Abstract
Colloidal suspensions of EuCl2, EuBr2, and EuSO4 nanoparticles (<50 nm) in dodecane and EuSO4 in 70% H2SO4 were synthesized. Moving single-bubble sonoluminescence (m-SBSL) spectra were obtained for a bubble performing radial oscillations in these suspensions and translational motions at the antinode of a standing ultrasonic wave with a frequency of about 27 kHz. In these spectra (at a spectral resolution of 10 nm), the sono-excited luminescence bands of the Eu2+ ion were detected for the first time, coinciding in the shape and position of the maxima (404, 413, and 377 nm for EuCl2, EuBr2, and EuSO4, respectively) with the bands of Eu2+ located in a crystalline environment in the photoluminescence spectra of nanoparticles of europium salts in suspensions. The detected sonoluminescence of Eu2+ arises due to the injection of nanoparticles into a bubble deformed during motion and excitation of a lanthanide ion at the periphery of the bubble volume during collisions of nanoparticles with charged particles, mainly electrons, coming from a hot nonequilibrium plasma, which periodically arises during bubble compression. Evidence for the excitation of the europium ion in the bubble is the absence of its luminescence bands in the SBSL spectra of the translationally immobile bubble, in which nanoparticles are unlikely to enter. The nanoparticles that enter the bubble also undergo decomposition in the plasma into fragments, in particular, with the formation of Eu, Eu+ in the excited state. The atomic lines of these fragments were recorded for the first time in the m-SBSL spectrum with a resolution of 1 nm for a suspension of EuSO4 nanoparticles in 70% H2SO4. The resulting m-SBSL spectra will add to the library of characteristic spectra of objects of sonoluminescent spectroscopic analysis and will make it possible to identify and determine the content of Eu or Eu2+ in these objects.
This is a visual representation of the abstract.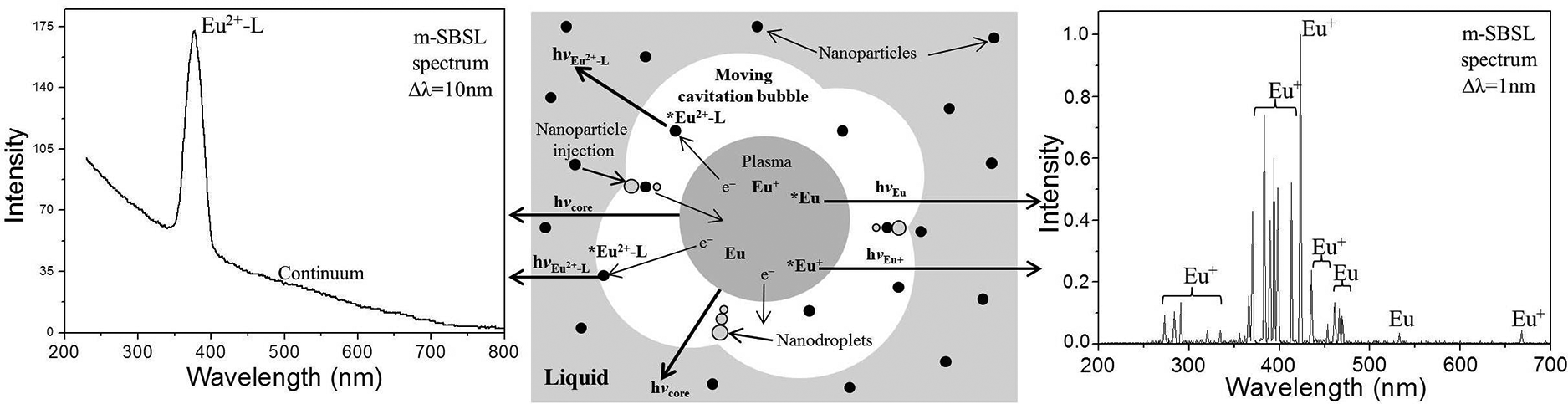
Keywords
Introduction
When considering objects for luminescent analysis, objects containing lanthanide ions (Ln n +) are of particular interest, as their luminescence is widely used in chemistry, biology, and medicine for the diagnosis of various processes.1,2 The luminescence of Ln2+ and Ln3+ ions with characteristic spectra occurs during their transitions to electronically excited states under the action of various sources of energy: photo-, radiation exposure, chemical reactions, electric and ultrasonic fields. Accordingly, in this case, the photoluminescence (PL), radioluminescence (RL), chemiluminescence (CL), electroluminescence (EL), and sonoluminescence (SL) of these ions are observed, each of which can be used for luminescence analysis. Among them, the first three types of luminescence of lanthanide ions are well studied, at least in solutions, and research in this field is summarized in a number of monographs and review papers.3–8 The SL of lanthanide ions belongs to the least studied type. However, Ln3+ SL has been considered repeatedly to date, and the characteristic spectral bands of a number of Ln3+ ions were recorded for multibubble SL,9–11 single-bubble SL (SBSL),10,12,13 and moving SBSL (m-SBSL)13,14 in aqueous solutions, as well as for m-SBSL colloidal suspensions of compounds of these ions. 15 At the same time, for Ln2+ compounds, only sonochemiluminescence of *Eu2+ and *Sm2+ ions were found, which appear during the reduction of Eu3+ and Sm3+, respectively, by the solvated electron sonogenerated in ethylene glycol.16,17 Examples of SL of Ln2+ ions arising by the mechanism of emitter formation as a result of their collisional excitation in the bubble plasma of cavitation bubbles during collisions, for example, with electrons: Ln2+ + e– → *Ln2+ + (e–)ʹ are not known to date.
This paper is devoted to the detection and identification of previously unknown examples of just such m-SBSL for the Eu2+ ion, as well as Eu atomic luminescence arising in colloidal suspensions in dodecane and 70% sulfuric acid of EuCl2, EuBr2, and EuSO4 nanoparticles. Sonoluminescent spectroscopy is a new type of luminescence analysis that has been developed recently.18–20 The recorded SL spectra of these nanoparticles will add to the library of characteristic spectra of objects of sonoluminescent spectroscopic analysis and will allow identifying and determining the content of Eu2+ ions, and Eu as an element, in these objects. It is the focus on revealing the SL of Eu and specifically Eu2+ that distinguishes this work from other works15,20,21 devoted to similar research topics.
Experimental
Materials and Methods
Commercial crystalline salts EuCl2 and EuBr2 were chemically pure grade (99.99%) and purchased from Lanhit Ltd. The EuSO4 was synthesized from EuCl3·6H2O crystalline hydrate (99.99%, Lanhit Ltd.) by reducing Eu3+ → Eu2+ in the Jones reducer according to the known method. 22 Suspensions of europium salt nanoparticles were prepared by adding 0.1 g of large-sized (10–300 μm) crystals of Eu2+ salts to dodecane (Acros Organics, 20 mL) or to bidistilled water (in the case of water-insoluble EuSO4) followed by ultrasonic treatment on a UZDN-2T disperser (ultrasound frequency 22 kHz, power 30 W, immersed titanium waveguide) for 1.5 h. The final fractions of suspensions containing nanoparticles no larger than 50 nm were obtained by filtration through an MFAS-1B membrane filter. The size distribution of nanoparticles was recorded by laser scattering on a Shimadzu Sald-7101 instrument. The viscosity of suspensions and solutions was measured on a rotational modular rheometer Haake Mars III. The resulting suspensions are quite stable, and the size distribution of nanoparticles was maintained for at least two to three weeks (1–2 days for suspension in 70% H2SO4), which is sufficient for all experiments, without fear of dissolution or aggregation of nanoparticles. For registration of SL spectra, the volume of the suspensions was diluted to 100 mL by adding dodecane or concentrated H2SO4 (in the case of EuSO4 suspension in water) followed by evacuation (0.01 Torr, 30 min) for removal of dissolved gases. The SBSL of a stationary bubble and m-SBSL were recorded on an original ultrasonic installation in a spherical glass flask resonator (V = 100 mL) operating in the glowing bubble levitation mode in the central antinode of a standing wave at a frequency of about 27 kHz. The procedure measurement of SBSL and m-SBSL is described in more detail in Sharipov et al. 14 and Gareev et al. 15 The photon emission from the cavitation bubble was delivered to the entrance slit of the monochromator using the quartz fiber (d = 0.2 mm). Its starting point was located 5 mm from the bubble. The temperature of the studied suspensions during the SL recording was maintained at 0 °С by blowing the resonator with liquid nitrogen vapor. The acoustic pressure (pa) in the middle of the resonator flask required to maintain the SBSL (pa = 1.07 bar in dodecane) and m-SBSL (pa = 1.29 bar in dodecane, 2.19 and 2.31 bar in 70% H2SO4) modes was measured using a miniature hydrophone (8103, Brüel & Kjær).
The PL spectra at a spectral resolution Δλ = 2–10 nm were recorded using a Fluorolog-3 Horiba Jobin Yvon spectrofluorimeter with a PMT Hamamatsu R928P detector. The SL spectra with a resolution of 10 or 1 nm were recorded on an Aminco–Bowman J4-8202 spectrofluorimeter or an original spectrofluorimeter based on an MDR-206 scanning monochromator. In both cases, the PMT Hamamatsu R3896 was used as the detector. The sweep rate for recording SL spectra varied (for spectra with different spectral resolutions) from 50 to 200 nm/min. Luminescence stability during registration was controlled by a separate PMT. The spectra are averaged over the registration data of five to eight individual spectra. All PL and SL spectra were corrected for the spectral sensitivity of the recording systems and light absorption by suspensions. The absorption spectra were recorded on a Shimadzu UV-1800 spectrophotometer.
Results and Discussion
Figures 1a–3a show the size distributions of Eu(II) salt nanoparticles for colloidal suspensions in dodecane prepared for the study. The average size of nanoparticles was 20, 25, and 30 nm in suspensions of EuCl2, EuBr2, and EuSO4, respectively. The absorption spectra of these suspensions are shown in Figures 1b–3b (curve 3). All absorption spectra have an edge of a not-very-intense band in the region of 200–300 nm. During photoirradiation at the long-wavelength tail of this absorption band and further at λex from 290 to 365 nm, the PL spectra of EuCl2, EuBr2, and EuSO4 nanoparticles in the corresponding suspensions are recorded (Figure 1b–3b, curve 4). The observed structureless luminescence bands with a half-width of 45–55 nm and maxima at 404 (EuCl2), 413 (EuBr2), and 377 nm (EuSO4) are due to the emission of light by excited Eu2+ ions (electronic transition 4f65d1 → 4f7) located in the solid in the corresponding nanocrystalline environment.
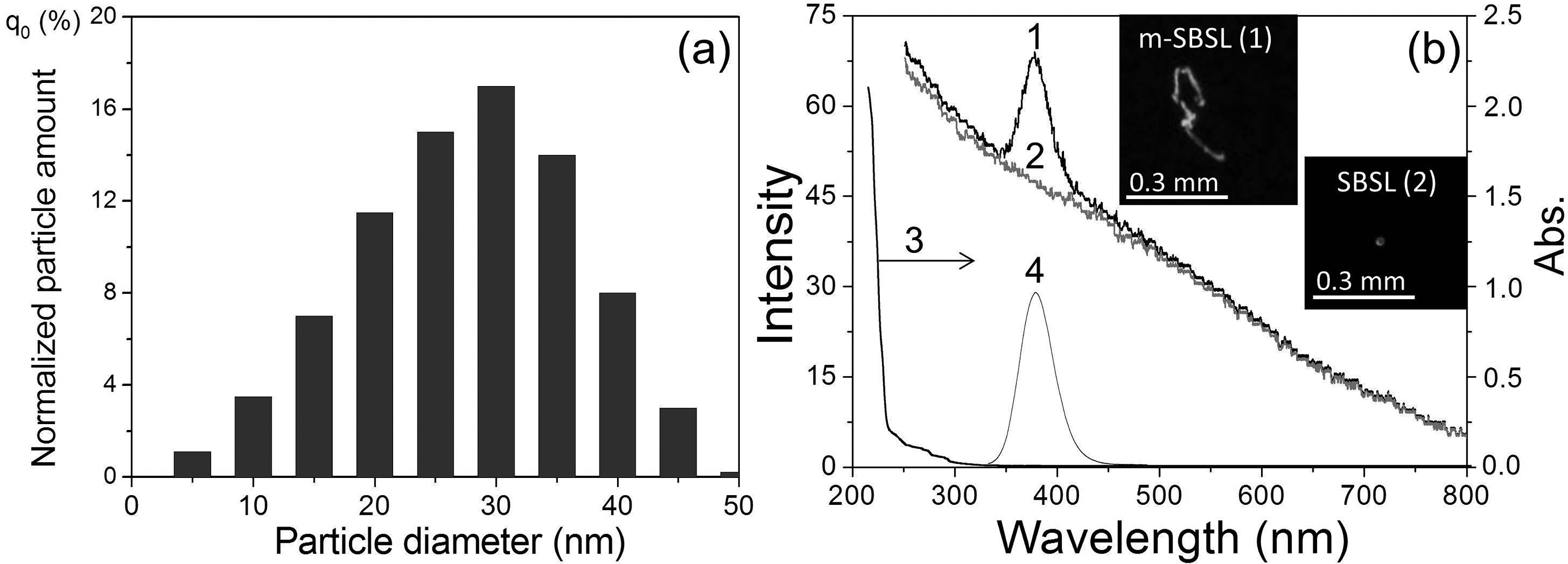
(a) Size distribution and (b) spectra of suspended EuSO4 nanoparticles in dodecane: (1) m-SBSL spectrum, (2) SBSL spectrum (Aminco–Bowman, Δλ = 10 nm), (3) absorption spectrum (ℓ = 1 cm), and (4) PL spectrum (λexc = 290 nm). The insets show photographs of bubbles in the m-SBSL (1) and SBSL (2) modes at an exposure of 0.1 s.
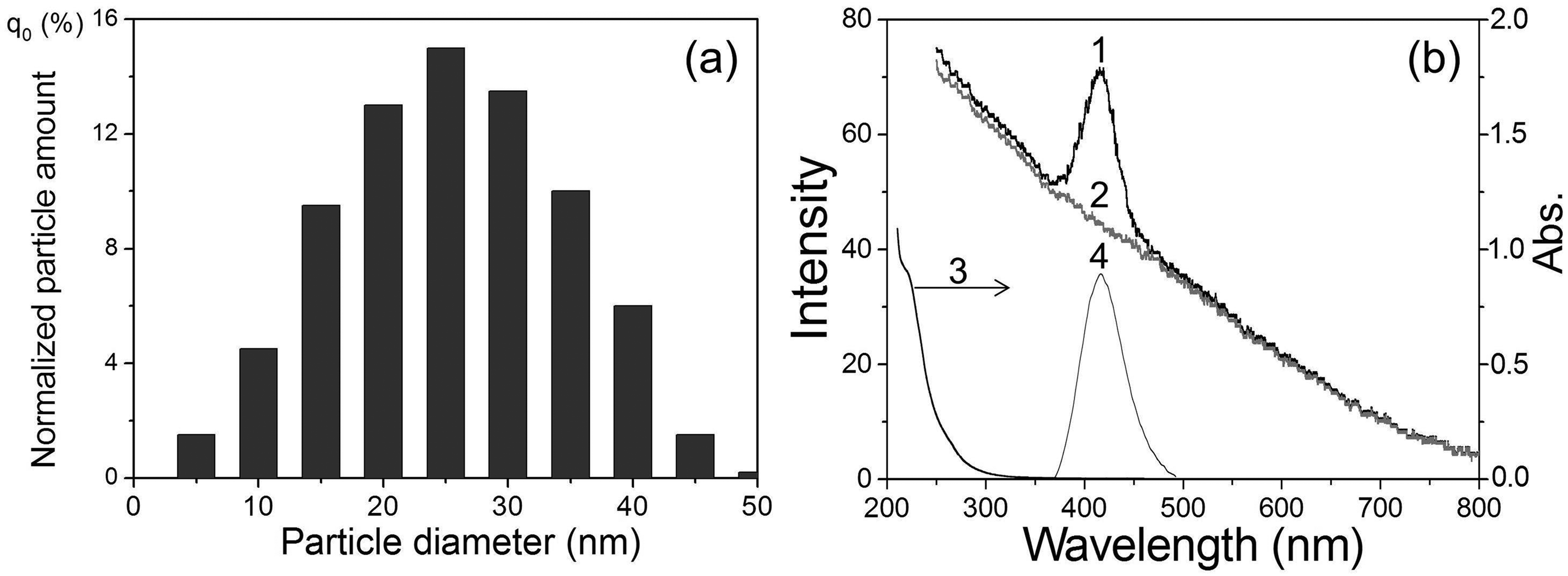
(a) Size distribution and (b) spectra of suspended EuBr2 nanoparticles in dodecane: (1) m-SBSL spectrum, (2) SBSL spectrum (Aminco–Bowman, Δλ = 10 nm), (3) absorption spectrum (ℓ = 1 cm), and (4) PL spectrum (λexc = 350 nm).
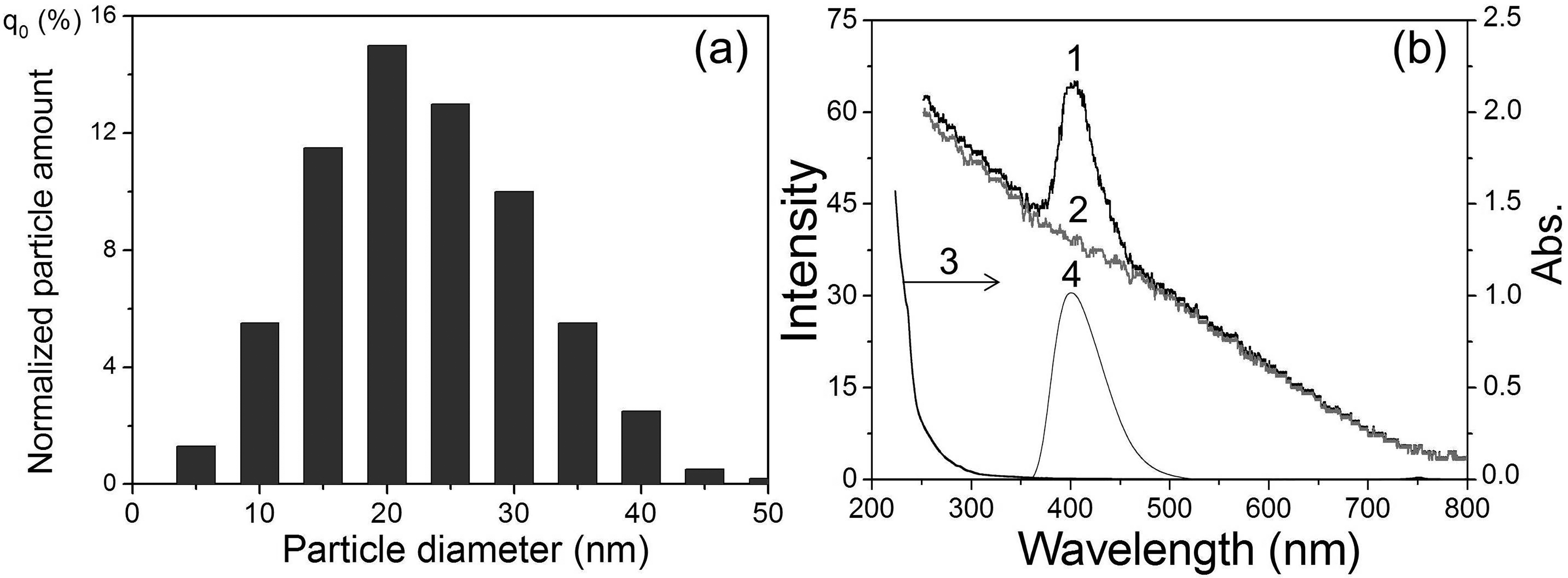
(a) Size distribution and (b) spectra of suspended EuCl2 nanoparticles in dodecane. (1) m-SBSL spectrum, (2) SBSL spectrum (Aminco–Bowman, Δλ = 10 nm), (3) absorption spectrum (
The PL maxima of Eu2+ in suspensions are shifted to shorter wavelengths by 3–12 nm relative to the PL spectra of solid powders: EuCl2 (407 nm), EuBr2 (425 nm), and EuSO4 (384 nm). Apparently, this hypochromic shift is a manifestation of the quantum size effect, i.e., the influence of the size of nanocrystals on their optical parameters, in particular, on the position of the maximum in the PL spectra. 23 In addition, the position of the maxima of the Eu2+ (Figure 1b–3b) luminescence bands also significantly depends on the nature of the anion (L=SO42–, Cl–, Br–) associated with the europium cation. This effect of the spectral shift, previously found in the series EuCl2, EuBr2, and EuI2 in crystals and complexes of Eu2+ with organic and inorganic ligands in a solution of tetrahydrofuran, was explained by an increase in the polarizability of the anion ligands and the degree of covalence of the Eu2+–L bonds as the Eu2+–L bond lengths increased, leading to a decrease in the energy of the excited level of the 4f65d1 electronic configuration of the europium ion. 24 As can be seen from our data, SO42– provides the shortest wavelength luminescence maximum among Eu2+ compounds with the studied ligands. Considering the bidentality of the sulfate ion and shorter bond lengths Eu–O (2.521 and 2.657 Å) 22 compared to Eu–Cl (3.095 Å) or Eu–Br (3.163 Å), the effect of SO42– on the PL spectrum can also be explained in terms of the approach proposed in Galimov et al. 24
Figures 1b–3b show the obtained SBSL (curve 2) and m-SBSL (curve 1) spectra of colloidal suspensions of europium salts in dodecane. These spectra contain a structureless wide band of the emission continuum of a nonequilibrium bubble plasma with a monotonically decreasing intensity from the ultraviolet (UV) to the infrared region of the spectrum. The plasma state of the gas content of the bubble periodically occurs with the frequency of the acting ultrasound at the moments of the greatest compression during the ultrasonic vibrations of the bubble. The formation of a plasma is facilitated by a decrease in the ionization potential in the bubble.25,26 The resulting flashes of light in pure liquids with a spectral continuum similar to that shown in Figures 1–3b (curve 2), were interpreted as thermal radiation of the plasma in the form of a black body, 27 and/or bremsstrahlung of moving plasma particles. 28 The state of the plasma in the region of the greatest bubble compression changes rapidly. It has been shown that for some time the plasma core can be optically opaque and, indeed, emit light as a black body, being in local thermodynamic equilibrium, but, in addition, the plasma also passes through the transparency stage.29–31 In these conditions, especially in solutions of various substances, in addition to the continuum, SL in the form of lines or bands of excited emitters occurring during collisions of various particles in the plasma takes place in the ultrasonic emission spectra. This mechanism of collisional excitation 32 is implemented not only in solutions, but also in colloidal suspensions, 20 where small nanoparticles (<50 nm; nanoparticles >100 nm destroy the bubble and SL disappears) enter a moving bubble with a deformed surface together with the smallest liquid drops (the sonochemical model of nonvolatile substances injection into a bubble in nanodroplets of liquid 33 ). Indeed, this ensures the delivery of the nanoparticles of europium salts into the bubble, and the corresponding Eu2+ luminescence bands are recorded in the m-SBSL spectra against the continuum background (Figures 1–3b, curve 1). It should be noted that the viscosity of the investigated colloidal suspensions of EuSO4 nanoparticles measured by us at 298 K (1.42 mPa·s) differs little from the viscosity of dodecane (1.40 mPa·s). Such an insignificant increase in viscosity does not have a significant effect on the dynamics of bubble motion and the glow generated in it. The shape and intensity of the continuum in dodecane and in the suspension of nanoparticles are practically the same. The continuum does not change significantly when passing from the SBSL regime for a stationary bubble (no nanoparticles enter the bubble, Figures 1–3, curve 2) to the m-SBSL regime (nanoparticles penetrate the bubble, Figures 1–3, curve 1). In addition, as noted above, in the latter case, Eu2+ bands appear against the background of the continuum. As shown in Figures 1b–3b, the positions of the maxima and the shape of these bands coincide with those in the PL spectra of the suspensions. Based on this coincidence, it can be assumed that the reason for the appearance of Eu2+ bands in the m-SBSL spectra is sonophotoluminescence (SPL). The latter is the re-emission by emitters (in our case, nanocrystals of europium salts) located in the bulk of the liquid, the part they absorb, the glow emitted by the bubbles, namely, the re-emission of a part of the continuum. Such SPL actually takes place and has been repeatedly observed for solutions of some luminophores, which have significant light absorption in the UV region of the spectrum and high (close to unity) PL quantum yields.34–36 However, this phenomenon is not recorded in the studied suspensions. If SPL contributes to the m-SBSL spectrum of suspensions of europium(II) nanoparticles, then it should also be recorded in the SBSL spectra (for a translationally motionless bubble), since the SBSL and m-SBSL continuums of colloidal suspensions, as shown by our experiments, have the same shape and intensity, and the conditions for absorption and re-emission of light by a bubble in these two regimes are almost identical. However, it turned out that the Eu2+ bands against the background of the continuum were absent in the SBSL spectra of the studied suspensions, despite the possibility of SPL (Figures 1b–3b, curve 2). Apparently, the existing small contribution of SPL to the luminescence of nanoparticles of Eu(II) salts, which does not exceed the detection threshold under the conditions of our experiments, is due to the following: insignificant absorption of the continuum only in the far UV region (<230 nm) in the presence of suppressing SPL, a noticeable absorption of light in dodecane, a small optical layer for absorbing the re-emitted light of the continuum, a small aperture of its light collection through the end of the fiber (despite the fact that after the act of absorption, the light directed to this end is re-emitted into the 4π sphere), and finally, not the highest quantum yields of the PL of salt nanoparticles europium in suspensions (according to our measurements, ∼0.1 for EuSO4 in sulfuric acid).
Since the absence of the SPL contribution to the SBSL spectra proves the absence of this contribution to the m-SBSL spectra, it is very likely that the complete identity of the Eu(II) bands in the PL spectra of the suspensions and the m-SBSL spectra indicates the luminescence in the bubbles of precisely undecomposed nanoparticles. Obviously, the strong inhomogeneity of the bubble gas content predetermines the possibility of the simultaneous short-term coexistence of both these nanoparticles at the bubble periphery and hot plasma, the fastest particles from which (more likely, these are electrons) excite nanoparticles during collisions. The excitonic excitation arising in a nanocrystal is localized at the Eu(II) luminescent center, followed by the emission of a sonoluminescent photon. In this case, as is typical for other cases of collisional excitation, for example, RL, the luminescence spectrum of these centers coincides with their spectrum upon photoexcitation.2,4,5 The larger cross-section of nanoparticles compared to the cross-section of many individual luminescence centers located in it increases the probability of collisional excitation.
It should be noted that nanoparticles entering the bubble do not remain unchanged in it. They are not only collisionally excited at the periphery of the bubble under the action of the plasma, but also undergo disintegration, with the formation of fragmentary particles: individual molecules, radicals, atoms, ions, and up to electrons. These particles of the plasma also appear in an excited form and emit photons of the characteristic SL. Indeed, this is shown, for example, for SiO2 nanoparticles containing a DyCl3 salt adsorbed from solution. The m-SBSL spectrum of colloidal suspensions of these nanoparticles, along with the continuum and bands of the initial emitter-phosphor Dy3+, contains line spectra of fragments of such decomposition: SiO, Dy+, and Dy. 15 These line spectra are observed when recording with a sufficiently high-resolution Δλ, on the order of 1 nm, at which the continuum and other broadband components of the spectra are subtracted after recording, to leave only the line component of the spectrum. 15 On the contrary, the line component of the spectra, even if it exists, becomes indistinguishable when recording with a low spectral resolution Δλ = 10 nm, at which the spectra in Figures 1–3. In order to search for the line spectra of possible products of the reduction in a bubble of the initial Eu2+ ions to Eu+ and Eu, we recorded the m-SBSL spectra of suspensions of Eu(II) salts in dodecane with a spectral resolution of Δλ = 1 nm. However, the Eu+ and Eu lines were not detected in these spectra.
It should be noted that the atomic lines of most lanthanides are very weak compared to the lines of other metals. 37 For their reliable detection, it is necessary to use more efficient excitation of SL than is the case for suspensions in dodecane. In order to intensify, it was decided to carry out sonolysis of suspensions in a liquid medium, which provides a high degree of bubble compression during acoustic vibrations and the achievement of the highest temperature indicators, providing a high degree of plasma ionization, a high kinetic energy of colliding particles, and a high frequency of their collisions. Sulfuric38,39 and phosphoric 40 acids are among the media with the highest electronic temperatures achieved with m-SBSL, and it is these liquids that can provide the highest sensitivity in the search and registration of low-intensity lines for luminescence analysis. Indeed, the use of a suspension of a 70% H2SO4 solution instead of dodecane as a dispersing medium made it possible to register Eu+ lines (273, 281, 291, 321, 333, 369, 373, 382, 369, 373, 382, 391, 393, 397, 413, 421, 444, and 665 nm) and Eu (459, 463, 466, and 536 nm) identified according to the data of Zaidel et al. 37 and Kramida et al. 41 (Figure 4a).
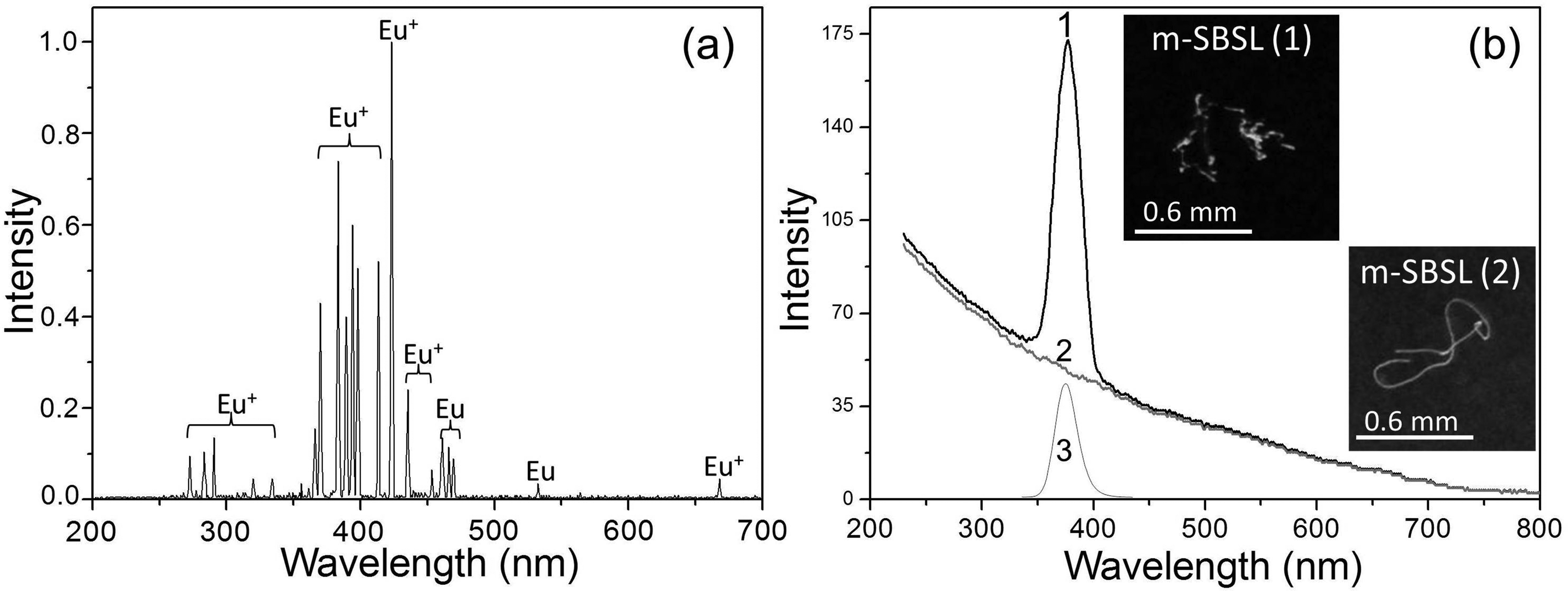
Suspension of EuSO4 in 70% H2SO4: (a) m-SBSL spectrum with a nonsmooth bubble trajectory (pa = 2.31 bar, MDR-206, Δλ = 1 nm, continuum background subtracted); (b) m-SBSL spectrum (pa = 2.31 bar, MDR-206, Δλ = 10 nm) with a nonsmooth bubble trajectory (1), m-SBSL spectrum (pa = 2.19 bar, MDR-206, Δλ = 10 nm) with a smooth bubble trajectory (2), PL spectrum at λexс = 290 nm, Δλ = 10 nm (3). Insets are photographs of bubble trajectories in two m-SBSL modes at pa = 2.31 bar (sub-panel 1) and 2.19 bar (sub-panel 2), with an exposure of 1/30 s.
When comparing the low-resolution spectra (Figures 1b and 4b), not only a strong increase in the absolute intensity of the continuum is noticeable, but also an increase in the intensity of the Eu2+ band relative to the intensity of the continuum, which occurs when dodecane is replaced by sulfuric acid. This also shows an increase in the detection sensitivity of the Eu(II) spectral band with this replacement. Also note the demonstration in Figure 4b spectra for two modes of m-SBSL, i.e., with a smooth bubble trajectory, without sharp changes in the direction of its movement and strong deformations of the spherical surface of the bubble (Figure 4b, curve 2), and with a strongly broken trajectory, which causes such deformations and the entry of nanodroplets and nanoparticles into the bubble (Figure 4b, curve 1). Here we see, as in Figures 1–3b for suspension in dodecane, the presence of only the continuum in the SBSL mode (not realized in sulfuric acid at any pa), and the presence of the Eu2+ band against the background of the continuum in the m-SBSL mode.
Conclusion
For the first time, luminescence bands of the Eu2+ ion were recorded in the m-SBSL spectra of colloidal suspensions of EuCl2, EuBr2, and EuSO4 nanoparticles in dodecane, which are identical to the bands of this ion during the 4f65d1 → 4f7 electronic transition in the PL spectra, with maxima at 404, 413, and 377 nm, respectively. These SL bands are due to the injection of nanoparticles into a bubble that is deformed during motion and the excitation of Eu2+ luminescence centers in them during collisions of nanoparticles at the periphery of the bubble volume with charged particles, mainly electrons coming from a hot nonequilibrium plasma, which periodically arises when the bubble is compressed. The atomic lines of Eu and Eu+ were also first detected as fragmentary products of decomposition in the plasma of nanoparticles trapped in a bubble in the m-SBSL spectrum of a colloidal suspension of EuSO4 nanoparticles in 70% H2SO4. The resulting spectra will add to the library of characteristic spectra of objects of SL spectroscopic analysis and will allow identifying and determining the content of Eu or Eu2+ in these objects, as shown in the examples of determining the concentration of some other metal elements in solutions of their salts. 20
Footnotes
Acknowledgments
This research was performed within the budget theme of the Institute of Petrochemistry and Catalysis UFRC RAS (No. FRMS-2022-0077) on unique equipment at the “Agidel” Collective Usage Center UFRC RAS.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
