Abstract
This study introduces two novel sandwich-type tungsten–oxygen cluster compounds synthesized by hydrothermal methods, H4(C6H12N2H2)3{Na(H2O)2[Mn2(H2O)(GeW9O34)]}2 (Compound 1) and H2(C6H12N2H2)3.5{Na3(H2O)4[Co2(H2O)(GeW9O34)]2}·17H2O (Compound 2). The two compounds comprise cluster anions [GeW9O34]10– coordinated with transition metal atoms, either Mn or Co, and are stabilized by organic ligands. These compounds are crystallized in the hexagonal crystal system and P63/m space group. The two compounds were characterized through various techniques. Fourier transform infrared (IR) spectroscopy showed absorption peaks of anionic backbone vibrations of the Keggin cluster at 500–1000 cm–1, IR spectral peaks of δ(N–H) and νas(C–N) of the ligand triethylenediamine at 1000–2000 cm–1, and IR spectral peaks of the ligand νas(N–H) and νas(O–H) of water at 3000–3500 cm–1. Despite similar one-dimensional (1D) IR spectra due to the same cluster anions and similar molecular structures, the two compounds exhibited distinct responses in two-dimensional correlation spectroscopy with IR under magnetic and thermal perturbations. Under magnetic perturbation, Compound 1 showed a strong response peak for νas(W–Ob–W), while Compound 2 exhibited a strong response peak for νas(W=Od), possibly linked to differing magnetic particles. Similarly, Compound 1 displayed a strong response peak under thermal perturbation for νas(W–Oc–W). In contrast, Compound 2 showed a strong response peak for νas(W=Od); these results may be attributed to the different hydrogen bonding connections between the two compounds, which affect the groups in distinct ways through vibration and transmit these vibrations to the W–O bonds. The research presented in this paper expands the theoretical and experimental data of 2D correlation IR spectroscopy.
This is a visual representation of the abstract.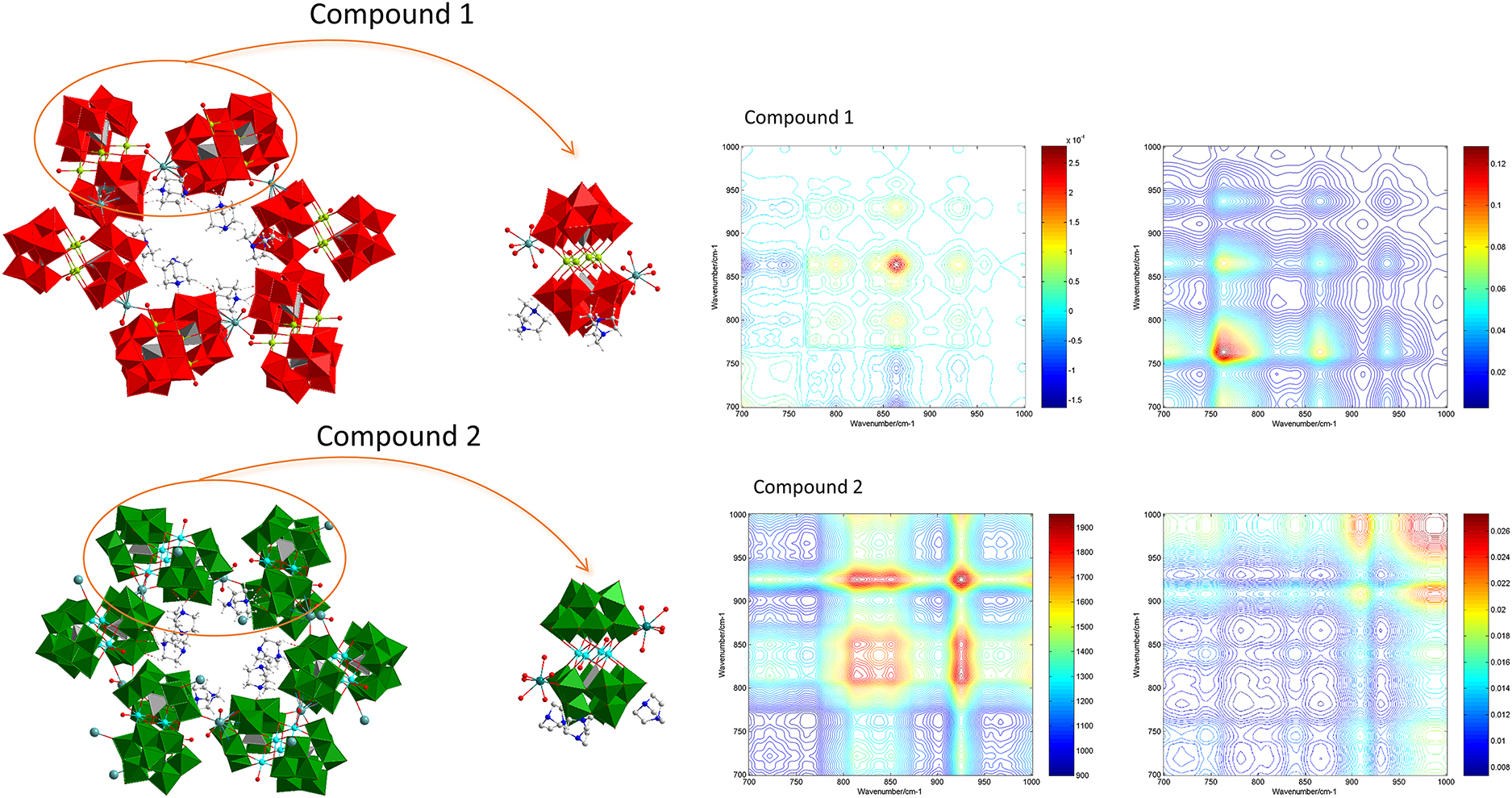
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
