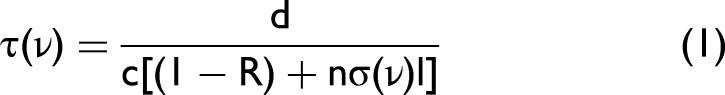Abstract
We have applied cavity ring-down spectroscopy (CRDS) to the study of the 166Er isotope in an atomic beam. These measurements were realized with an external cavity diode laser tuned to the 400.9 nm atomic transition of erbium and a customized high-finesse ring-down optical cavity under vacuum. Erbium atomic beams of different number densities were generated in a tantalum foil micro-crucible within the cavity. Absorbance values of the 166Er isotope between 3 × 10−6 and 7 × 10−5 were measured with a best-case precision on the order of 10−6, which is remarkable when considering the extreme temperatures at which the measurements were conducted, and the short detection path which is characteristic of collimated atomic beams. Number densities of erbium atoms were inferred to be between 2 × 106 and 6 × 107 cm−3. This work demonstrates for the first time the ability of studying dilute atomic beams of refractory materials with high accuracy utilizing CRDS. In these initial studies, we used erbium as a model system, but we expect to extend the proposed approach to the measurement of isotopes of uranium and plutonium for nuclear non-proliferation applications.
Introduction
The generation and study of atomic beams have made important contributions to many scientific fields. The main advantage of employing atomic beams as research tools as opposed to isotropic gases is that the former is free from inter-atomic collisions. Beams can be used to study the interaction of atoms with all kinds of charged or neutral species, with electric or magnetic fields, and, most importantly, with photons, a process that reveals the electronic structure and spectroscopic properties of these atoms. In this latter case, the most salient advantage is that Doppler broadening of atomic transitions can be practically eliminated. Atomic spectroscopy in beams is important in the very active field of cold atomic physics, where atoms are subsequently cooled, trapped, and manipulated in order to study fundamental and applied quantum science. 1 These experiments allow for the measurement of electronic transition energies and rates, which determine a wide range of laser cooling parameters, such as saturation intensity, Rabi frequency, Doppler temperature, capture limit, and maximum deceleration. 2 The accurate determination of absolute frequencies of optical transitions also helps to improve the precision of optical standards based on laser-cooled atom systems, which are emerging as the most stable and accurate frequency sources for optical clocks. 3
We have developed techniques for the efficient generation of collimated atomic beams of highly refractory elements,4,5 allowing, for example, the study of the electronic hyperfine structure of uranium isotopes by laser spectroscopy. 6 Our previous experiments demonstrated a best-case absorbance sensitivity of the order of 10−2. With the goal of increasing the sensitivity of our isotopic analysis methods, we have incorporated cavity ring-down spectroscopy (CRDS) as the atomic detection method.
Among the various existing laser spectroscopy techniques, CRDS is one of the most efficient high-sensitivity absorption methods.7,8 In CRDS, laser light is coupled into a high-finesse optical cavity, where it remains mostly trapped. In vacuum, the intensity of the trapped light decreases by a fixed fraction during each round trip only due to mirror reflectivity losses. If the laser frequency is resonant with one of the cavity modes, the intensity inside the cavity increases due to constructive interference up to a steady-state value. If the input laser light is suddenly interrupted, the trapped light will bounce back and forth several thousand times between the mirrors, effectively yielding an optical detection path of several kilometers. The exponential decay of the light intensity, i.e., the “ring-down” time, depends mainly on the cavity length and the reflectivity of the mirrors. If an absorbing species is placed within the cavity, the ringdown time will decrease, as light is absorbed before exiting the cavity. This ringdown time can be used to extract the concentration of the absorbing species with very high sensitivity. Since first proposed as a means to measure the reflectance of high-quality mirrors by Herbelin and McKay, 9 CRDS has experienced a rapid rate of development. O’Keefe and Deacon 10 used CRDS to conduct absorption spectroscopy and to quantify several properties of test gases. Most CRDS experiments at earlier times utilized pulsed lasers,11–13 which exhibit some disadvantages, such as multi-exponential decays and mode-beating. Romanini et al.14–16 first demonstrated trace gas measurement by continuous wave CRDS. This modality not only greatly simplifies the experimental setup and data analysis process, but also improves the sensitivity of CRDS systems, by exciting a single longitudinal mode of the ring-down cavity. 17
An important advantage of CRDS is that absorption by the target is determined by measuring the time dependence of the light instead of the total intensity exiting the cavity. Thus, neither absolute intensity values nor decay-to-decay intensity fluctuations influence the measured absorbance. Additionally, compared to other detection schemes, especially those involving frequency scanning or amplitude modulation, absorbance by CRDS is measured on an absolute scale and no prior calibration is required. Most importantly, the effective absorption path, which depends on the cavity length and the reflectivity of the cavity mirrors, can be very long. As a result, the interaction probability between photons and target atoms or molecules is greatly increased, and detection sensitivity is largely improved.
Here, we present our work using CRDS to detect a single isotope in an atomic beam for the first time. This work is a further step toward improving the sensitivity of our previously developed techniques for the isotopic analysis of refractory elements.4–6,18 For this purpose, we demonstrate the determination of the number density of the 166Er isotope in an atomic beam. We show a large increase in sensitivity by generating a much smaller number of densities of atoms in the atomic beam. This is accomplished by performing experiments at much lower temperatures (as low as 932 °C) when compared to our previous erbium experiments at 1400 °C to 1800 °C, since the atomic beam number density is determined by the vapor pressure of the sample, which decreases with temperature. Erbium is selected for this demonstration because atomic spectroscopy studies of this isotope are important for laser cooling applications,19,20 but we expect to extend these studies to the analysis of uranium and plutonium for nuclear non-proliferation applications.
Experimental
Figure 1a shows a schematic diagram of the experimental setup for atomic beam CRDS. A custom ring-down cavity (Stable Laser Systems) is formed by two mirrors of reflectivity of about 99.99% at the target wavelength at a distance of 15.8 cm, and assembled on top of a vacuum chamber, as shown in Figure 1b. Details of the method for generating atomic beams of refractory metals have been described in great detail previously. 4 A small amount (5 mg) of erbium metal is placed in a tantalum foil micro-crucible, which is resistively heated to 932 °C to 1333 °C in rough vacuum (30 mTorr). The crucible temperature was measured by a fiber-optic two-color pyrometer (iR2C, Omega Engineering). The crucible is a cylinder 10 mm tall by 3 mm diameter, and only atoms that freely evaporate from the crucible form the conical atomic beam with a half-angle divergence of 16°. The output from an external cavity diode laser (ECDL; model TLB7100, New Focus) intersects the atomic beam orthogonally at a distance of 43 mm from the mouth of the crucible. This laser is tuned to the erbium 400.9 nm transition (4f126s2→4f126s6p) with a power output of 3 mW, and absorption is detected by a photomultiplier tube (PMT; model PMT1001, Thorlabs) after exiting the ring-down cavity. A 638 nm diode laser (L638P150, Thorlabs) is coaligned with the ECDL to assist in the alignment process. Flip mirror (FM) selects either the diode laser or the ECDL beam through the cavity. Because of its custom design, the whole CRDS vacuum chamber is compact (20 cm × 17 cm × 13 cm) and light (3 kg) and can be integrated into portable instruments. The laser beam is coupled into the cavity and the wavelength is tuned and fixed to the transition peak of the 166Er isotope at 24 943.257 cm−1, which was determined previously by us 4 and others. 21 Since the linewidth of the laser (300 kHz) is much smaller than the width of the atomic absorption band (150 MHz), 4 the absorption of 166Er can be treated as constant within the laser linewidth, which avoids the necessity to use multi-exponential decay data analysis. Note also that negligible contributions to the signal from neighboring isotopes are expected. Our CRDS system utilizes a similar strategy as that used by Hahn et al., 22 where photons are accumulated in the cavity by sweeping the cavity length through a resonance mode at a given laser frequency. As a result, this configuration does not require any optical switching device such as an acousto-optic modulator or Q switch to extinguish the laser beam, greatly simplifying the system. The cavity length is modulated piezoelectrically at 50 Hz by a triangle driving signal, thereby ensuring that at least one strong cavity resonance occurs in each piezoelectric transducer (PZT) sweep half-cycle. The output signal of the PMT is acquired at a 1 GHz sampling rate by a data acquisition system (PXIe-5764, National Instruments, USA) using a custom LabVIEW program. Continuous sweeping of the cavity length while holding the laser frequency at the peak of the 166Er absorption band yields a series of resonant modes that decay with a characteristic exponential time related to the absorbance of the atomic beam sample.
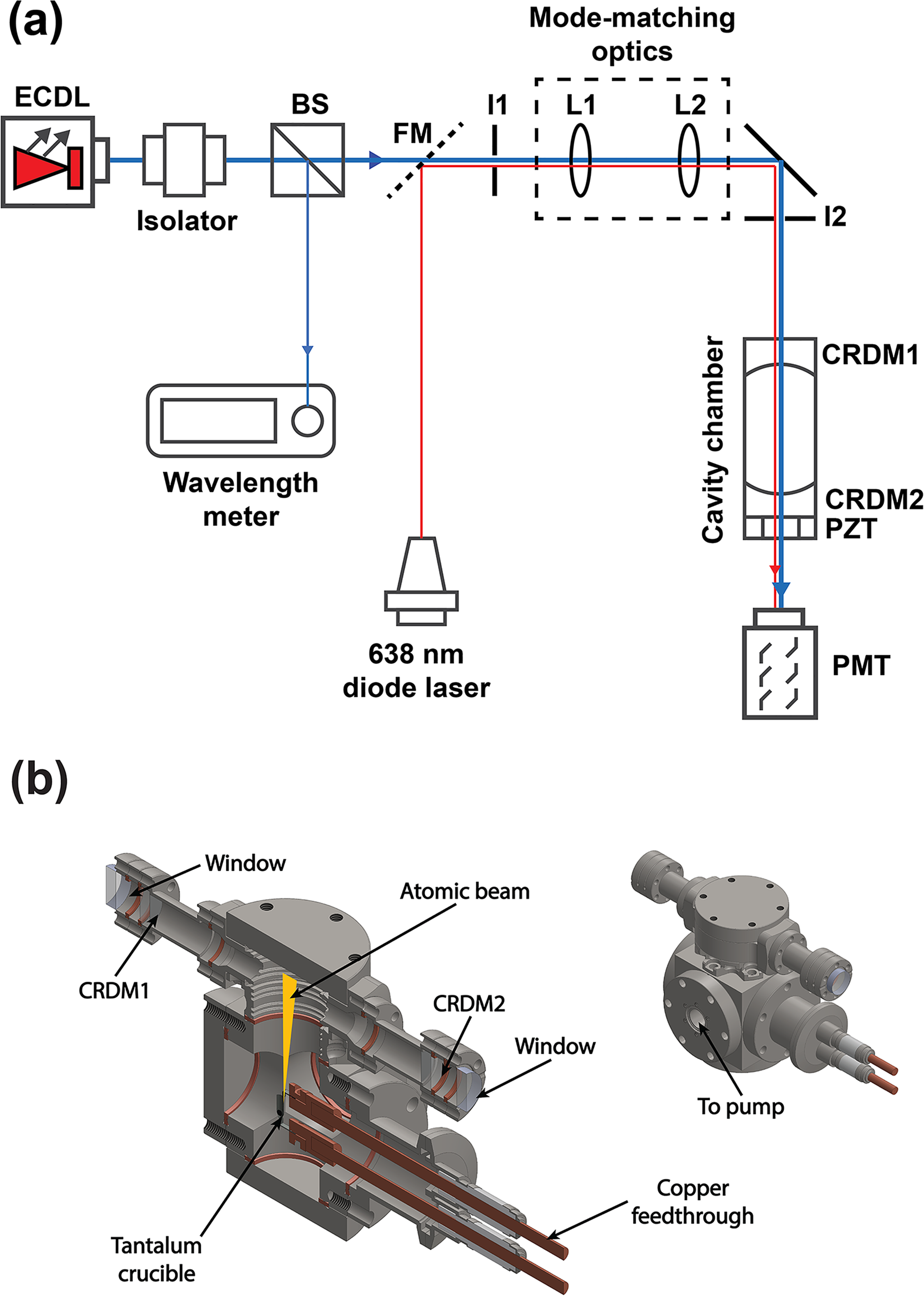
(a) Schematic of the experimental setup. The frequency of the ECDL is current and temperature controlled. A 42 dB optical isolator is used to protect the laser from back reflections. A beamsplitter (BS) directs 10% of the light to a wavemeter to monitor the laser wavelength and 90% to mode-matching lenses L1 and L2, which match the main cavity TEM00 eigenmode. Two irises, I1 and I2, are used to co-align the ECDL beam and the 638 nm alignment laser diode beam, either one being selected by FM. The CRDS cavity is made of a fixed mirror (CRDM1) and a mirror (CRDM2) mounted on a PZT. (b) Sectional and exterior views of the ring-down cavity mounted atop of the vacuum chamber.
Results and Discussion
Ring-down times were obtained from the tail of the transmitted intensity profiles, as shown in Figure 2. The region close to the ring-down peak was omitted from the exponential fitting because of the presence of ringing modulation, a phenomenon described in the literature.22,23 A Levenberg–Marquardt fit algorithm was applied to obtain approximations to each of the individually stored experimental ring-down decay curves. The red solid line shows a decay ring-down of the laser measured at a crucible driving current of 50 A, corresponding to a crucible temperature of 1168 °C, while the blue dashed curve shows the best fit. Mono-exponential curves were found to fit well in the target region, which demonstrates the single-mode excitation characteristic of CRDS. In a series of experiments, the driving current of the crucible was varied to change its temperature in the range of 932 °C to 1333 °C. The relationship between the ring-down decay time and the crucible temperature is shown in Figure 3. Each decay time shown was obtained by averaging 200–300 individual decays, and the error bars are obtained by calculating the standard deviations of the individual decay times. An increase in crucible temperature causes a decrease in decay times due to the increase in number density, as expected.
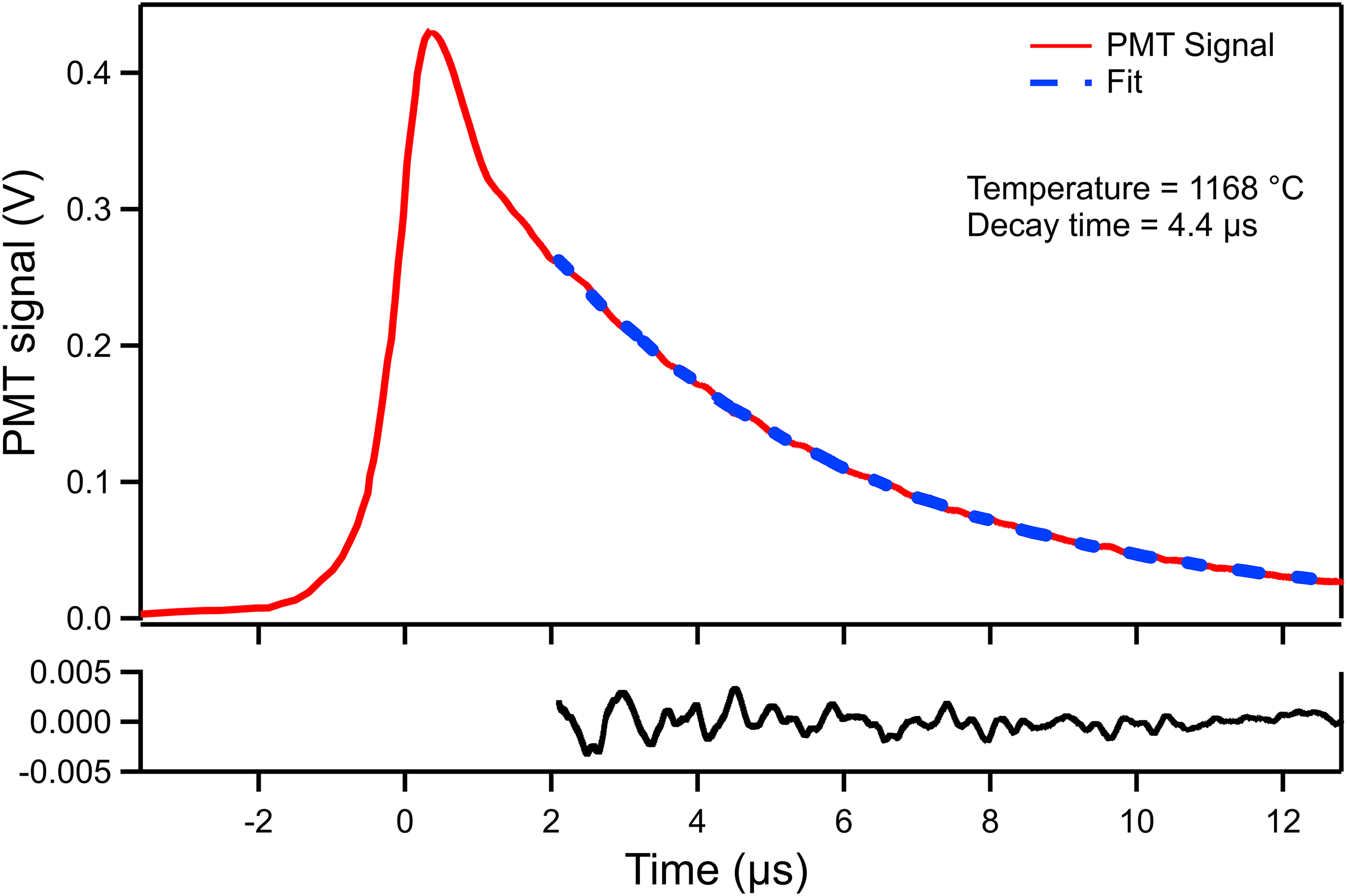
Experimental measurement and fitting of a typical ring-down exponential decay.
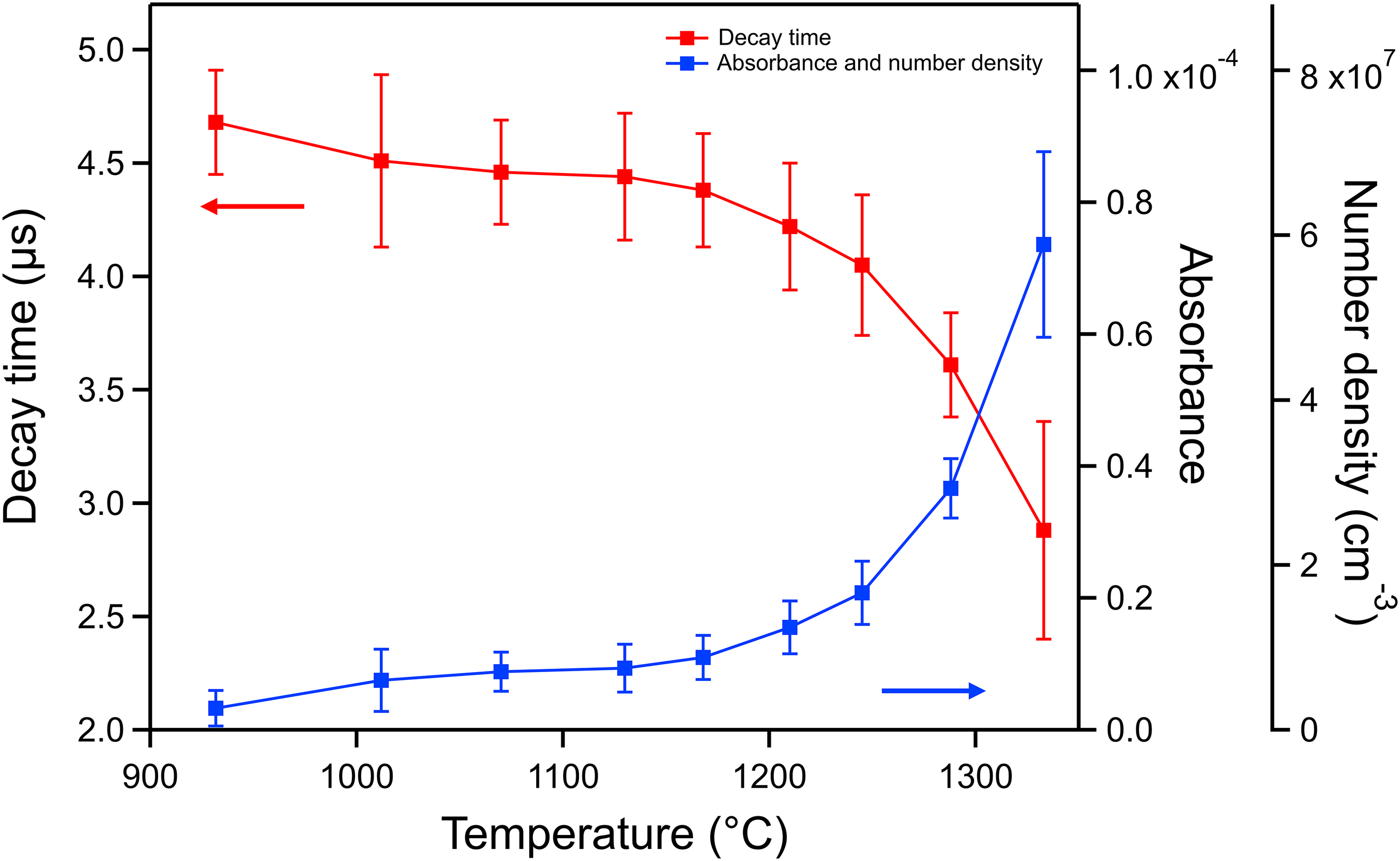
The relationship between the observed decay time, the atomic beam absorbance, the number density of 166Er atoms, and the temperature.
The number density of the absorbing species in the atomic beam
Conclusion
We have presented number density measurements of an atomic beam of the 166Er isotope using CRDS at the 400.9 nm transition. Decay times were measured over a crucible temperature range between 932 °C and 1333 °C. Absorbance values of the atomic beam at different temperatures were obtained between 3 × 10−6 and 7 × 10−5. The precision of the measured absorbance was in the range of 10−5 to 10−6 depending on temperature, which is remarkable when considering that these experiments were acquired at extreme temperatures where heat convection could significantly deteriorate the performance of the system. This effect is indeed observed by the larger measurement errors obtained at higher temperatures. Additionally, unlike most previous CRDS experiments that target isotropic gases, atomic beams occupy only a small portion of the detection path, which shortens the absorbing length of the system. Number densities of erbium atoms were estimated from the absorbance values and the lifetime of the transition, yielding values between 2 × 106 and 6 × 107 cm−3 with a precision of the order of 106 cm−3. This work demonstrates the ability of conducting spectroscopy measurements of individual isotopes of refractory elements using CRDS for the first time, with a 10 000× increase in sensitivity when compared to our previous experiments. Future work will include the study of isotopes of uranium and plutonium.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.