Abstract
In this work, we present studies on the effect of laser wavelengths on the laser-induced plasma characterization using a femtosecond (fs) ytterbium-doped potassium-gadolinium tungstate (Yb:KGW) laser. Plasma plumes of copper, steel, ceramics, and glass samples were induced using a multiple shot of 200 fs laser pulses with 1030 nm and 343 nm wavelengths at fixed laser fluence (
Keywords
Introduction
Laser-induced breakdown spectroscopy (LIBS) is a versatile tool widely used for the elemental analysis of various materials, both in situ and in the laboratory.1,2 The use of ultrashort femtosecond (fs) pulses for LIBS has many advantages compared to long pulses. One of them is the reduced heat-affected zone, which is proportional to the square root of the pulse duration.3,4 Other advantages of fs-LIBS are reduced optical continuum emission, generation of colder plasma, less susceptibility to sampling matrix and elemental fractional effects, non-gated detection, better spatial and depth resolution, lower ablation threshold, and better repeatability.2,5 Fs laser pulses are perfect for remote or stand-off detection because they can form filaments and are able to deliver high energies over long distances. They have been used in remote atmospheric sensing and pollution detection,6,7 analysis of chemical and biological agents, 8 cultural heritage monitoring, 9 metal analysis, 10 and the detection of explosives,11,12 etc.13,14
An important parameter of LIBS is the laser wavelength used for plasma generation on the sample surface. The dependence of laser-induced plasma parameters on wavelength has been studied before, but mainly using nanosecond laser pulses. Most of the studies presented below used nanosecond laser pulses.
Diaz et al. 15 found that the main parameters of LIBS (plasma temperature and electron density) were almost the same for both 1064 and 355 nm wavelengths of the neodymium-doped yttrium aluminum garnet (Nd:YAG) laser using 56 mJ pulse energy on gold- and silver-bearing minerals, but the repeatability of measurements increased by 50% with 355 nm wavelength. There were also other studies that have observed better repeatability with the shorter laser wavelengths.16–18
Amoruso et al. 19 evaluated absorption mechanisms in aluminum laser-ablated plasmas, and found that, at the visible laser wavelength (532 nm) of the Nd:YAG laser, in the first stages of the laser–vapor interaction, ionization is mainly caused by an inverse bremsstrahlung process due to electron scattering by neutrals and subsequent collisional ionization, whereas in the ultraviolet (UV), photoionization of excited species plays the most important role. In both cases, the plasma acts as a shield for the laser radiation, thus absorbing a part of the laser pulse. 20 However, shorter pulses (on the order of picoseconds rather than nanoseconds) have been reported to be more effective in ablating the mass, probably because plasma shielding does not have such a strong effect at shorter pulses. 21
Qaisar and Pert 22 investigated the effect of laser wavelength on the ablation process by comparing UV KrF excimer and infrared Nd:YAG lasers. It was discovered that plasma screening is more effective for longer-wavelength lasers, whereas shorter-wavelength lasers are more beneficial for ablation. Other studies confirm these results.16,21,23–25 Qaisar and Pert 22 also observed that a longer infrared (IR) wavelength produces a higher temperature but lower density plasma, as compared to shorter UV wavelength-induced plasma. However, Abdellatif and Imam 26 obtained different results, where the UV wavelength of an Nd:YAG laser produced both a higher temperature and higher electron density of the plasma on aluminum, as compared with the IR wavelength.
Kasem et al. 17 established that the use of the UV wavelength of the Nd:YAG laser with 22 mJ pulse energy results in more stable plasma, which gave more realistic elemental results of the archaeological bone plasma than longer IR wavelengths. They also observed that the use of UV laser radiation reduces fractionation effects, which can cause the ratios of the elements in a plume to differ from the stoichiometry of the sample. Other studies confirm these results.27,28 However, Huber et al. 29 concluded that the IR wavelength is better at finding heavy metal impurities in polymer materials.
Fornarini et al.
30
found that, when using Nd:YAG laser radiation in the UV range for LIBS on bronze samples, there is almost no dependence of the plasma parameters on fluence, which was varied from 50 to 250 J/cm
Aberkane et al. 31 investigated the influence of a 50 mJ Nd:YAG laser with wavelengths 1064, 532, and 355 nm at 7, 9, and 10 ns pulse durations, respectively, on the relationship between Te and surface hardness of Fe–V–C metallic alloys. They determined that employing an excitation laser in the IR range rather than in the UV range is more efficient for measuring surface hardness using LIBS.
Studies on the effects of an appropriate combination of laser wavelengths and pulse energies on the characterization of generated plasma on a pure copper sample using a single shot picosecond (170 ps) Nd:YAG laser were performed by Fikry et al. 32 It was found that the copper emission line intensities (SNR), plasma temperature, and density show a strong dependence on the laser wavelength and pulse energy. The electron temperature and electron density values gradually increase with the increase of the laser wavelength and pulse fluence. The researchers explained this tendency by the fact that using picosecond pulses enhances the plasma shielding effect, which is due to the inverse Bremsstrahlung being the dominant absorption mechanism, not photoionization. Therefore, most of the laser energy is absorbed by the free electrons of the target, and more energetic electrons are preserved, which causes an increase in the electron temperature and electron density with the increase of the picosecond laser wavelength and pulse energy.
A comparison of mid-infrared (2.05
The choice of wavelength for LIBS measurements is thought to be more important for longer pulses, closer to the regime of nanoseconds, but in the case of fs-LIBS, the pulse–matter interaction is different, the pulse duration is much shorter than the material heat transfer time, plasma forms after the pulse, eliminating the problem of plasma shielding. However, the dependence on fs UV and IR LIBS has not been extensively studied.
In this work, two different fs laser wavelengths of 1030 and 343 nm were used to determine the effect of wavelength on LIBS parameters. We found that using both IR and UV laser wavelengths results in plasma of roughly the same temperature, electron density, and intensity of spectral lines, but the SNR is significantly increased using UV laser wavelength for plasma generation.
Experimental Setup
The experimental scheme for our LIBS measurements is shown in Figure 1. In our experiment, we used the “Pharos” fs ytterbium-doped potassium-gadolinium tungstate (Yb:KGW) laser (Light Conversion Ltd.) with a pulse duration of 200 fs, average power up to 6 W at 1030 nm and pulse repetition rate of 6 kHz. The fundamental harmonic (1030 nm) was used for IR radiation. For the generation of UV radiation, the fundamental wavelength was first frequency doubled (515 nm), and then the sum frequency generated (343 nm) using nonlinear beta barium borate crystals. High reflectivity mirrors (for 343 nm) M4–M7 were used to filter out the first (1030 nm) and second (515 nm) harmonics, so that only the third harmonic (343 nm) remained. M1–M3 mirrors were used for the first harmonic and M2–M3 mirrors were used only during the IR laser wavelength experiment. The fluence on the sample using both UV and IR laser wavelengths was kept constant at
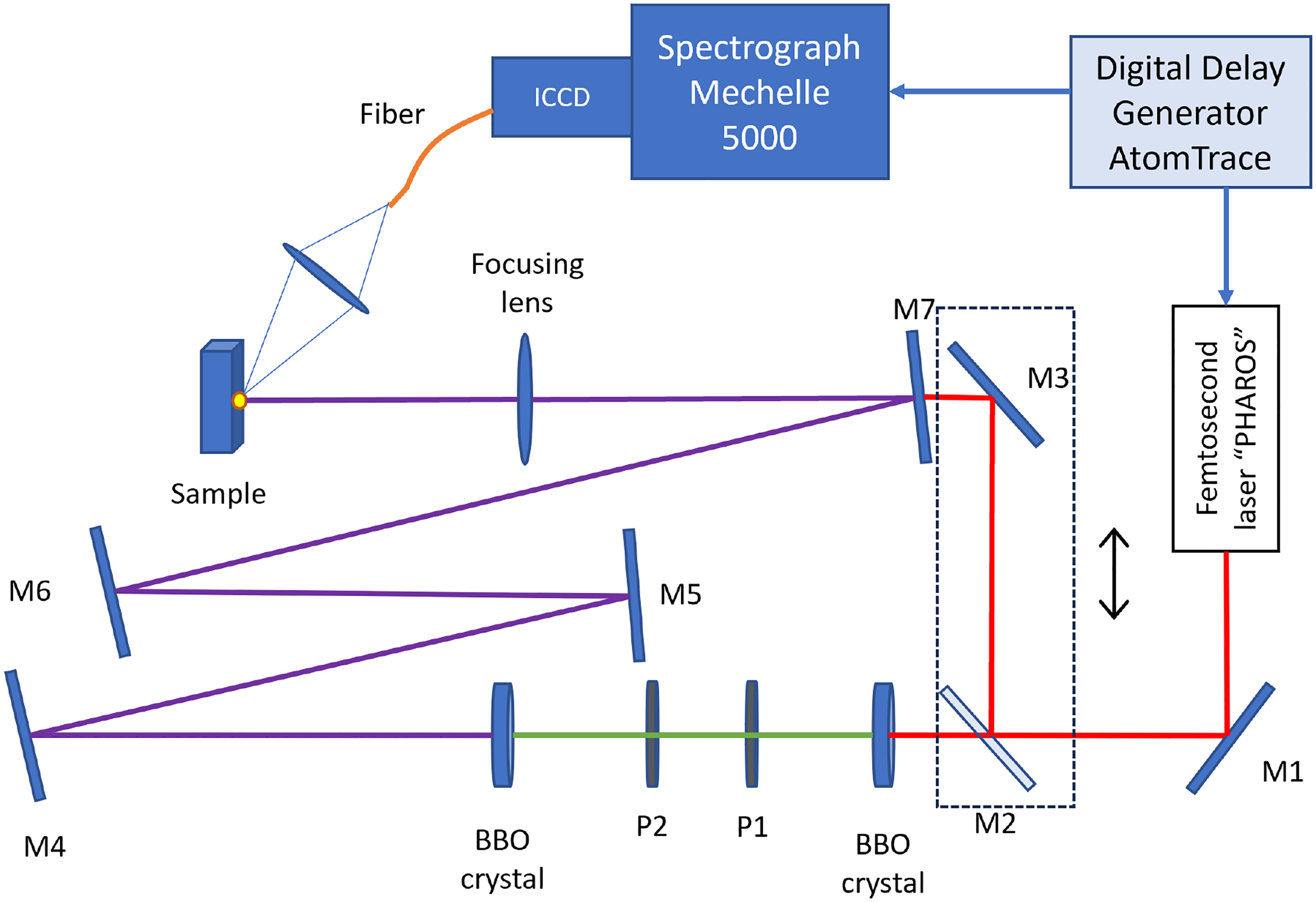
Experimental setup for time-resolved fs-LIBS measurements using 343 and 1030 nm wavelengths. M1–M3 are high-reflective mirrors for the first harmonic and M4–M7 are high-reflective mirrors for the third harmonic and high transmittance for the first and second harmonics (used to eliminate IR and visible laser radiation). P1 and P2 are half waveplate and dispersion compensation elements.
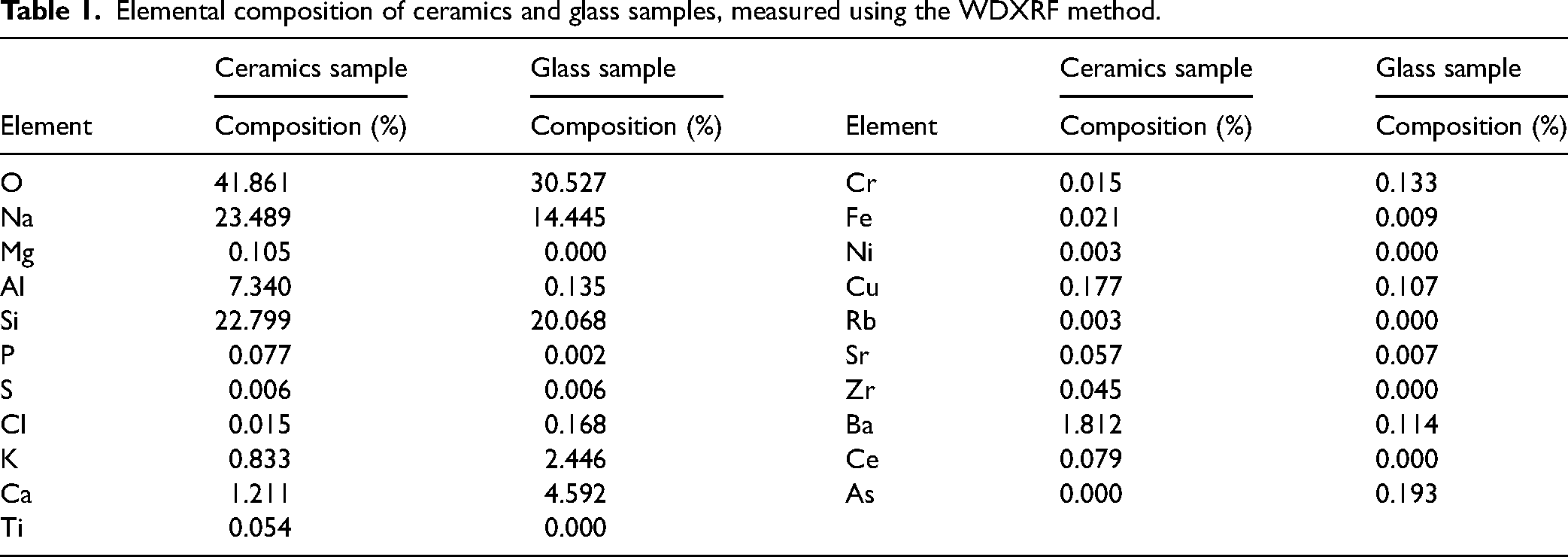
Our samples were copper, oxygen-free high thermal conductivity (99.95+% purity) (Goodfellow Cambridge Ltd.); stainless steel, AISI 301 (Goodfellow Cambridge Ltd.); glass (green optical glass filter; Izium); and a ceramics (Diwali light turquoise plate; Luminarc Ltd.). The elemental analysis of the glass and ceramics samples was performed with the wavelength dispersive X-ray fluorescence (WDXRF) method using the wavelength dispersive XRF spectrometer (Axios mAX) with a 4 kW STmax160 X-ray tube with an Rh anode. The composition is shown in Table I. A fresh spot on the sample surface was chosen for each accumulation after 3000 laser shots. The integration time for one spectrum was 0.5 s. Plasma emission was collected by a 400
Elemental composition of ceramics and glass samples, measured using the WDXRF method.
Results and Discussion
All plasma spectra were captured with a time gating window of 200 ns with a 50 ns delay step following the first registered plasma spectrum, until plasma emission was not present in the measured spectra at
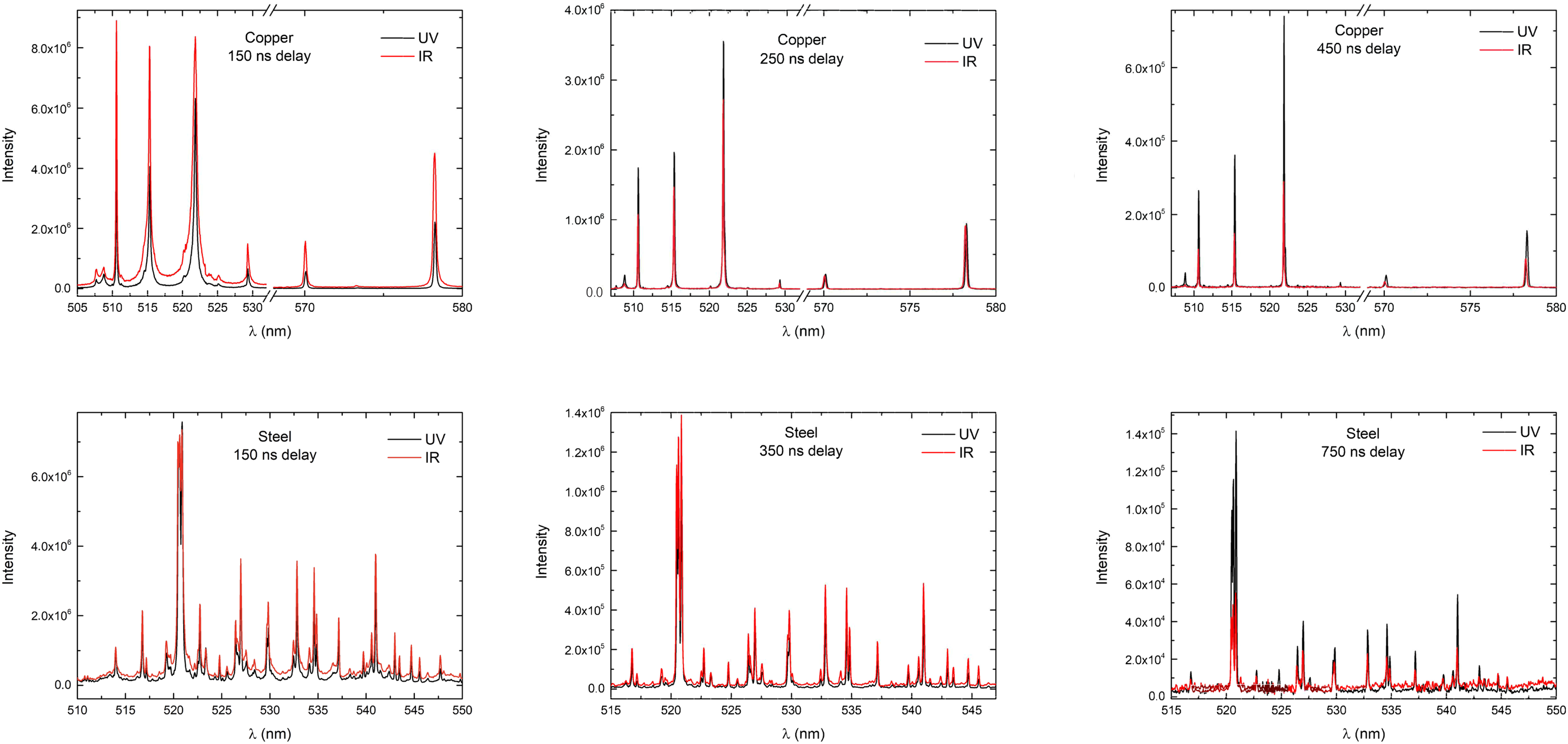
Comparison between the spectra of the plasma induced by the fs UV laser radiation (343 nm) and those induced by fs IR laser radiation (1030 nm) on copper (top) and steel (bottom) samples at different delay time.
Plasma spectra on ceramics and glass samples are shown in Figure 3. On the
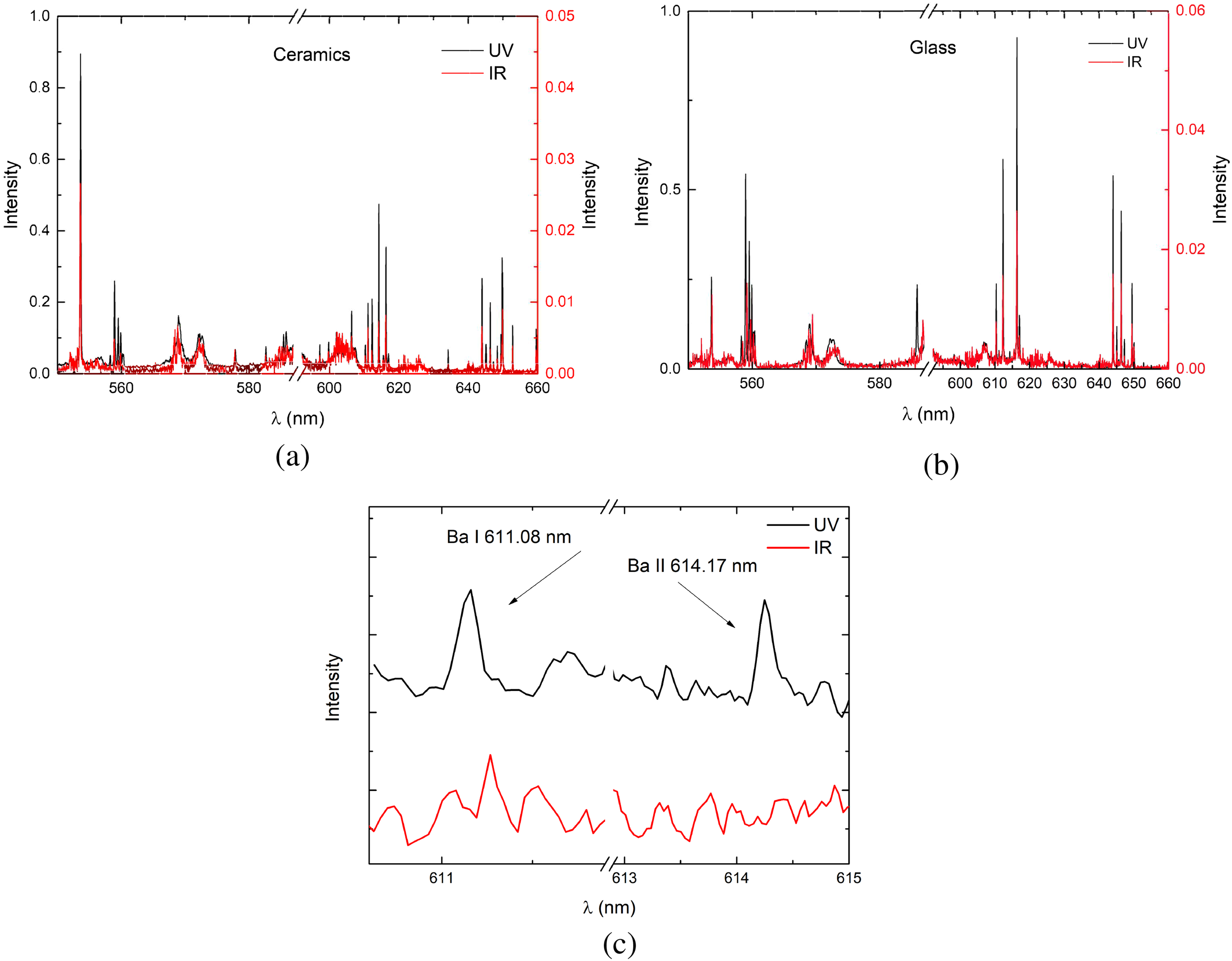
Comparison between the spectra of the plasma induced by the fs UV laser radiation (343 nm) and those induced by fs IR laser radiation (1030 nm) at 300 ns delay on a ceramics (a) and glass (b), and (c) shows a magnified spectrum region of interest on glass sample.
Plasma temperature and electron density were determined by analyzing Cu(I), Fe(I), Ca(I), and K(I) emission lines, their spectroscopic data were obtained from the National Institute of Standards and Technology (NIST) database
34
and is shown in Tables II to IV. The plasma temperature was calculated using the Boltzmann plot method,
35
under the assumption of local thermodynamic equilibrium (LTE), which was verified through the McWhirter criterion, presented in Eq. 1: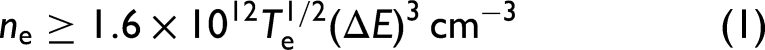
Spectroscopic parameters of Cu(I) emission lines used to calculate plasma parameters, taken from the NIST database. 33

Spectroscopic parameters of Fe(I) emission lines used to calculate plasma parameters, taken from the NIST database.33

Spectroscopic parameters of Ca(I) and K(I) emission lines used to calculate plasma parameters on glass and ceramics samples, taken from the NIST database. 33

Examples of Boltzmann plots obtained using 343 and 1030 nm laser wavelengths on copper, steel, glass, and ceramics samples are shown in Figure 4. The calculated excitation temperature values are the same, within error, for both used wavelengths on all samples investigated. Time-resolved plasma temperature is shown in Figure 5. The temperature on metal samples was in the range of 8000–11 000 K, on the glass sample it was in the vicinity of 4500–8000 K, and on ceramics it was 6000–11 000 K. The temperature uncertainty, according to El-Rabii et al.,
39
was expressed as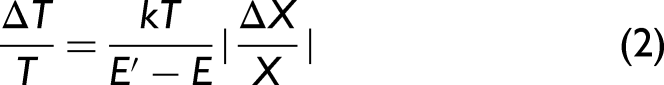
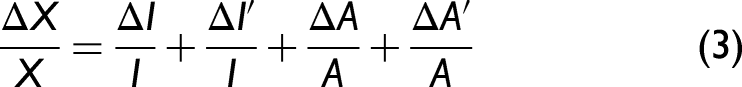
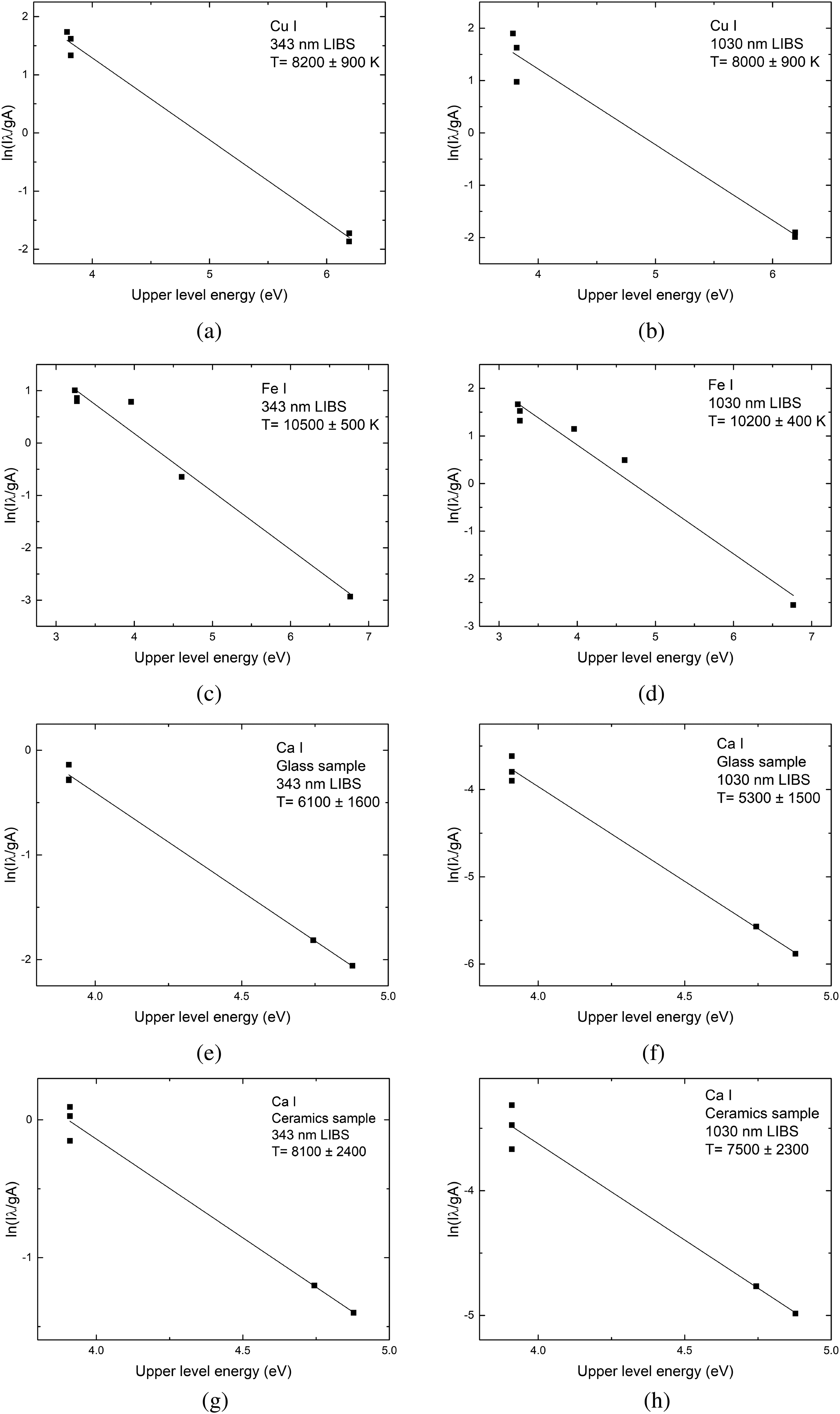
Boltzmann plots for the calculation of plasma temperature of Cu(I) (a, b), Fe(I) (c, d), Ca(I) on glass (e, f), and ceramics (g, h) samples.
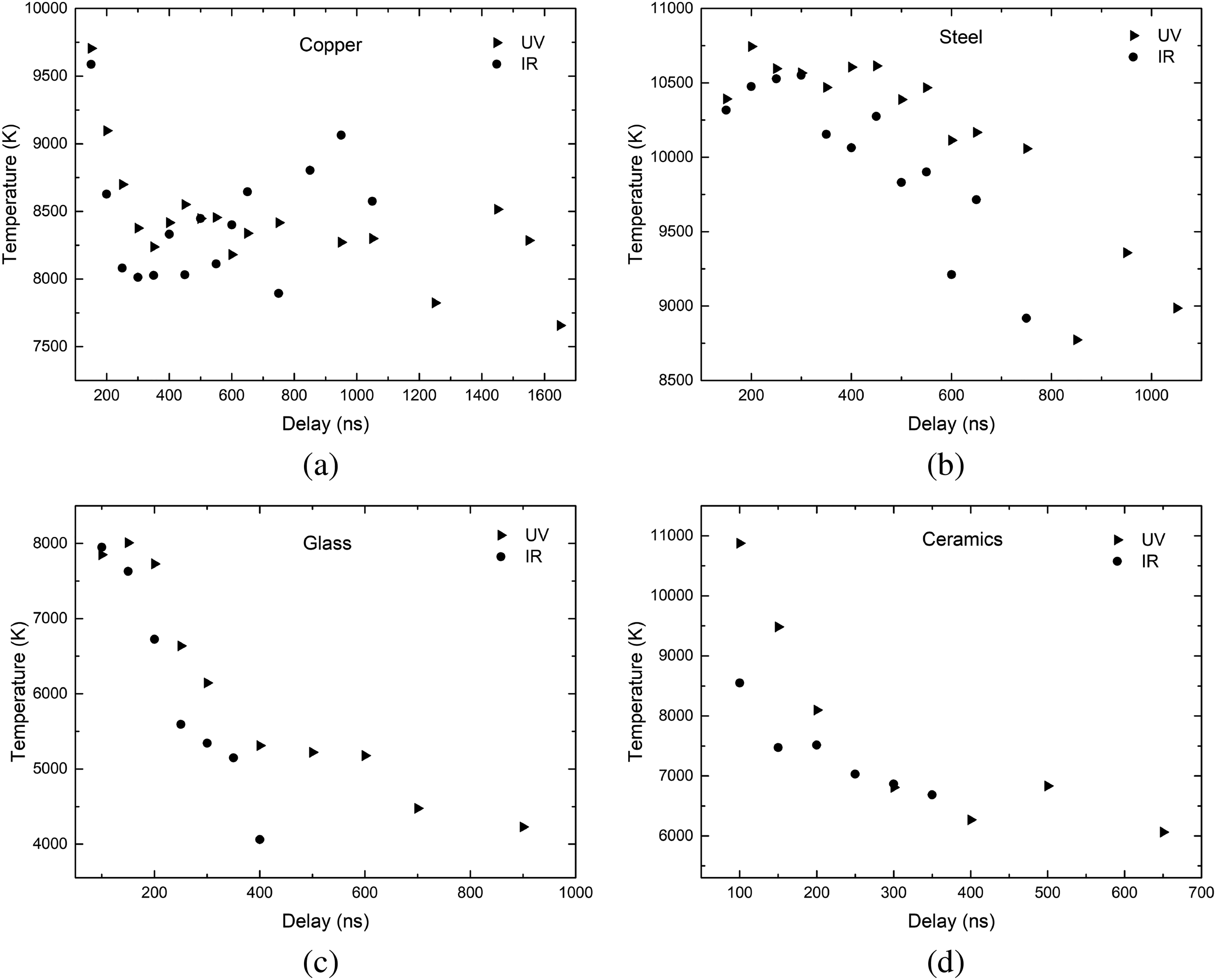
Time-resolved plasma temperature for fs-LIBS on copper (a), steel (b), glass (c), and ceramics (d) samples for 343 nm (UV) and 1030 nm (IR) wavelengths.
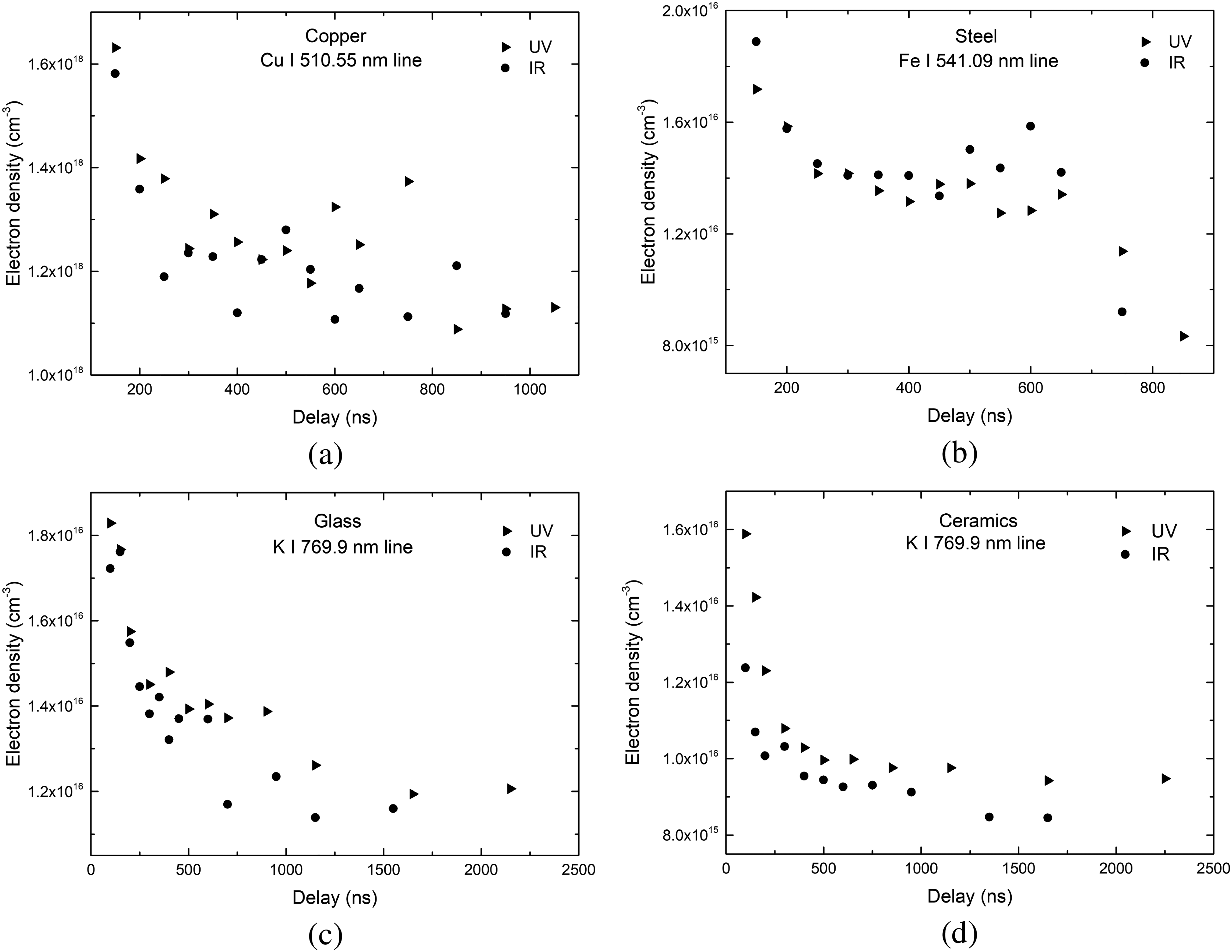
Time-resolved electron density for fs-LIBS for UV and IR laser wavelengths using Cu(I) (a) 510.5 nm, Fe(I) (b) 541.1 nm, and K(I) (c, d) spectral lines on copper, steel, glass, and ceramics samples, respectively.
Electron density was calculated from the Stark-broadened profile of Cu(I), Fe(I), and K(I) lines using Eq. 4:40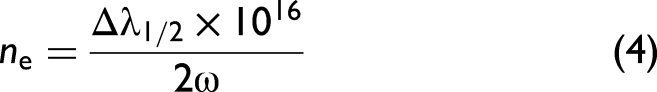
Signal-to-noise ratio (SNR) was also evaluated for both wavelengths used on all samples, i.e., Cu(I) 521.82 nm, Fe(I) 541.09 nm, and Ca(I) 612.22 nm on glass and ceramics samples, and is shown in Figure 7. The use of the UV laser wavelength resulted in significantly higher SNR on all samples: copper (
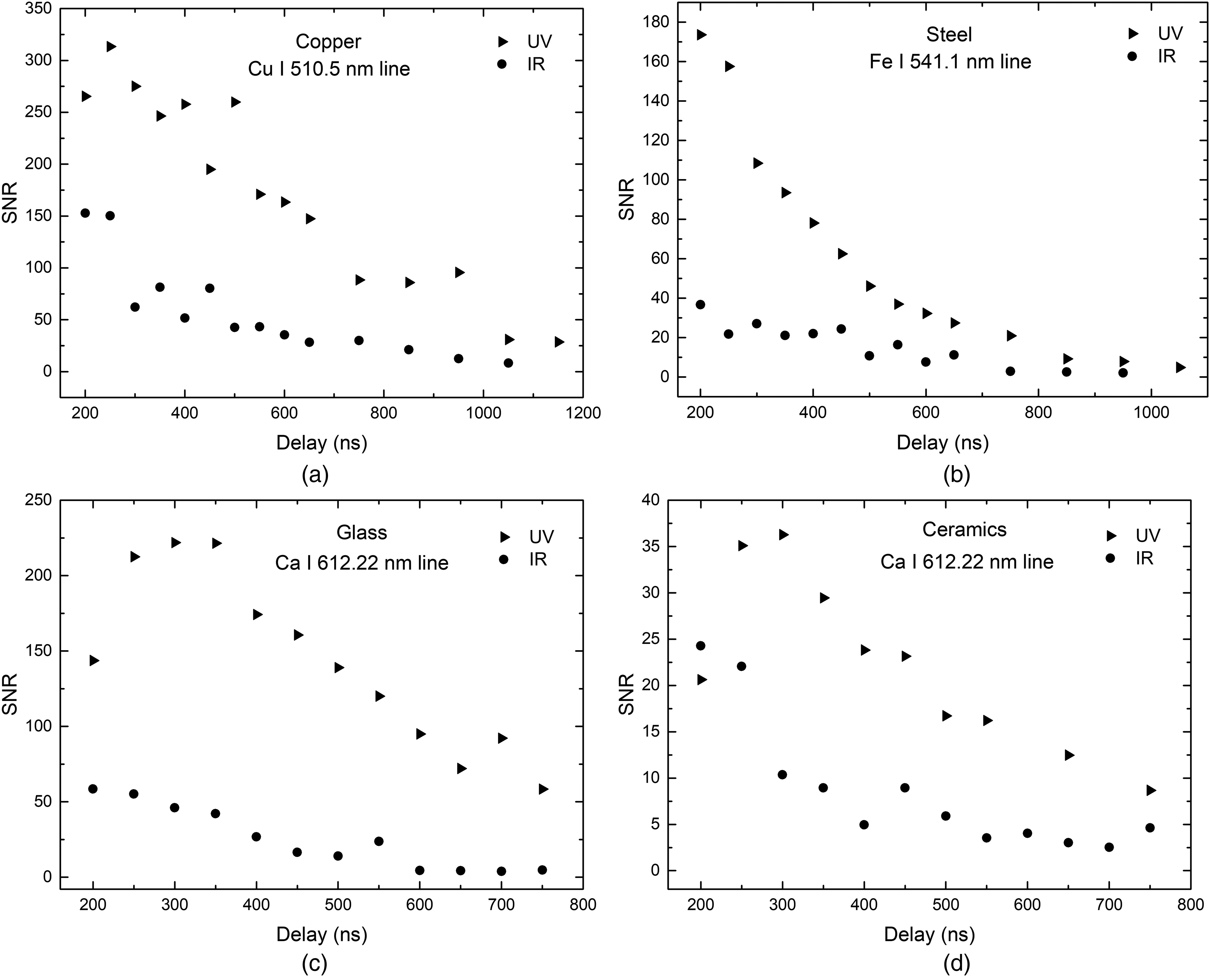
Signal-to-noise ratio (SNR) evolution as a function of delay for Cu(I) (a), Fe(I) (b), and Ca(I), (c, d) on copper, steel, glass, and ceramics samples, respectively.
The ablated volume on all samples was investigated using a 3D laser scanning microscope (Olympus OLS5100). On the copper sample, it was
Conclusion
In this work, we investigated the effect of a fs laser wavelength (1030 and 343 nm) at fixed energy fluence on the observed copper, steel, ceramics, and glass plasma parameters using the LIBS technique. The evaluated plasma temperature and electron density did not show any strong dependence on laser wavelength for all the samples and coincided with the limits of error. However, the SNR significantly increased using UV laser radiation for all samples: copper (
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has received funding from the European Regional Development Fund (project 01.2.2-LMT-K-718-03-0029) under a grant agreement with the Research Council of Lithuania (LMTLT).
