Abstract

Accurate identification of proteinuria is essential to the diagnosis and management of patients with kidney disease, as stated by Methven and MacGregor. 1 We can further agree that collection of 24 h urine samples is not necessary to measure protein loss and that reagent strip devices are generally unsuitable for proteinuria detection. However, these authors conclude that the urinary albumin to creatinine ratio (ACR) is less sensitive than urinary total protein to creatinine ratio (PCR) at predicting proteinuria and patient-relevant outcomes, fails to detect a significant minority of proteinuric patients and is more costly. We present an alternative view that although urinary albumin assays are marginally more expensive, they offer a more accurate and reliable test for proteinuria, especially at the low levels seen in the early stages of chronic kidney disease (CKD), the prognostic importance of which is becoming increasingly apparent. Further, urinary total protein assays are subject to interference which may give a false-positive indication of proteinuria potentially resulting in over-investigation and unnecessary patient anxiety.
If proteinuria in this context is defined as urinary loss of ‘total protein’, as historically it has been, then it is inevitable that ACR will appear worse at predicting proteinuria than PCR, since both PCR and urinary total protein loss are based on the same total protein measurement. But this presupposes that total protein loss should be the metric by which we judge the degree of kidney damage and glomerular permeability. The association between kidney disease and proteinuria goes back at least as far as the early 19th century, when Bright first described albuminous nephritis. 2 It was not until the 1960s that sensitive assays capable of specifically measuring urinary albumin became available. 3 For much of the intervening period, and beyond, the so-called total protein methods have been used to estimate urinary protein concentration albeit that, somewhat unknowingly, albumin has always been the main protein that has been measured. Although Bright and others 4 were able to recognize the critical importance of albumin in this context, it is not surprising that (total) ‘proteinuria’ has subsequently remained embedded in clinical nephrology practice as the sine qua non. But laboratory diagnostics does not stand still: increasing evidence suggests that albumin measurement is both a technically superior test and a better marker of kidney damage. 5
Methven et al. 6 state that ACR is less sensitive than PCR at predicting proteinuria. As noted above, this analysis was biased in favour of PCR, and in any case is not the key question. Let us ask instead: what is the best test at detecting a clinically significant increase in urinary protein loss? Urinary albumin measurement provides a more specific and sensitive measure of changes in glomerular permeability than total protein. 7,8 Albuminuria is the earliest marker of glomerular diseases, including diabetic glomerulosclerosis, where it generally appears before the reduction in glomerular filtration rate (GFR). Albumin is the predominant protein in the vast majority of proteinuric kidney diseases: generally proteinuria reflects albuminuria. In health, relatively small amounts of albumin (less than 30 mg/24 h) are lost in the urine.* Because total protein assays are imprecise and insensitive at low concentrations, relatively large increases in urine albumin loss (for example doubling) can occur without causing a significant measurable increase in urinary total protein. 8 Albumin immunoassays will detect such an increase. So in terms of sensitivity for detecting increased protein loss, there can be no doubt that ACR is superior.
Methven et al. 9,10 base much of their argument on interesting and important findings from their retrospective analysis of >5500 patients attending a nephrology clinic. In this cohort, they have failed to observe any advantage of ACR over PCR in predicting patient outcomes (death, renal replacement therapy (RRT), doubling of serum creatinine). We are unsurprised by this, since ACR and PCR are effectively two measures, albeit one cruder than the other, of the same thing; namely, albuminuria. As ACR increases, so does PCR – largely due to ACR. 11
Methven et al. 9 agree that their data are not representative of a primary care-based CKD population. 10 However, unfortunately they extrapolate their findings to this setting. In subsets of their cohort with PCRs ≥ 100 mg/mmol or ≥50 mg/mmol but ACRs < 70 mg/mmol or <30 mg/mmol, respectively (PCR versus ACR ‘discordance’) they observed an increased adjusted hazard ratio for doubling of serum creatinine compared to those with both ACR < 70 (or <30) mg/mmol and PCR < 100 (or <50) mg/mmol. Contrary to their interpretation, we believe that hazard ratios for death and RRT were not significantly different between these groups. Primarily on this basis they claim that screening with ACR alone will fail to identify 16% of patients with significant proteinuria and increased risk. This is rather disingenuous. The patients that they claimed would be missed using ACR testing actually had ACRs between 3 and 69 mg/mmol, i.e. they were albuminuric. 12 Furthermore, these patients were even less representative of a general population than their overall cohort, having a high prevalence of tubulointerstitial disease and relatively low prevalence of diabetic nephropathy. The data are of interest and suggest to us that, in this somewhat atypical subgroup, there may be urinary proteins, perhaps higher molecular weight proteins, 13 the specific measurement of which may give added prognostic information. However, it hardly seems a sound basis on which ‘Guideline recommendations on proteinuria screening in …CKD should be reconsidered’. 9
ACR represents a specific, quantifiable, continuous risk marker, which can be measured using standardized methodology
14,15
throughout the world and across different patient groups, including those most at risk of CKD. There is strong evidence linking increased albuminuria to all cause and cardiovascular mortality both in the general population,
16
amongst older people,
17
people with kidney disease
18
and amongst other people at high risk (e.g. those with diabetes
19,20
and hypertension
21
). Albuminuria also predicts the development
22
and progression
20,23
of kidney disease and risk of acute kidney injury.
24
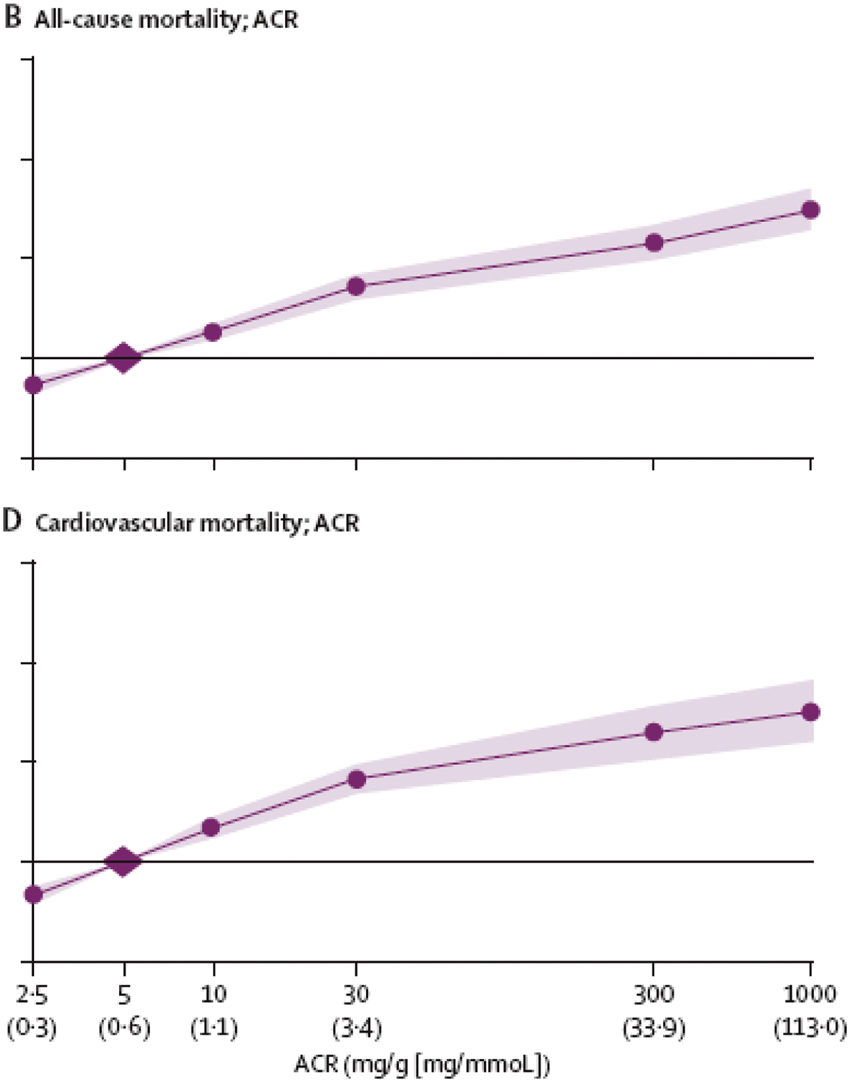
Some of the most powerful data come from meta-analyses undertaken by the CKD Prognosis Consortium demonstrating continuous associations between increasing ACR and subsequent risk of all-cause and cardiovascular mortality, kidney failure, acute kidney injury and CKD progression in the general population and in populations with increased risk for cardiovascular disease: these associations extend down to levels of albuminuria previously considered normal (Figure 1).
25
–27
Methven and MacGregor
1
are correct that, outside of diabetic nephropathy, there is no evidence that treatment of albuminuria< 30 mg/mmol is beneficial. But there is also no reason to suspect that such treatments, for example renin-angiotensin-aldosterone system (RAAS) blockade, could not be effective in this situation. Clearly, there is an urgent need to assemble this evidence.
Relationship of albuminuria with mortality. Hazard ratios (HR) and 95% CIs for all-cause (B) and cardiovascular mortality (D) according to albumin-to-creatinine ratio (ACR). The horizontal line crosses the y-axis at an HR of 1.0, with the axis being plotted on a doubling scale (i.e. 1, 2, 4 and 8). HRs (circles) and 95% CIs (shaded areas) are adjusted for age, gender, ethnic origin, history of cardiovascular disease, systolic blood pressure, diabetes, smoking, and total cholesterol and spline estimated glomerular filtration rate (eGFR). The reference (diamond) was ACR 0·6 mg/mmol (5 mg/g). To convert ACR in mg/g to mg/mmol multiply by 0·113. Approximate conversions to mg/mmol are shown in parentheses. Reprinted with permission from Matsushita et al.
25
As we have discussed previously, there are strong technical reasons for preferring albumin assays over total protein measurements, particularly at lower levels of proteinuria. 5 In 1696 individual 24 h urine collections Methven et al. 6 found, as others have before, 11 a poor relationship between ACR and PCR at lower levels of proteinuria. This was incorrectly interpreted as a criticism of ACR measurement rather than a reflection of the imprecision, non-specificity and susceptibility to interferences of the chemical and turbidimetric reactions they used to estimate total protein concentration. It is noteworthy that ACR and PCR had equal diagnostic performance at predicting proteinuria of 1 g/24 h amongst a subset of patients receiving RAAS blockade, amongst whom the proportion of albumin to total protein was higher. 6
There is concern, echoed by Methven and MacGregor, 1 that replacing urinary total protein with albumin measurement may cause non-albuminuric (effectively tubular and overflow) proteinuria to be missed. This is misplaced, as we have also discussed previously. 5 In our hands, amongst 569 patient urines obtained from primary care, we observed 36 (6%) to have a raised PCR (>23 mg/mmol) but normal ACR (<3.4 mg/mmol) by USA Kidney Disease Outcome Quality Initiative thresholds. 28 Was this due to tubular proteinuria? We subjected these ‘discrepant’ samples to repeat analysis plus further analyses for specific tubular proteins (cystatin C, alpha-1 microglobulin, β-trace protein) and qualitative examination using sodium dodecyl sulphate-agarose gel electrophoresis (SDS-AGE). ACR and PCR results were reproducible but we found no evidence of significant tubular proteinuria and only traces of albumin were visible following electrophoresis. 29 We conclude that the raised PCR was a false positive signal in these cases: it is tempting to speculate that this may have also been the case in the AusDiab study 11 referred to by Methven and MacGregor. 1 Testing for tubular proteinuria using a total protein approach almost certainly has very poor sensitivity for detecting tubular disease. When an isolated tubular lesion is suspected this is best investigated by measuring a specific tubular protein using an immunoassay.
Some have suggested that a significant proportion of urine albumin may be non-immunoreactive. 30 –33 Methven and MacGregor 1 also raise this issue, pointing to Australian studies where albumin measured by immunoassay was compared with that detected by HPLC. 34,35 Others have questioned this finding, however, suggesting that non-immunoreactive albumin is likely a co-eluting non-albumin protein fraction. 36 –38
The reagent cost of ACR is greater than PCR, although we do not agree with the up to 10-fold difference cited by Methven and MacGregor. 1 Comparative costs vary between different laboratories and health care systems. Although increased use of ACR may reduce the unit cost due to economies of scale, an antibody-based reagent will probably always be more expensive than a chemical one. In England, the National Institute for Health and Care Excellence (NICE) randomly sampled a small number of laboratories and estimated the average cost of an ACR to be £2.16 whereas a PCR cost £1.42. 39 In Canada, laboratory analysis costs (Canadian dollars) of $11.67 for PCR and $29.23 for ACR have been cited. 40 A full health-economic comparison would also need to take into account factors such as the impacts on treatment costs and patient outcomes following testing, including those arising from false positive or negative results. Furthermore, there are other factors that need to be considered when comparing diagnostic tests, for example clinician confidence in the test result, specificity and sensitivity for the target condition and frequency of indeterminate or uninterpretable results. 41 Heating urine in a teaspoon is cheaper than a colorimetric total protein assay, but no-one would advocate reverting to the detection of ‘coagulable urine’ on these grounds, nor of using Bradshaw’s test to detect Bence Jones protein. By comparison, troponin measurement is more expensive than creatine kinase; its introduction has facilitated a revolution in the management of acute coronary syndromes to the point where its measurement now defines the condition. Technology and clinical diagnostics improve with time and it is important we embrace such developments to allow for disease classification and interventional opportunities to emerge.
Use of urinary albumin measurement as the preferred test for proteinuria detection will improve the sensitivity, quality and consistency of approach to the early detection of CKD. By contrast, laboratory tests purporting to measure urinary total protein are flawed, often being standardized against, and predominantly sensitive to, albumin, having poor precision at low concentrations, demonstrating poor between-laboratory agreement, being insensitive, non-specific and subject to a range of false-positive and false-negative problems. In patients with established disease there may occasionally be clinical reasons for a specialist to use PCR instead of ACR to quantify and monitor higher levels of proteinuria (although, in truth, we have difficulty accepting this rationale). PCR testing may have a role, at marginal increased cost, as a prozone check on ACR assays, to guard against this relatively rare analytical issue. 42 But such matters are not the central concern of CKD guidelines intended for non-specialist use. An overwhelming body of evidence is accumulating pointing to the significance of low level protein loss in terms of morbidity and mortality: such data can only be gathered using urinary albumin. Recommendations from NICE, 39 and KDIGO 12 that ACR should replace PCR as the test of choice for proteinuria detection, were not based solely upon their relative abilities to estimate total protein loss, but on factors including that albumin measurement can be standardized and is more precise at lower levels of proteinuria, that it is already the test of choice in people with diabetes, and that it is the predominant protein in the vast majority of proteinuric kidney diseases. 5 To persevere in assigning primacy to total protein loss misses the point and hinders progress in this field.
Footnotes
Acknowledgements
The authors are grateful to Dr E.E. Bassett of the School of Mathematics, Statistics and Actuarial Science at the University of Kent for statistical advice.
Declaration of conflicting interests
EJL was a member of both the NICE and KDIGO CKD guideline development groups. PES is the chair of the NICE CKD guideline and co-chair of the KDIGO CKD guideline.
Funding
None.
Ethical approval
Not applicable.
Guarantor
EJL.
Contributorship
The article was prepared at the invitation of the Clinical Sciences Reviews Committee of the Association for Clinical Biochemistry and Laboratory Medicine. EJL prepared the first draft. All authors modified subsequent versions and approved the final version of this paper.
