Abstract
Discrete frequency infrared (IR) imaging is an exciting experimental technique that has shown promise in various applications in biomedical science. This technique often involves acquiring IR absorptive images at specific frequencies of interest that enable pathologically relevant chemical contrast. However, certain applications, such as tracking the spatial variations in protein secondary structure of tissue specimens, necessary for the characterization of neurodegenerative diseases, require deeper analysis of spectral data. In such cases, the conventional analytical approach involves band fitting the hyperspectral data to extract the relative populations of different structures through their fitted areas under the curve (AUC). While Gaussian spectral fitting for one spectrum is viable, expanding that to an image with millions of pixels, as often applicable for tissue specimens, becomes a computationally expensive process. Alternatives like principal component analysis (PCA) are less structurally interpretable and incompatible with sparsely sampled data. Furthermore, this detracts from the key advantages of discrete frequency imaging by necessitating the acquisition of more finely sampled spectral data that is optimal for curve fitting, resulting in significantly longer data acquisition times, larger datasets, and additional computational overhead. In this work, we demonstrate that a simple two-step regressive neural network model can be utilized to mitigate these challenges and employ discrete frequency imaging for retrieving the results from band fitting without significant loss of fidelity. Our model reduces the data acquisition time nearly six-fold by requiring only seven wavenumbers to accurately interpolate spectral information at a higher resolution and subsequently using the upscaled spectra to accurately predict the component AUCs, which is more than 3000 times faster than spectral fitting. Our approach thus drastically cuts down the data acquisition and analysis time and predicts key differences in protein structure that can be vital towards broadening potential applications of discrete frequency imaging.
This is a visual representation of the abstract.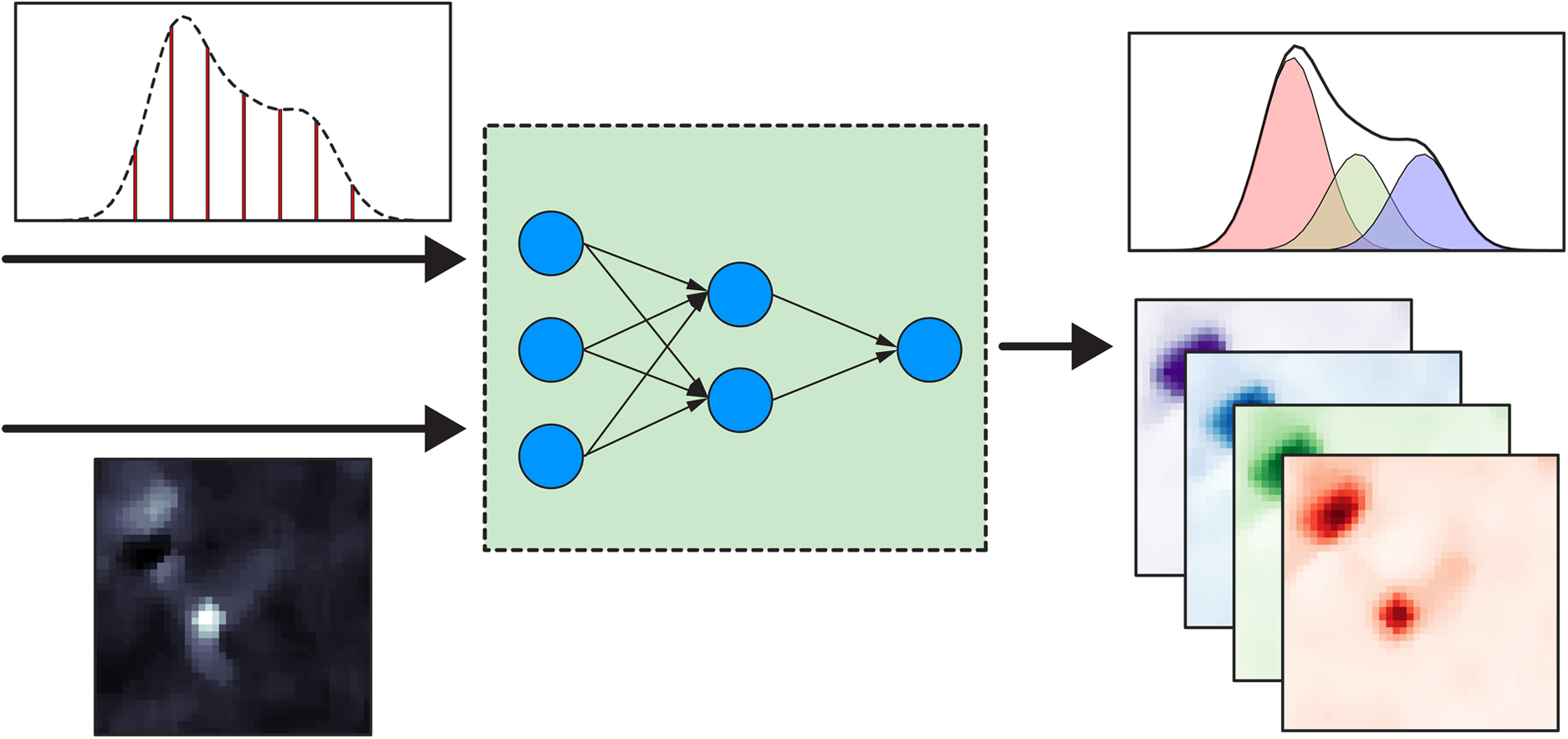
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
