Abstract
The coexistence of hypokalaemia and nephrocalcinosis poses a challenge in rapid diagnosis and appropriate management. We describe a 38-year-old woman who presented with thirst, intermittent carpopedal spasm, paresthaesia of both hands and progressive weakness of lower extremities for two years. She had a history of chronic hypokalaemia of unknown cause with intermittent potassium supplementation for 7-8 y and bilateral nephrocalcinosis notable for one year. She denied vomiting, diarrhoea or use of laxatives, alcohol or diuretics. Her blood pressure was normal. Laboratory investigations showed hypokalaemia (2.7 mmol/L) and metabolic alkalosis (HCO3− 32.6 mmol/L, pH 7.46). Two random urine samples both showed a consistently high urine K+ excretion but with excretion rates of Na+, Cl− and divalent cations which were high in one sample but not the other. Ingestion of furosemide 120 mg daily for body image for 7-8 y was uncovered. With furosemide cessation and potassium supplementation, her hypokalaemia with neuromuscular symptoms was corrected but nephrocalcinosis persisted. Surreptitious use of diuretics for various purposes should be kept in mind as an important cause of hypokalaemia and/or nephrocalcinosis. Measurement of electrolyte concentrations in at least two random urine samples is warranted to distinguish it from true renal tubular disorders and extrarenal causes.
Introduction
Hypokalaemia is one of the most common electrolyte disorders in clinical practice.1,2 The most significant effects of hypokalaemia are cardiovascular, neuromuscular, renal and metabolic and it may therefore be associated with morbidity and mortality. Hypokalaemia may have a variety of causes. Prompt elucidation of the cause along with appropriate management of the hypokalaemia will help avoid unnecessary investigation and potential complications. The serum potassium (K+) concentration is dependent on the total body K+ content and the distribution of K+ between intracellular fluid (ICF) and extracellular fluid (ECF) compartments. Hypokalaemia usually arises from re-distribution of K+ from ECF to ICF stores and/or a consequence of total body K+ depletion.3,4 In addition to a detailed history and physical examination, the evaluation of urinary K+ excretion, blood acid–base status and blood pressure help identify the underlying causes. 5
Nephrocalcinosis defined as the deposition of calcium in the kidney results from various metabolic defects such as disturbance of calcium metabolism, renal tubulopathy, medullary sponge kidney and medications. 6 The assessment of the calcium, intact parathyroid hormone and 1,25 (OH)2 vitamin D concentrations in blood, together with calcium, oxalate, citrate excretion in urine helps discriminate the underlying causes.7,8 In clinical practice, nephrocalcinosis associated with chronic hypokalaemia is often unrecognized and a correct diagnosis cannot be achieved without timely biochemical analysis of the urine. In this report, we describe an adult woman who presented with chronic hypokalaemia and nephrocalcinosis initially misdiagnosed as inherited Bartter's syndrome, a lesion in the loop of Henle. Surreptitious use of furosemide was unveiled based on two random urine electrolyte measurements.
Case report
A 38-year-old woman was referred for the evaluation of chronic hypokalaemia of unknown cause for 7–8 y and bilateral nephrocalcinosis that was present for one year, and that was absent 10 years previously. She described thirst, intermittent carpopedal spasm, paresthaesia in both hands and progressive muscular weakness over bilateral lower extremities of onset two years previously. She denied vomiting, diarrhoea or use of laxatives, alcohol or diuretics. Her family and past medical history were unremarkable. She was treated with oral potassium chloride (KCl) of 24–48 mmol daily but hypokalaemia persisted. A presumptive diagnosis of Bartter's syndrome was made.
Her height was 163 cm, weight 45 kg (body mass index 16.9 kg/m2), pulse rate 92/min and blood pressure 102/66 mmHg. The thyroid gland was not enlarged and physical examination was otherwise normal. Abdominal plain film X-ray showed bilateral nephrocalcinosis (Figure 1). Laboratory investigations revealed persistent hypokalaemia and hypochloraemic metabolic alkalosis (K+ 2.7,
Plain abdominal radiograph showing bilateral nephrocalcinosis in a medullary distribution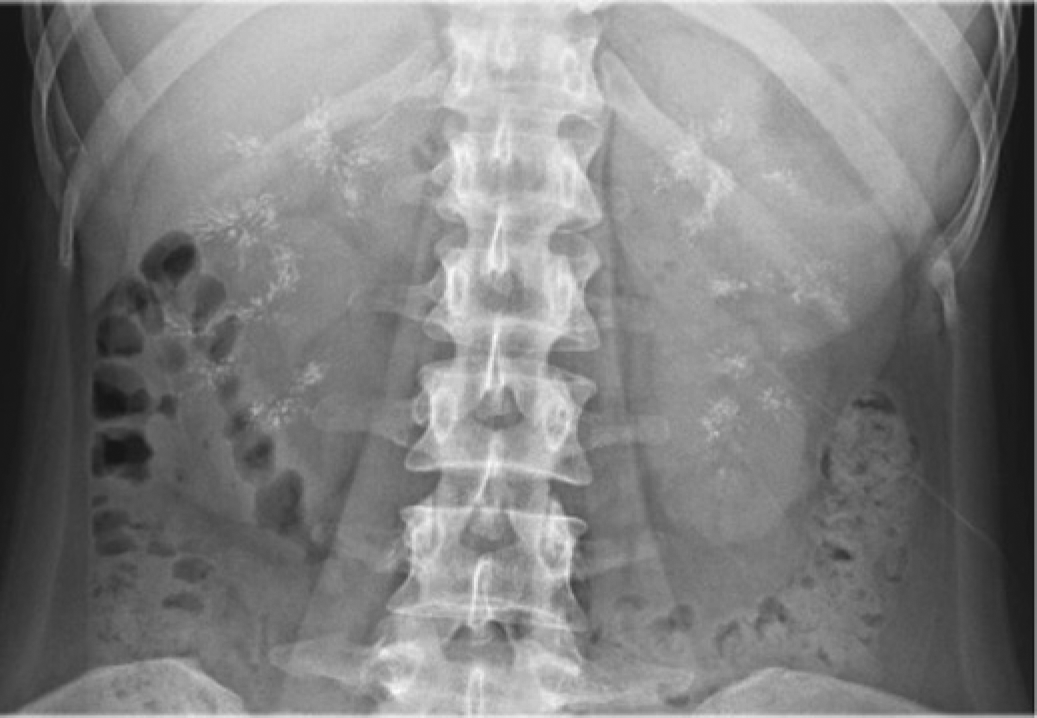
Laboratory studies
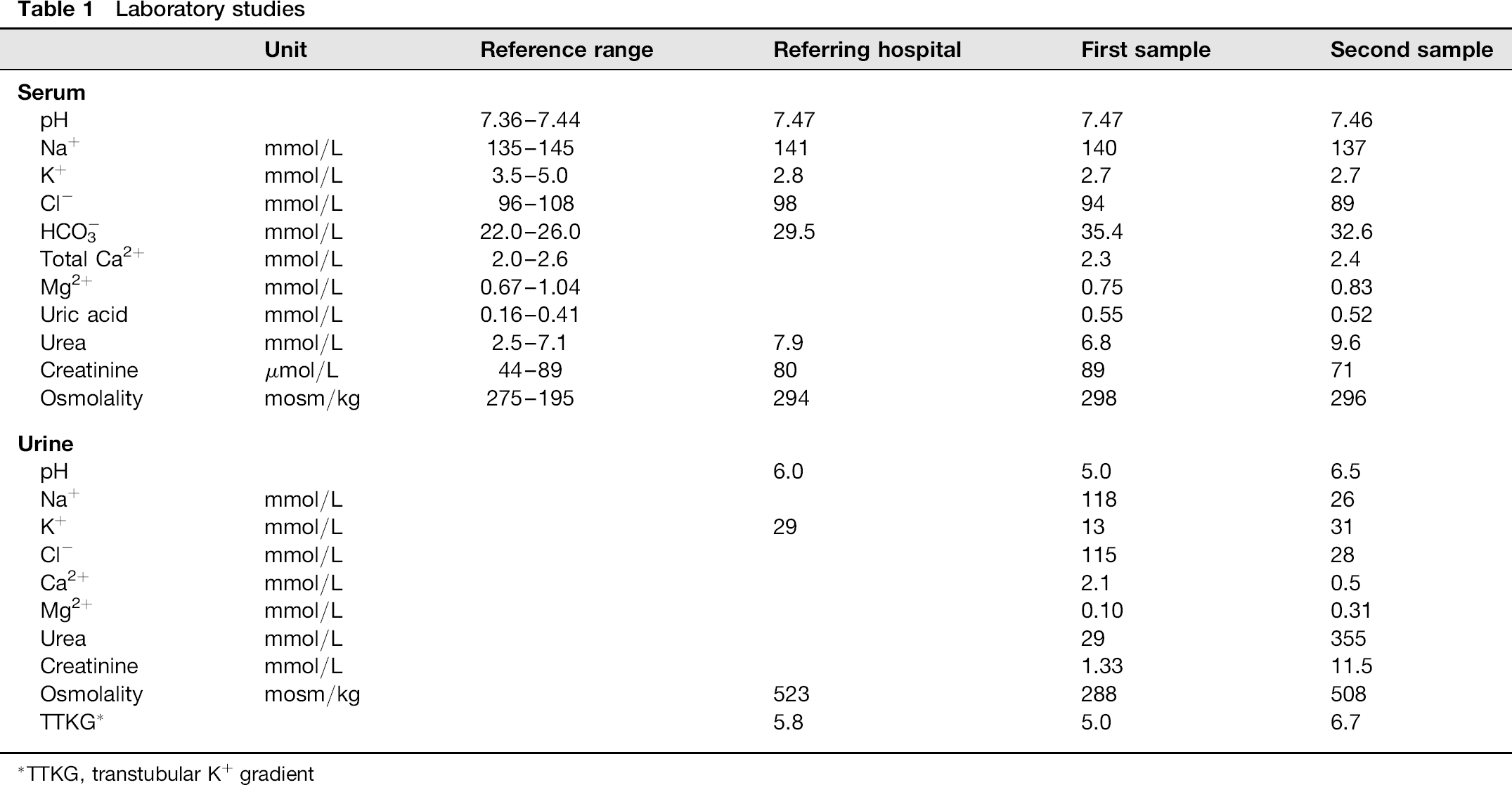
TTKG, transtubular K+ gradient
Discussion
This woman had chronic hypokalaemia with a presumptive diagnosis of inherited Bartter's syndrome and nephrocalcinosis. Her renal K+ wasting in response to hypokalaemia with hypochloraemic metabolic alkalosis and normal blood pressure indicated urine K+ loss together with Na+ and Cl−. Further evaluation of the urine concentrations of Na+, Cl− and divalent cations helped to discriminate renal from extrarenal and previous K+ loss.5,9 The first urine sample showed high urine Na+, Cl− and divalent cation excretion, suggesting dysfunction of the thick ascending loop of Henle (TALH), suggestive of Bartter's syndrome. In contrast, low concentrations of urine Na+, Cl− and divalent cation excretion in the second urine suggested the presence of vomiting, diuretic use, low salt diet or excessive sweating.5,9–11 In light of the inconsistent urine biochemical features, the case history was reviewed and the patient subsequently admitted to ingesting high doses furosemide over seven to eight years for body image purposes.
Diuretic-induced hypokalaemia is commonly encountered in clinical practice. This diagnosis largely depends on history and laboratory findings. However, physicians may easily ignore the diagnosis if patients deny the use of diuretics. In this setting, several random urine samples rather than one spot or 24 h urine may be very helpful in pointing to diuretics as the cause. A single spot urine sample might not unveil the use of diuretics because the renal tubular response to diuretics depends on its duration of action. For example, furosemide, a widely-used loop diuretic, has a half-life of approximately two hours and duration of action of 6-8 h. The excretion of urine Na+, Cl− and divalent cations is dramatically enhanced through action on the TALH but declines rapidly as diuretic action falls off. Serum divalent cation concentrations usually remain normal in furosemide ingestion (in the absence of poor oral intake or gastrointestinal malabsorption) because their re-absorption is enhanced in the upstream proximal tubule and downstream distal convoluted tubule. However, renal K+ loss is persistent because of early increased urine flow and then late secondary increased aldosterone concentration in response to ECF depletion to augment renal K+ secretion. Additionally, urine is more acidic when furosemide action is at its peak. 12 This ‘on-and-off effect on urine electrolyte concentrations and pH is characteristic and useful in distinguishing diuretic use from inherited renal tubular and extrarenal disorders. 9 It is worth mentioning that the measurement of diuretics in the urine also helps the diagnosis but is usually valid during the ‘on’ action of furosemide (high urine excretion of Na+ and Cl−). A false ‘negative’ result is often evident when the action of furosemide is ‘off’ (low urine Na+ and Cl− excretion).
Medullary nephrocalcinosis, another apparent feature in this patient, can be divided into two groups based on serum Ca2+ concentration. 8 Nephrocalcinosis associated with hypercalcaemia primarily results from hyperparathyroidism, excessive vitamin D, and milk-alkali syndrome. Mineralocorticoid excess-related disorders, hyperoxaluria, renal tubulopathy and the use of diuretics (loop diuretics and acetazolamide) will cause nephrocalcinosis without hypercalacemia. 7 Our patient did not have hypercalcaemia, hypertension or chronic diarrhoea, suggesting an inherited or acquired renal tubular dysfunction. The absence of nephrocalcinosis before the abuse of furosemide in conjunction with no identifiable cause of renal tubular dysfunction suggested that the furosemide abuse was associated with nephrocalcinosis. In fact, furosemide has been reported as an important cause of nephrocalcinosis in any age group.
The risk of developing nephrocalcinosis has been correlated with larger furosemide doses and longer duration of therapy and is generally a late finding.13–15 In our patient, ingestion of large dose (120 mg daily) and prolonged use of furosemide (7–8 y) supported this finding. The super-saturation of Ca2+ at TALH caused by loop diuretics has been reported to predispose to the formation of nephrocalcinosis, especially in the presence of hypokalaemia-induced hypocitraturia.15–17
In addition to diuretics, patients who are concerned about their body image may abuse laxatives or thyroid hormone-containing drugs that may lead to hypokalaemia. Laxative abuse usually causes hypokalaemia with hypochloraemic metabolic alkalosis and low urine K+, Na+, but high Cl−and ammonium excretion. 18 Patients with surreptitious ingestion of thyroid hormone may present with thyrotoxic periodic paralysis which is characterized by hypokalaemia with low urine K+ excretion and relatively normal acid-base status. 19 Of note, chronic hypokalaemia (regardless of aetiology) may also be associated with nephrocalcinosis and can be divided into two broad categories based on urine K+ excretion rate (Figure 2).6,20–23
Algorithm for the investigation of hypokalaemic patients with nephrocalcinosis or nephrolithiasis. AME, apparent mineralocorticoid excess; BS, Bartter's syndrome; dRTA, distal renal tubular acidosis; TTKG, transtubular K+ gradient; FEK, fractional excretion of K+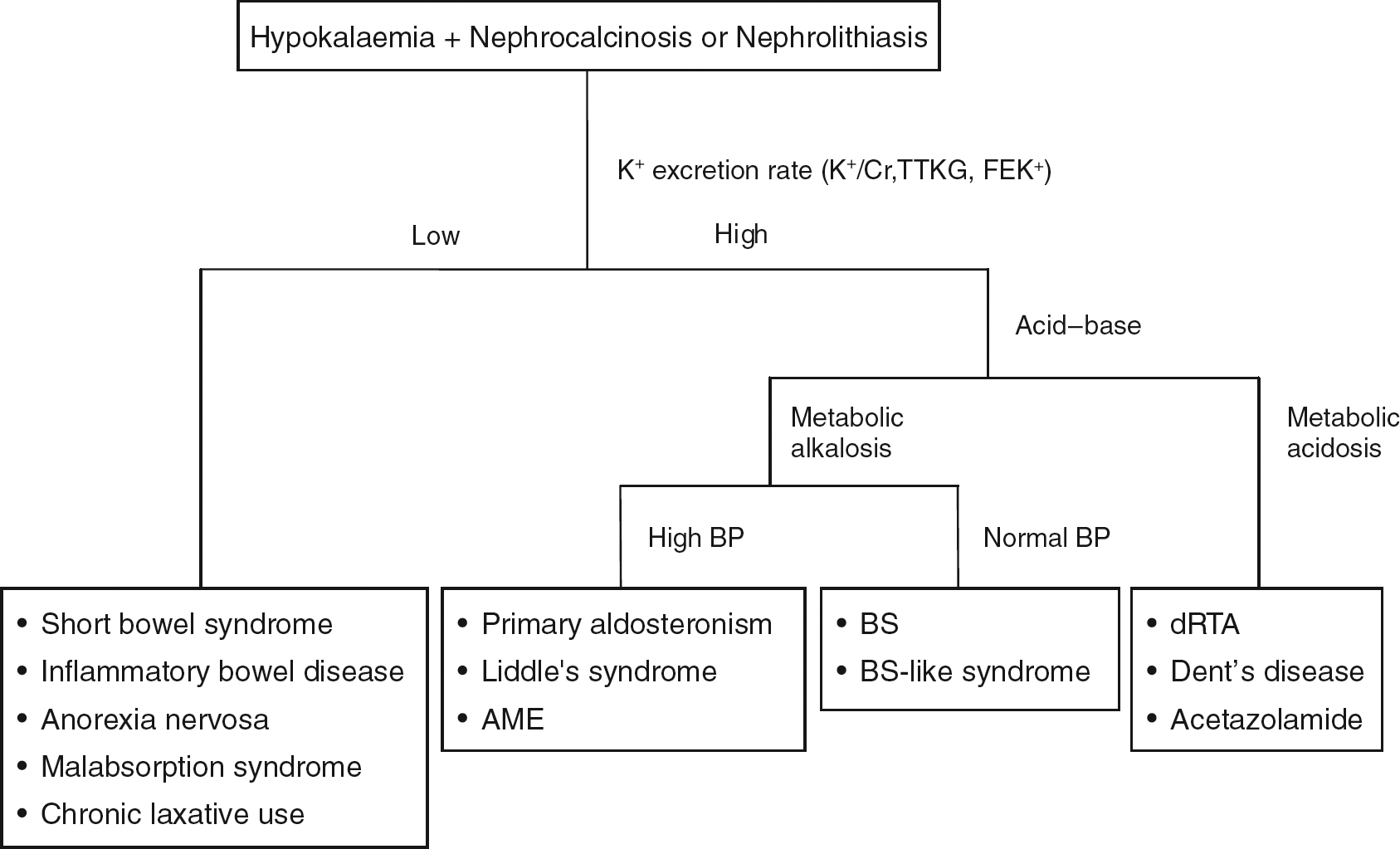
The adverse effects of diuretic abuse must be emphasized. Diuretic-induced hypokalaemia can lead to neuromuscular features including fatigue, myalgias, muscular weakness or cramps, and even complete paralysis. Cardiovascular complications such as electrocardiographic changes, ventricular and atrial arrhythmias may occur. Hypokalaemia also affects intestinal muscle cell and results in constipation or paralytic ileus. In the kidney, it may cause functional and structural defects including renal tubular vacuolization, renal cyst, interstitial nephritis, impaired concentration ability, decreased GFR and even end stage renal disease.9,11 Aside from K+ imbalance, other electrolytes including Na+ and divalent cations may be affected variably depending on the type of diuretics. 24 Furthermore, metabolic derangement such as dyslipidaemia (thiazides), nephrocalcinosis (loop diuretics), insulin resistance, hyper-uricaemia and acute gout are also commonly associated with the diuretic abuse. 24 Nephrocalcinosis may cause renal function impairment and may remain affected after withdrawal of loop diuretics and correction of hypokalaemia. The persistent microscopic calcification found on histology may account for the irreversible renal impairment. 25
In conclusion, diuretic-induced hypokalaemia and nephrocalcinosis may be overlooked and misdiagnosed, leading to inappropriate management. Nephrocalcinosis is a late finding associated with long-term use of loop diuretics. The analysis of two random urine samples rather than a single random or 24-hour urine samples often provides the diagnosis. Early recognition with prompt withdrawal of diuretics is mandatory to avoid unnecessary investigation and untoward complications.
Declarations
Footnotes
Acknowledgements:
None.
