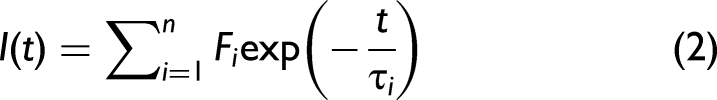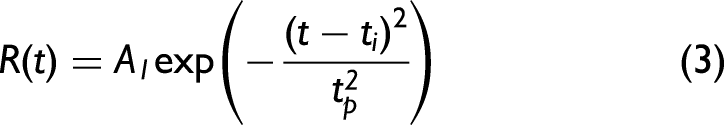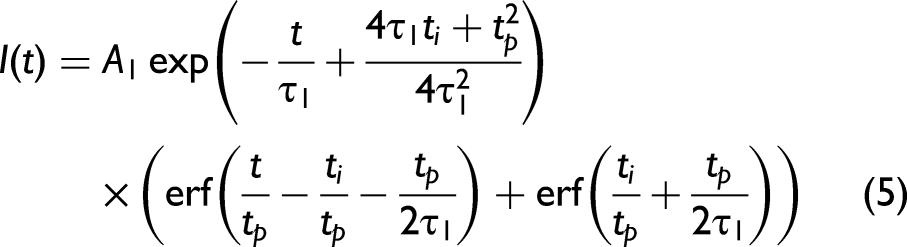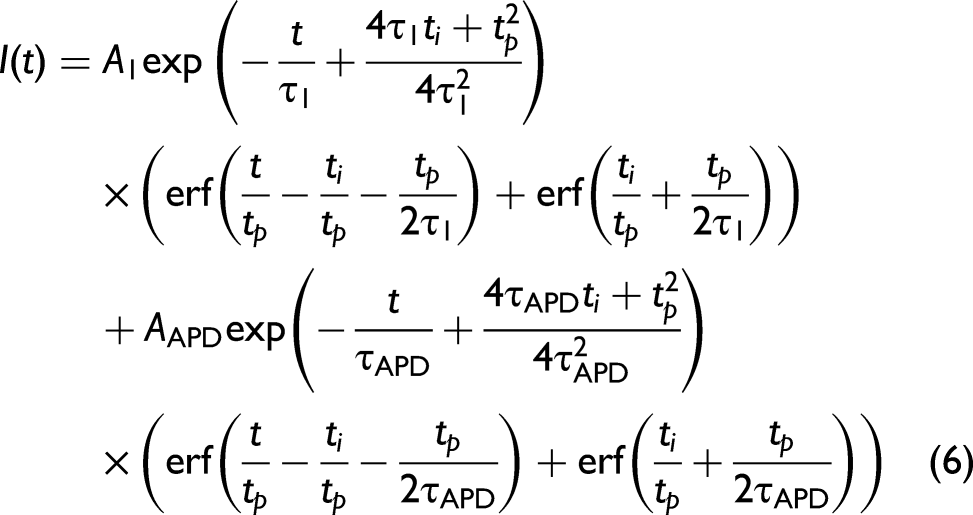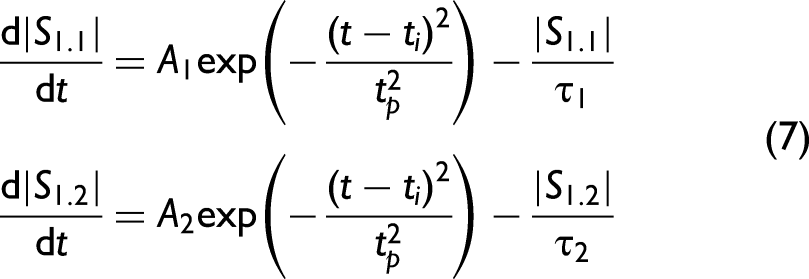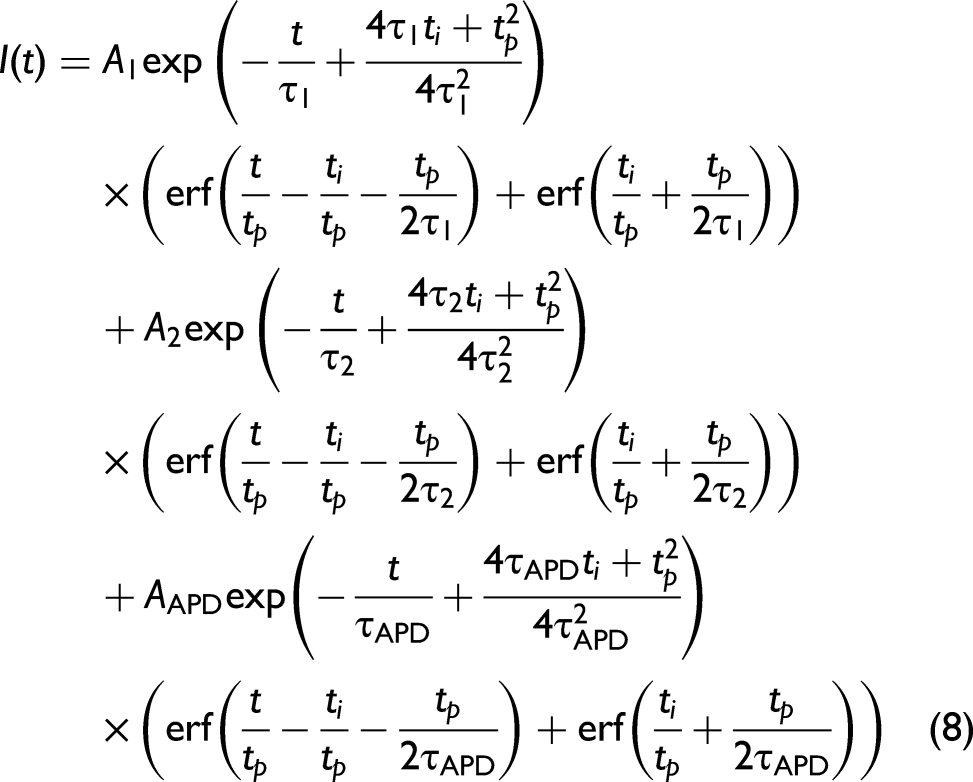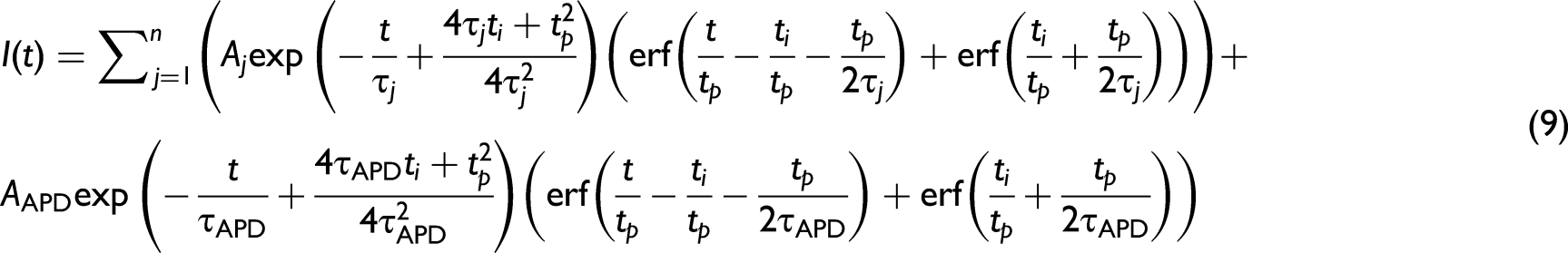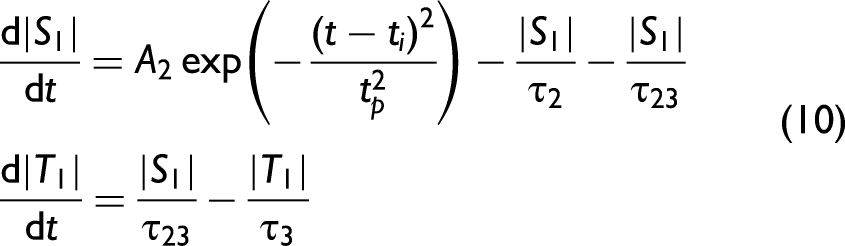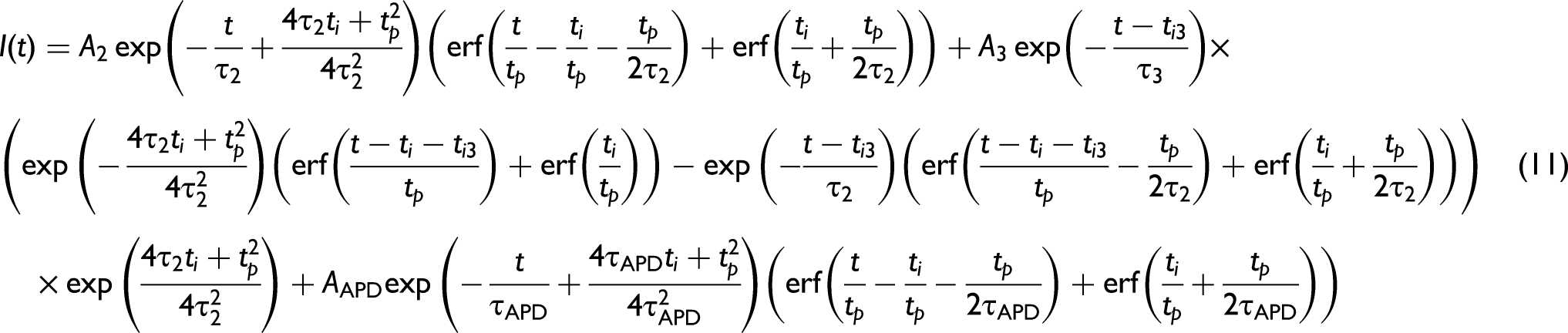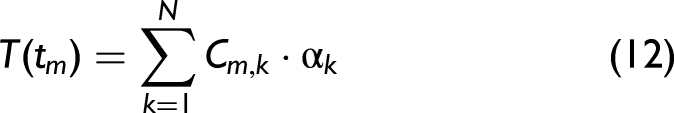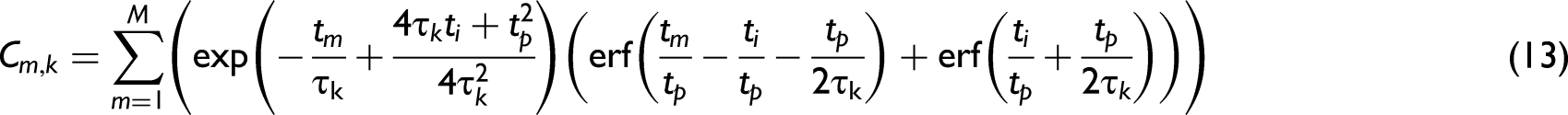Abstract
This paper presents a novel analytical technique for evaluating fluorescence lifetimes excited by a nanosecond pulsed laser using a linearized rate equation approach that accounts for the incident pulse temporal distribution, an equivalent instrument response function, and non-exponential fluorescence decay which limits the application of traditional fluorescence lifetime techniques in stand-off applications. The approach is applied to model the fluorescence of a variety of pharmaceutical powders and phosphorescing samples exhibiting non-exponential decay and compared to results obtained with the maximum entropy method. Fluorescence lifetimes are found to be 3–5 ns, typical for organic fluorescent powders, and phosphorescence lifetimes were on the order of hundreds of nanoseconds. The approach also shows potential for determining the composition of mixed samples and can be readily extended to model increasingly complex scenarios with additional fluorescing or phosphorescing components.
This is a visual representation of the abstract.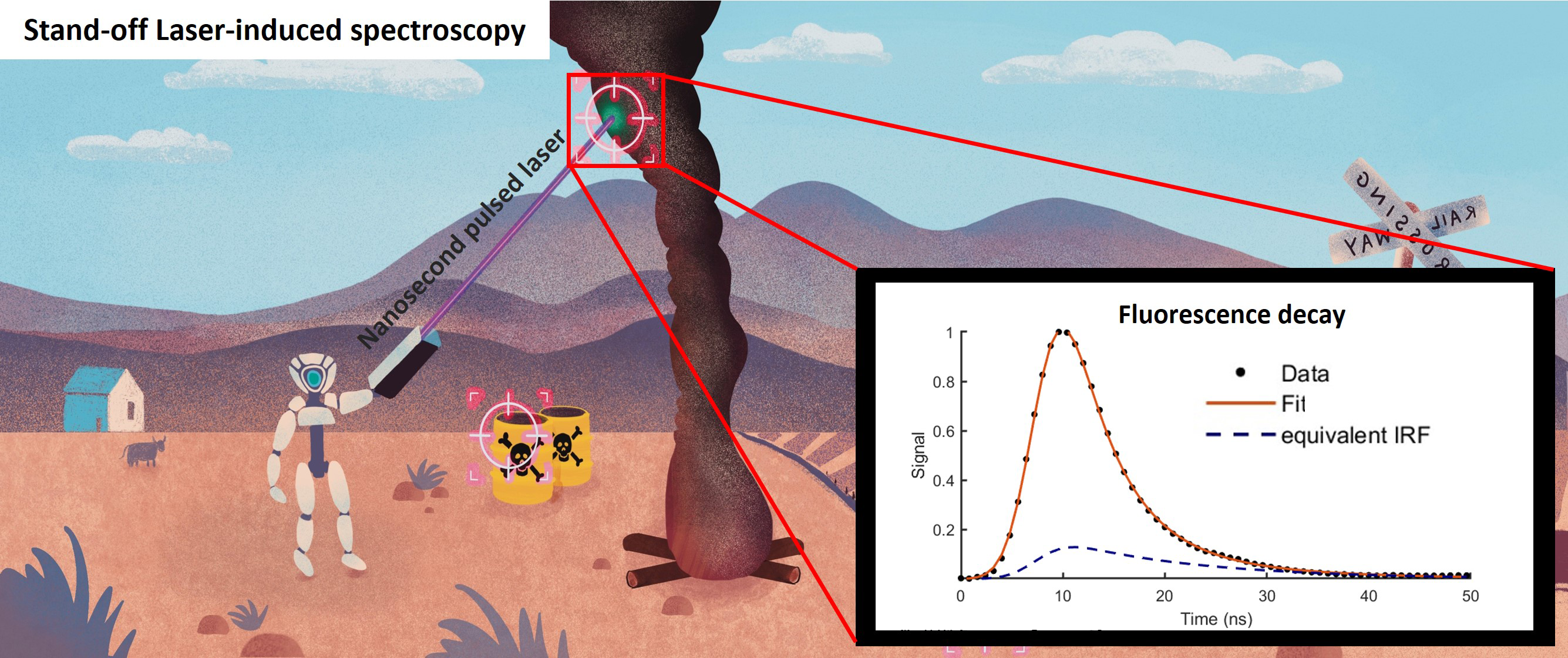
Keywords
Introduction
Laser-induced fluorescence (LIF) has been used since the advent of the laser in the 1960s to identify molecules through a measure of the number, wavelength, and time which photons are emitted after excitation. While it is a relatively mature diagnostic technique, over the last 10 years LIF has generated significant interest in security and bioterrorism applications1–3 such as aerosol monitoring4–11 and the identification of pharmaceutical products and drugs.12–23 It is also extensively used for food quality monitoring,24,25 the detection of crude oils,26–28 and biological materials in planetary exploration. 29
Intrinsic or autofluorescence is a type of fluorescence which results from fluorophores often found in biological materials which fluoresce naturally without requiring the addition of fluorescent dyes or enhancers. 30 Fluorescence occurs when a molecule is excited by incident light, with the relaxation process resulting in photon emission at a shifted wavelength. Materials and compounds can be identified by analyzing the spectral composition or temporal decay of fluorescence, or a combination of the two which has been shown to improve classification accuracy. 3
Although the fluorescence lifetime of bulk materials such as clinical or pharmaceutical samples has often been determined by fitting a single exponential to the emitted fluorescence,23,31–33 it has also been shown that the lifetime of fluorophores such as tryptophan or samples with multiple fluorescing components are better determined using double or triple exponentials.25,34–40 However, studies as early as the 1980s have shown that although adding additional exponential terms can provide a more complete description each added term introduces a greater margin of error, and furthermore there exists samples which exhibit non-exponential decay.34,35 Some alternative strategies that have been used to model fluorescence lifetime include the use of stretched exponentials, 41 limiting the measurement to capture decay from only short-lived fluorophores in a particular time range, 42 or making use of computational analytical procedures which aim for identification rather than lifetime measurements. 43 Furthermore, gated methods have seen some success, 44 as well as convolving the profile of an excitation source for a laser with a theoretical model function and then comparing to the experimental data.35–37,45 The maximum entropy method (MEM) was developed to more accurately determine multiple lifetime components simultaneously when the number of decay components present are unknown.46–49 This is achieved by modelling the fluorescence decay as a weighted sum of exponentials and determining a weight distribution of lifetime components which best satisfies the observed data. However, as with other techniques, MEM can be sensitive to the initial parameters and initialization values. Furthermore, this method works best when experimental data contains discrete lifetime values rather than a continuous distribution.
A challenge with many fluorescence lifetime measurement techniques such as time correlated single photon counting (TCSPC)38,39,50 is the determination of an instrument response function (IRF) to a zero-lifetime sample. The IRF must be considered in signal analysis to ensure accurate fluorescence lifetime determination and avoid introducing systematic errors that may confuse material identification.44,51 Deconvolutional analysis which is commonly used to account for the IRF is only valid when the excitation pulse width is much shorter than the fastest measured decay time. 44 The use of pico- or femto-second lasers can make IRF deconvolution more practical, however such laser sources are expensive and impractical for field use or when stand-off detection is required. Also, current deconvolution methods are numerical techniques, which can limit accuracy and can make it difficult to correlate results with underlying environmental conditions such as sample composition and age. 51
For high accuracy laboratory-based measurement of fluorescence, photomultiplier tubes (PMTs) or single photon avalanche diodes (SPADs) are employed due to their high sensitivity. However, SPADs are not practical for stand-off applications as they are easily saturated. 52 PMTs have excellent detection efficiency in the ultraviolet (UV) region, however this decreases in the visible and infrared regions. 53 Thus, for a sensor which aims to identify materials over a broad range of wavelengths, compact avalanche photodiodes (APDs) that can operate under ambient conditions in the field are often used.54,55 However, a drawback with APDs is that they have a wavelength dependent temporal response due to the diffusion of carriers below the active junction which results in a wavelength dependent exponential tail in the IRF.44,45,55 While this can be corrected to some degree by approximating the IRF using the temporal profile of a scattered laser pulse, or calibration using a fluorophore with a known lifetime, systematic errors can be difficult to remove completely.45,55
In this paper we present an analytical model based on linearized population rate equations that describes the temporal evolution of sample fluorescence from the time that the laser pulse first interacts with the sample. This allows accurate determination of fluorescence lifetimes including for samples that exhibit non-exponential fluorescence decay. This approach conveniently accounts for the wavelength dependent IRF of the APD making it suitable in stand-off LIF devices to identify materials where convolution or deconvolution methods are impractical. We demonstrate the effectiveness of the method by modelling the fluorescence decay of various pharmaceutical powders and phosphorescent samples which demonstrate non-exponential decay when excited by nanosecond pulsed light. Our model also has the potential to allow the composition of mixed samples in some situations to be determined and can be expanded to reveal lifetimes when multiple fluorophores or phosphorescence is present. Our technique provides a practical approach to fluorescence lifetime determination for stand-off material identification which can be utilized in field settings.
Theoretical Framework
Fluorescence lifetime is a measure of how long a fluorophore emits light after excitation and offers a means to differentiate and identify materials. Electron pairs in the ground singlet state (S0) with opposite spins can be excited to an excited singlet state (S1). In some cases, an excited triplet state (T1) is also possible where the molecule must undergo intersystem crossing (ISC) to produce unpaired electrons with the same spin, although this process is relatively rare as this transition is symmetry forbidden. Emission from other excited states (S2) is also possible though less common in biological molecules. 44 These fluorescence and phosphorescence pathways are shown in Figure 1.
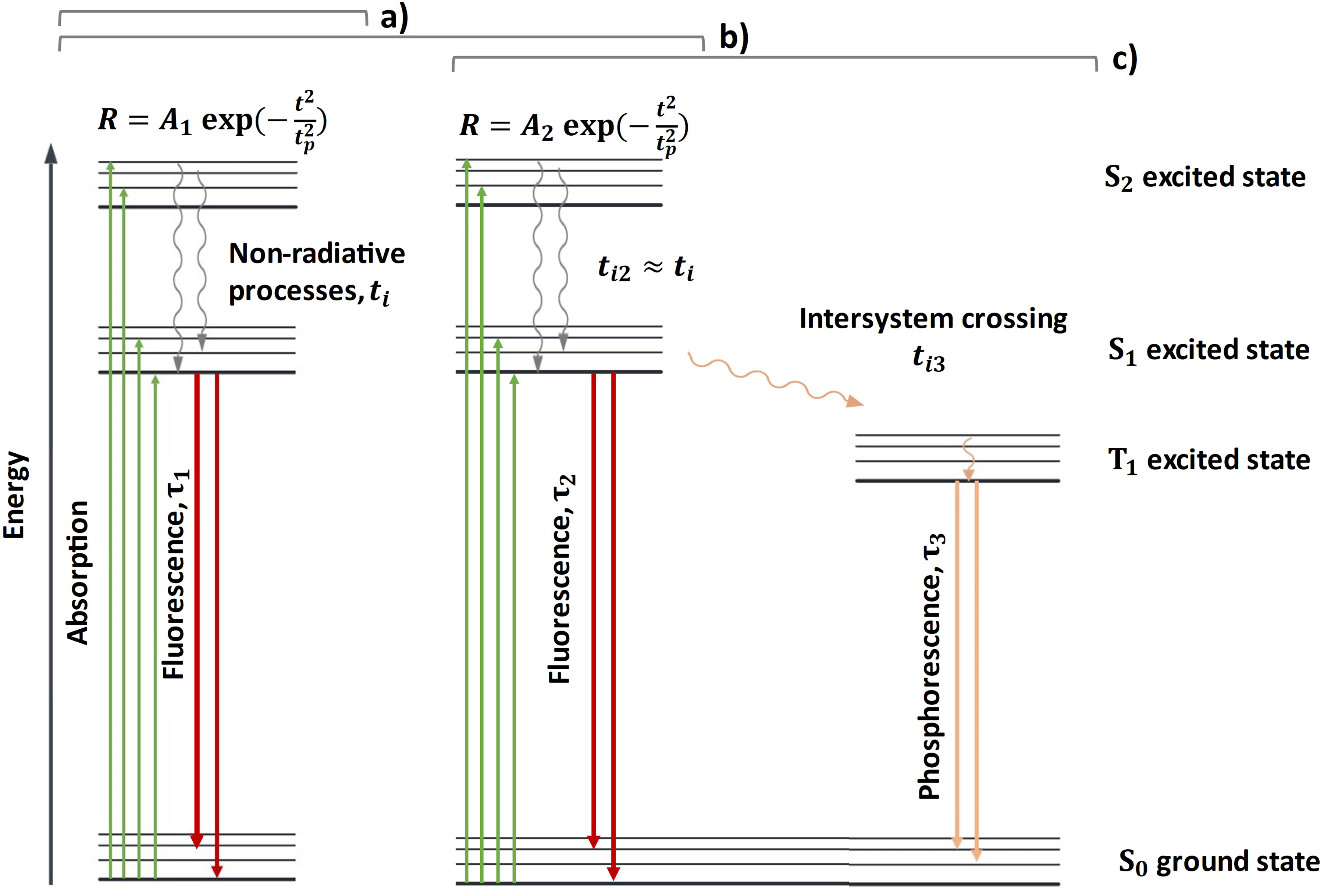
Energy diagram showing a molecule containing (a) a single fluorescing component, (b) multiple fluorescing components, and (c) demonstrating phosphorescence.
There is some contradicting evidence regarding typical decay factors and how best to model them. Fluorescence is inherently multiexponential as substances generally contain a combination of fluorophores which each have distinct lifetimes contributing fractional amounts to the total lifetime.
30
A typical model which can be used to broadly characterize molecules as organic or inorganic is given by Eq. 1.
In the case where multiple, n, fluorophores may be present, Eq. 1 can be rewritten with individual lifetimes
If a nanosecond pulsed laser is used as the excitation source, fluorescence from the sample will have commenced while the pulse is still pumping more of the population into the upper state. This impacts the temporal distribution of the emitted fluorescence which will not be strictly exponential and depend on the incident pulse duration and temporal distribution, often assumed to be Gaussian.
In principle, the fluorescence lifetime could be extracted by determining the IRF and numerically deconvolving it with the raw data or equivalent. However as discussed, these approaches have practical limitations for both stand-off measurements undertaken in the field and lab-scale measurements, arising from noise in the detection scheme and distorted signals. Hence, here we present a model which does not utilize deconvolution and accounts for the temporal distribution of the laser pulse and the APD IRF and can be applied to various fluorescing and phosphorescing systems.
Single Fluorescing Systems
Figure 1 demonstrates some of the relaxation pathways after a population has been optically pumped at a rate R into an upper state. In the simplest case, when a single fluorophore is present without an accessible triplet state, the number of molecules entering an excited state is proportional to the laser pumping rate equation derived from a Gaussian temporal pulse, as given by Eq. 3.
For a single fluorescing system as shown in Figure 1a, the pumped population will decay via fast non-radiative decay to the lowest excited level of the fluorescence band
Solving Eq. 4 and re-writing in terms of fluorescence signal intensity
Equation 5 can be further adjusted to incorporate the APD response through the inclusion of a second exponential component as given by Eq. 6. This approximates the average effect of multiple wavelengths interacting with the APD due to the sample fluorescence, where it is known that the wavelength dependent tail for an APD tends to be exponential for each wavelength.
44
Multiple Fluorophores
This approach can be readily extended to model a material which contains multiple fluorophores. The fluorescence intensities of these fluorophores are independent of each other and occur simultaneously as separate processes. In this situation the linearized rate equations for the lower excited level of each of the two fluorescence bands are given by:
This can be further expanded to fit any number of fluorescing components,
Fluorescing and Phosphorescing Systems
The approach can also be applied to the situation where ISC to a lower energy triplet state is available, and thus a molecule exhibits both fluorescence and phosphorescence. In this case, the rate equations describing the fluorescence and the phosphorescence can be written as
Determining Lifetimes Using Least Squares Fitting and Maximum Entropy Method
Least squares fitting can be used with Eqs. 6, 9, and 11 to determine suitable values for lifetimes and other parameters which fit with the experimental data. This can be easily achieved using various software packages. However, fitting a large number of parameters may result in large uncertainties and results that are not physically meaningful. Furthermore, there could be multiple solutions that provide a good fit. Thus here, MEM was used to confirm results.
A detailed outline of the MEM method is described by Esposito et al.
46
In brief, for each data point, m, in the time resolved fluorescence decay data a theoretical function
The MEM method then aims to select a distribution of lifetimes and weights which maximizes the Skilling entropy function, as outlined by Esposito et al. 46
Experimental
Materials and Methods
A schematic of the experimental setup used is shown in Figure 2. Fluorescence data was obtained using a nanosecond pulsed laser (Quantel Ultra, 355 nm, 20 Hz, 4 ns pulse duration,
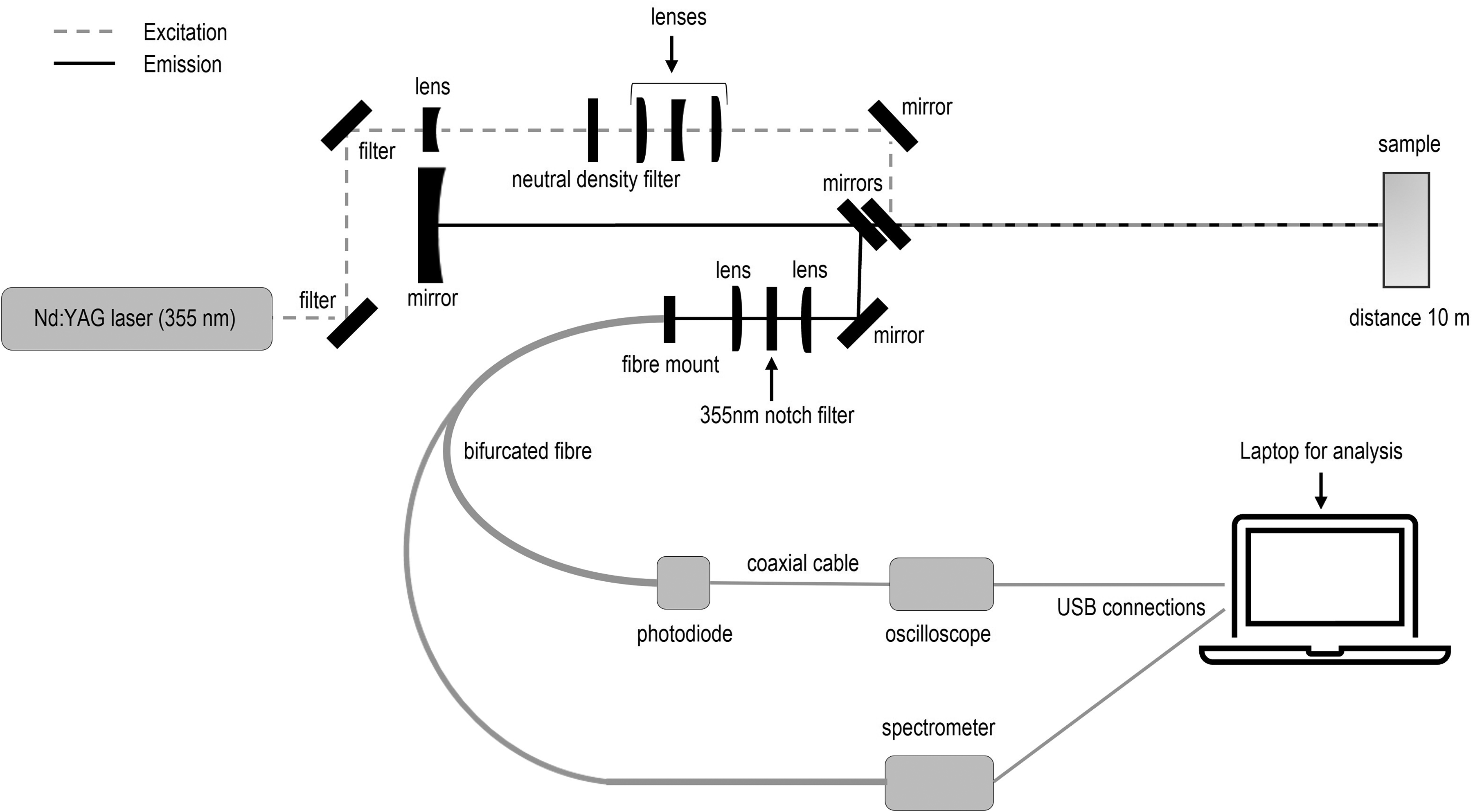
The experimental setup used to obtain fluorescence spectra and lifetime decay profiles. It includes a dedicated optical unit for stand-off spectroscopy that combines light delivery and capture.
In this investigation the temporal fluorescence decay signal captured was the sample emission across the entire 200 nm to 900 nm spectral range of the APD. To demonstrate that the approach presented here can negate the impact of the wavelength dependent IRF regardless of the spectral composition of a material’s fluorescence, the collected signal was split using a bifurcated fiber to simultaneously capture the temporal fluorescence decay and the fluorescence spectra resolved with a QE Pro (Ocean Optics) spectrometer. For very precise determination of individual fluorophore lifetimes in a laboratory setting it may be useful to make use of filters to capture the decay for a smaller region of wavelengths which can be more readily contributed to a single fluorophore. However, this requires prior knowledge of the material being investigated. While this is possible in laboratory settings where the aim is to measure and perhaps quantify a specific sample, here no spectral filters were used so as not to limit the range of materials that can be identified in stand-off mode.
The fluorescence decay for a variety of commercially available pharmaceutical samples were modelled using an average of 1000–2000 laser pulses. These included a pure aspirin sample, two types of coated aspirin (duentric and enteric), paracetamol, rash powder with maize starch, and a talcum powder. A summary of their excipients can be found in Table I. These were all prepared in powder form and placed in low-fluorescence quartz cuvettes. Commercially obtained phosphorescing water-based textile ink containing pigmented polymers, commercial zinc sulphide powder with dopants, and vinyl plastic containing phosphors were also used to model phosphorescence and non-exponential decay.
Summary of pharmaceutical samples and their lifetimes.

Results and Discussion
Table I shows the lifetimes for the bulk pharmaceutical powders determined from experimental data using least-squares fitting, which are within the range expected for similar samples.
Figure 3 shows the temporal variation of the fluorescence excited at 355 nm predicted by Eq. 6 for the aspirin sample using least squares fitting. The oscillations observed in the residuals have been previously reported when using APDs and are the result of high signal and APD gain
54
but since their net average is zero they do not introduce systematic errors to the fit. Previously it has been shown that a visual inspection of the fit and residuals often provides a good determination on the quality of the fit to given data
44
in situations where it is not possible to use the
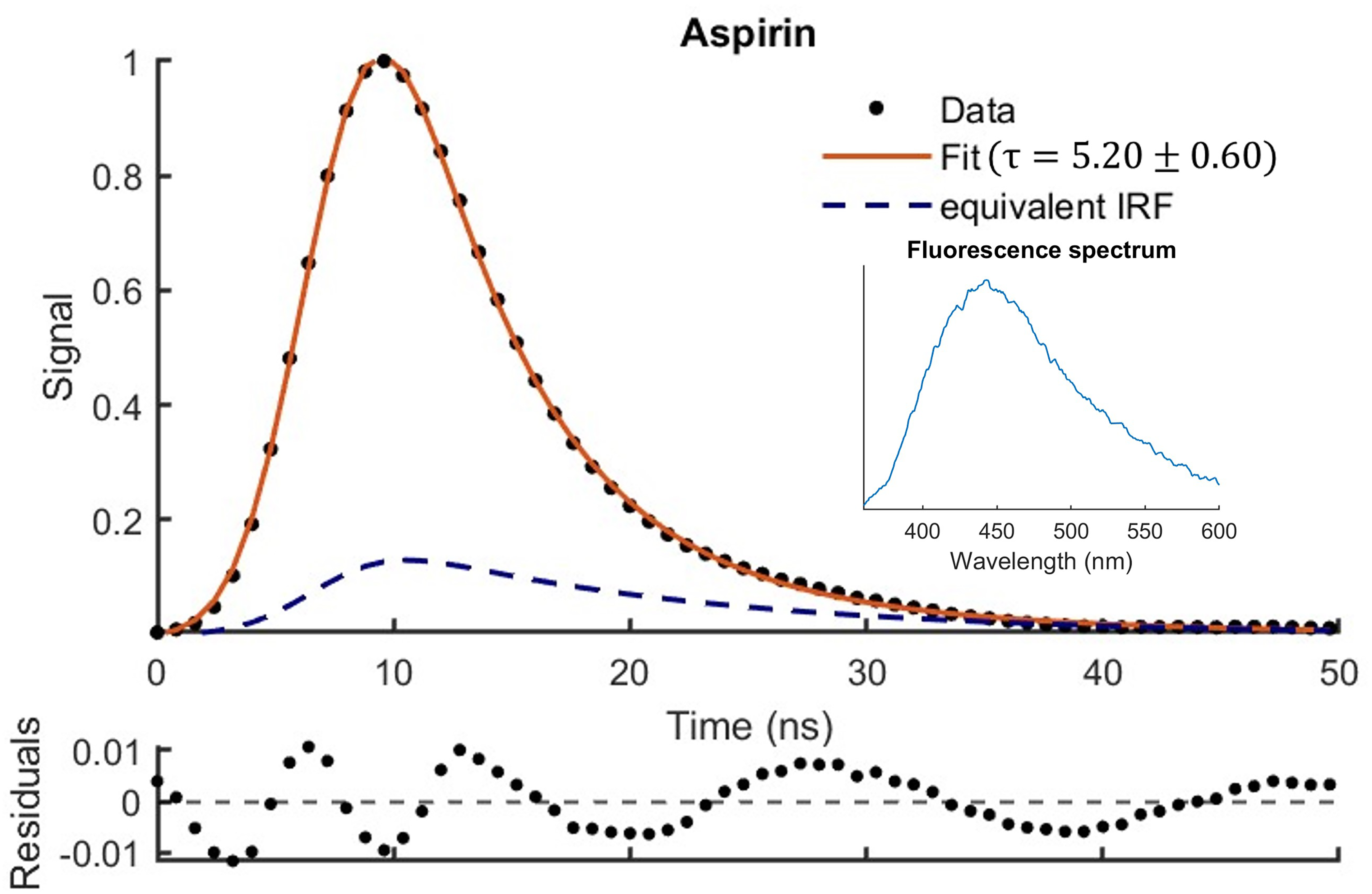
Fluorescence decay for the aspirin sample modelled using Eq. 6. The fluorescence spectrum is also shown.
Equation 6 can also be used to fit the fluorescence decay of samples with more complex compositions, such as the enteric and paracetamol samples with the results given in Table I. Paracetamol, with acetaminophen as the active ingredient, had a lifetime of
Equation 9 was used to model the decay of mixed fluorescing samples. This proved somewhat difficult if all parameters are unconstrained as the complexity of the model results in several possible solutions, and furthermore previous studies have shown difficulty in constraining fit parameters for complex models such as triple exponentials.25,50 The number of possible solutions were greatly reduced by constraining the parameters
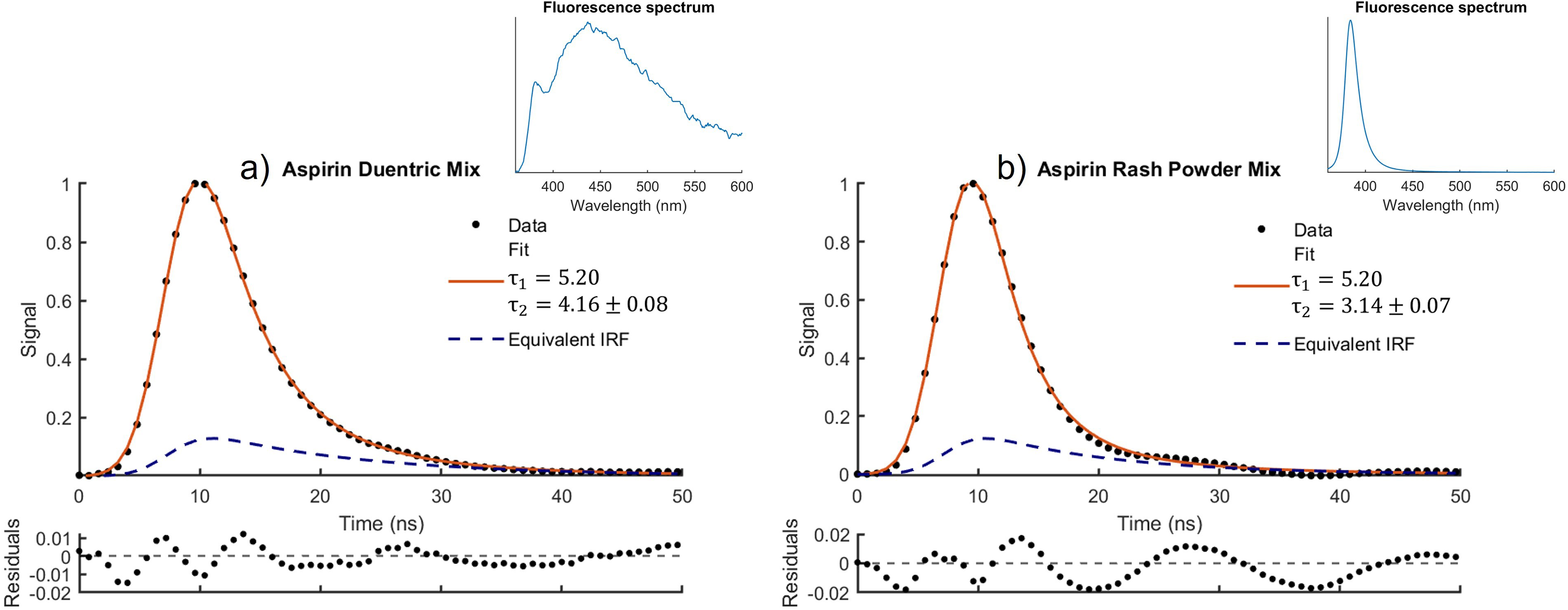
Fluorescence spectra and decay for mixtures of (a) aspirin and duentric and (b) aspirin and rash powder modelled using Eq. 9.
MEM was used to verify the results obtained through least-squares fitting. Using this method for the aspirin sample, the lifetime probability density value peaked at
Using MEM did not prove successful for separating the mixtures, once again likely due to the continuous nature of the lifetime distribution and the broad range of wavelengths over which the emission was captured, as well as the short separation between the expected lifetimes for the mixed components. However, the maximum probability density for each of the mixtures ranged from 4–6 ns. This is slightly longer than the lifetimes obtained using Eq. 9, where the artificial lengthening likely comes from the APD wavelength dependent tail. A sample of olive oil was found to fluoresce at both 355 nm and 532 nm with differing lifetimes. By exciting the sample with both wavelengths simultaneously we were able to use MEM to obtain two lifetimes of approximately 3.2 ns and 9.8 ns. This shows that under some circumstances MEM can be used to separate lifetime components over a broad range of wavelengths.
The spectra and temporal decay of simultaneously fluorescing and phosphorescing samples of water-based textile ink containing pigmented polymers, commercial zinc sulphide powder with dopants, and vinyl plastic containing phosphors are shown in Figure 5. Eq. 11 was used to fit to the combined fluorescence and phosphorescence of each of these samples. The phosphorescence and fluorescence lifetimes were found to be
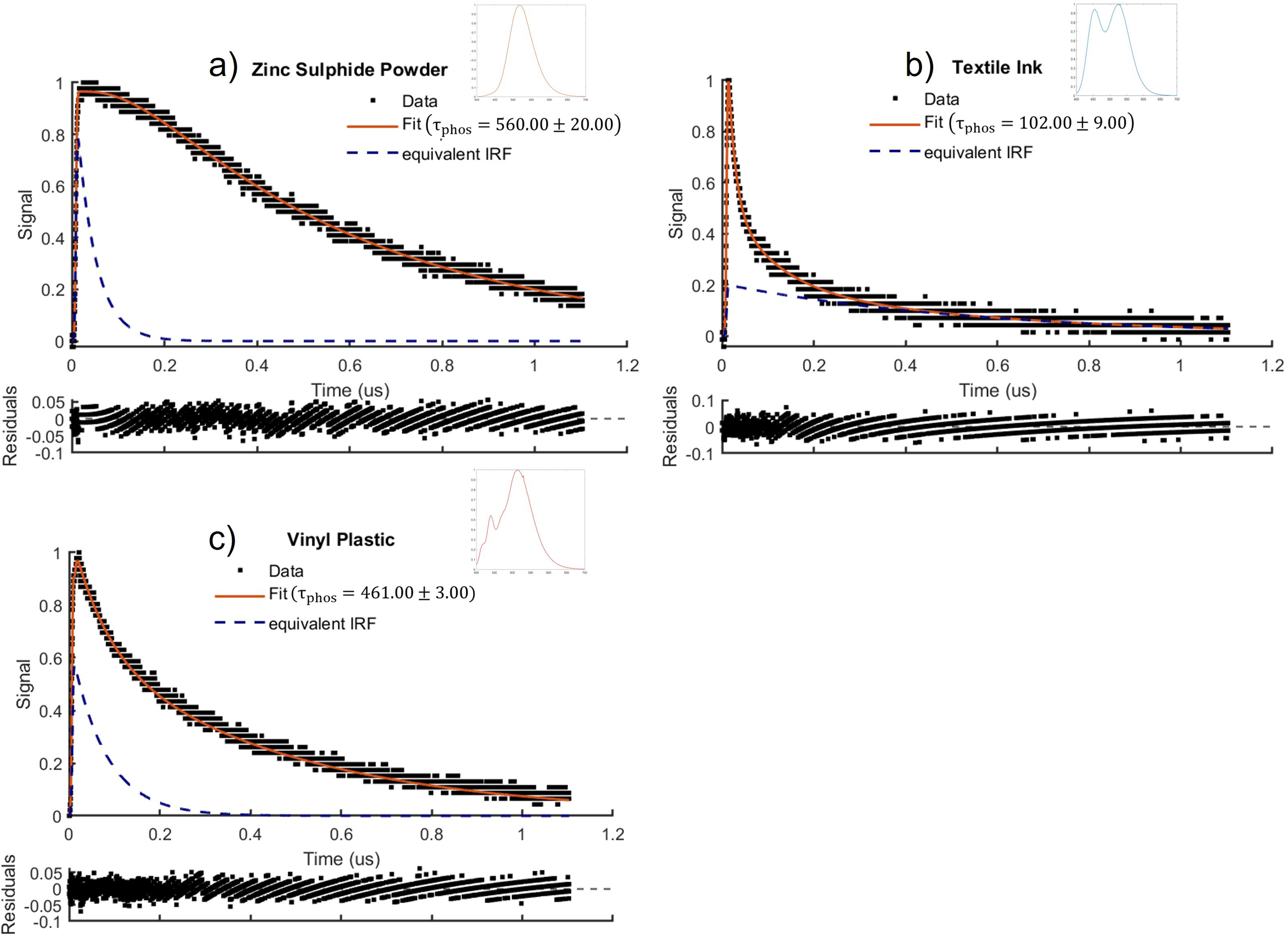
Phosphorescence decay modelled by Eq. 11 and fluorescence spectra for (a) zinc sulphide powder, (b) textile ink, (c) vinyl plastic.
Equation 6 was used to fit to a variety of bulk samples using each individual pulse to observe the spread of calculated lifetimes. This can be seen in Figure 6, where the signal has not been normalized. An equal volume of each sample was used, and as such the amplitude values
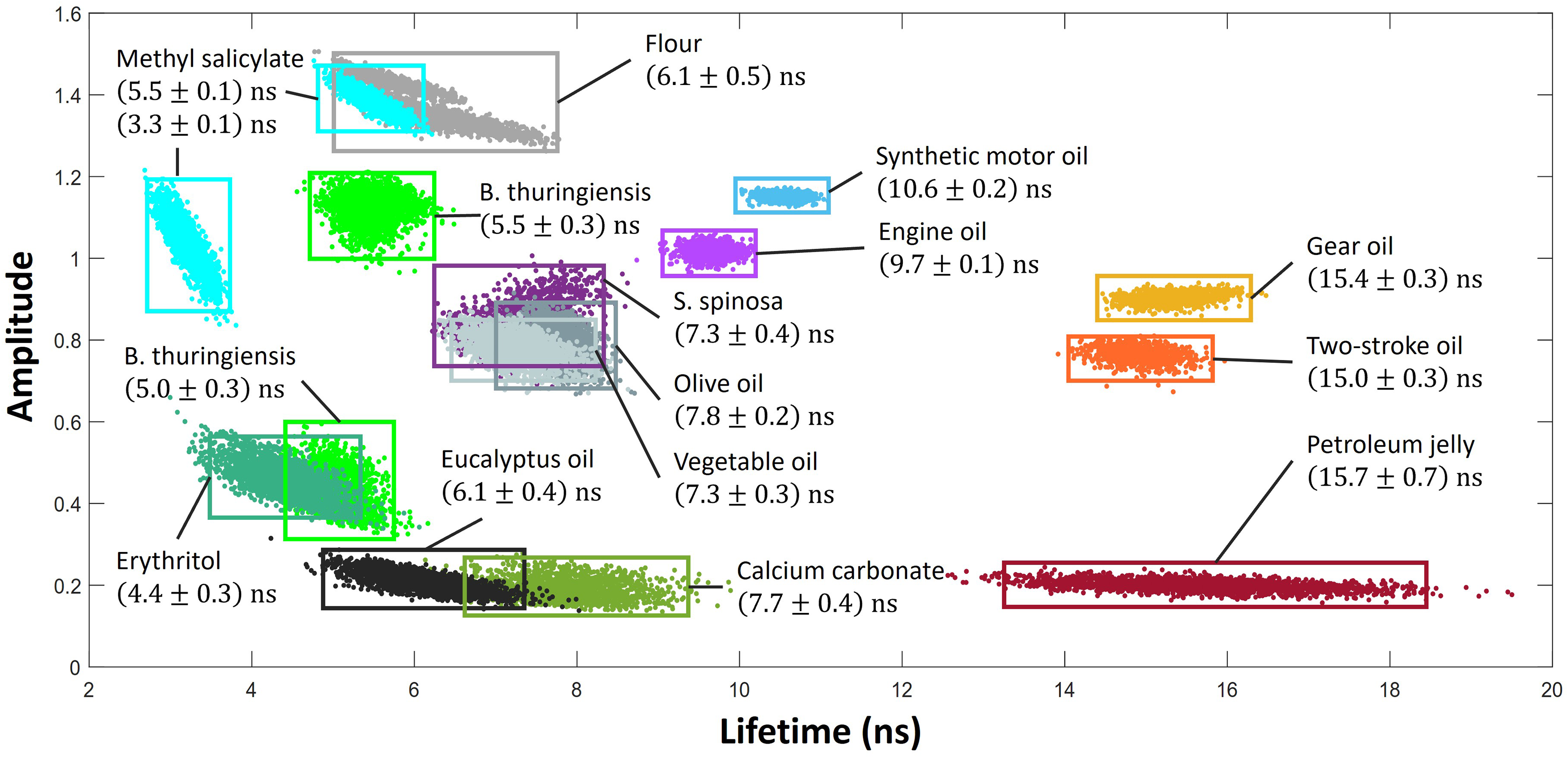
A selection of samples treated as bulk material with fluorescence decay curves modelled using Eq. 6 with least squares fitting, with the quoted lifetimes calculated from an average of 2000 pulses.
Conclusion
We have shown that our presented analytical model can be used to fit to raw temporal fluorescence data to determine lifetime values
In future work, we aim to use fluorescence lifetime determination with an APD as a supplementary method to spectrometers for sample classification and identification in situations where fluorescence spectra are difficult to interpret, and additional information is required to aid in identification, without significantly increasing the footprint of the sensor. There is also potential to use this as an alternative, stand-alone technique for situations where the size, weight, and cost of the sensor is of utmost importance. Furthermore, using this set-up as a stand-alone technique is beneficial as it consists of a simple circuit attached to a diode which does not require specialized software to interpret spectra. This will increase the deployability and robustness of stand-off LIF sensors that are used to identify possible hazards in the field.
Footnotes
Consent to Participate
There are no human participants in this article and informed consent is not required.
Consent for Publication
Not applicable.
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was conducted with the support of the Australian Commonwealth Department of Defence and the Operating in a CBRN Environments STaR Shot Academy.
Ethical Considerations
This article does not contain any studies with human or animal participants.