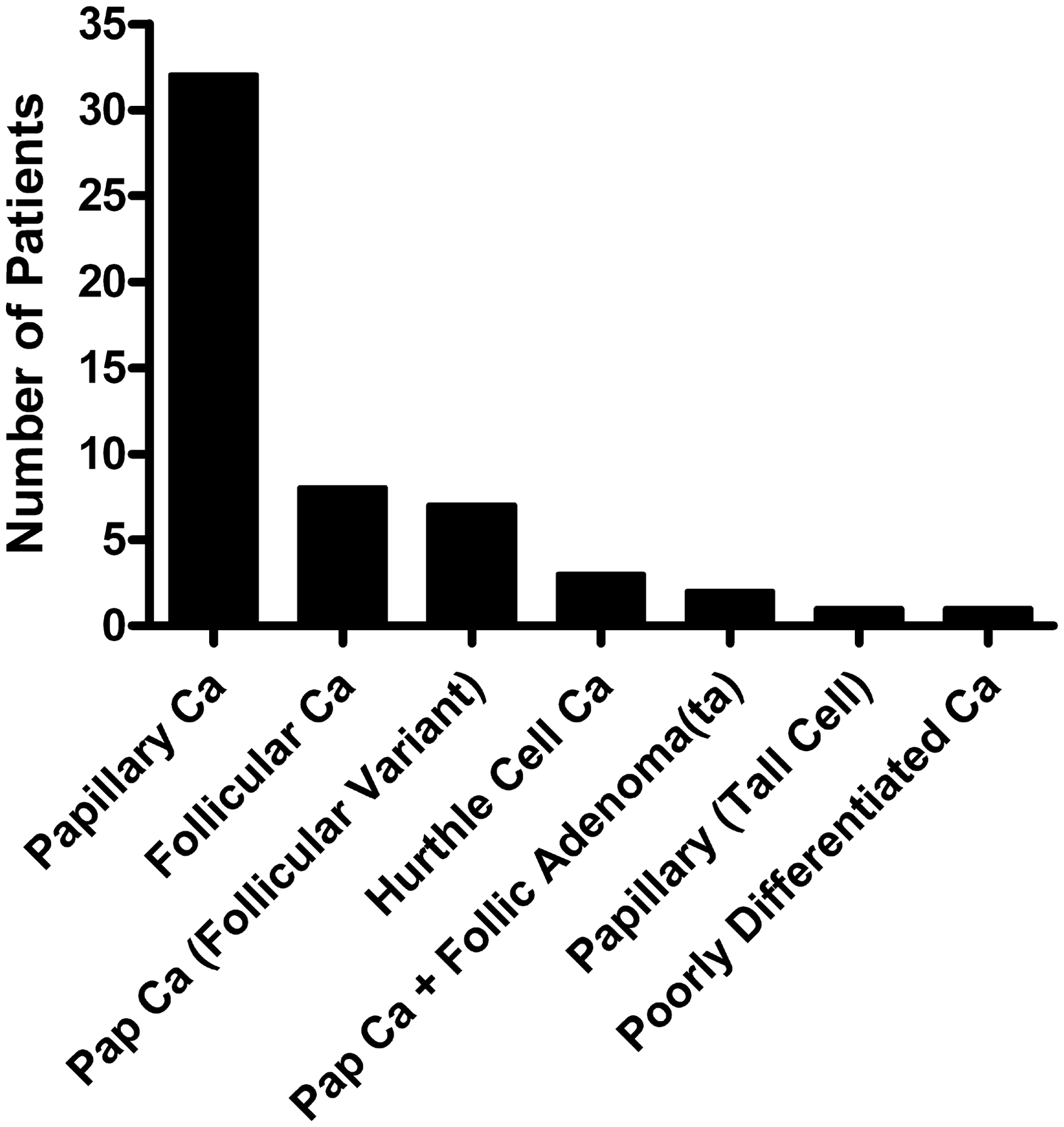
Over the period of the study, a total of 576 patients had serum Tg measured, of which 143 were excluded from the study because of less than one year of follow-up data, or no evidence of DTC as a diagnosis (Figure 2). Of the remaining 433 patients, 380 had Tg results that were concordant on both IMA and RIA methods. Conversely, 53 patients (12.2%) were identified as having discordant Tg results. The distribution of thyroid malignancy type in the patients demonstrating discordant Tg results is shown in Figure 3.
Clinical outcome of patients with discordant Tg results attending RIE between April 2000 and 31 January 2010. Distribution of malignancy type among patients demonstrating discordant Tg results.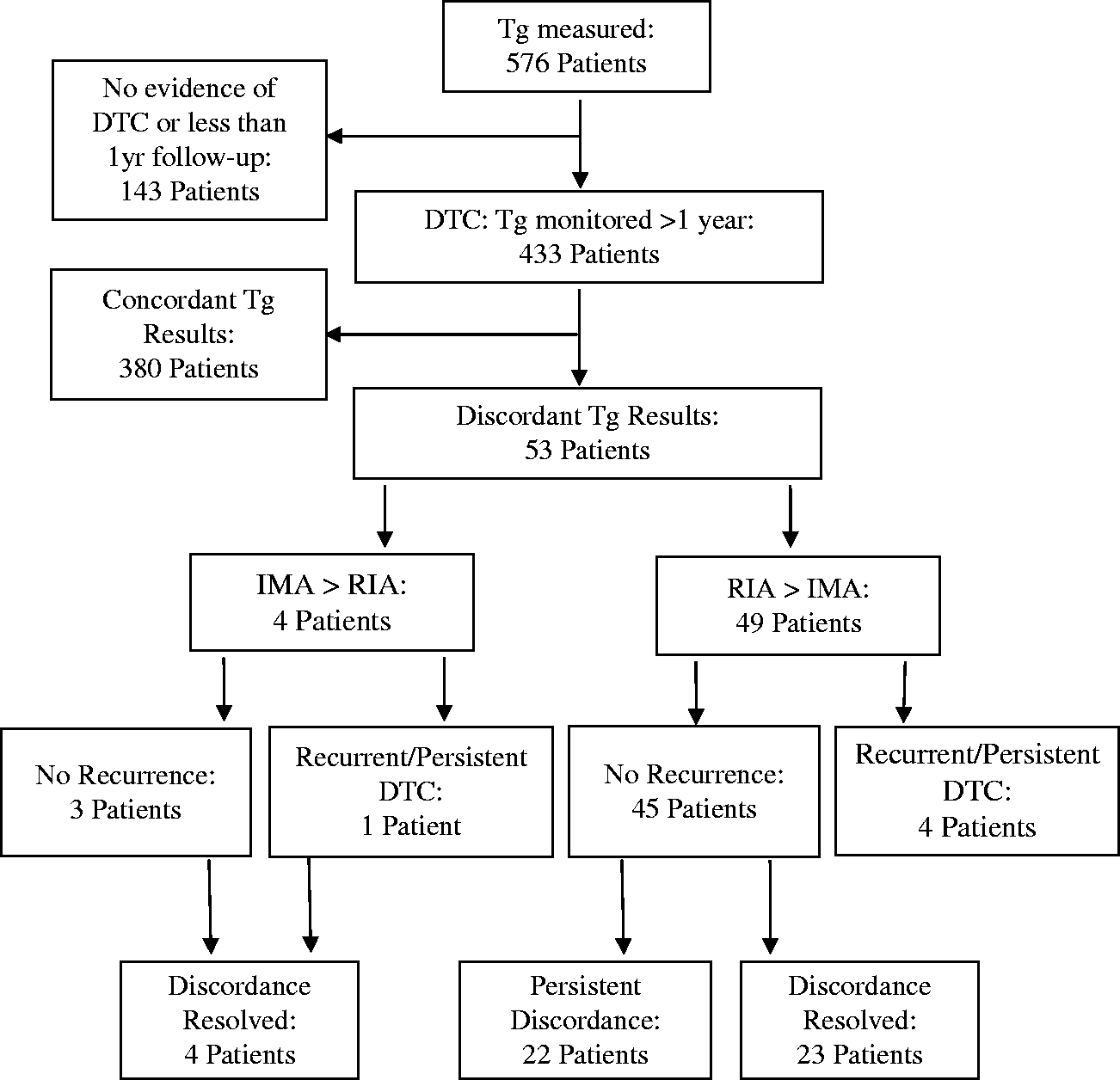
Discordant Tg results: IMA:RIA ratio greater than expected
Clinical data on patients demonstrating discordant (IMA > RIA) Tg results with negative TgAb.
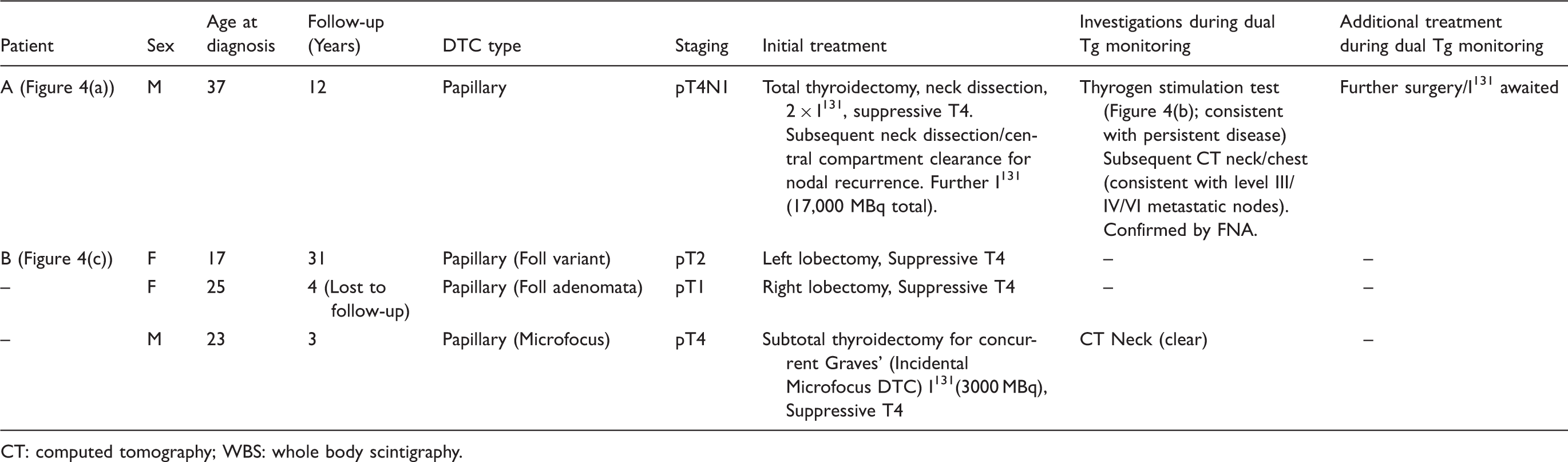
CT: computed tomography; WBS: whole body scintigraphy.
Patient A (Table 1; Figure 4(a)) underwent total thyroidectomy with neck dissection for papillary DTC. The patient had demonstrated recurrence prior to the study period that was previously treated by further neck dissection/central compartment clearance and I131 ablation. During the study period, the patient demonstrated further recurrence 13 years post-thyroidectomy following new onset lymphadenopathy, confirmed by imaging (CT Neck) indicating the presence of level III/IV and VI nodes, and fine needle aspirate cytology (FNA). The patient had previously demonstrated a positive thyrogen stimulation test (Figure 4(b)), consistent with persistent disease. On Tg monitoring, two points were outside the discordance criteria, with the pattern of Tg results on the different assays following a similar trend (Figure 4(a)).
Discordant Tg results due to a disproportionately higher Tg IMA result (IMA > RIA). Asterisks indicate points of discordance; downward arrows indicate results that were below the limit of detection (LOD). Patient A (Figure 4(a) and (b)) had recurrence. Patient B (Figure 4(c)) underwent hemithyroidectomy only, but both discordant Tg results were associated with a higher baseline TSH (TSH 0.08 and 0.32 mU/L, respectively; subsequently < 0.01 mU/L).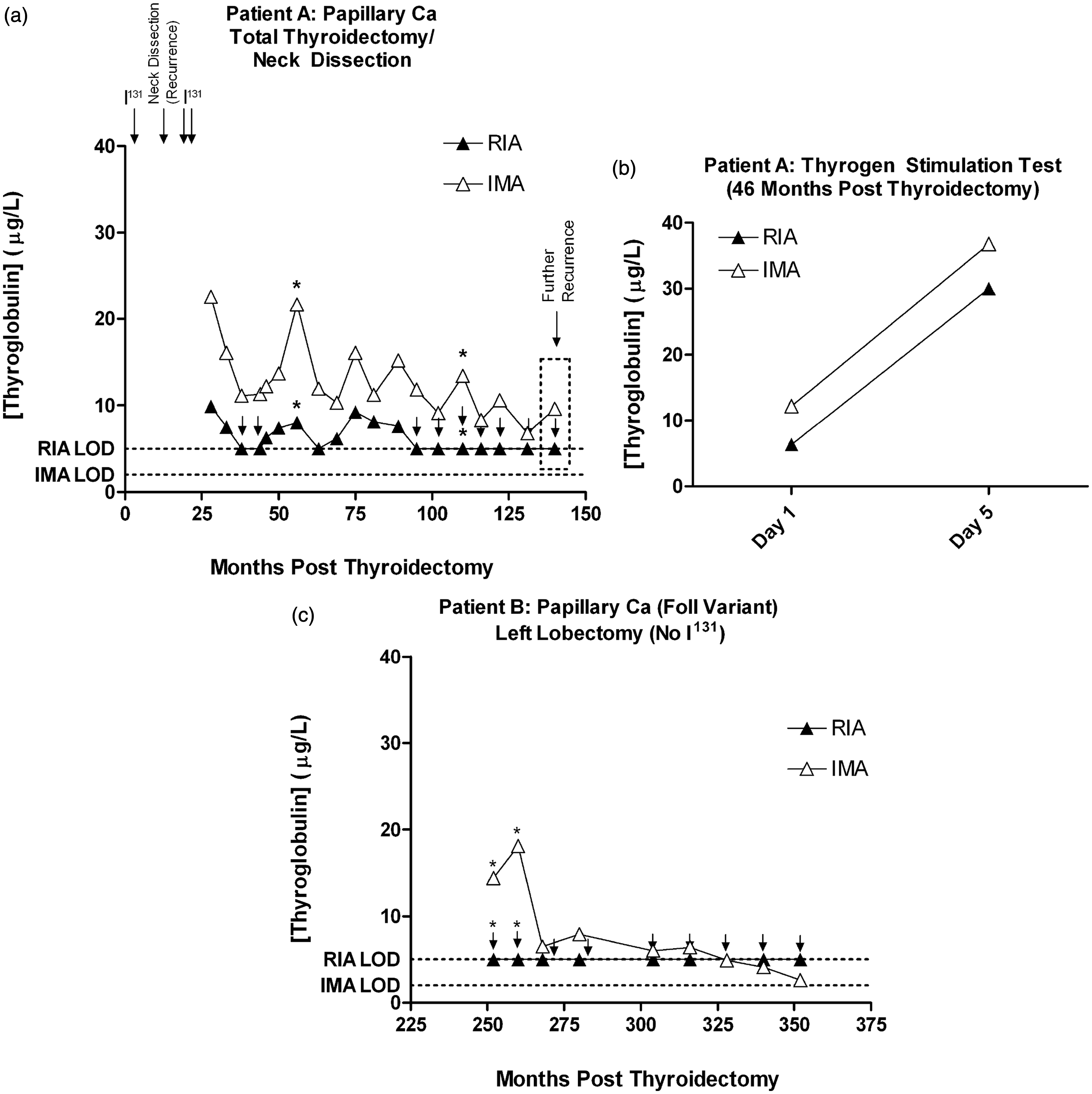
Of the remaining three patients with discordant Tg (IMA > RIA) results, one demonstrated a more pronounced difference between IMA and RIA methods on two data points (Patient B, Figure 4(c)). Patient B had undergone subtotal thyroidectomy, and the higher IMA results correlated with correspondingly higher TSH values. The remaining two patients underwent subtotal thyroidectomy, and demonstrated discordance on one data point only, with results that were only marginally out-with the discordance criteria (data not shown).
Discordant Tg results: RIA:IMA ratio greater than expected
A total of 49 patients were found to have discordance due to a disproportionately high RIA result. Four of these 49 patients were determined to have clinically recurrent and/or persisting disease over the study period, while the remaining 45 demonstrated no evidence of tumour recurrence.
Discordant Tg results (RIA > IMA) with persistent/recurrent DTC
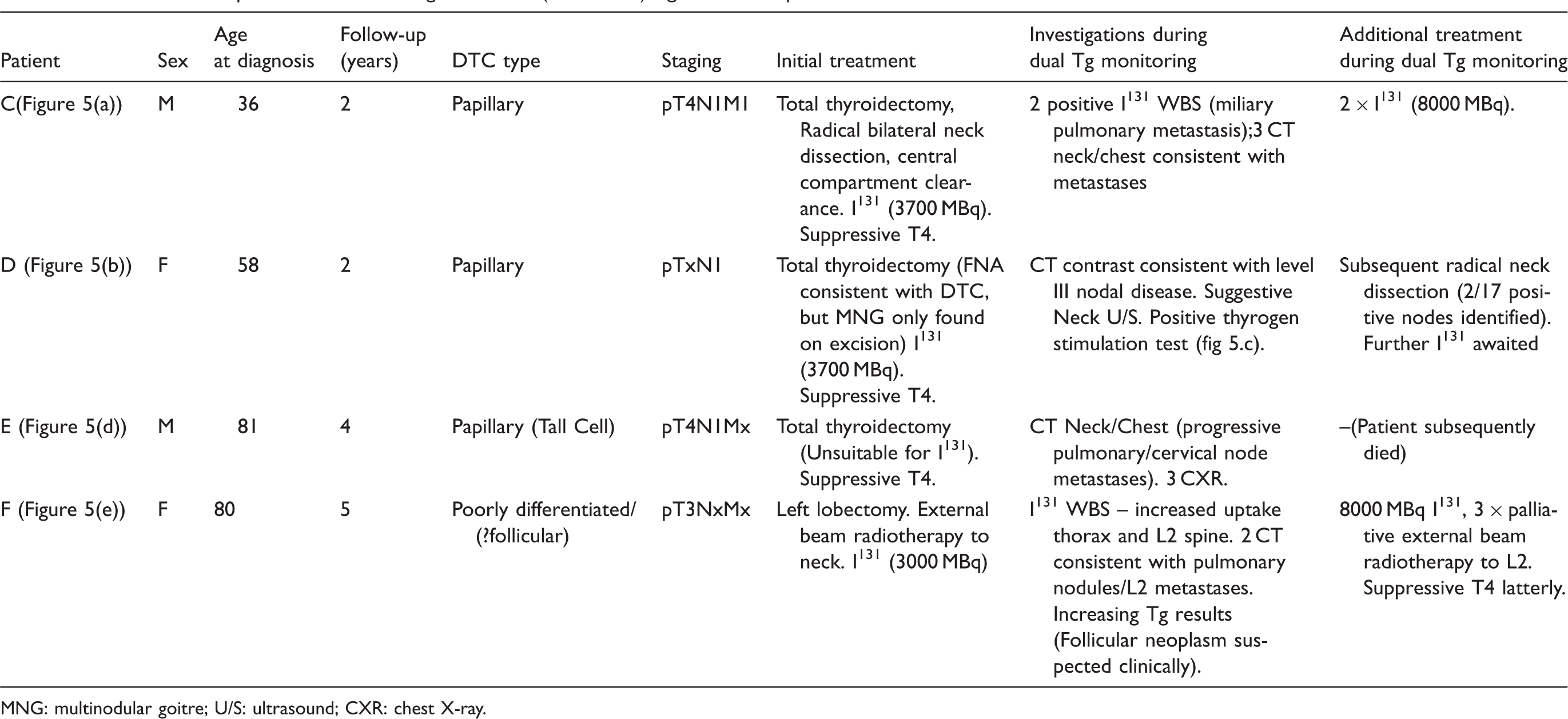
Table 2 summarizes the investigations and treatment of the four patients with discordant Tg results and evidence of recurrent/persistent DTC. Graphs demonstrating corresponding Tg results for each patient are shown in Figure 5. TgAb were detected in all four patients.
Tg monitoring in patients with discordant Tg results (RIA > IMA) and evidence of persistent or recurrent DTC. (a) Patient C. Although RIA/ICMA results are both detectable, Tg results are discordant given the inverted bias of RIA versus IMA. (b) Patient D underwent total thyroidectomy and radioiodine ablation, while 15 months later, a positive thyrogen stimulation test was demonstrated, (c) following suggestive neck U/S. Radical neck dissection was subsequently undertaken. (d) Patient E underwent total thyroidectomy, but was not suitable for I131 therapy. Radiologic imaging (CXR) was consistent with progressing metastatic DTC. Unfortunately, the patient died following a stroke. TgAb was only measured on the final data point (strongly positive). (e) Patient F was initially diagnosed with poorly differentiated thyroid cancer and underwent left lobectomy and cervical external beam radiotherapy. Tg discordance only manifest on the final data point following a marked increase in TgAb titre. Clinical data on patients demonstrating discordant (RIA > IMA) Tg results and persistent/recurrent DTC. MNG: multinodular goitre; U/S: ultrasound; CXR: chest X-ray.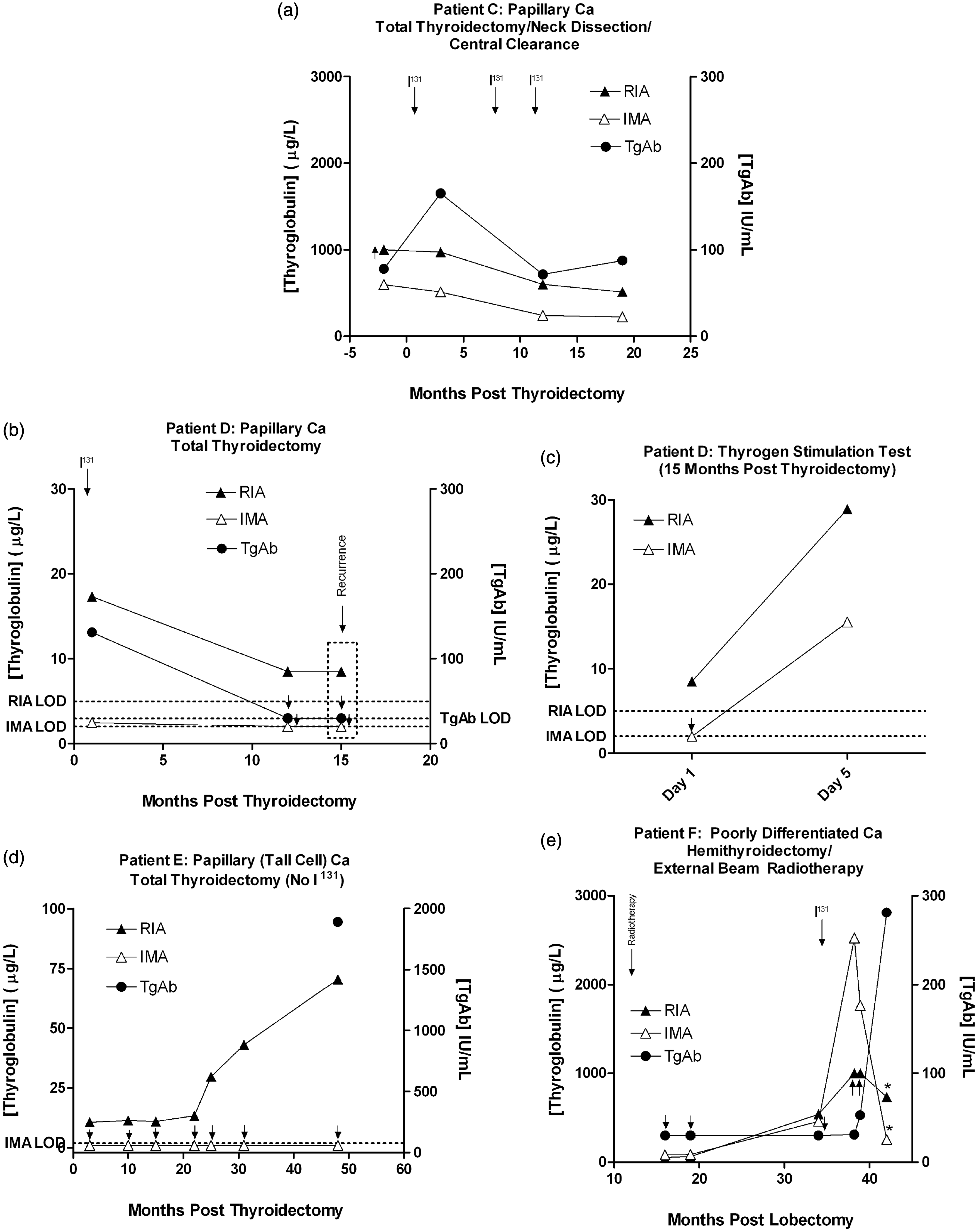
Patient C had persisting metastatic papillary cancer, with a clearly inverted Tg assay bias (Figure 5(a)). However, the profile of Tg results was consistent across both Tg assays, with decreasing Tg concentration presumably reflecting reduced tumour burden with treatment.
Patient D initially underwent total thyroidectomy following FNA of a multinodular goitre that indicated papillary DTC. However, on excision no obvious primary was located by histological examination. Following I131 therapy, some 15 months later the patient developed lymphadenopathy confirmed as likely recurrence by imaging (contrast CT; left level III node) and positive thyrogen stimulation (Figure 5(b) and (c)). Recurrence occurred at a point when both TgAb and Tg IMA were undetectable, but Tg RIA was detectable (Figure 5(b)). Subsequent neck dissection confirmed the presence of papillary DTC in 2/17 nodes.
Patient E underwent total thyroidectomy for Tall Cell carcinoma, although was not suitable for I131 ablation. This patient had clearly discordant Tg results (Figure 5(d)), with increasing RIA Tg results corresponding with radiological evidence of progressive metastatic papillary DTC. Corresponding Tg IMA results were persistently undetectable, consistent with interference in this assay. Although only measured on the final data point, TgAb were strongly positive. The patient subsequently died following a stroke.
Patient F initially underwent lobectomy with external beam radiotherapy for poorly differentiated thyroid carcinoma. During follow-up, Tg measured by both IMA and RIA methods demonstrated an increase consistent with a progressive neoplasm, with Tg assay discordance in this patient only becoming evident on the final time-point following a marked increase in TgAb titre (Figure 5(e)).
Discordant Tg results (RIA > IMA); no evidence of recurrence
Forty-five patients demonstrated a discordantly high RIA Tg result without demonstrating any evidence of tumour recurrence or progression. Twelve patients in this group underwent a thyrogen stimulation test, which was negative in all 12, indicating interference in the RIA Tg assay. Importantly, TgAb were detected in only 20 of the 45 patients with discordant (RIA > IMA) Tg results. However, in 14 of the remaining patients, TgAb were not measured at the time that assay discordance was apparent. Thus, a total of 11 patients were observed to have discordant Tg results (RIA > IMA) with no evidence of detectable TgAb.
Of the 45 patients with discordant Tg results and no evidence of recurrence, in 23 individuals (52%) the Tg assay discordance resolved over time, while in the remaining 22 patients the discordance persisted throughout the study period. The mean time taken for discordance to resolve in patients who did was 5.5 years post-thyroidectomy (range 1–19 years). However, discordant results appeared to resolve more quickly in patients who had undergone total thyroidectomy (mean 3.7 years, range 1–14, n = 19) compared to those who underwent subtotal thyroidectomy (mean 13.5 years, range 8–19, n = 4). One patient in whom the discordance resolved is shown in Figure 6(a).
Tg monitoring in a patient demonstrating discordant Tg results (RIA > IMA) that subsequently either resolved (a), or persisted (b–d).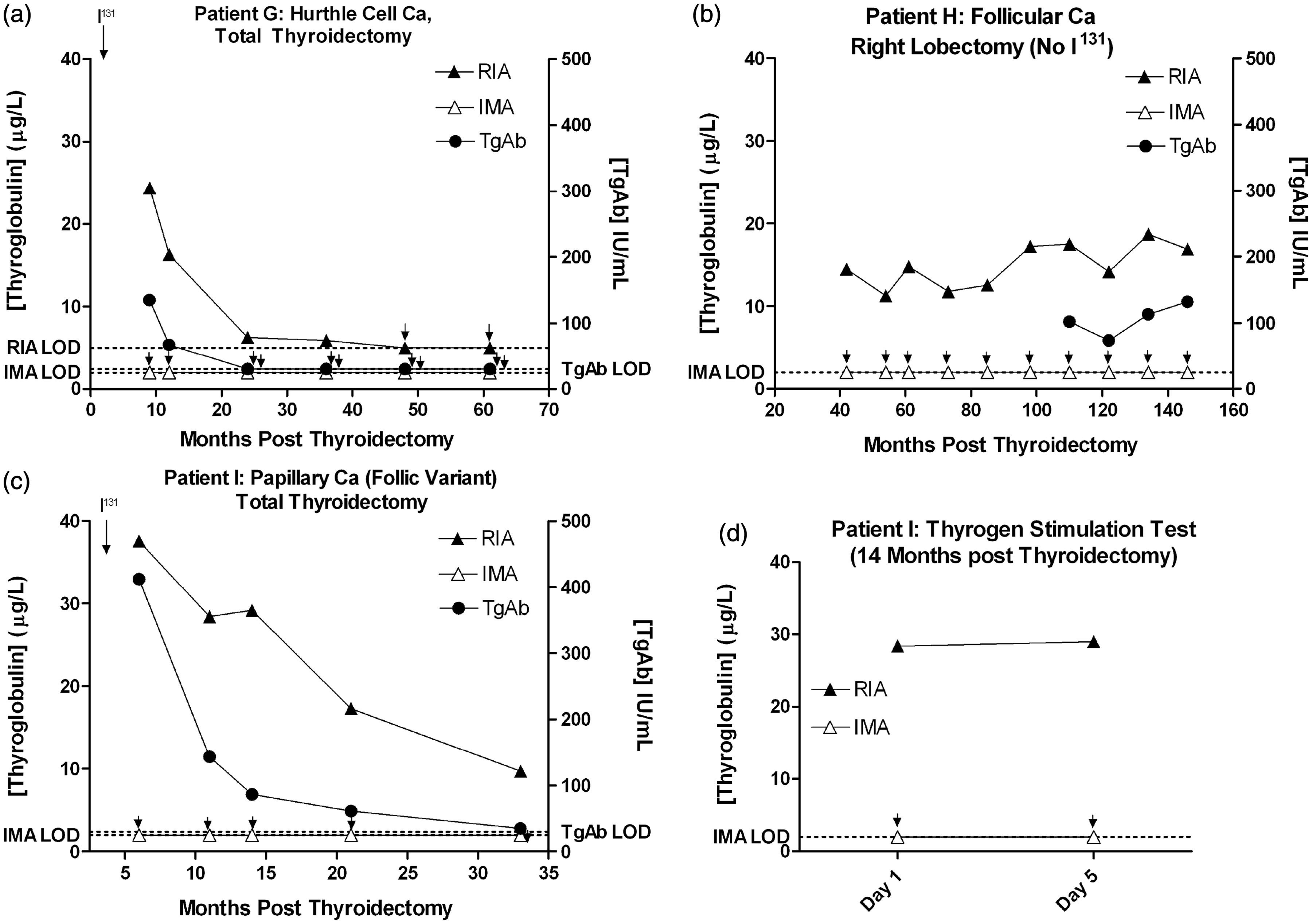
Of the 22 patients in which the Tg discordance persisted, 13 had undergone total thyroidectomy, while the remaining nine underwent subtotal thyroidectomy. These 22 patients were followed up for an average of 11 years post thyroidectomy (range 2–55 years). Graphs demonstrating two patients with persistent discordance are shown in Figure 6(b–d).
TgAb and concordant Tg results
Of the group of 433 patients who were included in the study, only three patients were found to be TgAb positive without demonstrating evidence of Tg assay discordance. No definitive histological evidence of tumour recurrence was reported in any of the cases, although equivocal FNA of a neck node with positive thyrogen stimulation was obtained in one. Irrespectively, in all cases TgAb titre subsequently declined to undetectable concentrations.
Discussion
The primary aim of this study was to determine the prevalence of discordant Tg results in a cohort of DTC patients attending the Royal Infirmary of Edinburgh over a 9-year period, examining clinical evidence of recurrence against the most commonly used RIA and IMA Tg assays employed in the UK. This was important against the context of observations that RIA methods may be more resistant to TgAb interference than IMA methods.7,9
The criteria used to define discordance in this study employed the use of 95% confidence interval. Although it is recognized that this method ensures that a defined proportion of patients will be classified as having discordant Tg results, the use of statistical means was important given that it represents the only objective way of identifying all potentially disparate results. In the absence of significant assay interference, the proportion of results expected to be classified as discordant in the study is expected to be less than 5%, given that the vast majority of samples included had undetectable Tg by both methods.
Results presented here indicate that discordance between Tg assay results affected 12.2% of patients undergoing Tg monitoring for DTC. Importantly, the presence of disparate Tg results did not appear to associate with a particular malignancy type (Figure 3), with papillary and follicular malignancy occurring with a similar frequency as expected in the wider population.
The most common orientation of discordant Tg results was a disproportionately high RIA result, occurring in 11.3% of patients undergoing Tg monitoring for DTC (and >90% of cases of patients with discordant Tg results). This figure is broadly within the range reported for the prevalence of TgAb in DTC patients (10–25%),9,16 the most likely source of this direction of Tg assay discordance. The propensity for this type of discordance was expected in view of the known interference of TgAb on Tg assays, causing falsely low Tg IMA results and/or potentially high RIA Tg results.6,7,13 Importantly, owing to a change in Tg IMA method during this study (from Sanofi to Immulite), some patients were monitored using two separate Tg IMA methods. However, criteria for discordant Tg results were determined separately for both Tg IMA assays. Moreover, we found no instances where the change in Tg IMA method affected the trend in Tg IMA result obtained, or prompted a change from a discordant to concordant category when compared to the RIA Tg result.
Only in a very small number of patients (4/434; 0.9% of total) did the IMA method generate a Tg result that was disproportionately elevated above the RIA-derived value. Importantly, in each specific case the discordance resolved in later samples. Of the four patients demonstrating this type of discordance, recurrence occurred only in one (patient A, Figure 4). However, the assay differences obtained on this patient were of minor significance given that the profile of Tg results on this patient were broadly similar, allowing for the clear positive bias of Tg IMA method. Similarly, in at least two of the other patients with discordant Tg results (IMA > RIA), the difference between the two Tg assays was only marginally out-with the 2 SD limit, implying that discordance in these patients may have been due to analytical imprecision at the lower limit of assay sensitivity.
In the remaining patient, the difference between the two assays was more prominent (Figure 4(c)), notably in which the IMA results correlated with TSH values in a patient who had undergone hemithyroidectomy only. Although the mechanism of discordance in these samples is unclear, it may reflect variants in Tg epitope that are preferentially detected by the IMA method above the RIA in these patients. This would be in keeping with the known heterogeneity of Tg expressed from different patients with DTC.19,20 Alternatively, the potential for HAMA interference in the RIA assay would have been useful to exclude at the time.
Of the 49 patients demonstrating discordance due to a higher RIA result, persistent and/or recurrent DTC was detected in only four patients. In two patients (patients C and F, Figure 5), the Tg assay discordance did not cause overt interpretative difficulty, but simply resulted in inversion of the expected bias between the two assays. Nevertheless, such RIA/IMA Tg assay differences may affect patient follow-up as Tg IMA values decrease to below the detectable range(s). In the remaining two patients, the difference between the Tg assays was more significant. In patient D (Figure 5(b) and (c)), detectable RIA Tg alongside suggestive neck ultrasound prompted further investigation with a thyrogen stimulation test, which demonstrated an increase in Tg measured using both assays. This finding suggests that thyrogen stimulation was sufficient to overcome residual TgAb-mediated suppression of the Tg IMA result in this patient. In patient E, there was clear radiological evidence (CXR and CT) of progressive metastatic DTC, despite continually suppressed Tg IMA results (Figure 5(d)). Although Tg RIA was clearly consistent with clinical findings, TgAb were only measured on the final sample and were strongly positive. Thus, increasing Tg RIA values may reflect either increased circulating Tg concentration and/or increasing positive interference with higher TgAb titres associated with progressive tumour growth.
Of the 45 patients who demonstrated Tg assay discordance (RIA > IMA) without evidence of recurrence, in more than half (23 patients) the discordance subsequently resolved. We anticipate that if followed up for a longer time period this number would likely increase. Unsurprisingly, discordance persisted for longer post-thyroidectomy in patients who had undergone subtotal thyroidectomy as compared to those who had undergone total thyroidectomy, presumably reflecting long-term persistence of Tg antigen and thus TgAb. Interestingly, in patients who had undergone total thyroidectomy, the mean time for the RIA and IMA results to resolve was 3.7 years, a period far exceeding any expected biological effects of I131 (radio-decay t½ ≈ 8 days) and Tg clearance from the blood (approx. 30 h). 21 Previous reports monitoring DTC patients have estimated the median time taken for TgAb to disappear post-thyroidectomy and ablative radioiodine at approximately 3 years.22,23 Observations here that the RIA Tg result paralleled TgAb concentrations in patients with no evidence of recurrence (Figure 6(a)), including those shown subsequently to have a negative thyrogen stimulation test (Figure 6(c) and (d)), strongly supports the hypothesis that the RIA assay is demonstrating positive interference. Thus, in patients with Tg assay discordance (RIA > IMA), or a demonstrable concentration of TgAb, the RIA Tg result appears to reflect at least a component of interference from circulating TgAb, and not Tg antigen.
Results presented here are informative towards devising a testing strategy for Tg monitoring in DTC patients. Current UK and American guidelines indicate that measurement of TgAb should be made alongside all Tg measurements,1,2 with most clinical laboratories presently operating automated Tg-IMA methods. In this study, although TgAb were only measured in samples collected after May 2006, we identified 11 patients with discordant (RIA > IMA) Tg results that were TgAb negative (TgAb < 30 IU/L), despite the use of a TgAb cut-off below the manufacturer-recommended positive cut-off (≥40 IU/L). In addition, we observed a number of cases where TgAb was detected, but the discordance between the RIA and IMA results persisted for longer than the presence of detectable TgAb. This was particularly notable in one patient who had recurrent DTC, where both the Tg IMA and TgAb results declined to undetectable concentrations despite recurrent nodal disease (Figure 5(b)). Although previous evidence indicates that tumour recurrence is rare in patients with detectable TgAb who subsequently become TgAb-negative (<1%), 24 these results suggest that assay discordance itself may be a more sensitive marker of TgAb presence than the measurement of TgAb itself. Interestingly, recent reports have demonstrated that the Siemens Immulite TgAb method used here may be less reliable than some other commercially available methods at detecting low titres of TgAb,11,12 raising the possibility that alternative TgAb assays may have produced greater agreement with discordant Tg results than observed in this study. The Immulite TgAb method is currently the most commonly used TgAb assay in UK Laboratories (UK NEQAS data), and in such cases dual Tg measurement with RIA and IMA methods may have the potential to identify recurrence earlier than patients monitored solely with Tg IMA and TgAb. However, we recognize that for the broader application of these findings it would be useful to compare alternative RIA assays with different IMA and TgAb methods to determine if the findings observed here are consistent across the different assay classes. We also acknowledge the potential fragility of a diagnostic service provided from a single UK provider. In any case, DTC monitoring using the RIA Tg alone has the potential for a significant false positive error rate.
Importantly, three TgAb-positive patients were identified who generated concordant Tg RIA/IMA results. In all three patients TgAb titre was relatively low (<50 IU/L; data not shown), and titres subsequently declined to an undetectable concentration without implying the presence of recurrent disease. Although unclear, these results may have been due to TgAb being directed to a different epitope on Tg, such that it prevented interference with either the RIA or the IMA Tg assays. Such a finding would be consistent with the known variation in TgAb epitope specificity that can be exhibited by different patients with DTC. 25
In summary, results from this study demonstrate that using the current commonly used RIA and IMA assay formats in the UK, clear instances of interference were demonstrable with both Tg assays, such that neither assay could be relied upon to generate the correct result. In a number of cases, the measurement of Tg assay discordance appeared to be a more sensitive marker of assay interference than the direct measurement of TgAb. The analysis of Tg by both RIA and IMA methods should be considered as an alternative strategy to Tg IMA/TgAb measurement for monitoring patients with DTC.
Footnotes
Declaration of conflicting interests
None.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
Not applicable.
Guarantor
MSC.
Contributorship
GB conceived the study, MSC analysed the data and wrote the first draft, ADT and MWJS reviewed the patients, aided in study design and provided clinical input. All authors reviewed and edited the manuscript and approved the final version.
