Abstract
Deep eutectic solvents (DESs) exhibit dynamic heterogeneity, where the intricate and dynamic hydrogen bonding within the DES mediates dynamic spatial variation in the DES local environment. The Type III DES composed of choline chloride and glycerol (ChCl:Gly) exhibits this effect prominently, and we report on the observed local organization and its dependence on system composition using the time-resolved reorientation dynamics of three illustrative chromophores of different polarities: perylene (neutral, nonpolar), oxazine 725 (cation, polar) and rose bengal (dianion, polar). Our findings demonstrate that the environments sensed by all three chromophores are markedly different than that predicted by the bulk viscosity of the DES, and that these local environments exhibit remarkably little change as the mole ratio of the DES constituents is varied. Taken collectively, these data provide clear evidence of short-range organization that bears very little resemblance to the longer-range structural organization that determines DES bulk properties.
This is a visual representation of the abstract.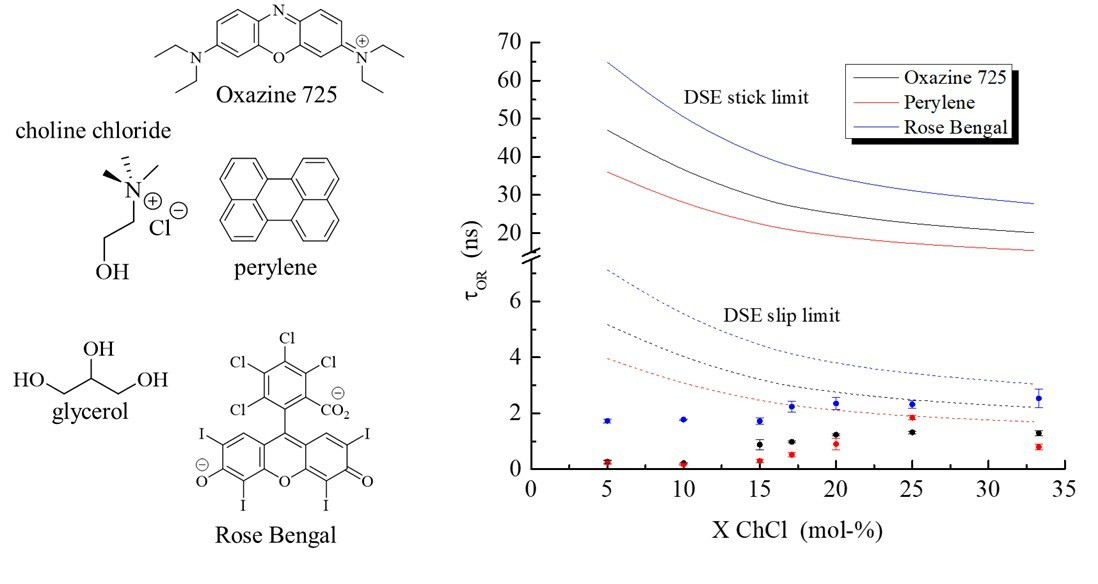
Get full access to this article
View all access options for this article.
