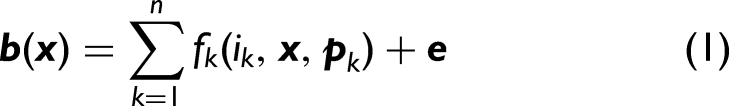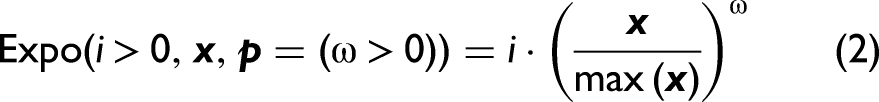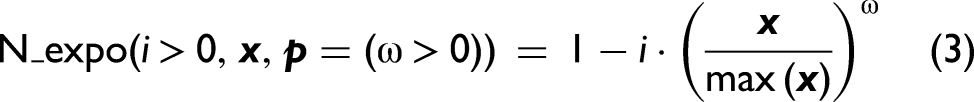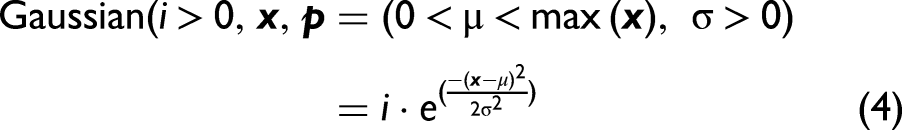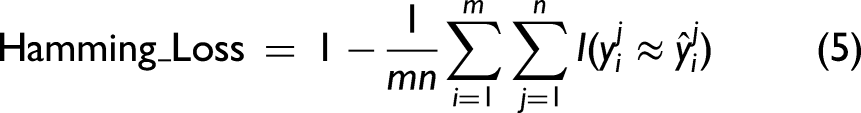Abstract
The development of measurement methodologies to detect and monitor nuclear-relevant materials remains a consistent and significant interest across the nuclear energy, nonproliferation, safeguards, and forensics communities. Optical spectroscopy of laser-produced plasmas is becoming an increasingly popular diagnostic technique to measure radiological and nuclear materials in the field without sample preparation, where current capabilities encompass the standoff, isotopically resolved and phase-identifiable (e.g., UO and UO
Keywords
Introduction
Since the achievement of the first controlled nuclear chain reaction in the mid-20th century, advancements in nuclear technology have ultimately led to diverse energy, industrial, medical, and defense applications.1–3 The first three of these applications rely on the use of nuclear materials for the generation of electrical power and the development of radioactive sources applicable in oil and gas exploration and medical procedures, while the latter involves the development of weapons and other technologies for national security applications. Due to an increasing global interest in nuclear energy and technologies, it is critical to establish new approaches that can minimize risk associated with next-generation nuclear reactors and the spread of “dual use” nuclear materials and technologies, thus supporting global and domestic nuclear security and nonproliferation missions.4–7
To support the safe operation of next-generation nuclear reactors, there is significant interest in the development of new sensor technology for characterizing in-core reactor operating conditions, spent nuclear fuel, and chemical reprocessing processes, as these sensors could facilitate real-time operational knowledge and address materials control and accountability requirements. Online monitoring allows operators to assess, in real-time, the state of key reactor structural components (fuel rods, pressure vessel, etc.) exposed to extreme environments (high radiation exposures, high temperatures, corrosive materials, etc.).8,9 Owing to a renewed global interest in nuclear energy to address energy access, curb climate change, and continue global investment in research and development activities involving nuclear materials, an ever-increasing burden is being placed on international organizations such as the International Atomic Energy Agency (IAEA), which is charged with verifying the non-diversion of nuclear materials and technologies for nefarious purposes. 10 Due to the need to characterize in-core reactor conditions, chemical processes and facilities, and nuclear materials relevant to the nuclear fuel cycle (NFC) and weapons proliferation activities, significant international and domestic efforts have focused on developing new detection technologies capable of performing non-contact, in situ elemental and isotopic analyses of a wide range of materials in a variety of ambient environments.
Several current efforts have focused on optical spectroscopy methods in conjunction with the use of pulsed laser ablation (LA) to perform standoff, near real-time isotopically resolved measurements of nuclear materials without the need for tedious sample preparation and lengthy laboratory-based analysis time frames, which are common drawbacks experienced in conventional chemical and radiometric methods of characterization.11–13 The incorporation of a high-powered pulsed laser allows for the vaporization and excitation of a sample to produce a luminous microplasma. Spectroscopic measurements (i.e., optical emission, absorption, and fluorescence spectroscopy) are then applied to quantify the properties of that plasma and provide measurements including the plasma temperature, electron number density, degree of ionization, and elemental, isotopic, and chemical composition of the sample. Common optical imaging techniques such as shadowgraphy, Schlieren imaging, interferometry, and velocimetry may also be applied to probe the spatiotemporal hydrodynamic evolution of the laser-produced plasma (LPP). 14 By coupling LA to these spectroscopic methods, hyphenated techniques such as laser-induced breakdown spectroscopy (LIBS), laser absorption spectroscopy (LAS), and laser-induced fluorescence (LIF) spectroscopy have been developed to characterize materials by assessing the LPP. Several recent works have focused on the use of either one or a combination of these LA-based optical spectroscopy techniques (e.g., LAS, LIF, and LIBS) and have further documented the advantages and disadvantages of their use.14–18
Laser-induced breakdown spectroscopy (LIBS) is a popular technique due to its lack of sample preparation, experimental simplicity, and ease of analysis leading it to be widely implemented across industry and research fields to address material characterization needs.19–22 In light of its versatility, LIBS has found application in a wide range of global industries, including the metallurgical industry23,24 (e.g., aluminum,25,26 steel,27,28 and other alloys29–31), pharmaceutical industry,32,33 wastes management industry,34,35 mining industry, 36 and food industry. 37 Several impactful review articles are present in the literature that cover recent efforts in LIBS as a state-of-the-art technique for material characterization as well as its application in specific industries.38,39 Significant efforts to enhance the sensitivity of the method have led to the development of hyphenated LIBS methodologies such as double-pulsed LIBS (DP-LIBS), nanosecond (ns)- and femtosecond (fs)-LIBS coupled with LIF (LIF–LIBS), spark-discharge-assisted LIBS, and microwave-assisted LIBS. 40 Additionally, significant efforts over the last two decades have focused on improving the technique to perform substantial standoff analyses (e.g., tens of meters) by overcoming laser focusing limitations observed in ns-LIBS through the development of filament-induced breakdown spectroscopy (FIBS).41–43
Sustained interest in LIBS-based analysis methodologies has resulted in significant efforts to further develop the technique toward a diverse set of applications within the nuclear community, with several review articles reporting broadly on the growing interest in characterizing nuclear-relevant and radiological materials.6,38,44 A query of the Web of Science database utilizing the keywords “laser induced breakdown spectroscopy” or “LIBS” occurring with either of the following terms: “nuclear fuel*,” “nuclear fusion,” “nuclear fission,” “nuclear industr*,” “nuclear material*,” “nuclear forensic*,” “nuclear reactor*,” “nuclear monitor*,” “nuclear waste*,” “nuclear security,” “nuclear nonproliferation,” “uranium,” or “plutonium” was performed and resulted in an initial set of 257 articles appearing in the available literature. Articles unrelated to LA plasmas and spectroscopy for nuclear applications were manually excluded, resulting in
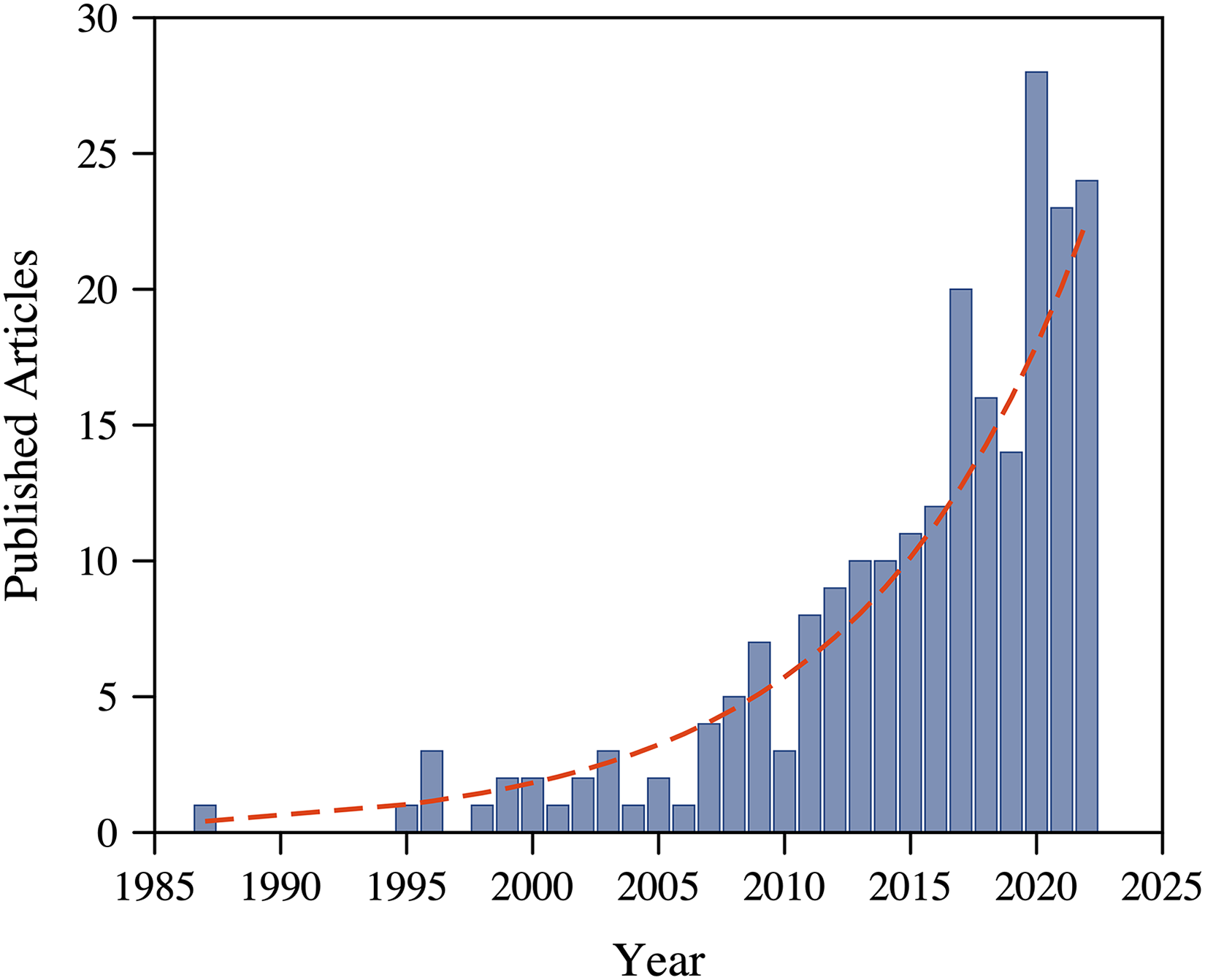
Number of publications per year within the last 30 years (1992–2022) available within the Web of Science database exhibiting a near exponential trend of research related to LIBS for nuclear applications. An exponential curve is fit to the data to guide the reader’s eye. Original figure produced by the authors.
The authors present this review article to critically discuss the role of LA plasmas and spectroscopy in light of the overwhelming applicability of these techniques for nuclear applications by providing an extensive examination of the available literature along with perspectives on future research and development directions. Recent review articles summarizing LA optical spectroscopy capabilities toward performing isotopic analyses, 5 assessing vapor-phase uranium oxidation chemistry, 57 and monitoring applications in nuclear reactor safety and security 9 are not comprehensive, and this work aims to support and expand upon these related efforts. This review will provide a comprehensive insight into fundamental research associated with and application of LA plasmas and spectroscopy to the broader field of nuclear energy and security, thus filling a topical gap in these previous compelling works and updating the discussion to include the substantial works that have occurred in the past several years. The topics covered in this review may appear peripheral to traditional nuclear energy-related applications; however, they were chosen to represent the wide-ranging field of nuclear energy and security.
The broad scope of this review article ranges from the principles of laser–matter interactions to implementations of machine learning to quantify or classify materials. We open with a summary of the physics involved in the formation of LA plasmas, followed by a discussion on the chemical dynamics and oxidation characteristics of reactive LPPs in addition to the gas dynamics of the laser-induced shockwave. Parting from the physical and chemical properties of the LPP, the multi-elemental and isotopic detection of nuclear-relevant materials is then discussed by first examining the utility of LAS, LIF, and LIBS for various applications. The long-range detection and atmospheric monitoring of radioactive plumes and aerosols is covered in detail, where an assessment of measured signal properties using ns-LIBS, fs-LIBS, and FIBS is provided. A synopsis of LIBS with NFC activities and reactor monitoring is also included, followed by a summary of various laser spectroscopy methods applied toward broader applications in nuclear safeguards, security, and forensics. A final discussion is then provided on automatic quantitative and qualitative analyses of LIBS measurements via the exploration of multivariate machine learning methods to facilitate the post-processing and analysis of complex spectra. Lastly, the review article concludes with a critical focus on the future outlook and impact of laser spectroscopy in the nuclear industry, as well as suggestions for improved understanding of the complex spatiotemporal properties of LA plasmas to further develop spectroscopic signatures of actinides and optical detection methods for nuclear materials and in unique nuclear applications.
Description of a Laser Ablation Plasma
Laser ablation (LA) is a multidimensional event that involves the formation of a luminous microplasma following the excitation of vaporized mass, leaving a sample in the form of atoms, electrons, ions, particles, and molten material. Understanding the mechanisms of laser–matter interactions is key to effectively driving the sample into a stoichiometric vapor such that the plasma composition and, ultimately, the optical emission spectrum is representative of that of the sample. The objective of this section is to introduce readers to the key LA and plasma plume phenomenology that will be covered in greater detail throughout the review. This section is not meant to be a thorough or entirely complete description of the LA process and readers are directed toward several excellent reviews19,65,66 and books67–70 available in the literature for more extensive discussions on these topics.
Typically, LPPs are considered to be a collisional plasma due to their high number density and ionization degree, which can be described through Maxwell’s electron distribution function.71,72 LPPs are spatially heterogeneous and are transient in nature, resulting in significant observable variation in both temperature and electron, ion, and atom densities. As a consequence of the dynamic plasma formation process, it is conventional to study the ablation event and associated laser–matter interactions in various timescales in order to fully understand the underlying physics. 73
Plasma formation is a complex process that exhibits several key phenomena occurring over a large dynamic temporal range from
The initial formation of the LPP occurs generally in a two-step process. The first step involves the initial generation of free electrons through direct interactions between the laser field and bound electrons.79,83 For ns-pulses, multi-photon ionization predominates and results when the combined energy of the absorbed photons exceeds the ionization potential of the atom.83,84 The second step forms the bulk of the plasma through electron cascade growth, which is characterized by an increasing frequency of collisional ionization. The laser field transfers energy to the free electrons through inverse Bremsstrahlung, initiating a chain of collisions and forming an ionization cascade.
81
At this point, the electron density and plasma temperature rapidly increase, causing the material to melt and further vaporize. This increase in plasma density forms an optically thick plasma that absorbs the remaining laser pulse, further heating the plasma and effectively shielding the sample surface from further material ablation.73,79,82 Immediately following LA, the resulting plasma temperature can reach up to
Laser–matter interactions exhibit substantial differences depending on the pulse duration of the laser used for material ablation due to their difference in energy deposition rate, which is particularly clear for the extreme case of ns- versus fs-LA often reported in the LA plasma and spectroscopy literature.92–94 LA using ns-pulsed lasers is characterized by a linear absorption process described by the Beer–Lambert law driven by thermal processes, while thermal mechanisms induced by fs-pulsed lasers are largely suppressed. 5 During ns-LA, various processes such as ionization, heating, and vaporization occur, which then lead to a transient change in the ablated material from a solid, to a liquid, and to a vapor phase. Moreover, with ns-LA and plasma formation, the trailing end of the pulse heats the plasma instead of targeting the ablating material itself. 95 However, this is not observed with fs-LA, where these processes tend to occur toward the end of the pulse. In fs-LA, the pulse duration is shorter than most major relaxation processes (e.g., electron-to-lattice energy transfer, heat diffusion, hydrodynamic motion, and free electron collisions) such that the majority of the laser pulse interacts with the sample material prior to a dense plasma being formed.5,96 As a result, large ionization cascades and plasma shielding are not observed during fs-LA, resulting in a colder plasma compared to ns-LA plasmas that exhibit lower emissions lifetimes and lower mean energies as depicted in Figure 2.90,97 Multiple efforts have focused on elucidating the plasma dynamics by characterizing LPPs produced by lasers that generate ns or ultrashort pulses (i.e., fs and ps).87,89,90,97,98 Some of the key advantages of using fs-pulsed lasers in LIBS includes reducing the thermal damage at the sample surface, obtaining consistent laser–sample interaction with the absence of plasma shielding, minimizing matrix effects, and producing lower continuum emissions that arise from colder plasma conditions.89,90,99 However, despite these advantages, commercial off-the-shelf fs-pulsed lasers typically come at a premium due to their complexity and higher pulse energies that are produced using chirped pulse amplification, which results in a bulkiness not yet suited for portable applications.
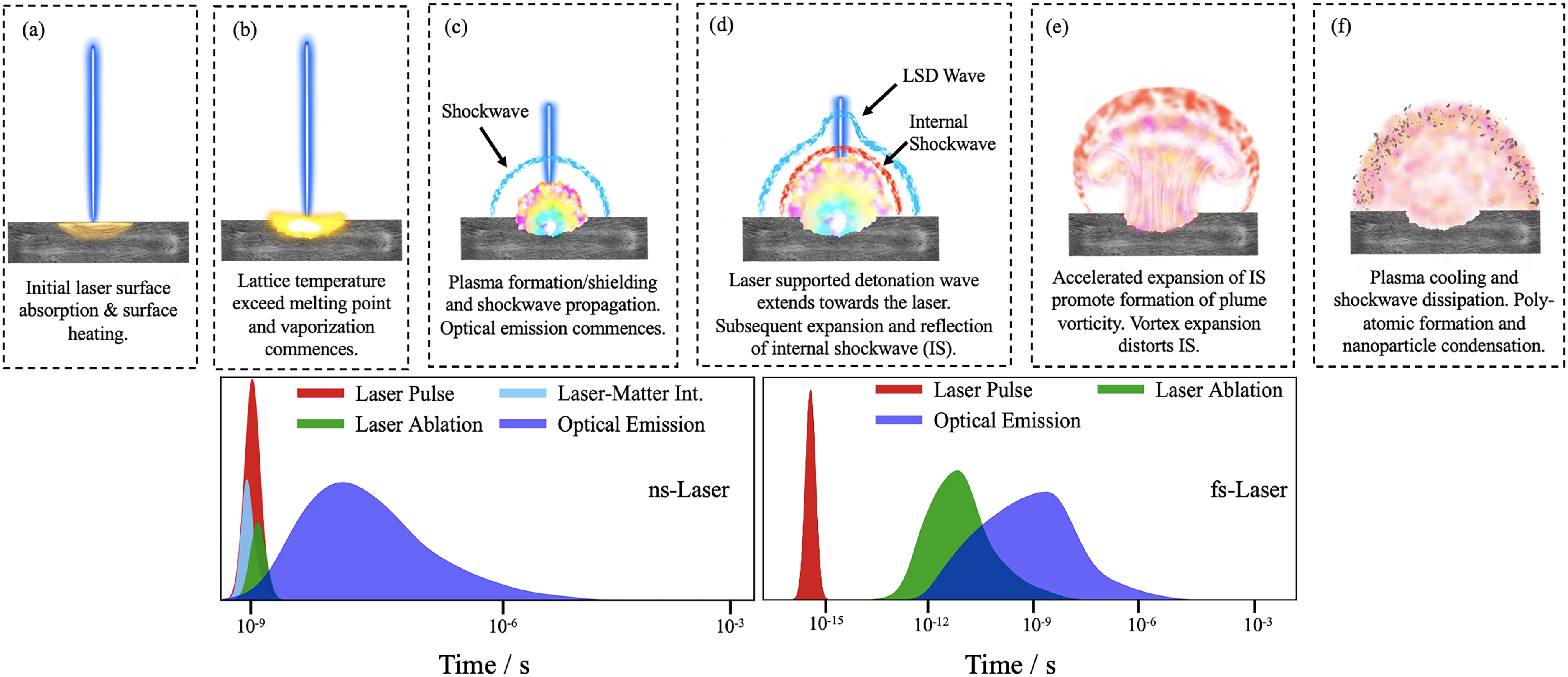
Depiction of the LA process ranging from (a) surface laser coupling to (f) shockwave dissipation and nanoparticle condensation. The timescale comparison between ns- and fs-LA starting with the onset of laser–matter interactions to optical emission is provided at the bottom for ns-LA (left) and fs-LA (right) adapted from Ref. 74, American Institute of Physics, 2012.
The specific mechanism governing the laser–matter interaction process is highly dependent upon the laser parameters, as energy absorption depends on the type of material ablated and the surrounding media where the plasma evolves. The plasma formation and laser–matter interaction mechanism discussed above is mainly related to the ablation of solid samples, and the ablation process and the dependencies related to plasma formation for solid samples differ from gaseous and aerosol samples. LIBS measurements of aerosols have recently become an attractive method for tracking atmospheric particle monitoring from radiological plumes as well as assessing volatile fission products and salts within the cover gas medium found in MSRs.62,100 A common observation during aerosol LIBS has been a lower ablation threshold for gases containing aerosols and particles compared to their pure gas counterpart.101,102 Early work has demonstrated that when utilizing infrared wavelengths, this reduction in ablation threshold is attributed to intense heating of the particle, which then serves as a site to initiate an ionization cascade. 103 However, other than the direct laser–particle interaction, the plasma–particle interaction that follows is a critical step as the majority of the measured emissions result from the dissociation of the particles that interact in the expanding plasma. Here, particle size is reported to be proportional to the emission intensity as long as the particles are fully dissociated in the plasma, where disproportionate emission yields indicate the onset of the partial dissociation of larger particles (2–10 µm) in the plasma volume.104,105 Moreover, similar to solid LIBS, the aerosol ablation mechanism highly depends on wavelength, pulse duration, and laser energy; however, three additional factors greatly influence the breakdown of aerosols: particle density within the stream, particle size distribution, and the focal volume of the beam.102,103,105,106 In short, gas sample ablation follows a similar, but not identical, mechanism to that of an aerosol system. For example, as with solid targets, a breakdown is initiated by photoionization followed by cascade ionization; however, pure gases achieve more consistent breakdown behavior compared to particle-laden gases as particles can influence where early free electrons are generated, which consequently dictates the position of the following ionization cascade.103,107 That being said, aerosols are particle ensembles residing in a gas media, and as such, many of the same trends observed in pure gases transfer to aerosolized systems. 103
Ablation that leads to a bright flash of light followed rapidly by a loud acoustic wave is indicative of plasma formation and subsequent generation of the shockwave caused by the high pressure and acoustic compression of the background gas. When the ablation laser delivers sufficient energy, the material reaches a critical point where it experiences a rapid phase transition from a solid to a super-heated molten liquid phase that undergoes vaporization and explosively expands outward.73,79 The plasma initially expands within an order of 10
The life cycle and certain properties of the plasma, such as reaction kinetics, opacity, pressure of the external shockwave, temperature, and electron density, are all properties directly and indirectly affected by the parameters of the LA. 14 Additionally, the laser wavelength can widely affect the plasma temperature, electron density, and absorption rate, which in turn affects the propagation of the resulting plume. For shorter wavelengths, the background gas is expelled by the expansion of the plume, while for longer wavelengths the ejected plasma is effectively mixed with the background gas at least hundreds of nanoseconds after the onset of ablation.95,109 The ablation and plasma formation depend on the laser pulse energy irradiating the material, where varying the laser irradiance has been observed to directly impact both the number density at the target’s surface and the plasma temperature. It is expected that since the amount of mass ablated increases with irradiance the number density would increase with energy as well, which is also accompanied by an increase in plasma temperature.95,110,111 Moreover, various reports offer insight into the different ablation regimes that can be observed, where these reports describe distinct ablation processes and laser penetration depth as functions of laser irradiance.112–114 Closely connected to the laser pulse energy, the pulse duration also greatly contributes to the LPP properties by altering the mechanism of energy dissipation onto the sample. Refer to Figure 2 for a visual overview of the high-temperature hydrodynamic phenomena associated with LA plasma plumes and relative timescale of the key process involved with ns-pulsed and fs-pulsed LA.
Laser Ablation Plasma Chemistry
Optical signatures measured using laser spectroscopy methods such as LIBS, LAS, and LIF are highly sensitive to the complex and rapidly evolving physicochemical conditions of the measured LA plasma.56,115 Plasmas generated in background gases containing reactive species such as oxygen readily undergo chemical reactions, leading to the formation of molecular species that alter the composition of the plasma plume.116,117 As the population of molecules in the plume grows over time, molecular signatures emerge in the optical spectrum to overlap and obscure atomic transitions.118,119 Understanding the underlying physicochemical processes that define transient optical signatures of LPPs is especially relevant with the lanthanide and actinide elements, which are characterized by heavily crowded spectra consisting of more than 30,000 atomic transition lines at energies between 200 and 3500 nm.120–123 The formation of simple diatomic molecules such as monoxides significantly changes their optical spectra. This phenomenon is demonstrated in Figure 3, where LA cerium plasmas generated in atmospheres containing either pure nitrogen or air produce distinctly different emission spectra for identical collection parameters (i.e., gate delay and width) as a consequence of CeO formation. Strong oxidation and the formation of metal oxides tend to be highly favored over the formation of nitrides. 117 Furthermore, the concentration of oxygen available to react with the plasma plume has also been shown to affect reaction pathway preferences and kinetics, leading to the formation of different thermodynamically stable end states between oxygen-deficient and oxygen-rich environments.124,125
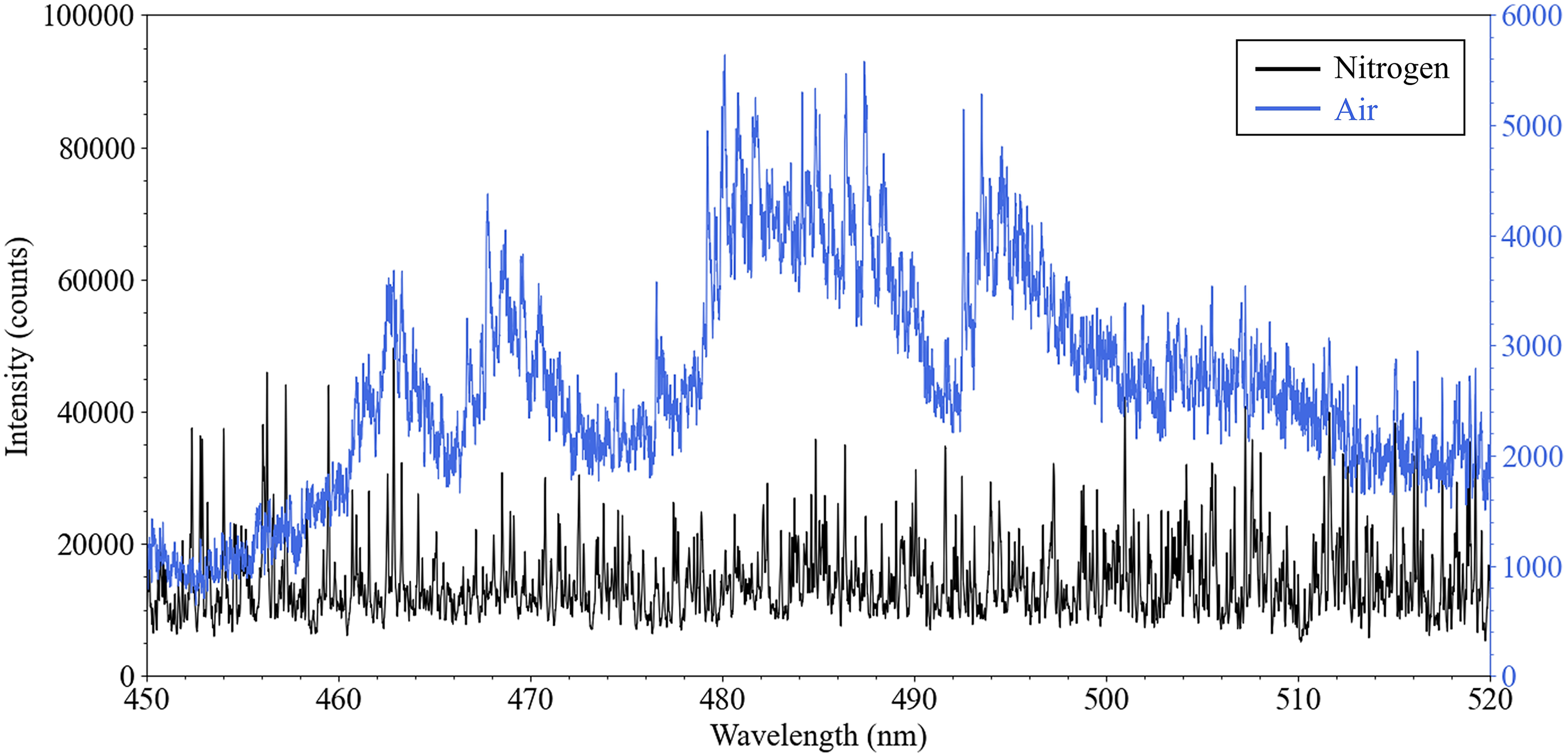
Demonstration of the effect of atmospheric oxygen on spectroscopic signatures of reactive ns-LA plasmas measured using LIBS with a CeO
The timescale for molecular formation in LA plasmas is strongly correlated with plasma temperatures and intermixing between plasma species and atmospheric oxygen. Generally, the plasma plume is dominated by atomic and ionic species at early times after LA when temperatures are typically measured to be greater than 10 000 K.86,88,126 Initial experimental observations of gas-phase molecular formation in ns-LA plasmas commonly occur on the microsecond timescale, where several publications have recorded monoxide emission signals as early as 2 µs over a range of ablation conditions.53,127,128 For fs-LA plasmas and filament-induced plasmas,
a
chemical reactions are reported to occur at earlier times (
Hydrodynamical mixing processes and spatial distributions of reacting species in the plasma plume also influence chemical dynamics of LA plasmas. At a fundamental level, collision theory states that reactants must undergo collisions with favorable conditions for chemical reactions to occur. This requires the plasma and background gas species to coexist or mix together over a shared spatial structure, or chemical reaction zone, within the plume. Experimental and computational measurements of plume hydrodynamics indicate that intermixing between plasma–gas species is primarily driven by diffusion due to large concentration and temperature gradients along the periphery of the plasma plume,128,133–135 which closely resembles physical and chemical processes observed with non-premixed diffusion flames.136,137 Shabanov and Gornushkin
133
computationally model the formation of several diatomic molecules at the plume–gas interface in the presence of large density and temperature gradients (thousands of Kelvin), while the hot central region of the plasma consists solely of atoms and ions. Wainwright et al.
128
experimentally measure the thickness of this thin plasma-gas boundary layer in LPPs generated from μm-Al powders, reporting a thickness of
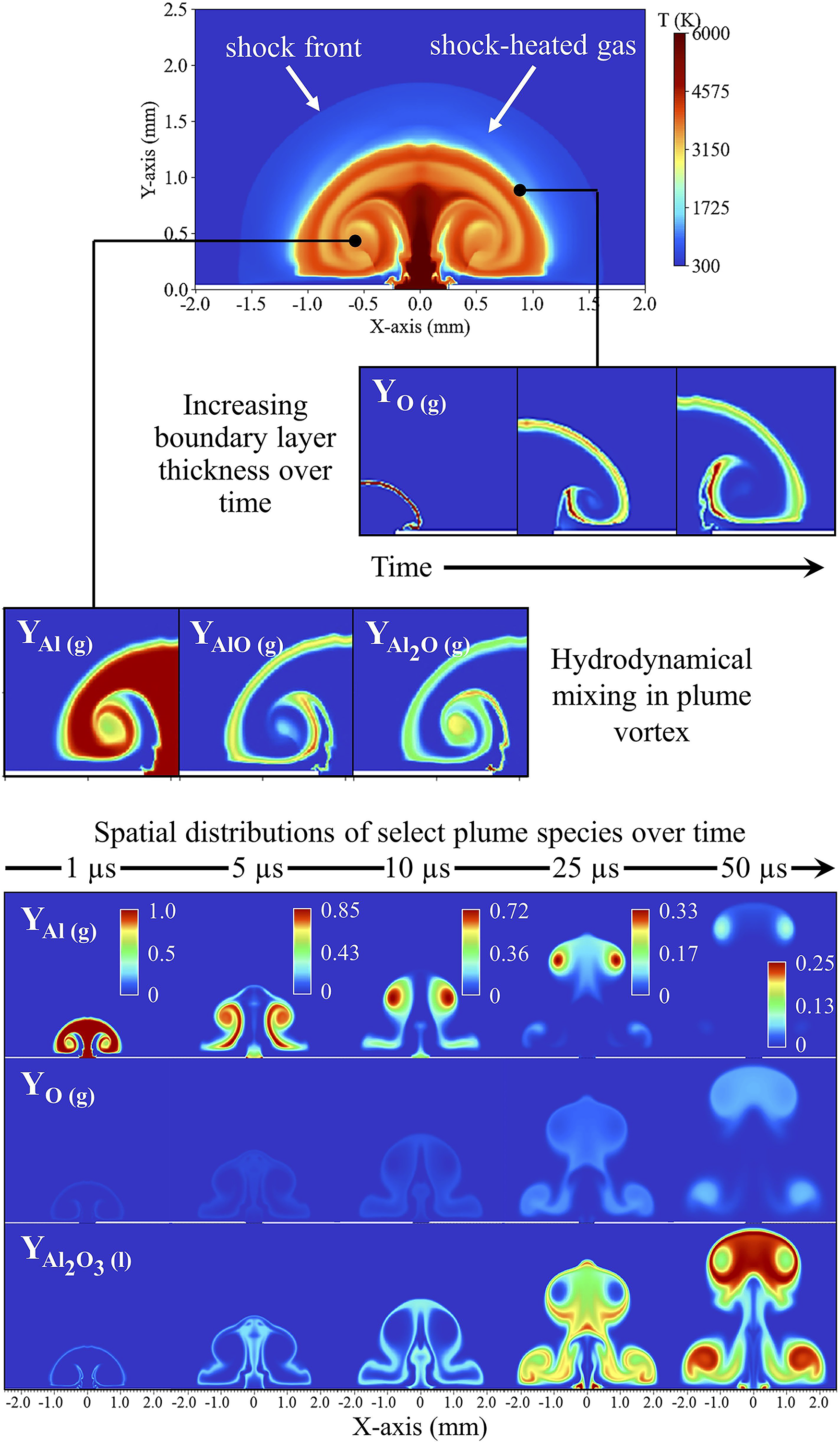
Simulation of Al vapor plume hydrodynamics and thermochemistry using the multi-physics and reactive multi-phase CFD code HyBurn. Original figure produced by the authors.
Beyond the spatial characteristics and thermal properties of the plasma plume, chemical reactions also depend on the bond strengths of the molecules themselves. Molecules with lower dissociation energies (3 eV> D
The oxidation chemistry of uranium LA plasmas is of special interest to the nuclear nonproliferation and safeguards community in regard to the in-field standoff detection of uranium signatures associated with NFC activities or weaponization programs. In addition, the high-temperature multi-phase chemistry of uranium LPPs is studied as a surrogate for nuclear fireballs to explain fallout particle formation and debris distributions subsequent to the detonation of a nuclear device. Presently, the development of uranium–oxygen plasma signatures has predominantly focused on early gas-phase chemical reactions by studying the formation and depletion of UO. UO is believed to serve as a precursor and reactant in the formation of polyatomic uranium oxides, such as in the following simplified description of gas-phase uranium oxidation pathways:141,142
Further investigations into uranium LPP chemistry involve experimental studies on the formation of polyatomic uranium oxides and nanoparticles. The electronic spectrum for UO
Uranium chemistry comparisons between ns-LA, fs-LA, and filament ablation plasmas have also been performed, where plasmas generated by ultrafast pulsed ablation are consistently characterized by higher UO/U I emission intensity ratios, lower excitation temperatures, and shorter lifetimes compared to ns-LA plasmas (Figure 5).
129
These observations are attributed to energy absorption and plasma shielding that occur during the ns-LA process that remains absent with fs pulses.
129
Furthermore, intensity ratios recorded for the fs-LA and filament-induced plasmas plateau over time, while in contrast ns-LA plasma ratios continue to rise at increasing rates. This behavior may be a result of different timescales and chemistry between fs and nanosecond plasmas. Over the times measured, nanoparticle formation was reported for the plasmas generated using focused fs pulses and filaments, yet not for the case of nanosecond pulses.
129
The spatiotemporal chemical dynamics of the filament-induced uranium plasma is further investigated using time-resolved, fast-gated imaging, where plume splitting was observed for time delays
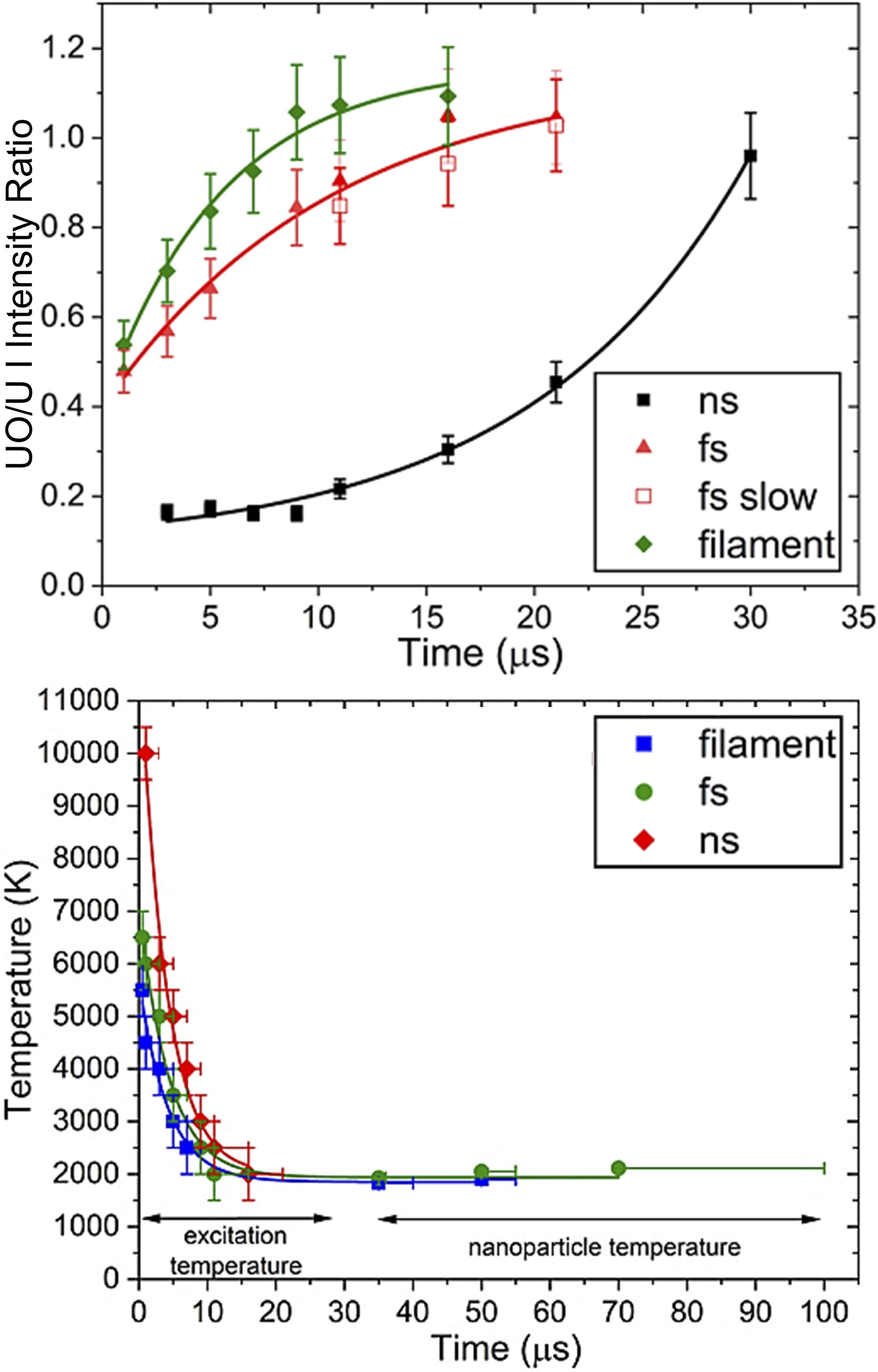
Uranium oxidation and plasma temperature comparison between ns-LA, fs-LA, and filament-induced plasmas. Emission intensity ratios use the transitions 591.54 nm U I and 593.55 nm UO. The “fs slow” label in the top subplot indicates a fs-LA plume that has experienced plume splitting, where the slow component refers to the portion of the plume located closer to the target surface. Note that the colors and markers used between the two plots are inconsistent. Adapted from Ref. 129, American Chemical Society, 2020.
Computational modeling of the high-temperature gas-phase chemical dynamics of LA uranium plasmas has been attempted by Finko et al.59,159 in two separate studies, where the first focuses on developing the reaction kinetics using a 0-D global kinetic model, while the second expands on this work with the addition of two-dimensional fluid dynamics and shock physics models. Diffusion processes are neglected and the ideal gas equation-of-state is assumed to accurately describe the LA plasma. 159 Furthermore, there are several significant assumptions made in relation to the uranium–oxygen reaction mechanisms and kinetics, yet acknowledgment of these assumptions is commonly omitted when these models are cited in the literature. To keep this discussion brief, we will only emphasize two of these assumptions here, the first of which declares that uranium–oxygen reaction mechanisms are analogous to that of the well-characterized aluminum-oxygen system. 59 Presently, there is not enough measured data available on the high-temperature gas-phase oxidation chemistry of uranium to disprove this assumption, yet realistically, uranium plasma chemistry will differ from that of aluminum. Due to this lack of data on uranium oxidation reactions, the authors were also required to theoretically calculate reaction rates and Arrhenius coefficients for their model, which leads us to the second item of discussion.
To model reaction kinetics for eight unimolecular thermochemistry reactions, Finko et al. 59 pull on the classical derivation of transition state theory (TST), which is a well-known method used to define rate constants for elementary chemical reactions (i.e., unimolecular, bimolecular, and termolecular). 160 The theory centers on an intermediate transition state that exists at the peak of the energy barrier between the reactants and products in the activated complex reaction pathway formulation.160,161 The transition state is assumed to be at quasi-equilibrium with the reactants, 161 and is determined through computational chemistry techniques by analyzing the potential energy surface for the system at room temperature. 160 Theoretical investigations to determine transition states for uranium oxidation reactions are very limited and aim to describe surface oxidation mechanisms of uranium metal. 162 In response, Finko et al. 59 simply used the thermodynamic properties of the products in place of those for the unknown transition states, which will underestimate activation energies for the reactions. Furthermore, it needs to be stressed that TST is derived for temperatures around 300 K and is only valid when thermodynamic properties do not vary significantly with temperature. 163 For high-temperature conditions, molecules will experience complex motions that are not captured by the classical TST formulation. 163 Hence, the theory breaks down and is not valid at the high temperatures (thousands of Kelvin) that characterize LPPs. Therefore, we caution against treating the computational models provided by Finko et al. as a baseline to compare experimental measurements against without acknowledging inherent deviations of the model from physical reaction mechanisms and kinetics. Computational chemistry approaches relying on quantum mechanical calculations, such as those performed in the literature,142,164,165 are recommended over TST to determine the reaction mechanisms and kinetics of gas-phase uranium oxides. Additionally, experimental approaches such as LAS, LIF, or other laser pump–probe techniques may be implemented to measure systems characterized by rapid chemical kinetics.166,167 A plasma flow reactor has been developed to measure high-temperature gas-phase chemical reactions over slower timescales (tens of milliseconds) compared to LA, 168 and has also been combined with scanning and transmission electron microscopy to study the final states of the particulates formed as a result of condensation and agglomeration processes in the plasma. 155
Beyond uranium, investigations into the high-temperature reactive chemistry of actinide-element LPPs are very limited. To the best of the authors’ knowledge, there has only been a single attempt published on the oxidation characteristics of LA plutonium plasmas, where possible Pu
Laser-Supported Absorption Waves and Laser-Induced Shockwave Dynamics
Laser ablation (LA) in ambient gases generates a microexplosion consisting of a millimeter-scale fireball and blast wave. During the ns-LA of solid materials, the vapor plume (i.e., fireball) is formed first following phase explosion and rapidly expands outwards in the direction perpendicular to the sample surface.65,73,177 The vapor plume works as a piston to compress the surrounding background gas, consequently resulting in the formation of a strong shockwave
b
.179,180 When the electron density of the vapor plume exceeds the critical electron number density (
The different laser irradiance regimes over which these absorption waves are formed determine whether the incident laser radiation is absorbed at the front edge of the plasma or by the opaque shocked gas immediately behind the strong shockwave.184,185 Over the lower end of the irradiances (
Most LA plasmas produced as an excitation source with spectroscopy methods fall in the LSD regime,
185
which exists over irradiances

Shadowgraph image demonstrating the structure of the spherical laser-induced shockwave and cylindrical LSD wave subsequent to the LA of aluminum. Reproduced with permission from Ref. 194, AIP Publishing, 2011.
At high laser irradiances (
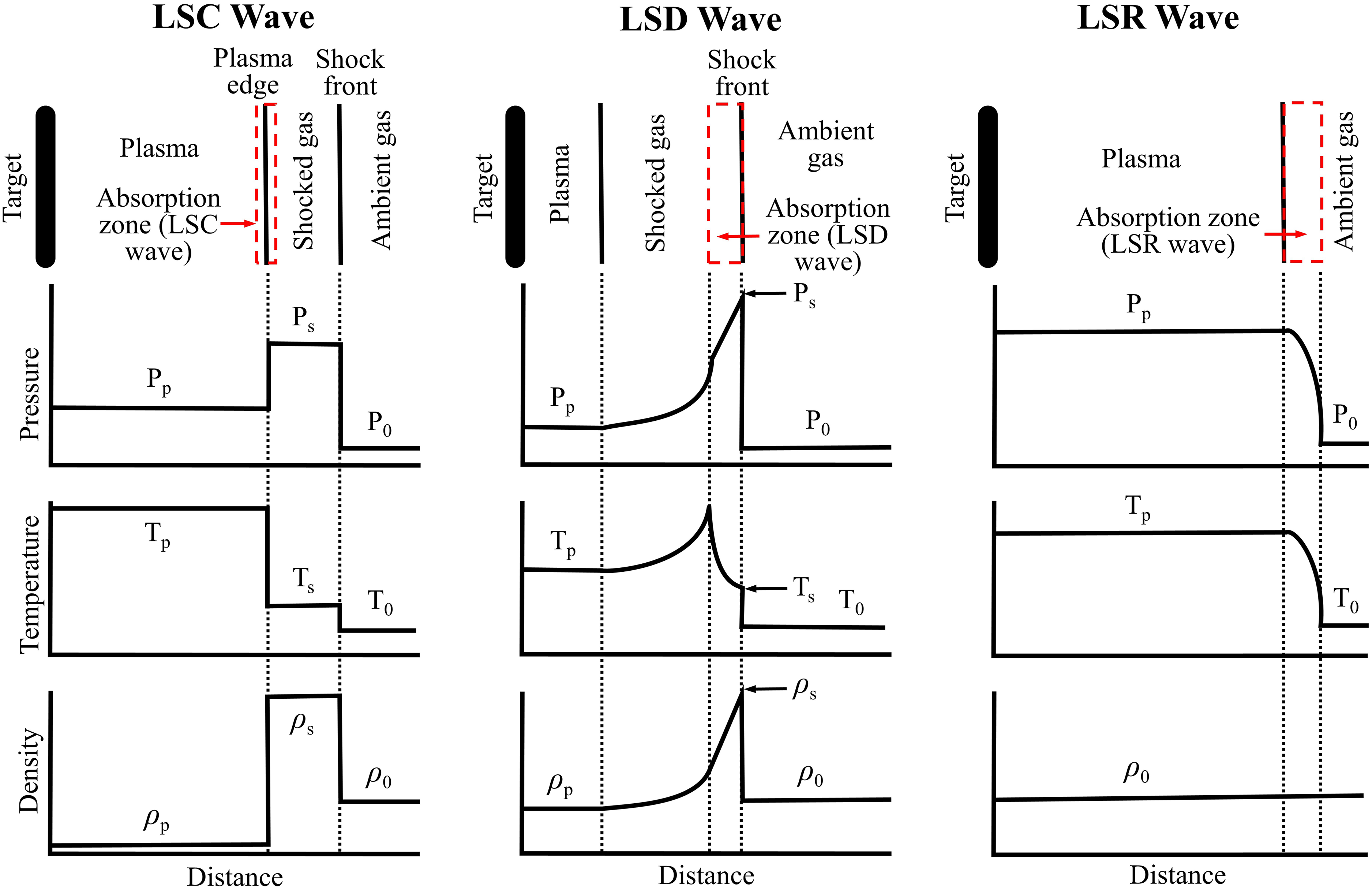
One-dimensional (1D) comparison on the structures of laser-supported absorption waves for pressure, temperature, and density. Adapted from Ref. 180, Marcel Dekker Inc., 1989.
Analysis of LA shock dynamics more frequently focuses on measuring and characterizing the expanding laser-induced shockwave as compared to the laser-supported absorption waves. Measurements are conducted using several different experimental methods including shadowgraphy,197–200 Schlieren imaging,201–203 interferometry,200,204,205 probe-beam deflection techniques,206–208 and Rayleigh scattering.209,210 Shadowgraphy is often applied as a qualitative analysis method, where individual shadowgraphic images of the shock front position are recorded as the shock expands outwards to provide snapshots of the shockwave trajectory through the propagation medium. Other methods such as interferometry and Thomson scattering are capable of providing a more quantitative analysis and have been used to measure electron densities of the laser-induced plasma located behind the shock front.209,211,212 The spatiotemporal evolution and expansion dynamics of both the plasma plume and laser-induced shockwave have been extensively studied in the literature, yet still remain a current research interest to explain the complex interaction of LA plasmas with their surrounding environment. Interests also exist in exploiting laser-induced shockwaves to compress and heat the plasma through shock reflections, thereby encouraging the population of higher energy levels and enhancing thermal emission signals (i.e., LIBS signals).213,214 Generally, larger shockwave propagation distances and velocities are routinely observed for atmospheric conditions consisting of lower gas pressures and densities.54,200,215–217 It has also been shown that shockwave velocities increase for decreasing irradiation wavelengths used during LA. 218 Furthermore, higher laser fluences produce stronger shockwaves with larger propagation velocities.54,200,219
Estimates of the initial energy supplied to produce the laser-induced shockwave are obtained using the Sedov-Taylor blast wave model,220–222 which has been implemented with various strong shock phenomena including atmospheric nuclear weapons testing to estimate device yields223,224 and supernova explosion events to describe expansion dynamics.
225
The blast wave model is an exact analytical solution that describes the distance an expanding blast wave travels through a homogeneous atmosphere with constant specific heat and density, and was derived using self-similar theory assuming a strong point source explosion.
226
The generalized formulation provided for different shockwave geometries is given by
The range of validity for the Sedov–Taylor model is based on assumptions made during its derivation and covers the following limiting dimensions of the spherical blast wave:226,229
Radioactive Plume and Atmospheric Monitoring
Real-time atmospheric monitoring of uncontained releases of a nuclear inventory with LIBS requires detecting signatures from mobilized nuclear material, commonly consisting of highly diffuse gaseous fission products or radioactive aerosols. In the event of a nuclear detonation, fission gases (e.g.,
Addressing challenges involved with measuring trace concentrations of airborne species requires examining the fundamental interactions occurring with each independent laser shot. In the case of ns-LIBS, the air plasma is generated by exceeding the laser-induced breakdown threshold of air, whereby the initial seed electrons liberated from multi-photon ionization are accelerated to form collisional ionization cascades. A typical neodymium-doped yttrium aluminum garnet (Nd:YAG) laser with a fundamental wavelength of 1064 nm requires an irradiance of
At this point, the LPP can be defined by a discrete sampling volume capable of characterizing constituents in air, which given the repetition rate of the pulse train corresponds to the volumetric sampling rate of air. Thus, it is optimal to maximize the volume of the LPP by increasing the pulse energy to several hundred mJ, which has also been shown to improve the signal-to-noise ratio (S/N) in aerosol measurements. 241 Trace species are sampled with respect to the volumetric sampling rate of the LPP where lower concentrations of airborne species require increasingly longer measurement times to resolve emission peaks. For aerosols at low concentrations, most laser shots will not interact with the trace number of particles present in air and thus will not result in any measurable emission signal. In cases such as these, single-shot conditional analysis is often used where only the laser shots that resulted in particle emissions are kept to form an averaged spectrum.240,242 An example is provided in Figure 8 for a low-concentration Cs aerosol, where conditional analysis is shown to be necessary to yield a resolved Cs emission peak at 852.11 nm. Only 5% of the total 10 000 laser shots in Figure 8a interacts with a Cs particle while Figure 8b demonstrates an average encompassing only 500 shots contributing to the analyte signal. Thus, it is understandable that to achieve an equivalent S/N at even lower concentrations, longer measurements are required to net a similar 500-shot average as the frequency of particle interactions decreases. 243 At higher concentrations, conditional analysis is not required and the shorter measurement times are ideal for real-time monitoring applications which have been demonstrated for Cs and Rb aerosols mixed with trace Xe and Kr gases, along with several other lanthanide elements.62,244,245
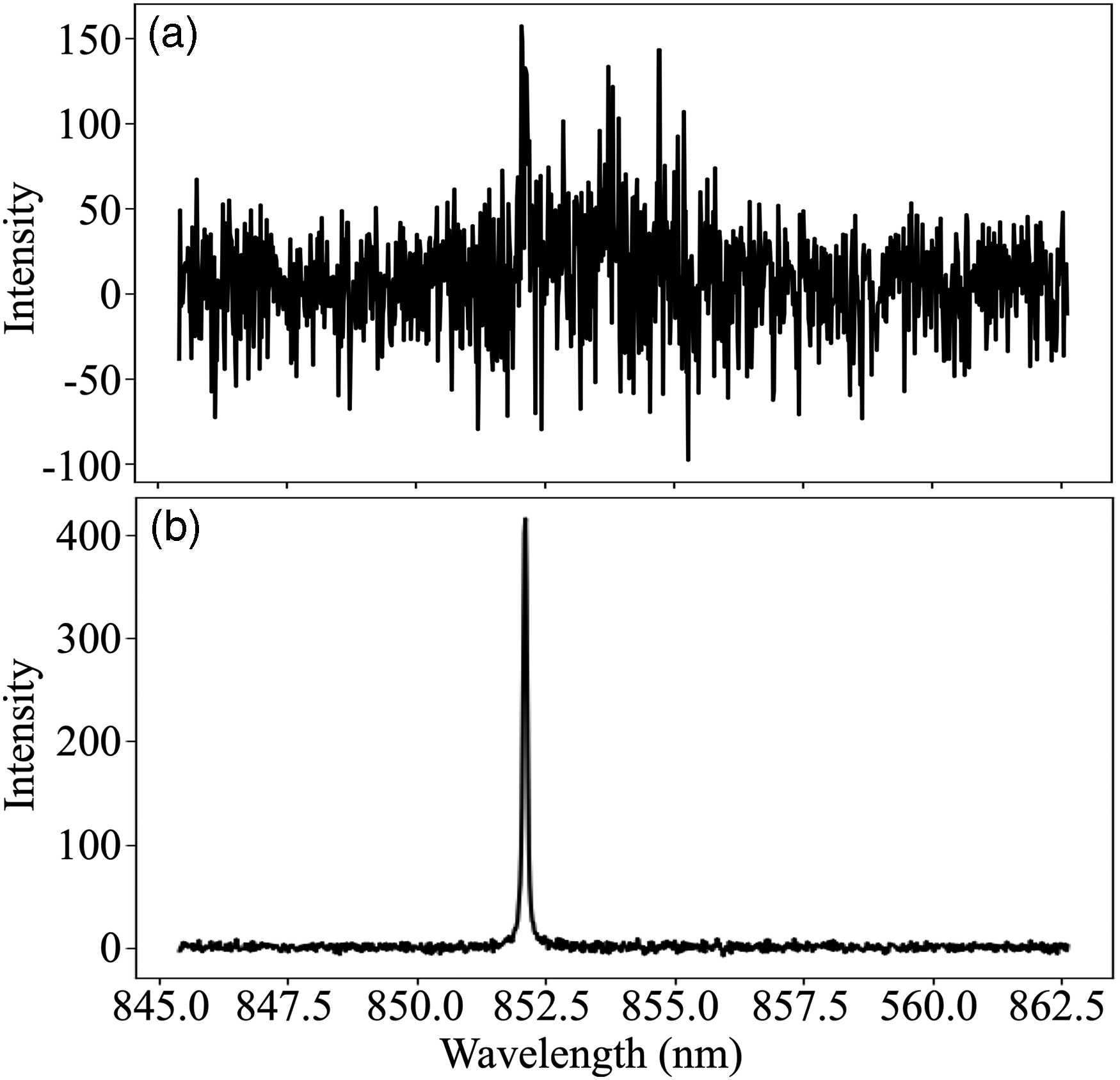
Conditional analysis of low-concentration Cs aerosol measurements using fs-filaments showing (a) 10 000-shot average without conditional analysis and (b) with conditional analysis, where only the 5% of the total shots showing Cs emissions are kept to yield a 500-shot average. Source: The authors.
The presence of particles and differences in their thermophysical properties can lead to matrix effects for both gas and aerosol species. Matrix effects refer to broad changes in emission characteristics influenced by the sample matrix, which in the case of air plasmas, is defined by the gas and particle species in the LPP volume. For aerosols, particles that are several µm in diameter are prone to undergo incomplete dissociation in the plasma, resulting in a particle-size matrix effect.239,246 Given that atmospheric radiation plumes consist mostly of sub-µm particles with slower settling velocities, particle-size matrix effects can be considered negligible. 247 However, for particles with diverse compositions, as in the case of fallout particles, differences in emission yields from particles with nonuniform thermophysical properties interacting in the LPP are inevitable, resulting in elemental fractionation. For example, Na particles generated from an aerosolized solution have been shown to yield up to 50% stronger Na emissions during specific times in the plasma evolution when Cu is included in the particle matrix without changing the concentration of Na. 248 Doping the particle matrix with other elements affects its thermophysical properties such that the local temperature of the particle in the plasma is directly influenced. Given that emission and excitation occur from the transfer of heat and mass during plasma–particle interactions, changing the plasma temperature corresponds to a shift in the population distribution of excited states, altering emission intensities depending on the upper energy level of the transition. 106 Particles can also be considered a contaminant when measuring gas species as they can introduce reactive elements found in the particle matrix that affect the plasma chemistry. This has been shown to influence atomic and molecular emissions from air species as elements in the particle matrix can react to form compounds with dissociated air molecules, thus introducing new reaction pathways that can potentially affect recombination rates.106,249,250 While elemental fractionation is unavoidable when monitoring airborne species, implementing time-resolved measurements can target specific windows during the plasma evolution where fractionation effects are minimal. Multivariate approaches have also been used in instances where the sample matrix is well-understood prior to measurements which has been successful in eliminating matrix effects. 251
Another optimal approach to minimizing temperature-driven matrix effects utilizes fs-lasers. Unlike air plasmas generated using ns-pulses, the plasma wave front is not fully developed on an fs-timescale, resulting in a colder plasma due to negligible heating from plasma shielding.
96
Thus, temperature-driven mechanisms such as elemental fractionation are de-emphasized and have been shown to be reduced by more than 50% compared to ns-LIBS.
106
Furthermore, the capability of ultrafast pulses to form laser filaments enables standoff measurements of radiation plumes, which is important to minimize exposure and avoid contamination. Plasma images comparing ns-LPP, fs-LPP, and fs-filament are shown in Figure 9 for 62 mJ ns-pulses and 7.1 mJ fs-pulses.
240
Typical fs-LPPs are smaller in comparison to ns-LPPs due to their lower temperature and density; however, fs-pulses form a highly stable plasma that is not influenced by nearby particles as the plasma is formed directly from photoionization processes, not by collisional cascades characteristic of air breakdown.
96
Conversely, ns-pulses can interact with particles with low ionization thresholds prior to reaching the breakdown threshold in air, and the generated free electrons can trigger an early breakdown event.
241
Early breakdown is seen in Figure 9b, where the ns-LPP formed in the aerosol is shifted
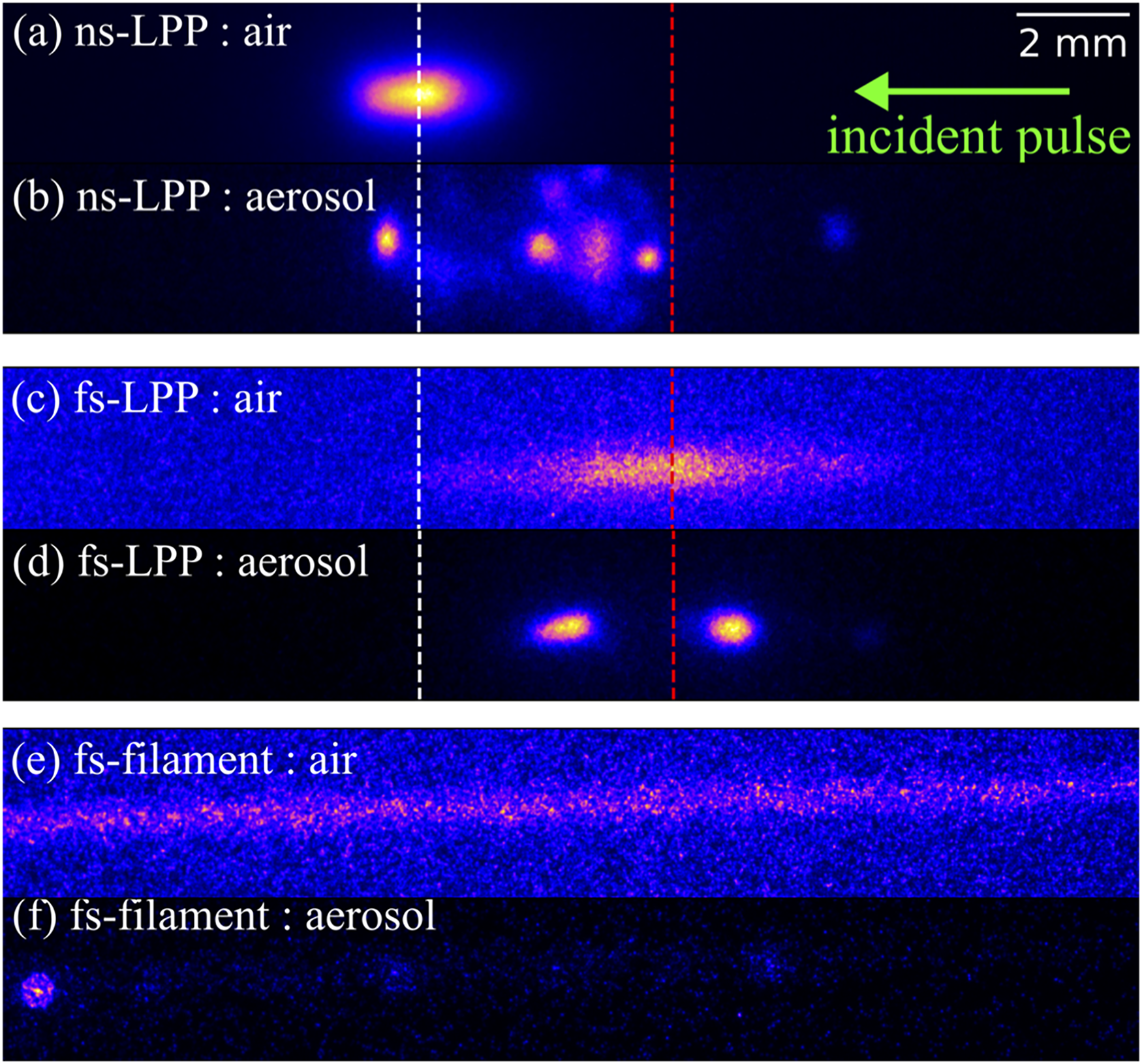
Images taken with a 590
Filaments have intrinsically unique properties, forming a thin plasma column approximately 70–100 µm in diameter that have been demonstrated to be tolerant to atmospheric turbulence and attenuation in aerosolized environments, allowing high pulse energies to be delivered under adverse conditions.254,255 Particle emissions can occur along the entire length of the filament as shown in Figure 9f, requiring an on-axis or near-on-axis light collection scheme for optimized light collection. Under optimized conditions, Na aerosol measurements have been conducted from up to 70 m away
256
and repeated at lower concentrations (1.4 µg/m
Laser-Induced Breakdown Spectroscopy (LIBS) Throughout the NFC
Laser-induced breakdown spectroscopy (LIBS) has been heavily investigated for a multitude of applications in the realm of nuclear energy production, extending throughout the entire NFC. The NFC concept refers to the lifecycle of nuclear fuel from fuel enrichment and fabrication, throughout its duration in a nuclear reactor, and up to either the recycling or disposal of the irradiated fuel in a spent nuclear fuel repository (Figure 10). The robust applicability of LA methods at standoff distances has continued to make LIBS of interest to researchers in the nuclear field, where standard measurement techniques can be hindered by the extreme, hazardous environments of nuclear reactors and radiological hot cells.
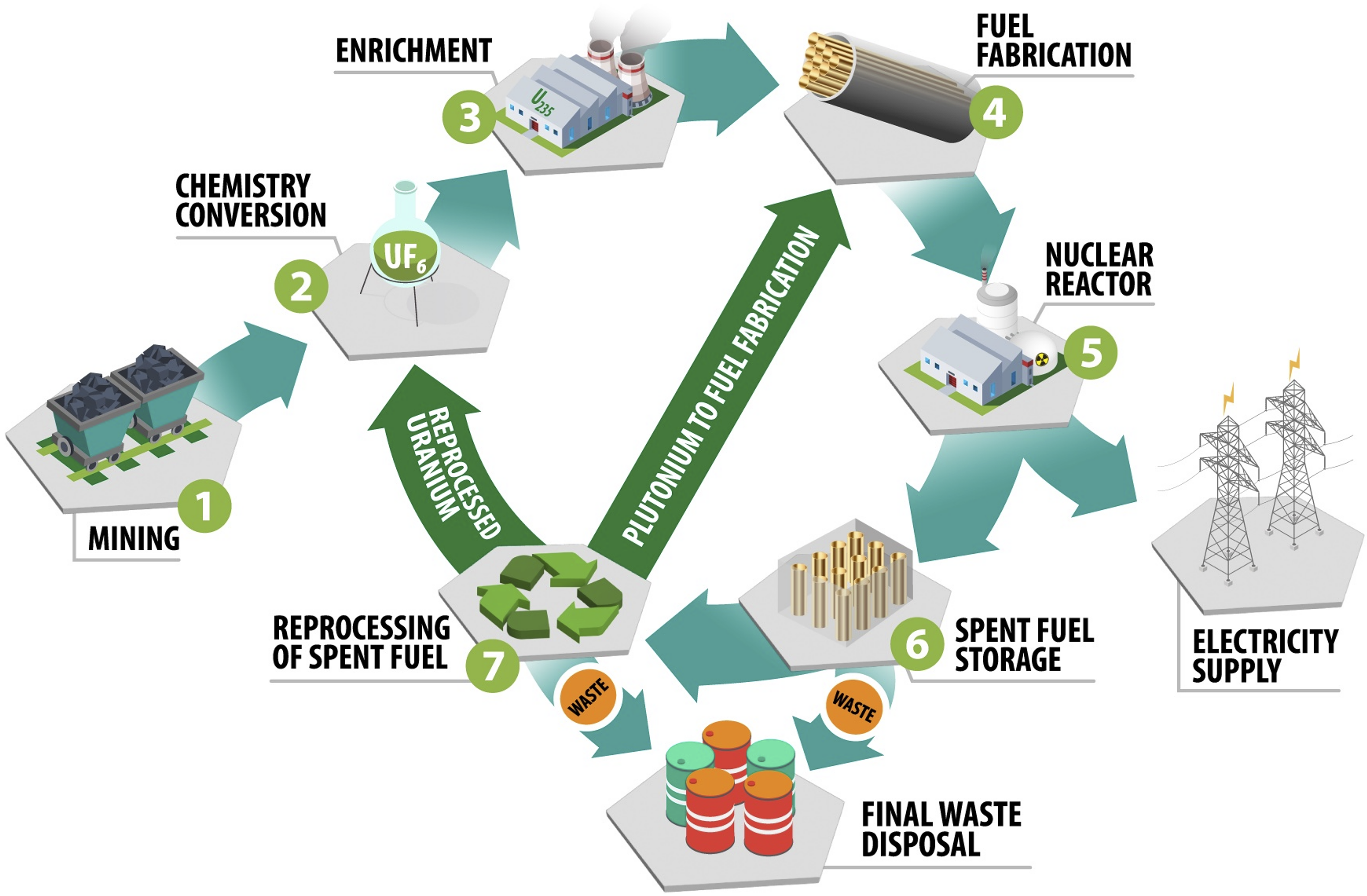
Stages of the NFC from ore mining to final waste disposal of spent fuel. Source: The authors.
Front End of NFC
The most prominent application of LIBS early in the NFC is on the direct assay of uranium enrichment in UF
Following the enrichment and fuel fabrication stages of the NFC lies the energy generation stage, where heat is generated from the splitting of atoms, or nuclear fission, and is then converted into electrical power. Nuclear power plants have traditionally employed oxide fuel surrounded by a metallic cladding with water serving as both the working fluid for heat transfer and neutron moderation. One of the main attributes of LIBS that makes it amenable for nuclear reactor monitoring is its ability to be fiber delivered, allowing sensitive equipment (e.g., laser, spectrometer, etc.) to be located in an area that is shielded and protected from radiation and heat. However, the fibers themselves are still exposed to ionizing radiation when using fiber optic (FO)–LIBS configurations. This poses a new challenge, as the optical fibers or other optical components (i.e., fused silica, sapphire, borosilicate glass, etc.), may darken with continued exposure to ionizing radiation. This darkening phenomenon is attributed to radiation-induced damage on atomic and molecular scales that impact material absorption properties. Davies et al. 271 investigated the impacts of radiation dose delivered to the optical fibers, finding that lower (shorter) wavelengths experienced more significant darkening and that the most significant darkening occurred during the first hour exposed under the 30 Gy/h dose c rate. 271 The fibers recovered from the darkening effects after removal from the gamma radiation, but this darkening behavior must still be accounted for in many nuclear applications of LIBS. Many of these FO–LIBS studies have focused on the detection of trace elements in various steels around nuclear power plants. Whitehouse et al. 272 implemented a 75 m FO–LIBS system to measure the Cu content in steam generator tubes during a reactor outage; they used the Cu content in the tubes as an indicator of the alloy manufacturer to locate at-risk components. Ernst et al. investigated Cu content in a reactor pressure vessel as an indicator of radiation embrittlement.272,273 FO–LIBS for nuclear research has also been employed by several research groups with systems ranging up to 100 m in length.271,272,274–277
Other elements that are commonly measured in-core as indicators of potential structural failures or material damage include Cr, Mn, Mo, Ni, Si, and V, where limits of detection range from around 100 to 200 parts per million (ppm).271,274,277–279 Crucial research efforts also include developing instrumentation to provide more affordable alternatives to Nd:YAG lasers that are typically implemented with the current detection systems. For example, Tamura et al.275,276 developed a microchip laser FO–LIBS system that could potentially offer a cheaper alternative to the typical Nd:YAG lasers used. This system was tested using radiation doses up to 10 kGy/h, where adequate signals were obtained under these conditions. 276
The ability of LIBS to detect and differentiate nearly every element in the periodic table in seconds makes it an appealing method for light element detection. Kautz et al.
280
investigated the use of LIBS for measuring H isotopes (D:
Reactor Monitoring
Recent decades have seen significant effort into the development of advanced reactor designs that use alternative fuels, moderators, and coolants to provide better efficiency and safety. Some of these advanced reactors include HTGRs, MSRs, and liquid sodium fast reactors (LSFRs). LIBS has been applied in several different roles to support these research and development efforts.
Advanced gas-cooled reactor designs use a He coolant with either fuel pins or a pebble fuel bed with graphite moderators for fast breeder reactors or thermal reactors, respectively. Design concerns involving the use of fuel pins include the leakage of fission gases into the coolant gas through cracks in the fuel cladding. In response, Burger et al. 245 have proposed a method using LIBS to detect trace Xe in a He atmosphere, where a limit of detection (LOD) as low as 0.2 µmol/mol was achieved. Vors and Salmon 285 have also applied LIBS to monitor the degradation of graphite moderators in gas-cooled reactors by measuring the amount of carbon present in the coolant gas. In a recent study, Manard et al. 350 performed elemental mapping and depth profiling of the aforementioned pebble fuel, or tri-structural isotropic fuel, using LIBS. Here, LIBS offered the benefit of distinguishing among metal-oxide, metal-carbide, and carbon layers, providing an avenue for quality assurance during fuel production.
Liquid sodium fast reactors (LSFRs) propose the use of liquid Na as the coolant, rather than a gas or water, due to its beneficial heat transport properties and its inability to moderate neutrons. This makes liquid Na a strong fit for fast reactors. 286 Similar to the work discussed earlier for HTGRs, Namitha et al. 61 investigated the use of LIBS to monitor for Xe and Kr fission gases as well as He buffer gas from a failed fuel pin. The calculated limits of detection for Xe, Kr, and He were 22 parts per billion (ppb), 40 ppb, and 2 ppm, respectively. Several studies have also focused on measuring liquid Na directly for impurities with limits of detection for Pb and In of 6 and 5 ppm, respectively.287–290 While these limits of detection (LODs) are low in regard to those found in many LIBS applications, Maury et al. 290 indicated LA-LIF approaches were being pursued for increased sensitivity to measure species with much lower solubility limits in the liquid Na. LIBS has also been applied to monitor Na aerosols outside of the reactor vessel to identify coolant leakages. 290
Molten salt reactors (MSRs) also use a high-temperature molten coolant such as LSFRs, but in these designs molten salts can serve as both the coolant and fuel form, eliminating the use of traditional cladding-based fuels and associated concerns regarding radiation damage to the fuel pellet materials. Instead, the use of molten salts presents unique challenges in material compatibility and chemistry (e.g., corrosion), salt monitoring, and the continuous generation of volatile species and fission gases. To address these challenges, LIBS has been employed with several efforts to determine the effectiveness of the method for quantifying salt-induced corrosion on structural materials, including a study on the interaction between light elements (e.g., F, Li, Be, Na, K, H, and O) with nuclear-grade graphite and stainless steels subsequent to their exposure to molten salts.291,292 Three-dimensional maps on the permeation of salt species into graphite samples have also been presented under controlled conditions, where effects such as salt hydration were absent. 292
In addition to the corrosion of graphite resulting from interactions with molten and aerosolized salts, other volatile species and noble gases generated during reactor operations must be monitored to maintain a clean cover gas for the reactor core. 60 In response, Andrews et al. developed a LIBS system to continuously monitor the off-gas stream from MSRs in real time, where off-gas constituents include aerosolized fission product species (Gd, Nd, and Sm), noble gases (Xe and Kr), and their respective daughter products (Cs and Rb).62,244,293,294,295 The system uses a sheath gas approach to contain the aerosols within a sample chamber, thereby protecting the optics from particle deposits and making it amenable to in situ deployment in industrial scale systems, where the system would need to operate for extended periods of time before requiring replacement of any optical components exposed to aerosols. LIBS has also been demonstrated as a useful tool for evaluating off-gas treatment components in real time, such as monitoring the Xe and Kr capture by a metal-organic framework filter in the vent stack of potential nuclear fuel reprocessing facilities. 295
Lastly, LIBS has been proposed as a strong candidate to directly monitor the reactor salt itself. Pedersen
296
proposed the use of an in-line LIBS system for monitoring fuel salt with the Copenhagen Atomics MSR design. Sarkar et al.
297
demonstrated the use of a partial least squares (PLS)–artificial neural network (ANN) hybrid model to monitor U concentrations in UF
Used Nuclear Fuel Disposal and Reprocessing
The back end of the NFC involves handling, recycling, and/or disposing of nuclear fuel after it has been irradiated over several cycles (typically 18–24 months per cycle) in a reactor. At this point, the fuel is considered to be “used” nuclear fuel (UNF) and is transferred to a spent fuel water-filled pool to allow for short-lived radionuclides to decay and the resultant decay heat production to be reduced. This UNF is then placed into an on-site dry cask for storage, pending transport to a geological repository or reprocessing facility to recover approximately 95% of the fissile material that remains viable for future irradiation and energy production. LIBS has been applied to ensure the safe storage of UNF in dry casks 299 as well as reprocessing of UNF. 300
Several studies have investigated the use of LIBS for measuring UNF, where the technique can measure a plethora of fission product elements without the dissolution of solid samples or diluted samples requiring removal from a hot cell for analysis.301–303 Again, FO–LIBS is widely proposed to allow in situ measurements of samples in hot cells while keeping equipment protected. Singh et al.302,303 demonstrated the ability to detect nine main fission product species in simulated fuel materials and several species in nuclear waste glass.
A multiyear project focused on the development of a LIBS system relying on robotic delivery of ablation and collection optics coupled to a singular fiber was conducted to routinely inspect the steel surfaces of dry cask storage systems exposed to ambient environmental conditions for salt contamination that may lead to salt-induced stress corrosion cracking.299,304–306 Early works demonstrated the applicability of LIBS techniques to monitor Cl on steel surfaces using DP–FO–LIBS, and resulted in the development of calibration curves for Cl concentration determination ranging 5–100 mg/m
Two primary methodologies for UNF reprocessing exist (aqueous reprocessing and pyroprocessing), both with the goal of removing fission products that interfere with the reuse of the UNF in a reactor and recovering the unused fissile material for subsequent use. Briefly, aqueous reprocessing utilizes liquid–liquid extractions where the UNF is dissolved into an aqueous product and then contacted with an organic solvent that extracts the uranium. In pyroprocessing, the UNF is dissolved into a molten (typically >800 K) salt solvent (e.g., LiCl–KCl eutectic) where the fissile materials can then be refined through electrochemical reduction onto a metal cathode or into a liquid metal cathode. The cathode can then be extracted to recover the fissile material. Both methods of reprocessing involve hazardous environments, radioactive samples, and caustic and corrosive materials, making them both ideal application spaces for LIBS research.
Much of the LIBS research for reprocessing applications has included measurements of lanthanide fission products and impurities.300,307–313 LIBS spectra of lanthanides typically exhibit thousands of emission lines, which provide a wealth of possible lines to choose from but also generate significant spectral interference issues due to the substantial broadening of these closely spaced lines characterizing the LA plasma. Many studies have focused on measuring several lanthanides simultaneously to identify key peaks of interest that exhibit minimal spectral interference. Several studies have also focused on estimating lanthanide and actinide transition probabilities to enable calibration-free-LIBS and Saha–Boltzmann measurements for these species where traditional calibrations may be hindered by lack of available materials.310,311,314 For many aqueous reprocessing applications, the direct measurements of liquids are a key interest.312,313 Ruas et al. 313 demonstrated the use of a liquid jet configuration to measure Zr in solutions with limits of detection of 4 mg/L. The liquid jet configuration was shown to mitigate issues that plague traditional liquid LIBS applications, such as splashing due to laser-induced cavitation and variable liquid surface location.
Online Monitoring and Pyroprocessing
Several LIBS systems have been set up in hot cells to enable the spectroscopic analysis of radioactive environments. 315 Singh et al. 316 demonstrated a standoff approach for measuring a Cs sample in a surrogate hot cell. A recent paper from Griffiths et al. 317 details two hot cell LIBS systems developed by Applied Photonics Limited (APL); one fires the laser through a port to focal lenses inside the cell, while the other uses an umbilical to connect the power source to the laser house in the LIBS probe inside the cell as the collected light is sent back through the umbilical to the spectrometer in the control room. This study also compared ultraviolet fused silica (UVFS) optics with BK7 glass, finding that BK7 components darkened from irradiation, whereas the UVFS components were far less affected. A similar FO–LIBS probe from APL was installed in a hot cell at the Korea Atomic Energy Research Institute. 318 Offey et al. 319 developed a robotic arm for hot cell deployment equipped with LIBS and Raman systems mainly for material classification and identification.
LIBS has been studied significantly in pyroprocessing research in the last decade, mainly in the pursuit of in situ salt monitoring for process control and safeguard purposes. Despite the ultimate goal of online monitoring of these processes, much of the work in this space has been completed with frozen salts due to the difficulties of liquid LIBS heightened with corrosive molten salts. Many studies have used siphoned salt samples frozen into cylindrical ingots for testing.320–324 Williams et al. 320 quantified Ce and Gd in frozen LiCl–KCl held inside small sample jars but had challenges due to salt dust accumulating on the jar and degrading light collection. Andrews and Phongikaroon321–323 used a similar approach in an inert glovebox, firing the laser into the glovebox through a quartz window to quantify Sm, U, Gd, and Mg in LiCl–KCl salt ingot. For the latter three species, a sensor fusion method that combined LIBS and electrochemical signatures was used to successfully quantify blind samples. 323 Hull et al. 324 tested salt ingots mounted inside an Ar-filled sample holder, which used vacuum fittings to ensure a sealed system to quantify Pr, Ho, and Er in LiCl–KCl. LIBS has recently been used to quantify O in frozen salt samples with a LOD of 236 ppm. 325
Relatively few studies have attempted to measure salts in their molten state.298,326–329 Hanson et al. 327 investigated the response of the LIBS signal to the temperature of the salt sample. The temperature was varied such that the sample was tested as a solid salt, in a transition state, and ultimately, in molten form. The results indicated that the molten salt sample provided the most repeatable data and the lowest self-absorption, but the signal intensity was lower with the liquid sample. Recent studies by Lee et al. have applied many of the lessons learned from the previously discussed studies and made improvements to their experimental system and their quantification models.298,328,329 These include a glovebox LIBS system with the laser being passed into the box through a quartz window and the use of acoustic sensors to account for variance in the ablation process due to shot-to-shot movement in the liquid sample. Additionally, a cover gas was used to prevent salt dust from accumulating on the optical lenses. These systematic improvements resulted in a system able to measure Ce in molten LiCl–KCl with a LOD of 360 ppm. 329 Lee et al. have also used machine learning to train a model with an intentionally imperfect collection of the emitted spectra, thus making the trained model more robust to variation in the liquid level. They were able to monitor Sr and Mo in LiCl–KCl at concentrations ranging from 500 to 7000 ppm using a machine learning algorithm trained on purposefully defocused LIBS spectra to account for fluctuations in salt surface level height variations. 328
Lastly, an aerosol system was developed by Williams and Phongikaroon330,100 for online monitoring of pyroprocessing salts. The aerosol system used a Collison nebulizer to mitigate clogging issues due to the presence of a high concentration of salts, where LIBS was performed directly on the aerosol stream. The system was demonstrated on Ce and U in LiCl–KCl molten salts at 773 K. The system’s ability to extract salt samples from a liquid sample pool and send the generated aerosol to another location for LIBS testing represents a very viable approach to deploying LIBS on an industrial pyroprocessing scale.
Fusion Reactors
While research on current and advanced fission reactor fuel cycles has continued, so too have efforts toward establishing nuclear fusion as a viable energy source. During nuclear fusion, two light nuclei (in many cases a deuteron and a triton) are forced together, overcoming their Coulombic repulsion to fuse and form a helium nucleus. During this process, small amounts of mass are converted into energy, which could be converted into electrical power. Unfortunately, to overcome the Coulombic repulsion of the two fuel elements, a significant amount of energy is needed to heat and confine the fuel as an ionized plasma for a sufficient time period to facilitate the fusion reactions occurring (i.e., the Lawson criteria). For nuclear fusion to become commercially viable, the net energy from the fusion reactor must be positive, and the fusion reactor must be able to sustain the reaction or pulse the reaction rapidly enough to produce power over long enough periods to enable predictable and controllable supplies of energy to an electric grid. A continual focus of research into nuclear fusion energy production has been the development of advanced materials for the PFCs, and it is in this area that LIBS has seen significant applicability.
The vast number of studies on LIBS as an in situ diagnostic tool for PFCs revolve around a few key subjects. Galmed et al. 331 correlated LIBS plasma temperatures to Vickers hardness changes related to C ion irradiation as expected in Tokamak PFCs. Many studies focus on using LIBS to monitor material deposition or H isotope penetration into PFCs.332–336 Mittelmann et al. 332 used fs-LIBS along with calibration-free approaches to quantify D in W materials. Zhao et al. 334 investigated ps-LIBS, finding that the lowest energy ablation regime resulted in the optimal W signal and a minimal ablation rate, providing very high-resolution depth profiling capabilities. Hai et al. 337 investigated the LIBS spectral response to sample temperature and found the signal to be enhanced at high temperatures, which would be advantageous for in situ monitoring of PFCs during operation. Hernandez et al.338,339 developed a remote-operated LIBS system for tritium and dust inventory control for Tokamak reactors that could be used under operational conditions. An initial set of LIBS measurements was performed using the remote-operated system; however, this work did not demonstrate measurement under Tokamak operating conditions or measurement of the deuterium and tritium contamination on the PFC.
Several proposed fusion reactor designs rely on the blanket of a fusion reactor serve to absorb and transfer the energy of the fusion reaction products and to produce the tritium (
It is clear from the above discussion that early successes have occurred for the measurement of H and Li isotopes including under ambient atmospheric conditions using relatively few laser shots and elegant LIBS variations such as LIBRIS. Each of the above-highlighted works has focused on the application of H and Li isotope measurement to nuclear security and forensics; thus, an opportunity exists to investigate applications of LA plasma spectroscopy techniques for molten salt blanket material in proposed fusion reactors where monitoring the production of tritium and depletion of
Nuclear Security, Safeguards, and Forensics
Nuclear security, safeguards, and forensics measures aim to stop the proliferation of nuclear weapons and illicit trafficking of nuclear and radiological materials as well as enable response to and attribution of nuclear detonation events. The discovery of fundamental nuclear physics processes such as radioactivity and fission resulted in the rapid advancement of nuclear technologies during the first half of the 20th century. This rapid advancement resulted in the establishment of significant commercial and military use of nuclear technology and materials, which culminated in a need to verify the peaceful use of these technologies and materials to limit the spread of nuclear weapons. Nuclear security broadly encompasses all activities related to the protection of people, property, society, and the environment from the harmful effects of ionizing radiation. 7 Nuclear safeguards are a set of technical measures that are applied by the IAEA on nuclear facilities and materials through which the agency seeks to independently verify a state’s legal obligation to ensure nuclear facilities and materials are not diverted from peaceful purposes. 345 Nuclear forensics is the examination of nuclear materials using analytical techniques to determine the origin and history of these materials in the context of law enforcement investigations. 346
The technological capability needed in nearly all nuclear security, safeguards, and forensics applications is the detection and detailed characterization of materials that occur in the nuclear fuel and weaponization cycles.7,347 Materials of interest for characterization include the minor and major actinides in the NFC (U and Pu particularly), compounds used in nuclear fuel fabrication and reprocessing, isotopic enrichment, and other structural or ancillary materials associated with nuclear power plants. Special nuclear material is a uniquely defined subset of nuclear materials by the U.S. Nuclear Regulatory Commission that can be directly used in a nuclear weapon, such as highly enriched
Historically, the approach to characterize nuclear materials is to detect the interaction of ionizing radiation (e.g., alpha, beta, gamma, X-ray, and neutron radiation) emitted through radioactive decay common to many nuclear materials. Similar to the spectral signatures of atomic spectroscopy described in detail in the sections above, the energy of emitted ionizing radiation commonly contains information about the unique source nuclide (i.e., isotope) that the radiation emanated from. Ionizing radiation exhibits a limited range in the ambient environment and relatively poor measurement statistics based on the long half-lives of many nuclides of interest for nuclear security, thereby requiring macroscopic amounts of material to be present and/or lengthy analysis timelines for traditional radiation detection systems.
Actinide Measurements
The significant interest in LIBS for analysis of actinide-containing samples (as highlighted earlier in this review in Figure 1) stems from the need for rapid, accurate, and sensitive field-deployable instruments and techniques capable of performing measurements at standoff distances in the field under potentially hazardous conditions that can overcome some of the challenges that arise in traditional ionizing radiation measurements.42,49,63 In this section, we review several recent applications of LPPs for the measurement of the major actinides and their isotopes, which is of substantial interest for the nuclear security, safeguards, and forensics communities.
Within the nuclear field, one of the first applications proposed for LIBS was in the area of process monitoring for nuclear fuel reprocessing and other NFC facilities to enable the safe operation of these facilities as well as nuclear safeguards. In the context of the NFC, this topic is discussed in detail in the previous section of this review. One of the first measurements of U using LIBS was performed in 1987 by Wachter et al., 348 who demonstrated the ability of LIBS measurements to not only identify U in a nitric acid solution, but also with sufficient precision and dynamic range to perform process control analysis of many streams within NFC facilities. Since this early work, many more studies have appeared within the literature that are focused on the detection of U for monitoring nuclear weaponization activity signatures in the environment using both laboratory-based and fieldable instruments.340,349,350 The primary difficulty with LIBS measurements of U is the analysis of the complex and congested spectrum characterizing the element, where an example is shown in Figure 11. The number of emissions lines present for the major actinides of interest adds the capability to utilize several signatures for identification and quantification of U in a sample, but also serve to reduce the intensity of the average emission line compared to simpler elements commonly measured and investigated with LIBS in the literature.19,119 Several other elements (mostly lanthanide, actinide, and some transition metal elements) show similar LIBS spectral complexity and, beyond the stronger impact of oxidation effects present for U and other major actinides, methods to develop this technique toward the quantification of elemental U in samples mirrors efforts more broadly in the community to do the same for these other elements like Fe.351–355 A more detailed discussion on recent advances in data science for the quantification and interpretation of LIBS measurements are provided in the next section of this review; however, in the context of measurements of the major actinides in LA plasma spectroscopy (namely LIBS for simple elemental detection), it is expected that advances in machine learning and other chemometric techniques for the lighter elements commonly applied toward applications on characterizing geological ores as well as metal recycling and refining will continue to benefit LIBS measurements of the actinides.
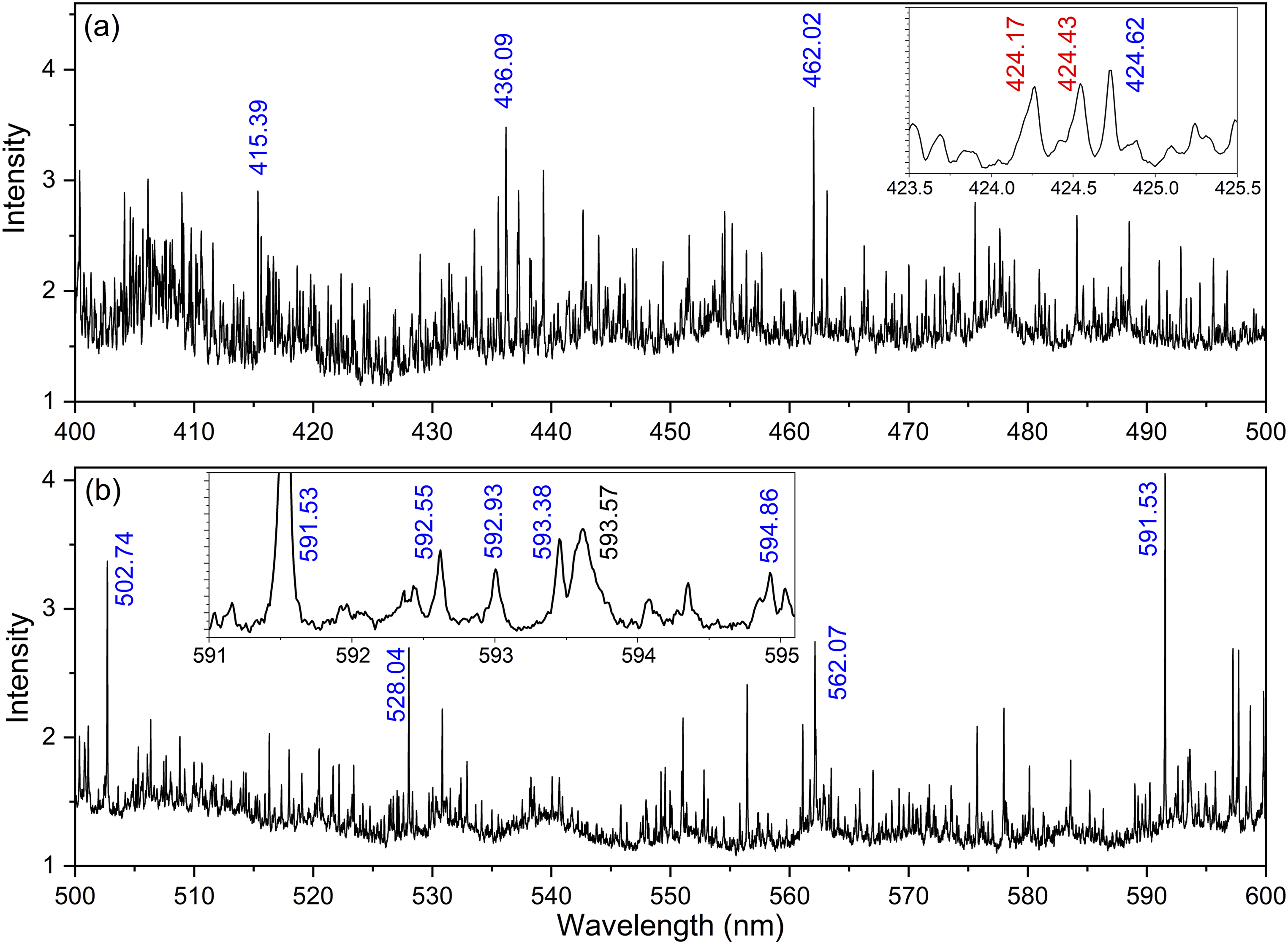
Intensity calibrated LIBS emission spectrum of natural uranium metal sample following fs-LA in the range of (a) 400–500 nm and (b) 500–600 nm with a 1.0 µs gate delay and 10 µs gate width averaged over 10 laser shots with an energy of 6 mJ. After the vacuum chamber was evacuated, it was filled with argon to a pressure of 82 Torr; however, 3 Torr of ambient air leaked into the chamber during the measurement. Strong and commonly referenced U emission lines are labeled in the spectrum for neutral (blue), ionic (red), and UO molecular (black) emissions. Reproduced with permission from Ref. 141, The Optical Society, 2017.
Laser-Induced Breakdown Spectroscopy (LIBS)
Observation of isotope shifts via optical emission spectroscopy is not a common application for LIBS, mainly due to the very high spectral resolution and spectral linewidth required to perform such measurements. One of the earliest applications of LIBS to the nuclear security field was performed by Pietsch et al.,
356
where the authors reported for the first time the isotopic splitting of a U emission line in a LA plasma. The reported isotopic splitting between the
Laser Ablation Molecular Isotopic spectrometry (LAMIS)
Laser ablation molecular isotopic spectrometry (LAMIS) is an LA plasma spectroscopy technique developed by Russo et al. 359 that relies on the chemistry that occurs in the LA plasma plume under ambient atmospheric conditions to determine the isotopic composition of a sample of interest through the exploitation of diatomic molecular emission spectra. LAMIS measures molecular emission as the plasma cools at late times (typically >10 µs) when free molecules are formed in the plasma plume via several mechanisms. At these later times, excited isotopologues consisting of diatomic dimers, oxides, nitrides, or halides are formed from chemical reactions between the atomized sample matter in the plasma plume and reactive species in the ambient atmosphere. These chemical species can typically be measured in the absence of spectral interference from atomic or ionic emission because molecules radiate at later times during the lifetime of the plasma.360,361 The electronic molecular transitions are isotope-dependent; however, the magnitude of the electronic isotope shift is significantly smaller compared to the rotational and vibrational transitions. While the Russo group may have popularized the LAMIS technique for nuclear applications, as early as 1998 work by Niki et al. 362 demonstrated the exploitation of molecular spectra observed in an LA plasma plume for the accurate B isotopic ratio measurement.
The presence of molecular isotope shifts in both the vibrational and rotational molecular spectra allows for the exploitation of multiple emission lines in a small spectral window for isotopic analysis.
359
Isotopologue molecules observed in a LAMIS measurement exhibit significantly larger isotopic splitting than those in atomic spectra; however, the magnitude of the observed molecular isotopic splitting is a function of the reduced mass of the molecule, which means that for a heavy element such as U that the splitting observed will be much less than that of a light element such as B.
359
For uranium-containing samples, the diatomic molecule UO is formed following LA under ambient atmospheric conditions. The molecular isotopic splitting is reported to be
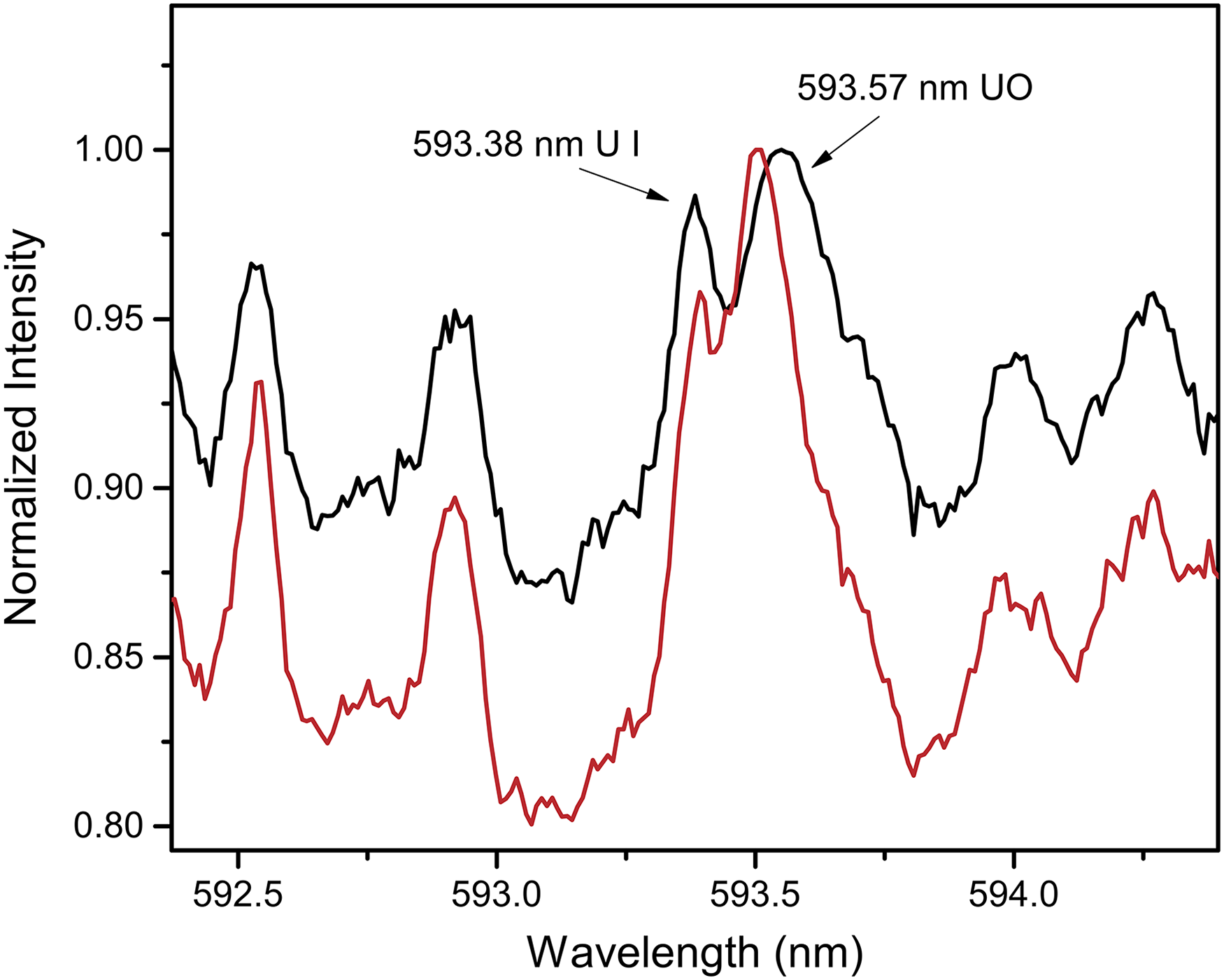
Standoff detection F2-LAMIS spectra at 1 m of the DU (black) and HEU (red) samples showing a molecular isotope shift of 0.05 nm for the 593.6 nm uranium molecular emission band head. Reproduced with permission from Ref. 42, Nature, 2017.
Both ns- and fs-pulsed lasers have been used for LAMIS measurements.
360
Femtosecond ablation yields significantly stronger molecular emission at a lower background as fs-LA generally results in a relatively cool and short-lived plasma versus ns-LA.363,364 The use of high-irradiance fs laser radiation offers the additional benefit for standoff detection applications using LAMIS because a self-focusing contracted (in the radial direction) plasma channel (“filament”) can be formed.
365
Laser filaments are able to propagate over substantial distances with a sufficiently small diameter (
Laser ablation molecular isotopic spectrometry (LAMIS) has been demonstrated for rapid optical analysis of isotopes of hydrogen, boron, carbon, nitrogen, oxygen, chlorine, strontium, and zirconium.
360
Advantages of this technique include the ability to measure isotope abundances with low-resolution spectrographs that exploit the large isotope shift observed in the spectra of diatomic molecules. Standoff ns LIBS measurements have been demonstrated at distances up to 120 m using a 400 mm aperture telescope to collect the LA plasma emission; it is possible to achieve similar standoff measurements using F
One method for improving the ability to resolve isotope shifts in LIBS measurement is through increasing the resolving power of the spectrometer; the use of large, high resolving-power spectrometers has been a constant in isotopically resolved measurements of atomic spectra. However, increasing the size of the spectrometer is not always feasible for in-field measurements. Effenberger and Scott 371 proposed an alternative method for performing high-resolution LIBS measurements by adding a low-cost Fabry–Perot etalon to the entrance slit of a 0.5 m focal length Czerny–Turner spectrometer. The performance of the instrument was demonstrated using the hyperfine doublet of the 313.2 nm Hg emission line, which exhibits a splitting of 29 pm, similar to the isotope shift for U. Ko et al. 372 further developed the hybrid interferometric/dispersive technique by developing a method to reconstruct the high-resolution spectrum for LIBS measurements and showed improvement in the spectral resolution and accurate reconstruction of the peak ratios compared to conventional dispersive only measurements. Ultimately, isotope selective measurement of U was demonstrated for the first time using the hybrid interferometric/dispersive instrument in a LIBS measurement. 373 Impressively, this measurement was performed under ambient atmospheric conditions, which was possible due to the efficiency of the collection optics placed close to the sample that was well coupled to the FO and the use of a moderately long gate delay of 10 µs and gate width of 5 µs that greatly reduced the broadening of the emission lines (similar to the earlier work by Chinni et al. 349 ). The development of a simple “add on” device to improve the resolution of manageable-sized spectrometers can help advance LIBS applications in the field where high resolutions and compact instrument sizes and portability are needed, such as in environmental or process sampling for nuclear safeguards. An alternative approach to retaining compact device sizes with high resolving powers is custom-made spectrometers that are designed to target limited spectral bandwidths over which emission lines and isotope shifts of interest are characterized.
Laser-Induced Fluorescence-Laser-Induced Breakdown Spectroscopy (LIF–LIBS)
The ability to perform sensitive (i.e., trace) and quantitative analysis of the elemental composition in samples is an important component, alongside isotope-resolved measurements, toward advancing the use of LA plasma spectroscopy-based techniques to nuclear applications. LIF–LIBS is an effective method for improving the analytical sensitivity and capability of LA plasma spectroscopy measurements for nuclear applications. First introduced by Kwong and Measures in 1979, 374 this technique involves tuning a second, in this case, pulsed, laser source to the specific atomic transition for the target analyte and propagating this tuned laser into the generated LA plasma plume. Typically, to avoid interference, the transition targeted by the tunable laser is one that has two or more strong emission lines that share the same upper energy level, so that the fluorescence emission (i.e., from rapid de-excitation of the upper energy level) can be observed at a different wavelength from that of the tunable laser that could interfere with measurement of the fluorescence emission. LIF allows for the selective excitation of emission lines associated with specific elements in the sample. The technique relies on a large population of atoms in the plume to exist in lower energy levels that will be excited by the tunable laser; thus, the technique exhibits substantial improvement in the emission intensity of targeted lines at later times when the plasma has substantially cooled and thermal emission has substantially decreased. The selective excitation of specific emission lines at late times in the plasma plume greatly increases the selectivity and sensitivity of LA plasma spectroscopy measurement as well. 375 Li et al. 376 compared the LOD and calibration curves for U measurement in ores using the LIF–LIBS technique and showed that both had been significantly improved over LIBS alone. The LOD for the LIF–LIBS technique was shown to be 35 µg/g, which is over five times smaller than that for LIBS alone at 199 µg/g. Additionally, the U transition lines in the ores were selectively enhanced to achieve the purpose of eliminating severe spectral line interference. Finally, Shen et al. 377 demonstrated that the LIF–LIBS technique has high selectivity and sensitivity and is suitable for real-time and in situ analysis of trace U in solid samples under ambient atmospheric conditions. Most of the tunable pulsed lasers on the market presently have the ability to automatically tune to specific target wavelengths of interest, allowing the operator to target many different transitions, and thus, elements of interest automatically. When coupled to a high repetition rate laser such selective and quantitative LIF–LIBS measurements can be done in near real time, which is useful for process monitoring in potentially hazardous facilities such as nuclear fuel reprocessing plants.
While the improvement in emission intensity in the LIF–LIBS technique is valuable, the pulsed tunable lasers used in such setups exhibit a bandwidth of 3 to 10 cm
Smith et al.
48
performed the first CW swept diode laser measurement of a U LA plasma plume in 1999 to perform LIF measurements of the
Laser Absorption Spectroscopy–Laser-Induced Breakdown Spectroscopy
In 2009, Bushaw and Anheier 379 expanded on the CW LIF–LIBS technique by developing a compact absorption-based technique termed LA-absorption ratio spectrometry (LAARS) for the rapid isotopically resolved measurement of micron-sized particles on swipe samples from potential safeguards enabled environmental samples in and around uranium enrichment plants. The authors prepared samples of Gd-bearing particles with different isotopic distributions as surrogates to demonstrate the feasibility of LAARS as an approach to isotope ratio analysis for characterizing environmental aerosol samples containing traces of enriched uranium. It was shown that by using two diode lasers to simultaneously monitor two different isotopes in different atomic transitions within the LA plasma plume isotope ratios can be accurately determined with good reproducibility on a shot-by-shot basis. The measurement sensitivity is also shown to be sufficient for measuring low ratios of <1% for individual micron-sized particles. Again, a rarefied (1.3 kPa) inert (Ar) atmosphere was used to reduce the linewidth of the absorption profile and suppress oxidation effects that act to reduce the ground state population, and correspondingly, the resulting LAS signal.
In 2012, Miyabe et al.
380
investigated the temporal evolution of the absorption signal for U I and U II transitions in the visible spectrum for the purpose of exploring the plume dynamics of U LA plasmas for the accurate determination of
Two-Dimensional Fluorescence Spectroscopy
Renewed interest in CW LIF–LIBS occurred following the work by Harilal et al.,
382
who used a two-dimensional fluorescence spectroscopy (2D-FS) LIF–LIBS technique to measure the coupled absorption and emission properties of atomic species in plasmas produced by the LA of Al targets at atmospheric pressure. The authors showed that time-delayed and gated detection of the emission spectrum could be used to isolate the resonantly excited fluorescence emission from the thermally excited (i.e., LIBS) emission from the plasma. Additionally, the tunable CW laser was able to resolve the intrinsic (physical) linewidth of the Al transition in the LA plasma plume by scanning the laser across the absorption spectrum of the Al transition, with ultra-high resolution at late times after the plasma has cooled, resulting in spectral linewidths that are not limited by instrumental broadening in the spectrometer system. Their result highlighted that fluorescence spectroscopy employing a CW laser re-excitation after LA combined benefits of both a spectrally resolved emission measurement with a swept CW tunable laser (i.e., two-dimensional). Harilal et al.
383
further developed the technique to show that by using the two-dimensional fluorescence spectroscopy technique on LA plumes, isotope shifts
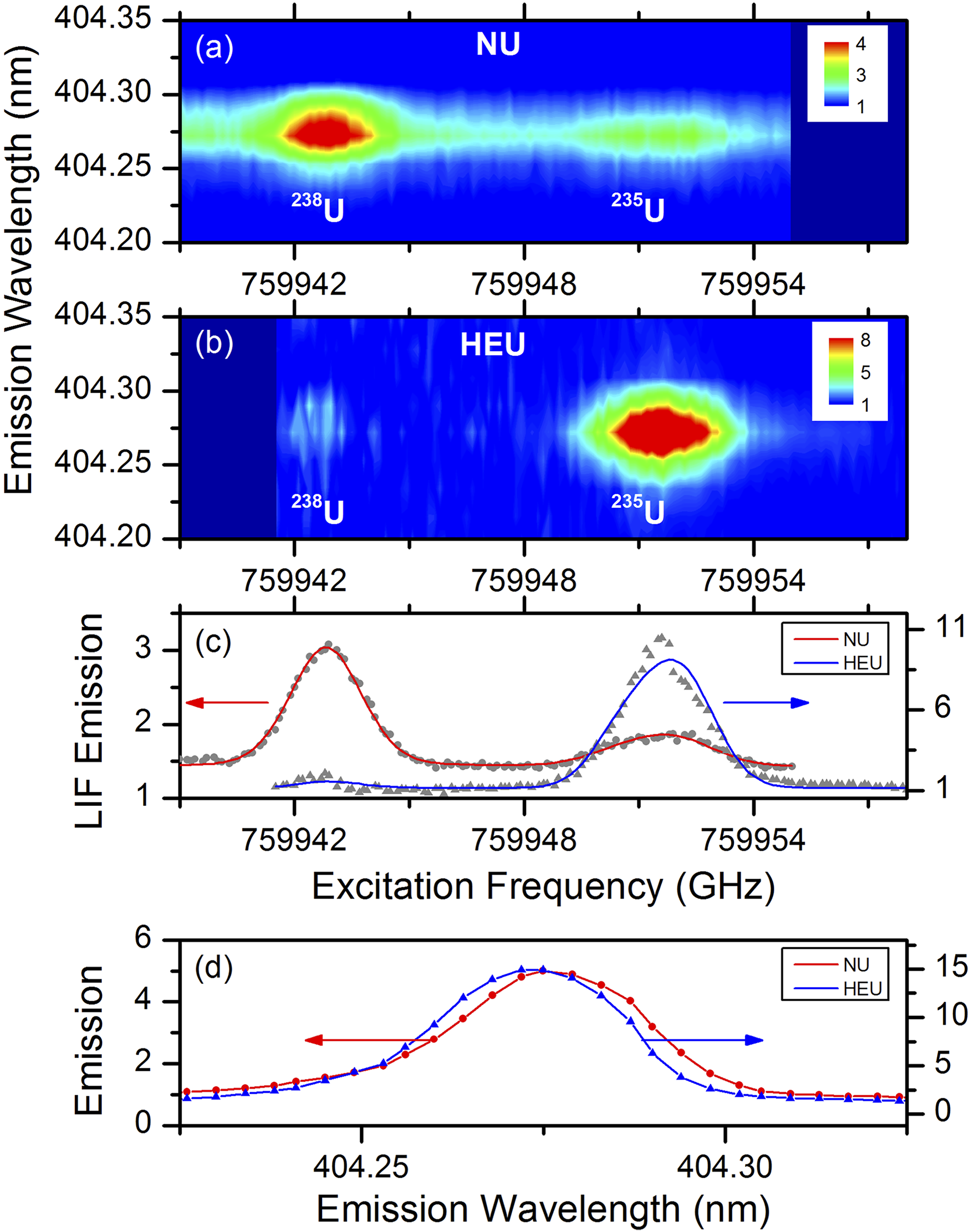
Two-dimensional fluorescence spectroscopy (2D-FS) of (a) natural uranium (NU) and (b) HEU uranium metal samples in 50 Torr Ar. The darker blue represents regions outside the scan range to align each measurement in the image. (c) LIF excitation spectrum for NU (red) and HEU (blue) samples. Solid lines show fits to the hyperfine structure and splitting of the U I 394.38 nm absorption line coupled into the emission of the 404.28 nm U I emission line. (d) Emission spectrum of the U I 404.28 nm transition for NU (red) and HEU (blue) samples with external cavity diode laser (ECDL) resonant with the
Filament-Induced Breakdown Spectroscopy (FIBS)
To conclude the discussion on LIF–LIBS techniques, Kautz et al.
43
demonstrated the application of LIF for increasing the emission intensity of standoff FIBS. The authors showed that ultrafast laser filament-produced plasmas generated from an Al target at varying standoff distances from 1 to 10 m, and LIF was observed to enhance the emission intensity by
Nuclear Forensics
While it is clear that measurement of isotope ratios of major actinides such as U and Pu are relevant for nuclear forensics, it is important to be clear on some definitions. Nuclear forensics is commonly defined as a suite of analytical techniques that are used to determine a materials composition, production history, and origin prior to being collected or intercepted primarily for law enforcement purposes. The techniques used in a nuclear forensics analysis must, typically, be admissible in a court of law, which requires substantial vetting and rigorous examination of each technique in the international community (i.e., published literature, conferences, societies, standards, etc.). 386 To put it simply, LIBS and related LA-based techniques have yet to show the LOD, accuracy, and precision necessary to be useful in a nuclear forensics analysis. The few works available in the literature related to nuclear forensics and LA plasma spectroscopy are focused on the quantification of a major actinides such as U in a mixed/complex sample, 12 methods for improving the emission intensity of species of interest,377,387,383 isotope ratio measurement,49,340,358 use of clustering and machine learning techniques for origin identification of ores and other materials,351,388 and screening/triaging of environmental samples in a post-detonation scenario.389,390 This is not to say that LA-based techniques have no place in nuclear forensics research as the opposite is very true.
Portable and fieldable LIBS instruments have been in development at government-associated laboratories and commercial entities for several years, where proposed use applications span the mining and recycling industries in addition to developing environmental sampling methods at nuclear facilities or following a nuclear detonation or release event. These portable instruments range from cart-portable and backpack-portable instruments to handheld gun-type instruments that offer a point-and-shoot level of complexity to taking a measurement. Shattan et al.
391
investigated a handheld LIBS (HH-LIBS) system (SciAps Z-500-ER hLIBS) to identify particles of UO
A more capable backpack-portable LIBS instrument was developed by LaMontagne et al. 390 for nuclear safeguards applications and environmental sampling as well as deployment following a potential post-detonation nuclear event or release. A primary design objective of the backpack-portable system was the real-time analysis and reporting of results while still in the field during a safeguards inspection. The components of the main system include a small Nd:YAG laser with an output energy of 25 mJ/pulse that is capable of producing one pulse every three seconds, as well as three Ocean Optics HR2000+ spectrometers, a power supply capable of supporting 6 h of continuous operation, signal processing electronics, and a Pocket PC to collect and analyze spectra. The prototype backpack LIBS system was demonstrated in 2008 to analyze several samples, including ore samples with U concentrations ranging from 450 to 7500 ppm.
Laser-ablation-based techniques are continuously making valuable contributions to the broader nuclear forensics research field in areas such as plasma surrogates for nuclear fireball chemistry discussed earlier in this review, as well as similarity of other phenomena between LA plasmas and nuclear fireballs, particularly radio-frequency generation, 392 particle formation, 75 and strong wave generation and interaction with the ambient environment that is partially discussed earlier in this review.54,55 As always, additional research and development will advance the capabilities of these techniques, and it is possible that these advances improve the applicability of these techniques for nuclear forensics analysis and not just for elucidating fundamental phenomena that occur during and following a nuclear detonation.
Machine Learning as an Analysis Approach to LIBS Measurements
Laser spectroscopy methods such as LIBS provide standoff multi-elemental detection capabilities in the field, yet post-processing and analysis of the data are still predominantly performed manually. To enable the robust, automatic real-time analysis of spectral measurements beyond the simple template-matching algorithms commonly implemented with HH-LIBS devices, we suggest consideration of methods in data science and machine learning.
The quantitative analysis of LIBS measurements begins, at the most basic level, with a univariate approach to produce calibration curves relating analyte signal intensities to their elemental concentrations based on a single peak for each element of interest. This technique leverages the direct linear relationship between the concentration of an element in a sample to its integrated peak intensity in the absence of self-absorption and other confounding effects. In practice, univariate calibration analyses work very well with simple, spectrally sparse samples and are known as standard methods for performing quantitative chemical analyses within the LIBS community. However, univariate calibration curves typically fail for complex samples where self-absorption and matrix effects (i.e., interference effects between different elements) are prevalent. For example, Gómez-Nubla et al. 393 demonstrated that univariate calibration curves lack robustness and are characterized by poor linearity for meteorite analogs containing Al, Ba, Ca, Fe, Mg, Na, and Si. They instead favor a multivariate approach based on the PLS method to perform an in situ analysis of the elemental concentrations of the samples. 393 Multivariate techniques are used to address and mitigate the detrimental influence of matrix effects on predictions by including information from multiple emission lines up to the entire visible spectrum. However, it has been demonstrated that better results can be achieved by selecting only a small fraction of the thousands of emission lines making up a typical LIBS spectrum.394–396 Multivariate algorithms related to predicting the analyte concentration in LIBS include principal component regression (PCR) and PLS, where PLS is more widely used.
Before examining PCR and PLS let’s first consider ordinary least squares (OLS) regression, which is the fundamental problem of fitting a curve to a set of data points. This is done by minimizing the least squares error between the predicted target (curve) and the true target (data points). PCR is essentially OLS with principal component analysis (PCA) as a preprocessing step. PCA is an unsupervised learning technique commonly used to reduce the dimensionality of a data set (i.e., throw out uninformative features) and/or to visualize high-dimensional data. This technique removes correlation between features through a linear transformation where the data is projected along directions of maximum variance onto a lower dimensional locally linear manifold. Important assumptions of this technique are that the data can be represented linearly and that classes are separated along directions of maximum variance. PCR is chosen over OLS regression when a data set is characterized by high multicollinearity, which means that two or more variables are highly related to each other in a linear manner. This is frequently the case for LIBS data sets, where intensities for adjacent emission lines are typically highly correlated.395,398 Therefore, applying PCR to LIBS spectra eliminates collinearity from the data set while simultaneously addressing overfitting through the reduction in dimensionality of the data set, where an example demonstrating the transformation of LIBS spectra collected on different samples into cluster-like distributions using two variables (or principal components) is shown in Figure 14. PLS is very closely related to PCR, yet additionally considers variability in the target variable as compared to only the features. Multiple separate PLS models have been developed to perform in situ online monitoring of elemental concentrations in molten salts, and aim to determine either the uranium content in the salt during the reprocessing of spent nuclear fuel297,330 or the amount of fission products (Sr and Mo) in the salt during reactor operations. 328
An example of PCA applied to (a) LIBS spectra of anti-personnel and anti-tank landmine casings, demonstrating the (b) structure of a single principal component (PC) and (c) cluster-like distributions formed over multiple dimensions (i.e., multiple PCs). Adapted from Ref. 397, SPIE Publishing, 2009.
Additional applications of regression algorithms in the nuclear LIBS community involve the development of multivariate spectral fitting algorithms to determine the isotopic composition of uranium. Current models require the manual selection of emission lines and are limited to narrow wavelength windows (<1 nm).266,269,399,400 For example, Chan et al.266,400 recently developed a spectral fitting algorithm based on an iterative, multivariate least-squares regression model with weights to predict the enrichment of UF
Qualitative analyses of spectral measurements, such as elemental identification, are also of high interest concerning defense and forensic applications. As an example, PLS discriminant analysis (PLS-DA) is a favored technique regarding the identification of hazardous materials using LIBS, where PLS-DA is an adaptation of PLS regression methods to address the problem of discrimination.352,396,401–404 Gottfried et al. 401 have demonstrated the standoff detection and discrimination of biological warfare agent surrogates, nerve agent simulants, and interferents using standoff LIBS and PLS-DA. Stearns et al. 403 have shown that PLS-DA can successfully identify 99% of LIBS spectra in a database consisting of vapor-phase chemical warfare agent simulants and rocket fuels in variable low-pressure environments.
Considering the case of binary classification, PLS-DA works to draw a straight line through space to separate the two variables (see visualization of this in Figure 15). In the presence of more than two classes, this discriminator would exist in multidimensional space and be represented by a hyperplane. 405 However, while PLS-DA was developed as a discrimination tool, it is often applied to classification problems in chemometrics in the literature, such as with two of the above six references (Myakalwar et al. 396 and Wang et al. 404 ). Based on the results in the literature, PLS-DA does appear to be capable and effective toward classification, yet these results can be misleading in many cases depending on the characteristics of the data set. Brereton and Lloyd 405 strongly caution against treating PLS-DA as a full classification technique and argue that the widespread availability of software packages has enabled scientists to implement PLS-DA on their data without understanding the underlying mathematics and assumptions of the method. PLS-DA was not developed to be a classification algorithm in its own right and is more so an informal extension on PLS methods. In fact, special cases of PLS-DA are equivalent to the well-known discrimination techniques Euclidean distance to centroids and linear discriminant analysis. 405 Pomerantsev and Rodionova 406 also warn against using PLS-DA as a hard classification tool and instead suggest that the technique may be used as a soft classification tool where the hard categorial rule that a sample must belong only to a single class is relaxed, allowing the sample to be labeled with multiple classes or to be left unclassified.
Discrimination between explosive and non-explosive materials using PLS-DA. Adapted from Ref. 407, The Optical Society, 2008.
Alternatively, PLS-DA may be thought of as a supervised version of PCA, where the target variable is defined as a continuous dummy variable of enumerated class labels. Both PLS-DA and PCA transform data to a lower dimensional space through a linear transformation. PLS-DA does this by preserving maximum covariance between the data and labels, while PCA does this by preserving maximum variance in the original data. 408 In other words, PLS-DA works to minimize intraclass variance while maximizing interclass variance. 352 This is particularly important concerning LIBS due to the inherent shot-to-shot variability between measurements, which may result in the intraclass variance exceeding that of the interclass variance. Barker and Rayens 409 favor the use of PLS-DA over PCA for dimensionality reduction when the objective is discrimination and class separation. Ruiz-Perez et al. 408 have shown that PLS-DA performs well when the signal features are clustered and perform much better at discriminating between clusters than PCA does. This behavior provides insights into the apparent success of Wang et al. 404 despite the misuse of PLS-DA as a classifier, where the data was shown to be clustered based on the provided figures.
To summarize, all machine learning techniques presented here that have been aimed at elemental identification are based on removing collinearity in the data by maximizing the variance between data points. An opposite approach can be taken with a method that searches for components that are statistically independent and uncorrelated, and then separates those independent sources from a mixed signal. This technique is called independent component analysis (ICA), where important assumptions of the method include that the signal sources are independent, that their mixing is linear, and that the independent components are characterized by a non-Gaussian distribution. Forni et al. 410 have reported great success with a combined ICA and clustering algorithm toward the classification of Martian rocks using the NASA ChemCam LIBS database. Their method was demonstrated to be more robust with higher sensitivity than PLS-DA on the same data set. 410 Even so, a disadvantage of ICA concerning its application to spectral data is that the technique predicts negative values in its transformed feature space, where this result was also observed with the PCA example shown in Figure 14. Negative values do not provide a physical interpretation regarding LIBS spectra, which are characterized by strict positivity. To eliminate this shortcoming, the reconstructed signals can be constrained to be non-negative, yielding the technique non-negative matrix factorization (NMF). NMF is an unsupervised machine learning technique that is based on the constrained factorization of a data matrix to provide an alternative representation of that data. 411 This method can also separate superimposed signals assuming that the cumulative signal can be represented by the summation of positive latent components. Different formulations of the NMF algorithm have been presented in the literature, yet this review will focus on the formulation given by Lee and Seung. 411 This formulation uses either additive or multiplicative update rules with the expectation–maximization algorithm to converge to a local factorization. 411 Convergence to a global minimum is not guaranteed.
Application of the NMF algorithm to LIBS data is currently limited. As of 2021, Rammelkamp et al.
412
demonstrated the successful classification of Martian soils and rocks in the NASA ChemCam LIBS database using a combined NMF and
Finally, it is important to provide at least a brief discussion regarding ANNs and their application to the analysis of LIBS data sets. ANNs are becoming increasingly popular within the scientific community; spanning across disciplines including neuroscience, astrophysics, and analytical chemistry; as scientists seek to implement machine learning techniques into the analysis of their data.418–421 However, as many experts cautioned against using PLS-DA without understanding the underlying mathematics and assumptions of the technique, we will make that same argument here concerning neural networks. While neural networks have become very appealing in the last few decades due to their impressive performance on complex data sets, they require very large training data sets that are composed of at least a few thousand samples; 395 Microsoft has developed a database for large-scale face recognition that contains millions of images. 422 This essential data requirement is largely misunderstood by novices who implement neural networks on data sets containing a few hundred samples or less, and then either claim state-of-the-art performance or report heavy overfitting without any attempt at mitigating the issue. For these reasons, we highly encourage spectroscopists and engineers to first consider the use of machine learning-based feature extraction and classification algorithms with their data sets prior to the implementation of deep learning algorithms and neural networks unless size-appropriate data sets are available. Potential feature extraction methods that may be considered in addition to those already discussed include genetic algorithms, support vector machines, ensemble learning, hierarchical learning, and others.
Conclusion
The need to detect and characterize nuclear materials and processes continues to expand and become more restrictive as advanced nuclear reactors are developed that limit the application of traditional detection techniques involving radiation detection or direct sampling involving lengthy and laborious laboratory analyses due to high temperatures and corrosive environments present in many of the proposed designs. Additionally, there is an ever-increasing need for new tools and techniques for wide-area environmental sampling for safeguards inspections, environmental monitoring, and in response to nuclear release or detonation events where triaging of samples and guiding of sampling campaigns will be critical to efficiently and effectively respond to these events.
Recent progress in the use of LA plasmas and spectroscopy techniques (i.e., LAS, LIF, and LIBS) toward nuclear energy and security applications have been thoroughly discussed in this review article. Particular emphasis was placed on recent advancements and activities supporting the development of these techniques in the areas of LA chemistry, laser-produced shockwaves, radioactive plume detection, nuclear security applications, and machine learning. These specific topic areas have value toward advancing the capability of LA plasma spectroscopy techniques to make measurements in the field, at standoff distances, and without sample preparation that are applicable for many nuclear applications.
Many recent successes have been highlighted in this review, such as the demonstration of the 2D-FS technique to reconstruct the isotopic enrichment of a HEU sample, the standoff detection of the isotopes of U using the F
While more work is still needed in the area of LA plasma chemistry, once the high-temperature plume hydrodynamics is well understood, new methodologies may be developed to improve the practicality of these techniques in ambient environments. What is needed to help advance this effort is pump–probe measurements of these oxidizing actinide plasma plumes to quantify the reaction mechanisms and rates that will inform methods to improve the analytical capability of LA plasma spectroscopy techniques. To advance the usefulness of LA plasma spectroscopy to post-detonation debris analysis and as surrogates for nuclear fireball chemistry measurements, high-resolution spectroscopic measurements to identify and label ro-vibrational transitions for PuO (and potentially polyatomic oxides as well) are needed.
Laser-induced breakdown spectroscopy (LIBS) has been demonstrated as being very valuable to the many parts of the NFC, but few studies have gone beyond proof of principle. The ability to consistently monitor processes over periods of time needs to be demonstrated. To further develop the application of LA plasma spectroscopy to the NFC, more advanced data analysis tools and calibration models will be needed as these techniques are bridged from a “complicated” lab study (e.g., quantifying foru to five species) to a system with real dynamic complexity (e.g., dozens of species which may only be present intermittently). Machine learning-based techniques will play a major role in the success of this effort. Calibration transfer methods, such as those leveraging machine learning, will be needed to account for effects such as optical degradation and/or spectrometer drift. Finally, the benefit of hyphenated LA techniques has been demonstrated in the available literature, yet constructing an in situ monitoring tool that uses these more capable and complex systems will be more challenging to deploy in the field under industrial and harsh environmental conditions. Ultimately, a jump from benchtop to engineering-scale systems is needed to see the continued growth of LIBS for real-time monitoring.
To advance the use of LA plasma spectroscopy techniques for application to tracking of radioactive contamination plumes, detailed studies that characterize LIBS matrix effects present in multi-element fallout particles for quantitative analyses are needed. Additionally, once characterized, methods to limit or correct for these matrix effects need to be developed, as it is not possible to correct for the matrix a priori through the use of calibration standards as the particle composition is understandably unknown prior to analysis. Improvement in single-shot conditional analysis to extract weak analytical signals arising from low-concentration aerosol measurements will help with measurements on heterogeneous plumes where ensemble averaging is disadvantageous as particle composition may vary drastically; thus, averaging across many varying particles would be deleterious to extracting accurate compositional information on the individual particles and vis à vis the plume. To enable standoff measurements, an improved understanding of fs pulse interactions with particles and aerosols in the ambient atmosphere for the purpose of emission spectroscopy is needed, based on recent work showing the sampling limitation of fs filaments for atmospheric sensing as well as the disruption induced by these particles to the filament for downrange solid target ablation. Ultimately, it is vital that LA plasma spectroscopy techniques continuously aim to achieve lower LOD measurements, especially for actinides.
In addition to the development of real-time standoff measurement technologies of radiological and nuclear materials is the need to perform real-time analyses of spectroscopic data. Data science presents great potential toward addressing this requirement, where several machine learning methods (e.g., PCA, PLS-DA, ICA, and NMF) have already been successfully demonstrated on LIBS data sets. The demonstrations discussed in this review include the determination of uranium enrichment levels in UF
It is evident that numerous opportunities exist for LA plasmas and spectroscopy to support measurement activities within the nuclear energy and security field. While examples of LA plasmas and spectroscopy measurements in the nuclear industry and community may be lacking, the renewed interest in the technique for this mission space over the past decade has set the stage for promising future advances in scientific understanding and measurement demonstrations at nuclear facilities alike.
Footnotes
Acknowledgments
The authors acknowledge undergraduate researchers Enrique Medici and Thiago Arnaud in the University of Florida Optical Science and Nonproliferation Group who have supported ongoing research activities that partially enabled our focused efforts on this review article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This document is the result of research funded partially by the Department of Defense Science, Mathematics, and Research for Transformation (SMART) Scholarship-for-Service Program; US Department of Energy (DOE) National Nuclear Security Administration Monitoring, Technology, and Verification (MTV) Consortium award number DE-NA0003920 and Consortium for Nuclear Forensics award number DE-NA0004142; Defense Threat Reduction Agency Interaction of Ionizing Radiation with Matter (IIRM) University Research Alliance award number HDTRA1-20-2-0002; and DOE’s Oak Ridge National Laboratory, which is operated by UT-Battelle LLC under contract number DE-AC05-00OR22725. This work was funded by the DOE’s Office of Nuclear Energy (NE), Advanced Reactor Development Program (ARDP), MSR Program. The views and opinions of authors expressed herein do not necessarily state or reflect those of the United States Government or any agency thereof.