Abstract
Physiological systems are not at equilibrium and undergo time-dependent fluctuations, making it challenging to relate in vitro studies to in vivo biomolecular behavior. To bridge this gap, enzyme dynamics can be studied in the presence of controlled perturbations that recapitulate the intracellular environment. Here, we report an approach to the study of reactive oxygen species (ROS) based on the in situ manipulation of hydrogen peroxide (H2O2) levels in functionalized nanopore-based electrochemical zero-mode waveguide (EZMW) arrays, with each nanopore presenting small numbers of immobilized horseradish peroxidase (HRP) enzyme molecules. H2O2 is generated or consumed within the attoliter volume of the EZMW nanopores by poising an embedded ring electrode to suitable potentials, and the resulting effect on apparent turnover of HRP under non-equilibrium conditions is monitored using the enzymatically accelerated conversion of the non-fluorescent probe Amplex Red to fluorescent resorufin. A Nafion membrane is placed on the top surface of the EZMW array, providing a cation permselective barrier to transport in, or out, of the EZMW nanopores, thereby improving the sensitivity of the experiment by sequestering enzymatically generated resorufin in the attoliter volume of the EZMW nanopores. By fabricating arrays presenting 441 individual reaction volumes in parallel, distinct changes in population dynamics in the presence of in situ H2O2 generation or consumption are characterized with respect to temporal evolution and magnitude of the H2O2 aliquot delivered. This approach presents a promising avenue for studying biomolecular reactions in spatiotemporally controlled chemical environments that can mimic the non-equilibrium conditions encountered in vivo.
This is a visual representation of the abstract.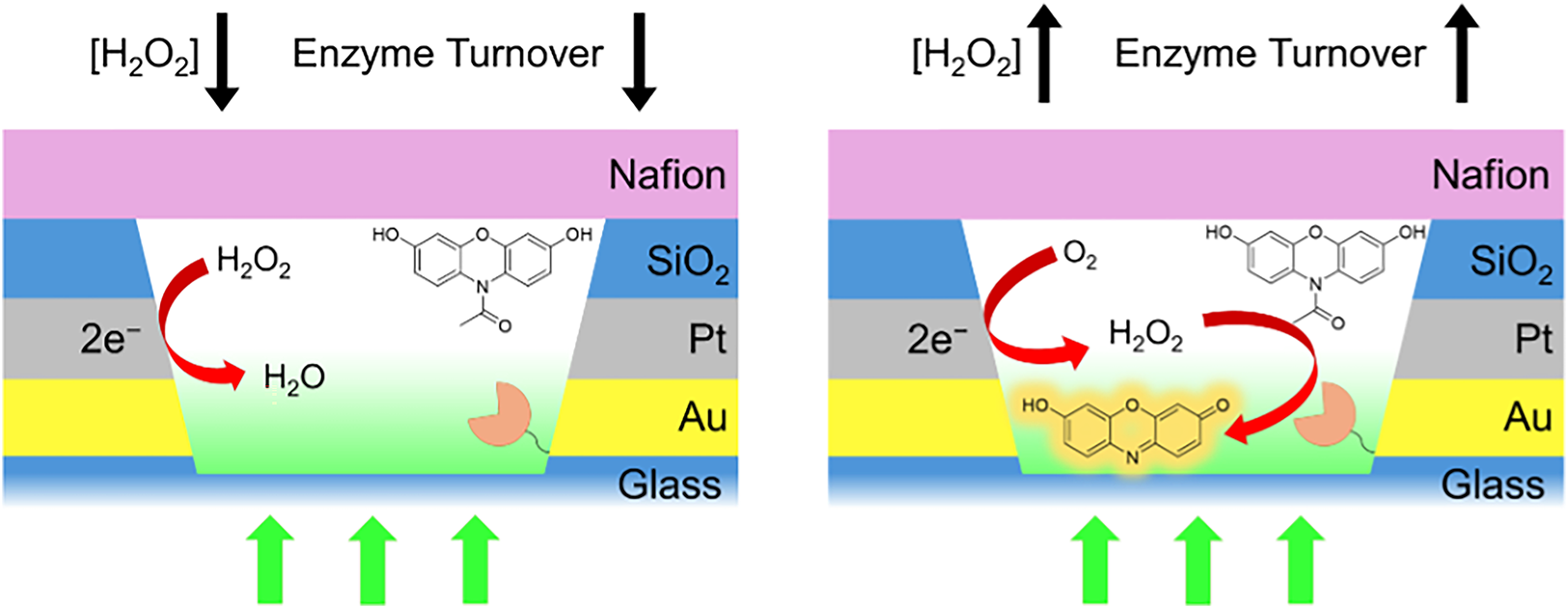
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
