Abstract
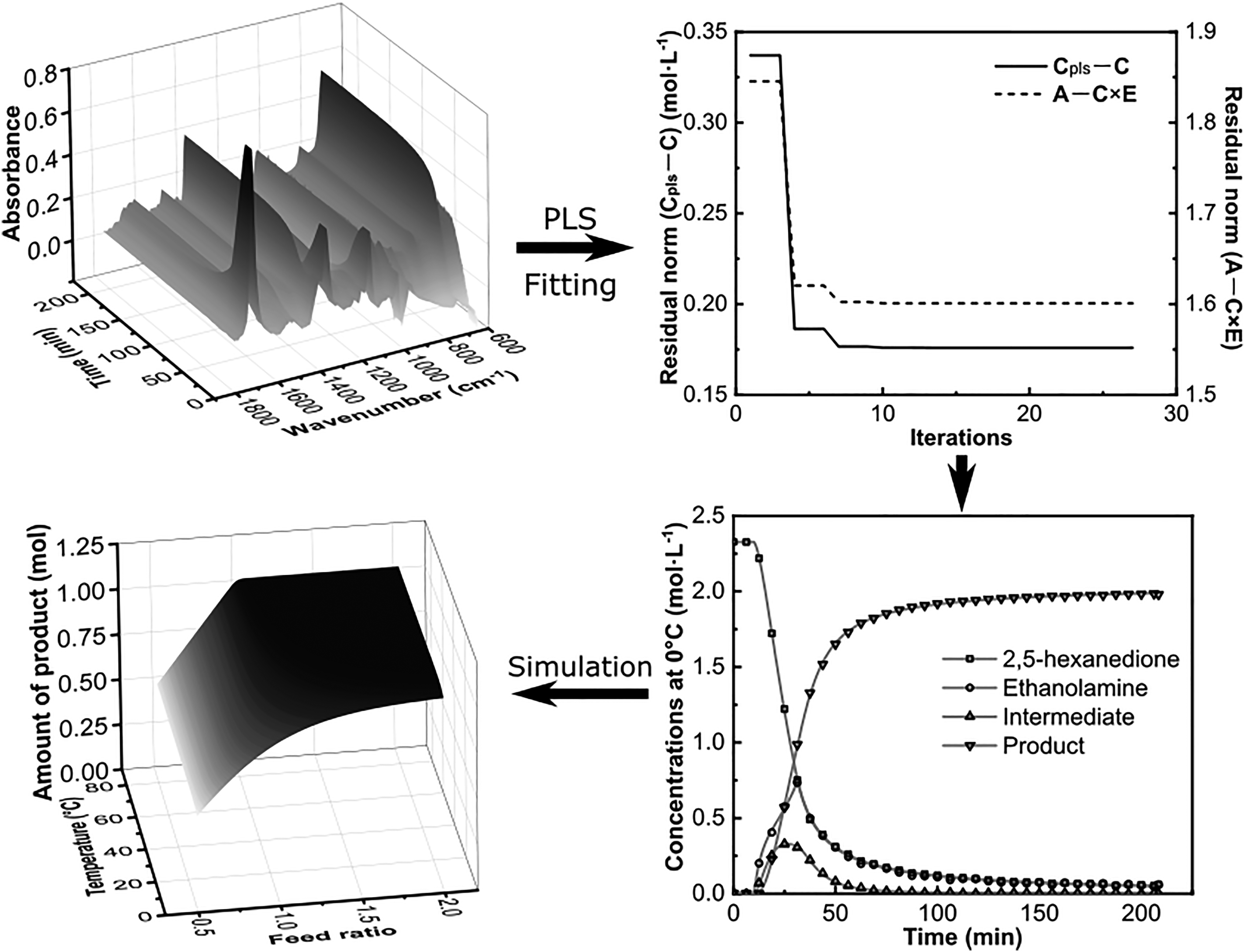
In-situ Fourier transform infrared (FT-IR) spectroscopy has been recognized as an important technology for online monitoring of chemical reactions. However, analysis of the real-time IR data for identification and quantification of uncertain reactants or intermediates is often ambiguous and difficult. Here, we propose an analysis algorithm based on reaction kinetic modeling and the chemometric method of partial least squares (PLS) to comprehensively and quantitatively study reaction processes. Concentration profiles and apparent kinetic parameters can be simultaneously calculated from the spectral data, without the demand of complicated analysis on characteristic absorbance peaks or tedious sampling efforts for multivariate modeling. Paal–Knorr reactions and glyoxylic acid synthesis reactions were selected as typical reactions to validate the algorithm. A lack of fit of the Paal–Knorr reaction spectra was less than 2.5% at various conditions, and the absolute errors between the predicted values and HPLC measurement of glyoxylic acid synthesis were less than 6% during the reaction process. Moreover, the reaction kinetic models extracted from FT-IR data were used to simulate reaction processes and optimize the conditions in order to maximize product yields, which proved that this analysis method could be used for process optimization.
Keywords
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
