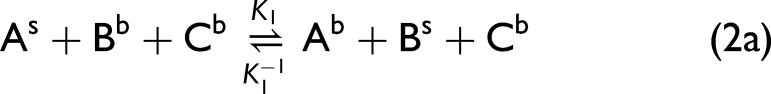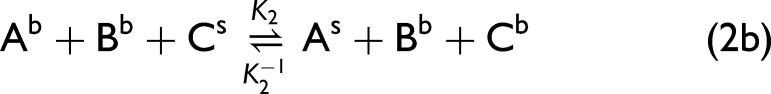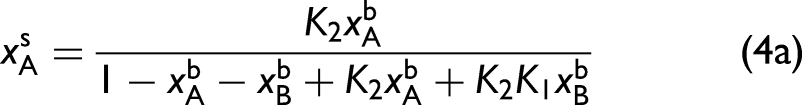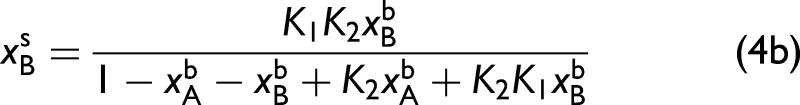Abstract
A method is proposed whereby the range of equilibrium constants that describe how a ternary mixture of molecules is adsorbed onto a solid substrate may be obtained. This information may then be related to the surface preference of a particular species. The technique utilizes infrared and/or Raman spectra of the bulk phase, together with sum frequency generation spectra of the surface in a heterospectral two-dimensional correlation analysis. The method consists of a series of up to three experiments in which the bulk concentrations are varied, followed by a set of rules for interpreting the resulting correlation maps to identify adsorption sequences and surface preference. A scheme is provided to direct the design of the experiments and analyses for such multi-component mixtures.
This is a visual representation of the abstract.
Keywords
Introduction
Ternary mixtures can exhibit complex phase behavior, surface affinities, and adsorption dynamics. Understanding adsorption behavior in ternary systems is crucial for various industrial, environmental, and scientific applications.1–4 These interactions govern the efficiency of chemical separations, catalysis, drug delivery, water treatment, and biofuel production.5–11 Surface affinity—an indicator of which component preferentially adsorbs at an interface—affects the stability, composition, and separation efficiency in many of these systems. In biodiesel production, for example, the adsorption of methanol, lipids, and catalyst influences phase separation, reaction efficiency, and product purification.12–14 Similarly, in water treatment, polyelectrolyte adsorption onto contaminants impacts flocculation and filtration processes.15,16
Recent studies have highlighted the intricate nature of adsorption in ternary systems, especially concerning competitive adsorption behaviors. For instance, the adsorption of heavy metals such as Cu2+, Cd2+, and Ni2+ onto biochar in the presence of microplastics demonstrated that the presence of additional components can significantly influence adsorption capacities and mechanisms.17,18 Such findings underscore the importance of understanding multi-component interactions in adsorption processes.
In general, the characterization of surface composition is challenging in cases where the same components are present in an adjacent bulk phase, primarily because most measurement methods would therefore be overwhelmed with signals from the bulk contributions. Spectroscopic methods are particularly valuable due to their ability to probe buried interfaces since they require only access by light. However, spectroscopic characterization of surfaces has an additional challenge in that the markers from surface species may coincide with those in the bulk phase. For this reason, second-order nonlinear optical techniques such as vibrational sum-frequency generation (SFG) spectroscopy are powerful in their ability to provide label-free fingerprints of molecules at surfaces, at sub-monolayer coverage, and inherently exclude contributions from the bulk due to the symmetry requirements of the underlying even-order response function.19–21
We have previously illustrated how a combination of infrared (IR) and/or Raman spectra of the bulk phase can be used together with surface SFG spectra to determine preferential adsorption in binary systems.22–27 This was based on the application of two-dimensional correlation analysis (2D-COS)28–31 that revealed synchronous and asynchronous changes between features (vibrational bands) in the surface and bulk spectra. We now seek to extend this method to study adsorption in ternary mixtures, investigating the role of surface preference in heterogeneous systems. We propose a rule-based approach derived from 2D-COS heterospectral analysis30,32–35 that can be used to characterize a particular class of ternary system. By systematically analyzing the interactions and adsorption behaviors within these mixtures, we aim to provide insights that can inform the design and optimization of processes in various applications, from environmental remediation to industrial separations.
Background and Methods
Surface Excess Determination From 2D-COS: Comparison With an Approach Developed for Binary Systems
We have previously described a method for determining the surface preference in the case of a binary system using 2D-COS.22–26 One of the inherent challenges is that the SFG peak amplitude is related to both the surface population and the orientation/conformation of the molecules at the surface. Furthermore, some species may reorient as they pack closer together. Therefore, before a particular band in the SFG spectra can be used for surface preference determination, one must first verify that the orientation of that particular moiety is not dependent on the surface population. We have previously shown that the way to determine this, without any spectral fitting, is to look at two different vibrational modes associated with the same moiety or that are connected to a rigid part of the molecule. 26 If there is no SFG homospectral asynchronous cross peak between these frequencies as the bulk concentration changes, then we can be assured that the SFG amplitude is directly proportional to the surface population. Although we previously noted that this then enables the sign of the same cross peak to be analyzed in the surface–bulk (e.g., SFG–IR) heterospectral asynchronous map, we now realize that there was a missed opportunity, as any peak could actually be used in the final step, including the surface–bulk “diagonal” peaks. Note that, although it is not customary to see diagonal peaks in asynchronous maps, these occur as we are performing a heterospectral correlation analysis. The relationship between the sign of a heterospectral asynchronous peak and the surface preference of that species simply follows Noda’s rules, 28 taking the direction of the perturbation into account, namely the direction in which the bulk concentration changes.
In binary systems, the experiment to be performed is obvious, as the bulk mole fractions of the components should be changed, and this becomes the perturbation in the subsequent 2D-COS analysis. However, in the case of the ternary systems we will describe here, the design of the necessary experiments requires some consideration.
Model Ternary System
Although there are many possibilities for how three components can interact with a surface in a ternary system, we have chosen to study the particular situation where there are three species A, B, and C with a superscript b designating their presence in the bulk solution phase and s when they present on the surface.
Infrared, Raman, and Sum-Frequency Generation Spectra
As an example, we have chosen to work with simulated vibrational spectra consisting of mixtures of glycerol, methanol, and toluene. Ternary mixtures of these molecules are of interest in the purification of glycerol recovered from biodiesel production.10,11 Simulated spectra provide a unique opportunity for this proof-of-concept since we can control the equilibrium constants
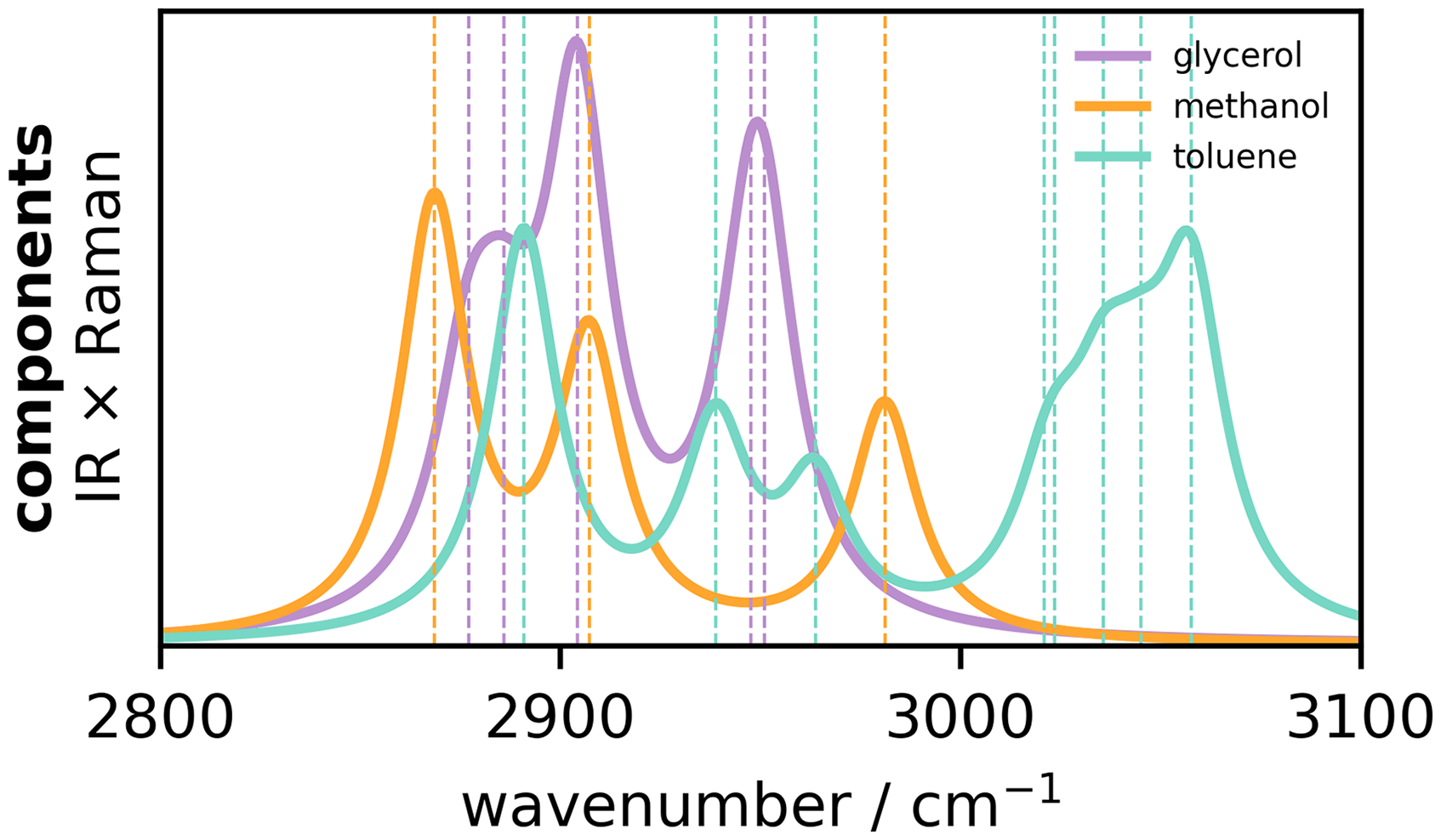
Bulk (IR–Raman hybrid) spectra of the three pure components considered in the ternary mixture. Vertical lines indicate the frequencies of the normal modes, color-matched to the spectra. The same colors will be used to mark frequencies of interest in the subsequent 2D-COS maps.
Results and Discussion
General Approach
In this particular ternary system, the surface populations are determined by
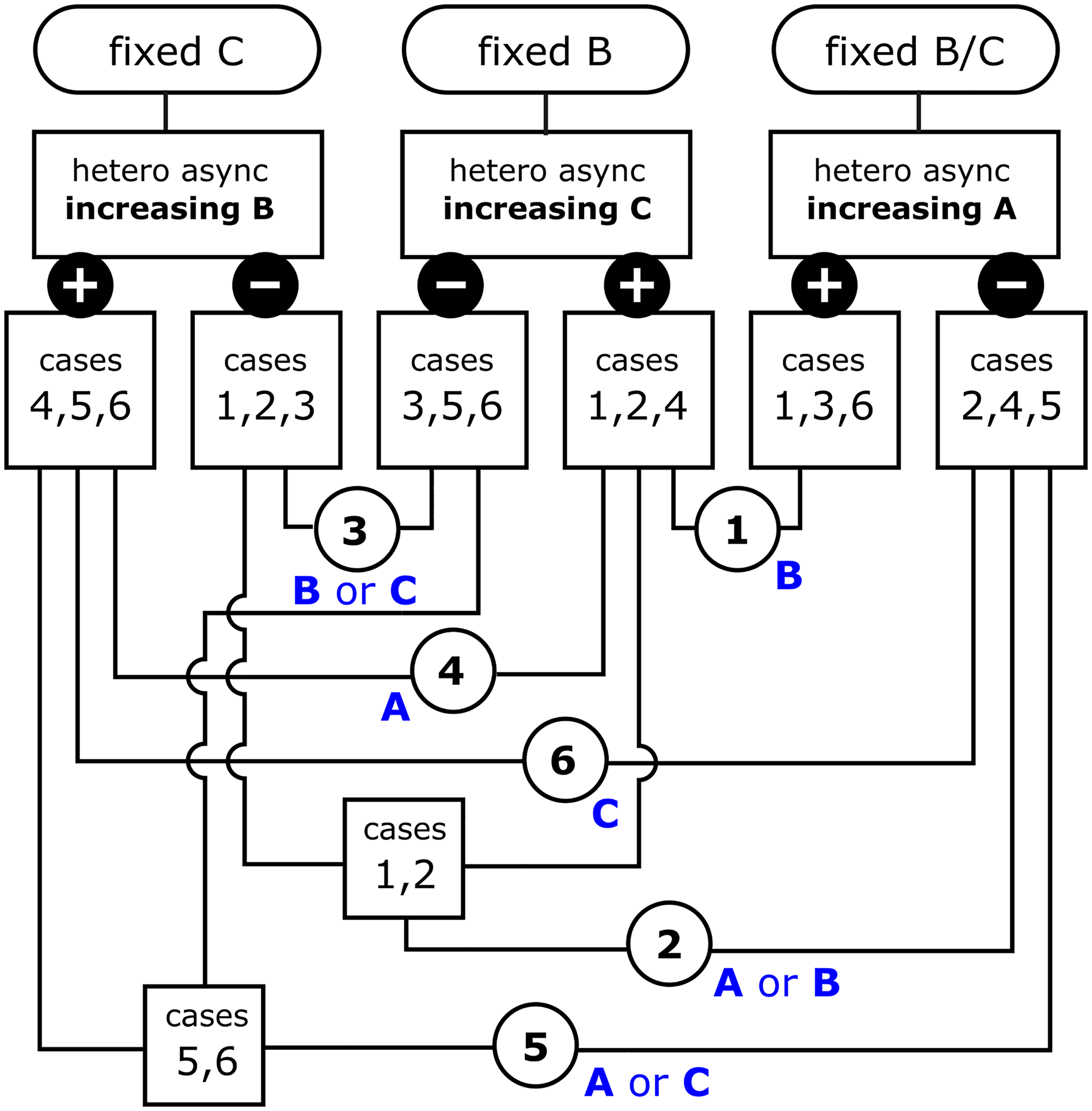
A flowchart illustrating the three potential perturbations required in order to determine which of the six cases (see Table I) characterize the ternary system under investigation. The resulting cases 1–6 must be arrived at using a Boolean AND operation. Only two of the three proposed experiments are required, but those may not be known in advance. Text in blue indicates the surface preference that results from that particular combination of equilibrium constants.
The ternary system shown in Eq. 1 is characterized by two equilibrium constants,
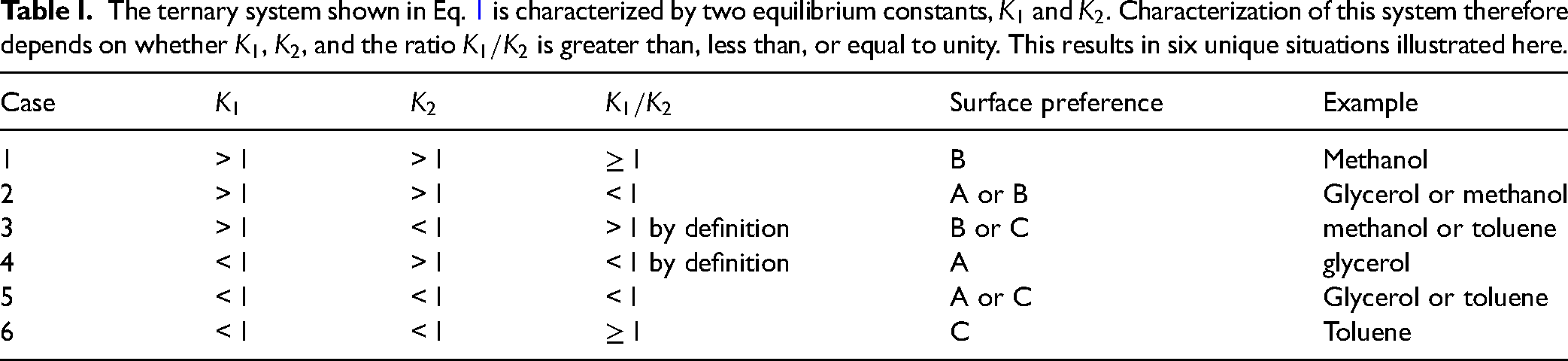
We therefore start with the two simplest experiments, where
Example
We consider a ternary mixture where species A is glycerol, species B is methanol, and species C is toluene. For the sake of this illustration, we have generated model spectra using
As we have mentioned, one normally needs to ensure that surface species do not reorient as their surface population changes, prior to the interpretation of the heterospectral correlation peaks. However, as this is a simulation, we have simplified the situation by ensuring that the surface orientation remains constant. We therefore do not need to examine the surface (SFG) homospectral correlation, as we know (and have verified) that there are no asynchronous cross peaks; all SFG signals can therefore readily be interpreted in terms of surface population changes.
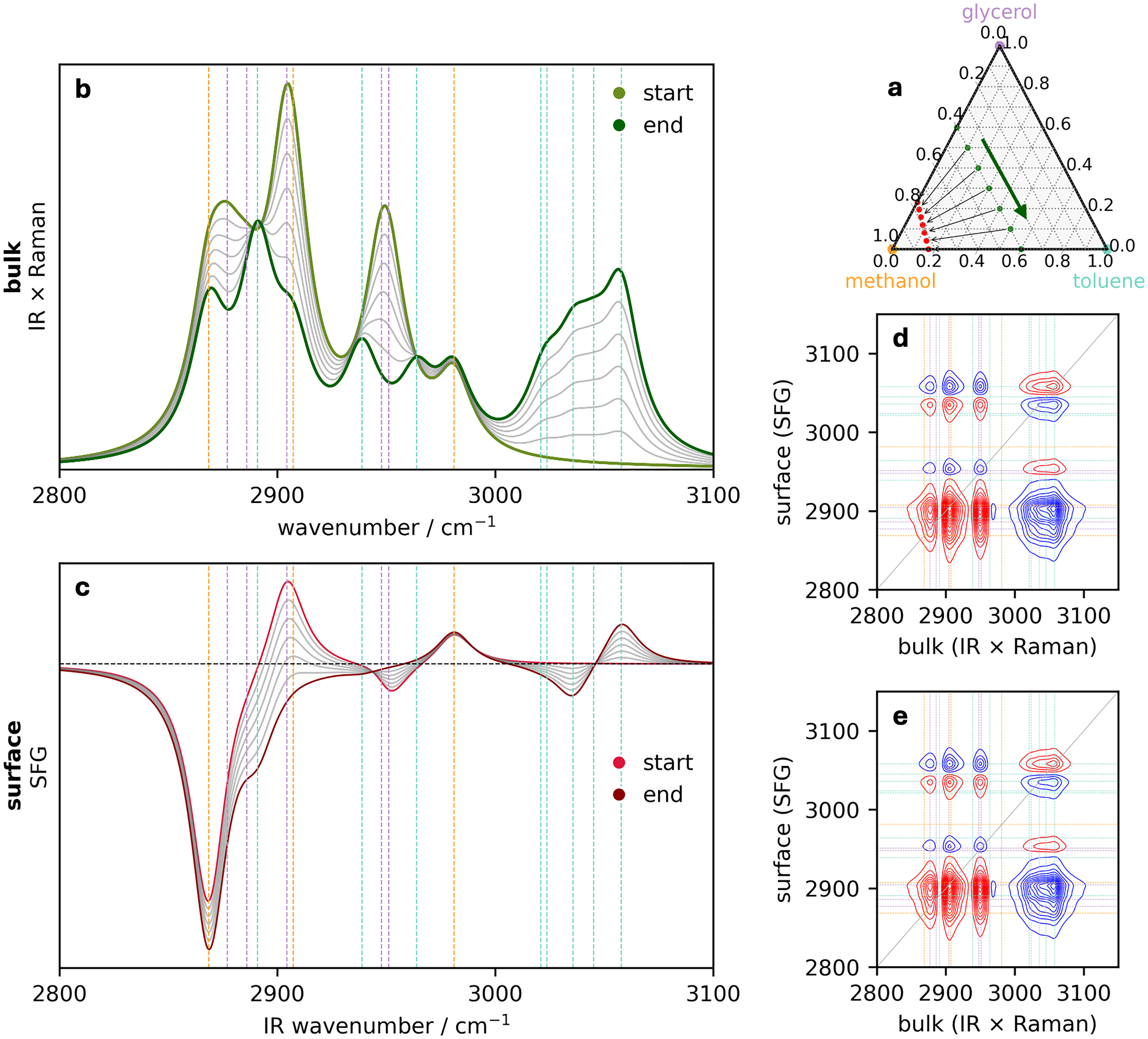
Perturbation 1: The bulk methanol mole fraction remains fixed. (a) Ternary diagram illustrating the initial bulk concentrations of the three components (green circles) that define the perturbation along the direction indicated by the arrow. The surface concentrations are provided by Eq. 4a and 4b and are indicated by the red circles. The corresponding series of (b) bulk (IR–Raman hybrid) and (c) surface (SFG) spectra. Surface–bulk heterospectral correlation analysis showing the (d) synchronous and (e) asynchronous maps. In all spectra, the vertical dashed lines are colored corresponding to the assignment of the pure spectral components in Figure 1.
For both bulk and surface spectra, vertical lines mark the wavenumbers of characteristic bands originating from the individual molecules, using colors corresponding to those shown in Figure 1. We now turn to the heterospectral 2D-COS analysis presented in Figures 3d and 3e, and first note that the unusual situation where the synchronous and asynchronous maps identical shapes for all bands. (In this case, the signs of the synchronous and asynchronous peaks are the same too, but we will later see an example where the signs are all opposite to each other.) This situation occurs because, in the absence of molecular reorientation, the SFG and bulk IR–Raman spectra are just scaled according to the corresponding surface and bulk populations. This is especially evident in our simulated spectra as there is no noise but in practice, the same would be observed experimentally if none of the species reoriented. We will come back to the point about surface reorientation later. But first, we seek to identify glycerol or toluene bands in relatively uncongested spectral regions where we can be more sure of the band assignment. In this case, it is straightforward to look at toluene aromatic C–H stretching, as those signals uniquely appear above 3000 cm−1. In the absence of surface reorientation, we can study any asynchronous toluene peak in Figure 3e, including diagonal peaks. We notice that the diagonal peak near 3040 cm−1 is negative (blue), and the corresponding synchronous peak is also negative. Note that the 3060 cm−1 peak is positive in both maps and has a different sign from the 3040 cm−1 feature due to the difference in sign in the SFG response. Either of those toluene modes can be used for this analysis, where Noda’s rules 28 indicate that the bulk toluene concentration increases before the surface toluene concentration increases. The surface avoids toluene, characterized by cases 1, 2, and 4. In order to narrow down the possibilities among these three cases, at least one additional perturbation is required.
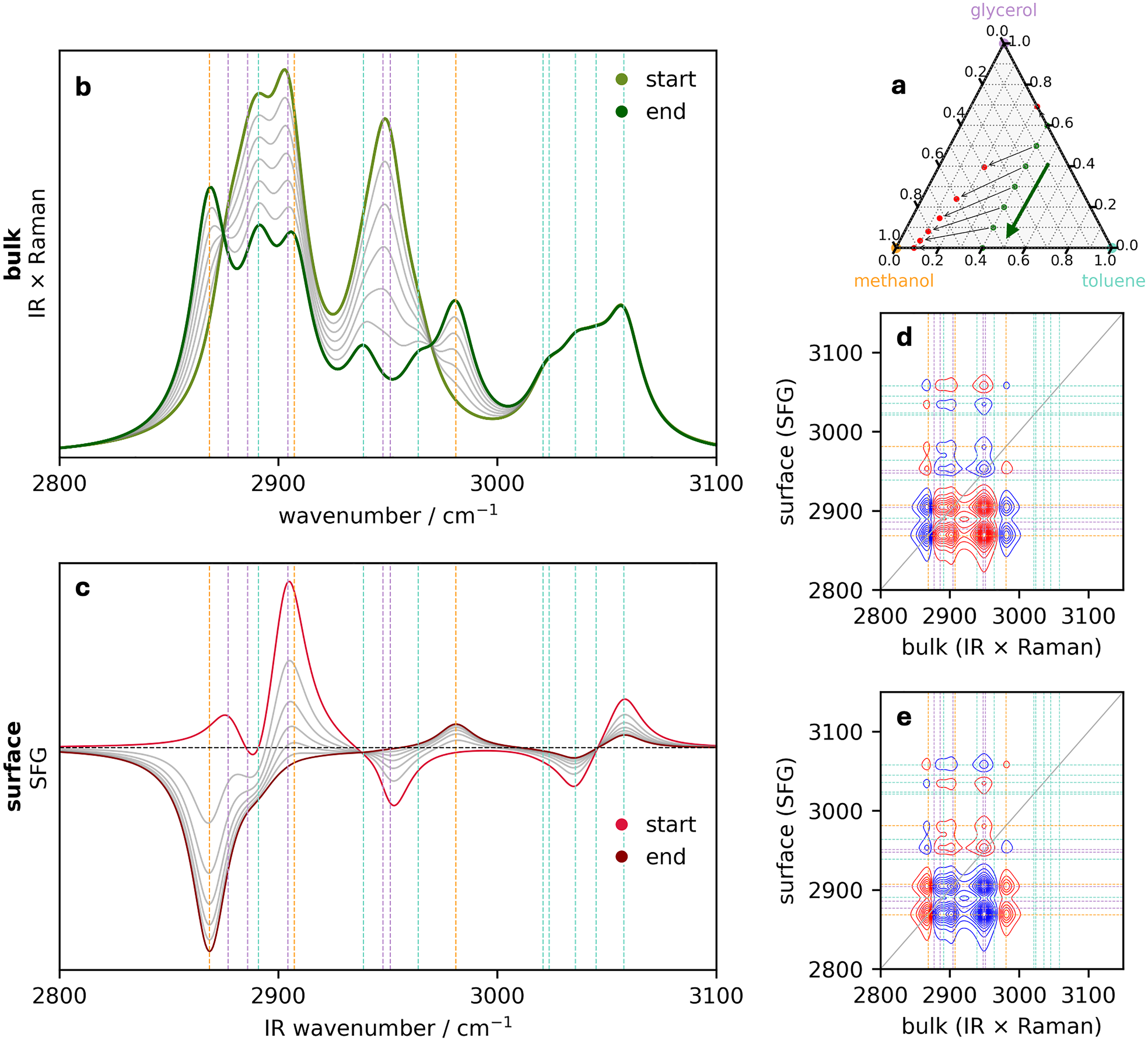
Perturbation 2: The bulk toluene mole fraction remains fixed. (a) Ternary diagram illustrating the initial bulk concentrations of the three components (green circles) that define the perturbation along the direction indicated by the arrow. The surface concentrations are provided by Eq. 4a and 4b and are indicated by the red circles. The corresponding series of (b) bulk (IR–Raman hybrid) and (c) surface (SFG) spectra. Surface–bulk heterospectral correlation analysis showing the (d) synchronous and (e) asynchronous maps. In all spectra, the vertical dashed lines are colored corresponding to the assignment of the pure spectral components in Figure 1.
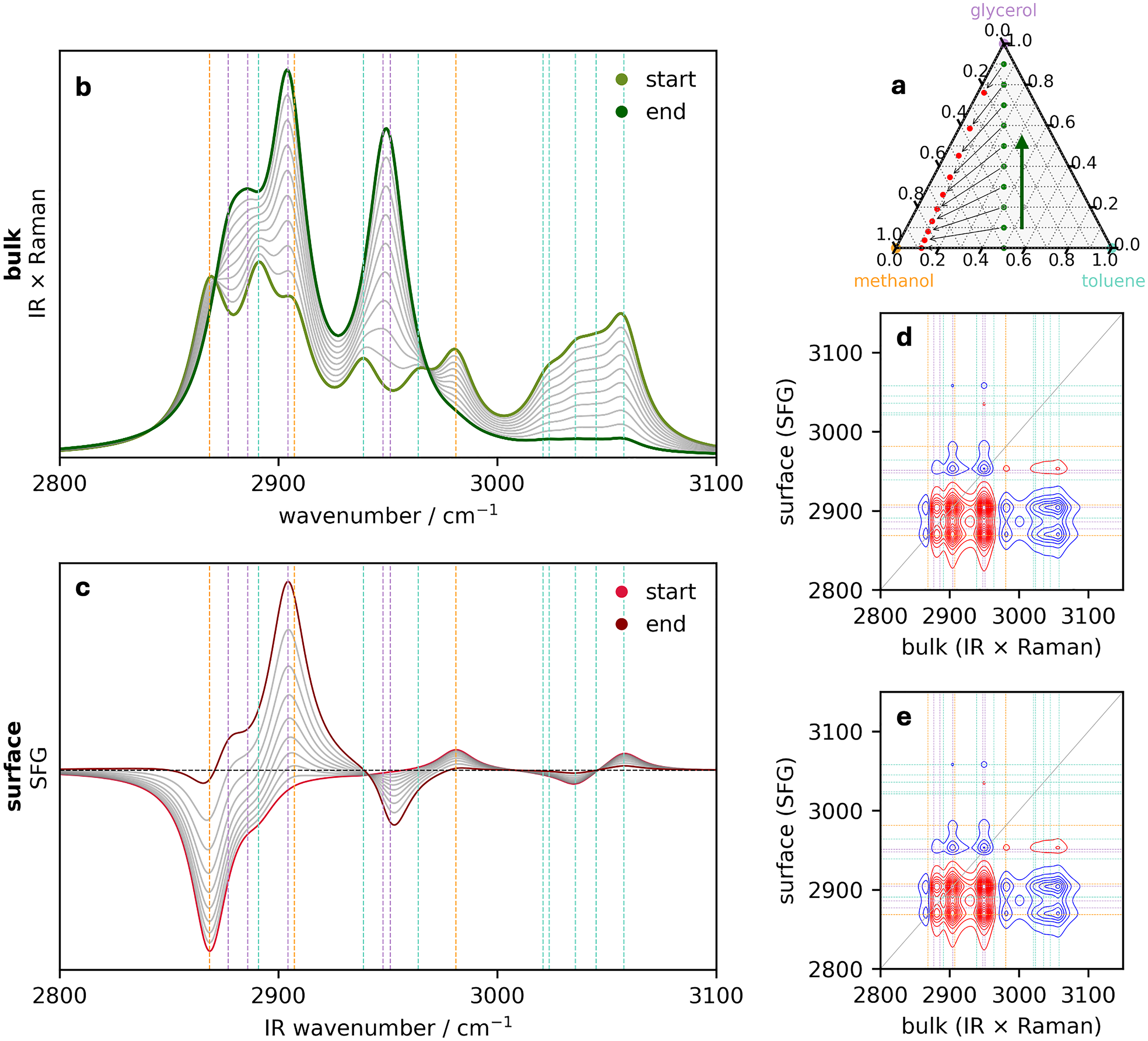
Perturbation 3: Methanol/toluene ratio remains fixed. (a) Ternary diagram illustrating the initial bulk concentrations of the three components (green circles) that define the perturbation along the direction indicated by the arrow. The surface concentrations are provided by Eqs. 4a and 4b and are indicated by the red circles. The corresponding series of (b) bulk (IR–Raman hybrid) and (c) surface (SFG) spectra. Surface–bulk heterospectral correlation analysis showing the (d) synchronous and (e) asynchronous maps. In all spectra, the vertical dashed lines are colored corresponding to the assignment of the pure spectral components in Figure 1.
Additional Considerations
Throughout the discussion, we have lumped together cases where the equilibrium constants or their ratio are greater/less than or equal to unity, but we have not described the situations where
One of the primary motivations of this work has been to understand the surface preference for selectively adsorbing specific molecules from a bulk liquid. In the case of a binary system, such a conclusion directly follows from the application of the rules we have discussed for interpreting heterospectral surface–bulk 2D-COS maps. However, there are many possibilities for how species are adsorbed/desorbed and interconverted in ternary systems. In our chosen system (Eq. 1), the characterization of the set of equilibrium constants resulted in six possibilities, and we have demonstrated how a judicious choice of up to three perturbations can be used to isolate one of the six combinations. However, some of those combinations do not yield conclusive information on the surface preference. Looking at Table I, we can see that the surface preference is determined for cases 1, 4, and 6. In other words, in those three combinations of
Another point in general concerns the reorientation of species when their surface population changes. This is relatively common, for example, as the packing density of adsorbed monolayers often moves their tilt angle to more upright orientations. In our earlier description of binary systems, 26 we noted that if one of the two species reoriented on the surface (as evidenced by SFG homospectral asynchronous features between two vibrational modes corresponding to the same rigid moiety), then it may still be possible to have a full surface preference determination based on the other species. In the case of ternary systems, this is still true. If one component does not reorient in each experiment, it may still be possible to navigate the flowchart in Figure 2 and arrive at a conclusion. Further details of this situation may be investigated in future work.
Conclusion
We have provided a framework where IR and/or Raman spectra can be used together with vibrational SFG spectra to characterize a ternary system with the aid of heterospectral 2D-COS. The primary merit of this approach is that the direction of the equilibria can be determined solely from the signs of the heterospectral asynchronous peaks. This work therefore provides an initial step toward the understanding of more complex adsorption phenomena and adsorption dynamics in ternary systems. This preliminary study provides a scientific foundation for optimizing material design, improving industrial separation techniques, and understanding reaction kinetics in complex mixtures.
Footnotes
Acknowledgments
This work was enabled by facilities at the University of Victoria Centre for Advanced Materials and Related Technologies (CAMTEC). We thank Harpreet Kaur for insightful discussions on the applications and utility of ternary liquid adsorption.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Discovery (RGPIN-2020-06030) and Discovery Accelerator (RGPAS-2020-00049) grants from the Natural Sciences and Engineering Research Council of Canada.