Abstract
Background
Cystic fibrosis (CF) is an autosomal recessive condition that has an incidence of 1:2500 live births in Northern Europe. Due to the large number of mutations that can result in classical or atypical CF phenotype, the sweat test, which quantifies the amount of chloride and sodium in sweat, is vital in supporting the diagnosis of CF. Patients with CF have raised concentrations of chloride and sodium in their sweat; however, it is the concentration of chloride in sweat which provides the greatest diagnostic sensitivity for CF.
Method
An inductively coupled plasma mass spectrometry (ICP-MS) method for the analysis of sweat chloride and sodium was evaluated for the routine measurement of sweat collected using the Wescor Macroduct® Sweat Collection System. The precision, linearity and agreement with the all laboratories trimmed means (ALTMs) and ‘weighed-in’ concentrations of sodium and chloride in samples supplied by the UK NEQAS external quality assessment (EQA) Sweat Testing Scheme were assessed.
Results
This ICP-MS method for the quantification of chloride and sodium in sweat samples was shown to be accurate, precise and suitable for the routine analysis of sweat chloride and sodium.
Conclusion
The method performs well and is now used in the routine analysis of sweat in this laboratory.
Introduction
The analysis of sweat forms part of the diagnosis of cystic fibrosis (CF). Typical CF mutations in the cystic fibrosis transmembrane conductance regulator (CFTR) result in an abnormally high concentration of chloride in the sweat of affected patients.
Examination of UK NEQAS EQA Sweat Testing data shows that there are currently four methods being employed for the analysis of sweat, with over 50% of the 175 UK NEQAS EQA Sweat Testing Scheme subscribers currently using coulometry to quantify sweat chloride. 1 The use of sweat sodium analysis in the diagnosis of CF is not recommended as sodium concentrations do not discriminate well between healthy and CF populations; 2 however, many laboratories continue to analyse sodium as very high concentrations may indicate sample contamination. In December 2011, 79 users reported sweat sodium results to UK NEQAS, whereas 179 users reported chloride. There are essentially two methods used for the collection of sweat, the most popular being the Wescor Macroduct® Sweat Collection System (Wescor, Inc., South Logan, UT, USA). Less than 15% of UK NEQAS participants use the original Gibson and Cooke 3 method to collect sweat.
This laboratory has traditionally used the Gibson and Cooke 3 method of sweat collection. This required an extraction step to remove sweat from the filter paper. Sweat chloride analysis was carried out using a coulometric method and sweat sodium was analysed using a flame photometer. The instrumentation used to perform this analysis had become dated and the laboratory wished to use a method that was more amenable to the analysis of sweat collected using the Wescor Macroduct® Sweat Collection System, and which ideally did not require a preanalytical sample extraction procedure. The method employed for the measurement of sweat chloride and sodium concentrations must be suitable for use with very small sample volumes; the minimum volume of sweat that must be collected over a 30 min collection period using the Wescor Macroduct® is 18 μL. Sample volumes of less than 18 μL indicate that the sweat production rate has been inadequate (<1 g/m2/min) and these samples are therefore not suitable for analysis. 2
A robust method for the analysis of sweat using ICP-MS was developed.
Method and results
An Agilent 7700x ICP-MS (Aglient Technologies UK Limited, Stockport, UK) was used. 23Na and 35Cl were selected for monitoring, with 45Sc monitored as the internal standard. Due to the relative ease of sodium ionization in relation to chlorine, 23Na was monitored with the collision cell mode on, with the aim of reducing the 23Na signal reaching the MS detector. With the collision cell mode on, the Agilent 7700x ICP-MS system uses an octopole reaction system with helium as the collision gas; this was developed by Agilent to aid the removal of matrix and polyatomic interferences. However, in this mode the analyte of interest is diluted in the collision gas; analysis of 23Na in this mode effectively dilutes the 23Na signal and prevents MS detector damage. The routine set-up of the analyser is quick (approximately 5 min) and simple; it involves checking that the argon and helium gas supplies and rinse solution (5% nitric acid) are sufficient before igniting the plasma and checking the tune parameters. With a typical work load of two patient samples per week, usually collected on the same day, there is adequate capacity on the analyser to allow sweat analysis to be run on the same day as collection. Sweat collected using the Wescor Macroduct® may be stored with the capillary ends sealed for up to 72 h in case of any analyser down time. 2 Samples, NaCl standards and commercially available QC material (Quantimetrix 3 level sweat QCs, supplied by Alere Limited, Stirling, UK) were prepared in singleton by being diluted 1 in 101 (15 μL into 1.5 mL) in 1% (v/v) nitric acid containing 10 μg/L internal standard mix (Agilent ICP-MS Internal Standard Mix part #5188-6525) prior to analysis. A total volume of 1515 μL is adequate for duplicate analysis of QC and patient samples. A 5% (v/v) nitric acid solution was used as a system rinse between samples. A set of three standards was prepared in-house by diluting Fluka (Sigma-Aldrich – Gillingham, UK) ICP-MS grade solutions of sodium (434.1 mmol/L) and chloride (282.0 mmol/L) in 1% (v/v) nitic acid. Standards 1, 2 and 3 contain sodium and chloride at the following concentrations: 1, Na 20.00 mmol/L and Cl 19.74 mmol/L; 2, Na 66.77 mmol/L and Cl 56.40 mmol/L; 3, Na 95.50 mmol/L and Cl 101.52 mmol/L. A series of three ‘blanks’ was run at the beginning of each batch, these comprised 1% (v/v) nitric acid containing 10 μg/L internal standard mix. The third ‘blank’ at the beginning of each run was designated as the zero calibrator. Standards 1, 2 and 3 then followed to produce a calibration curve for each batch. A ‘blank’ was analysed following both standard 3 and the high level QC sample as well as between samples to prevent the possibility of carry over. QC samples were run at the beginning and end of each batch. Including wash stages, each standard, QC or patient sample takes approximately three minutes to analyse.
For method evaluation studies statistical analyses (Student's paired t-tests) were performed using Microsoft Excel with the aid of ACB Method Verification Spreadsheets. 4 Unpaired t-tests were preformed using Microsoft Excel when comparing sweat sodium and chloride concentrations obtained in patient samples pre- and post-ICP-MS implementation.
Interbatch CVs for chloride and sodium measurement using QC material and ICP-MS analysis
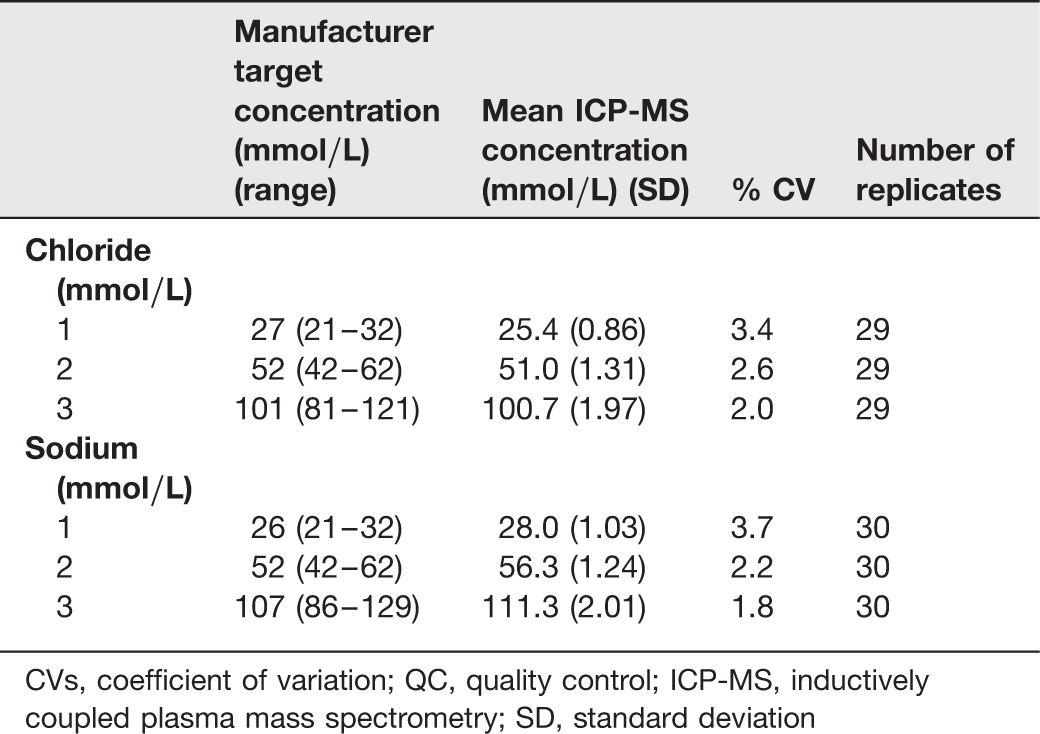
CVs, coefficient of variation; QC, quality control; ICP-MS, inductively coupled plasma mass spectrometry; SD, standard deviation
Samples from UKNEQAS EQA Sweat Testing distributions were used to compare this method with others in use (Figure 1). The ICP-MS chloride method had a mean bias of +0.25 (SD = 1.20) mmol/L in comparison to the all laboratory trimmed mean (ALTM). Mean bias against the weighed-in chloride concentration in the EQA samples was −0.32 (SD = 1.20) mmol/L. However, these biases were not significantly different (paired student's t-test, P = 0.190 and P = 0.139 respectively). The ICP-MS sodium method mean bias was +0.80 (SD = 1.53) mmol/L when compared with the UKNEQAS ALTM and +0.47 (SD = 1.68) mmol/L when compared with the UKNEQAS weighed-in sodium concentrations. Although a small and significant bias (P = 0.003) was seen between the ICP-MS and ALTM concentrations of sodium, this was deemed to be both analytically and clinically insignificant, especially as sweat sodium concentrations do not form part of the diagnostic criteria for CF. Furthermore, there was no significance in the bias observed between the ICP-MS sodium concentrations and the UKNEQAS weighed-in sodium concentrations (P = 0.104). Unfortunately due to the low volume of sweat typically collected from patients, it was not possible to directly compare the ICP-MS sweat chloride and sodium measurements with those obtained by coulometry and flame photometer respectively.
Comparison between UK NEQAS ALTMs and ICP-MS chloride results (n = 32). (a) Identity plot. (b) Bland–Altman difference plot to compare absolute chloride concentrations (SD = standard deviation). UK NEQAS ALTMs, UK national external quality assessment all laboratories trimmed means; ICP-MS, inductively coupled plasma mass spectrometry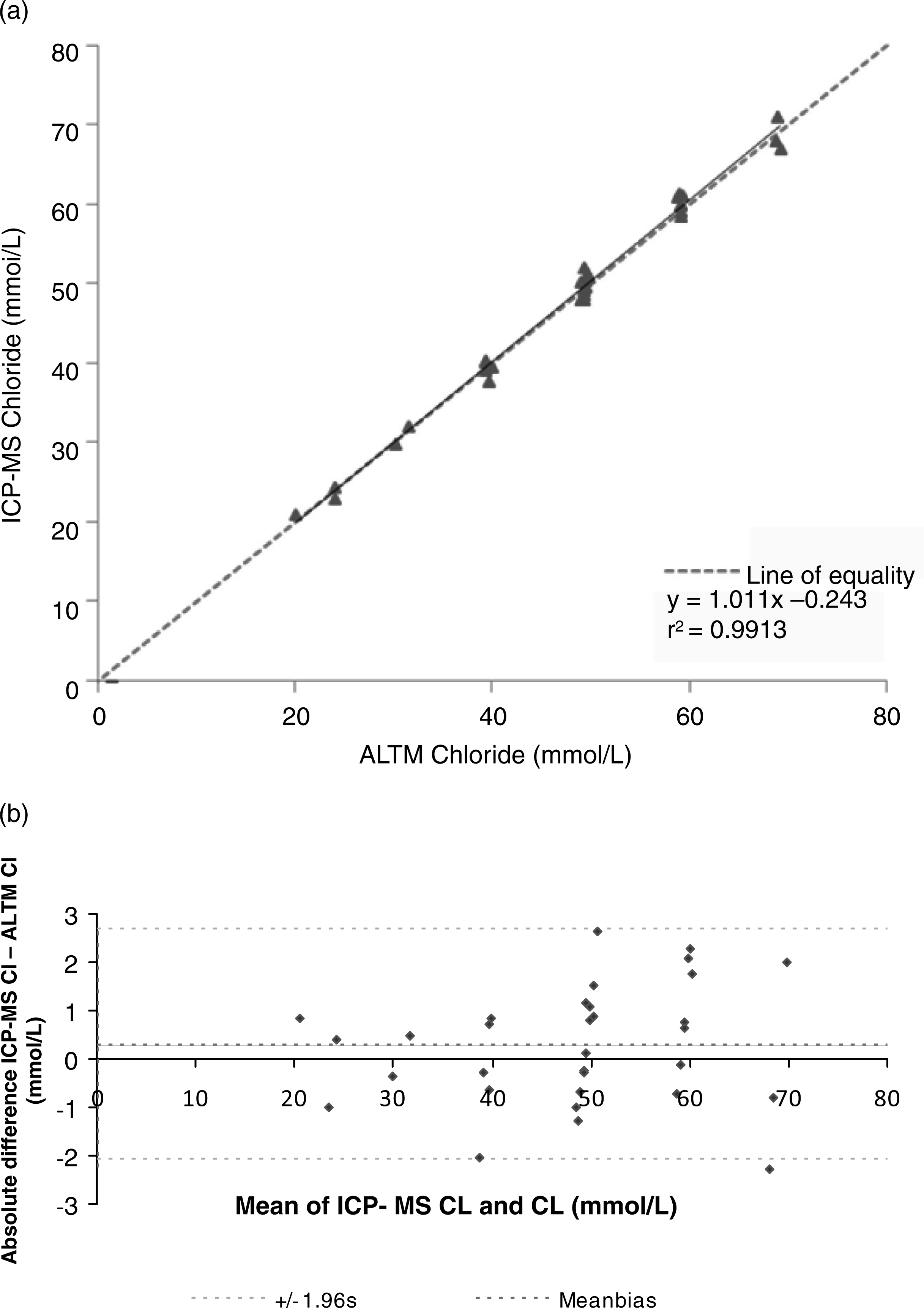
Number of patient sweat samples analysed and the range of chloride and sodium results reported in a ten-month period before and after ICP-MS introduction
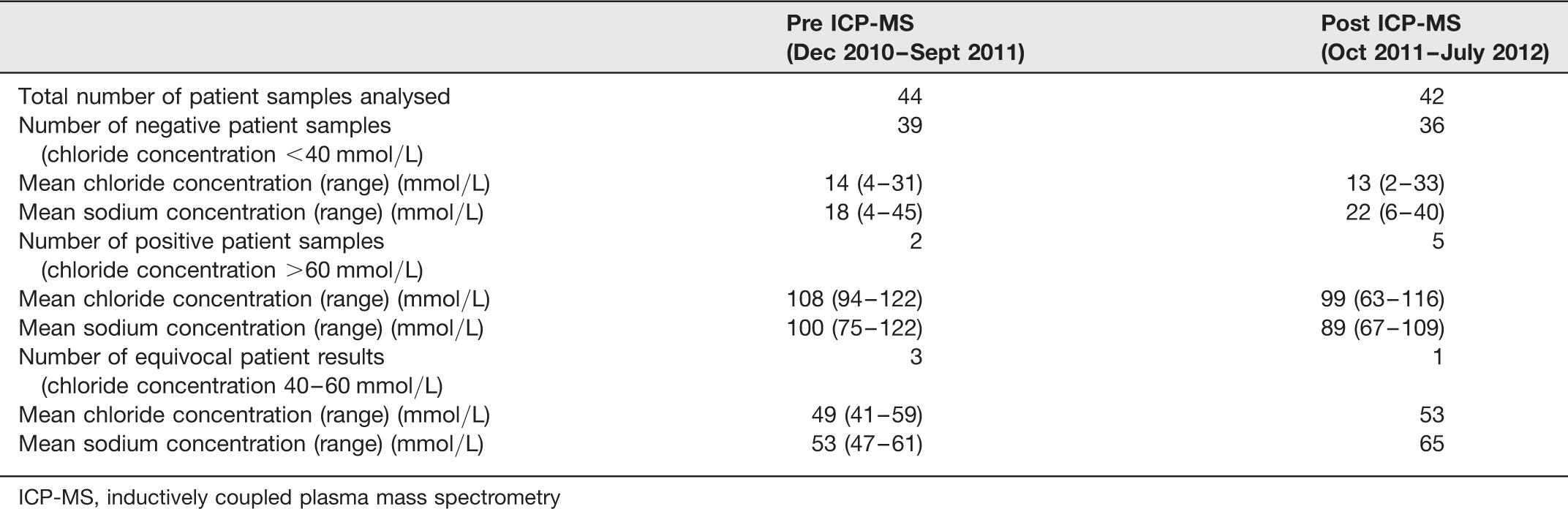
ICP-MS, inductively coupled plasma mass spectrometry
Conclusion
The ICP-MS method for the measurement of sweat chloride and sodium is compatible for use with the Wescor Macroduct® Sweat Collection System. The method is accurate, precise, has a wide linear range and fulfils the requirements of the National Guidelines. 2 Audit data pre- and post-ICP-MS implementation did not reveal that there have been any significant changes in the range of sweat sodium and chloride results reported in our tested population.
We believe that this is the first reported use of ICP-MS for sweat sodium/chloride measurement.
