Abstract
Objective:
Estrogen has been found to impact cochlear blood flow and auditory function, but epidemiological studies on menopause and hearing are inconsistent. This study investigates the associations of menopause, its age at onset, reproductive lifespan, and hormone replacement therapy (HRT) with hearing loss in adult women in the US.
Methods:
Study cohort includes 1778 adult women (40-69 years), from the National Health and Nutrition Examination Survey 2011 to 2012 and 2015 to 2016 who had complete data on audiometry and reproductive health. Hearing loss was defined based on speech-frequency pure-tone average (0.5, 1, 2, and 4 kHz) in better hearing ear. Menopause status was self-reported. Multivariable regression analyses were performed to explore the associations of menopause, age at onset, reproductive lifespan, and HRT with hearing loss.
Results:
After adjusting for age, demographics, medical comorbidities, and noise exposure, there was no significant association between binary postmenopausal status and hearing loss (β: 0.92 dB, [95% CI: −1.02 to 2.86]). When further considering age of menopause onset and reproductive lifespan, we found that late onset of menopause (vs early onset) was significantly associated with the better hearing (β: −4.60 dB, [95% CI: −8.42 to −0.79]) and longer reproductive lifespan was significantly associated with better hearing (β: −0.16 dB per year, [95% CI: −0.32 to −0.002]). Comparing reproductive lifespan quartiles, the fourth quartile was significantly associated with better hearing relative to the first quartile (β: −4.86 dB, [95% CI: −8.28 to −1.44]). Among post-menopausal women, there was no significant association between hearing loss and HRT (β: 0.45 dB, [95% CI: −1.69 to 2.58]).
Conclusion:
While menopausal status was not significantly associated with hearing loss, later onset of menopause and longer reproductive lifespan were associated with better hearing. Future studies should evaluate the clinical significance of the associations between hearing and reproductive health and potential causal relationships.
Level of evidence:
2b
Introduction
The burden of hearing loss is widespread amongst the American populace, affecting approximately 48 million Americans. 1 Hearing loss has significant negative effects on both health and the economy, posing a considerable burden on healthcare systems. 2 Hearing loss negatively contributes to various health conditions, including poorer quality of life, social isolation, and cognitive decline.3-5 Investigating associated risk factors could lead to preventable measures, reducing these burdens.
Estrogen, a hormone vital for various physiological processes, is primarily produced in the ovaries of premenopausal women and in adipose tissue after menopause. 6 It plays essential roles in vascular health, neural function, and bone maintenance through its interaction with 2 receptors: estrogen receptor alpha (ERα) and estrogen receptor beta (ERβ).6-8 Its protective effects on hearing are attributed to antioxidative, anti-inflammatory, and vasodilatory properties.9-15 Estradiol, a potent form of estrogen, has been shown to affect hearing sensitivity in postmenopausal women, with lower serum estradiol levels significantly associated with reduced bone density and an increased risk of hearing loss.16,17 Furthermore, studies highlight estrogen’s involvement in auditory function, as ERα and ERβ are expressed in inner ear tissues and neurons of the primary auditory cortex.18,19
Menopause is the permanent cessation of menstruation resulting from the loss of ovarian function, either due to natural causes or bilateral oophorectomy. 20 The natural decline in sex hormones associated with menopause and the use of hormone replacement therapy (HRT) on a subgroup of postmenopausal women provide an opportunity to explore the effects of estrogen on auditory function based on the epidemiological data. Currently, the Food and Drug Administration approves the use of HRT for explicit menopausal symptoms which include vasomotor disturbances, bone density loss, and genitourinary issues. 21 However, the relationship between otologic symptoms, menopause, and HRT remains unclear. Previous epidemiological studies on menopause and HRT have yielded mixed results. Some research suggests that menopause accelerates hearing decline,22,23 while other studies indicate that a later onset of menopause may increase the risk of hearing loss. 24
In this study, we examined the associations of menopause, menarche, reproductive lifespan and HRT with hearing loss using objective measures of hearing in a nationally representative cohort of U.S. women.
Methods
Study Population
The study cohort consists of 1778 adult women aged 40 to 69 years from the National Health and Nutrition Examination Survey (NHANES) 2011 to 2012 and 2015 to 2016 cycles, who had complete data on audiometry, reproductive health, and related demographic characteristics. NHANES, conducted by the Centers for Disease Control and Prevention (CDC), is a program designed to assess the health and nutritional status of adults and children in the United States. Each year, it employs a cross-sectional design with a complex, multistage probability sampling method, including the oversampling of underrepresented subgroups, to ensure a representative sample of the civilian, non-institutionalized U.S. population. Survey weights are applied to account for the complex sampling design, allowing analyses across the 2 cohorts to be generalizable to the U.S. population. The age range of the study cohort was chosen based on data availability and prior studies that have examined menopausal status in NHANES. All participants of the NHANES protocols were approved by the National Center for Health Statistics Research Ethics Review Board (Protocol #2011-17) and were provided with written informed consent. Additionally, the study protocol was reviewed and deemed exempt by the University of Southern California Institutional Review Board (UP-20-01447).
Menopause and Hormone Replacement Therapy Variables
Variables related to menopause and HRT were collected from the Reproductive Health Questionnaire Interview. Participants were asked if they had at least 1 menstrual period in the past 12 months and the reason they have not had a period in the last 12 months. Postmenopausal status was defined as those who had no menstrual period in the past 12 months due to natural menopause or had a bilateral oophorectomy (surgical menopause) as in previous literature. 20 Participants were also asked the age of when they had their first and last menstrual cycle and whether or not they have ever used female hormones such as estrogen and progesterone of any form. Menarche was categorized by their first menstrual period at <12 (early) and ≥12 (not early) years. 25 Age at menopause was categorized into early, normal, and late with ages <45, 45-54, ≥55 years respectively. 26 Reproductive lifespan was defined as the difference between ages at menopause and menarche as a continuous variable and as quartiles (<30, 30-35, 36-39, >40).
Audiometric Assessment
Audiometry examinations were performed by mobile examination center (MEC) health technicians that were professionally trained by certified audiologists from the National Institute for Occupational Safety & Health. 27 Air conduction hearing thresholds were determined in both ears of participants in a sound-isolating room in the MEC. Testing was conducted according to a modified Hughson Westlake procedure using the automated testing mode of the audiometer (Model AD 226; Interacoustics). Quality assurance and control were maintained by daily calibrations and background noise checks using a sound level meter. As an additional measure of reliability, thresholds at 1 kHz were measured twice in each ear. Speech-frequency pure-tone averages were calculated for each ear with thresholds at 0.5, 1, 2, and 4 kHz. Hearing loss was defined based on speech-frequency pure-tone average in the better hearing ear as a continuous variable. 28
Other Variables
Demographics, medical history, and noise exposure history provided by interviews were used as covariates in multivariable analysis. Age was limited to the population that received a hearing test, 40 to 69 years. Race/ethnicity was sorted into Hispanic (Mexican-American or other Hispanic), Non-Hispanic white, Non-Hispanic black, Non-Hispanic Asian, and other. Education was categorized into 3 groups: Less than 12th grade, high school graduate, or some college or more. Household income was split into 5 categories (<$20 000, $20 000-$44 999, $45 000-$74 999, ≥$75 000, unknown). Insurance status had 4 categories (none, private insurance, Medicare, Medicaid). Smoking was divided into never, former, or current. Diabetes, strokes, hypertension, cardiovascular diseases (congestive heart failure, coronary heart disease, angina pectoris, or heart attack) were determined by self-reported diagnosis. Three different variables addressed noise exposure with yes or no questions. Occupational noise exposure (“Have you ever had a job where you were exposed to loud noise for five or more hours a week?”), recreational noise exposure (“Outside of a job, have you ever been exposed to very loud noise or music for five or more hours a week?”), and firearm exposure (Ever use firearms for any reason?). Heavy alcohol consumption was defined based on the response to the question: “Was there ever a time or times in your life when you drank 4 (if female) or 5 (if male) or more drinks of any kind of alcoholic beverage almost every day?”
Statistical Analyses
Survey weights were used to account for the analysis of 2 cycles in accordance with NHANES’ complex survey design. 29 Univariable and multivariable linear regression was used to evaluate menopause, age of menopause onset, age of menarche, reproductive lifespan, and HRT usage and their individualistic associations with audiometry-measured hearing as a continuous variable. Multivariate models adjusted for the covariates as aforementioned. The association analysis between hearing loss and reproductive variables, age of menopause onset, reproductive lifespan, and HRT usage was conducted on a subgroup consisting of only postmenopausal women (n = 1145). An additional supplemental model was conducted including heavy alcohol use as an additional covariate, limited to participants with complete data on alcohol use. Analysis involving menopause status was performed in a subgroup (n = 1366). Analysis involving HRT use and age of menopause onset was conducted in a further subgroup of postmenopausal women (n = 875). Statistical significance was set at P < .05, two-tailed. All statistical analyses were conducted using The R Project for Statistical Computing software version 4.4.1.
Results
Table 1 summarizes the characteristics of the study cohort. Among U.S. women aged 40 to 69 years, 65.9% (95% CI: 62.7-69.1) reported being postmenopausal, and 42.5% (95% CI: 38.1-46.7) of these postmenopausal women reported using HRT.
Characteristics of the Study Cohort, NHANES 2011 to 2012 and 2015 to 2016 (n = 1778).
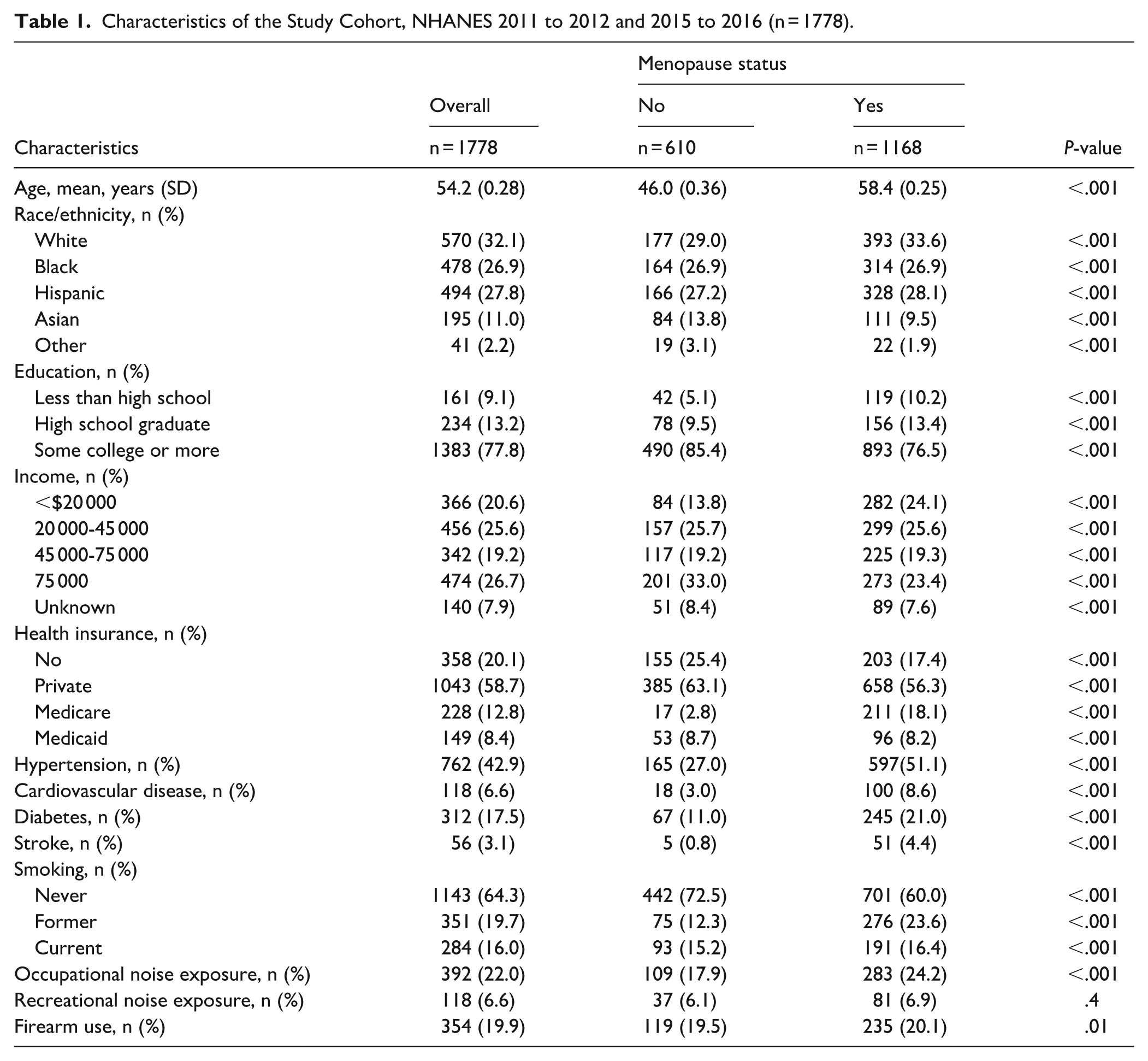
Table 2 summarizes the association of menopause status and menarche with audiometry-measured hearing. Sequential models incorporate additional adjustments for demographics, medical comorbidities, and noise exposure history. In a multivariable model adjusting for demographics, medical comorbidities, and noise exposure history, there was no significant association between postmenopausal status and hearing loss (β: 0.92 dB, [95% CI: −1.02 to 2.86]). Comparing early versus later menarche, no significant associations were found between menarche and hearing loss (β: 0.22 dB, [95% CI: −1.38 to 1.82]).
Linear Regression Analyses Examining the Association of Menopause Status and Menarche with Audiometry-Measured Hearing (Change in Speech-Frequency Pure-Tone Average (PTA)).
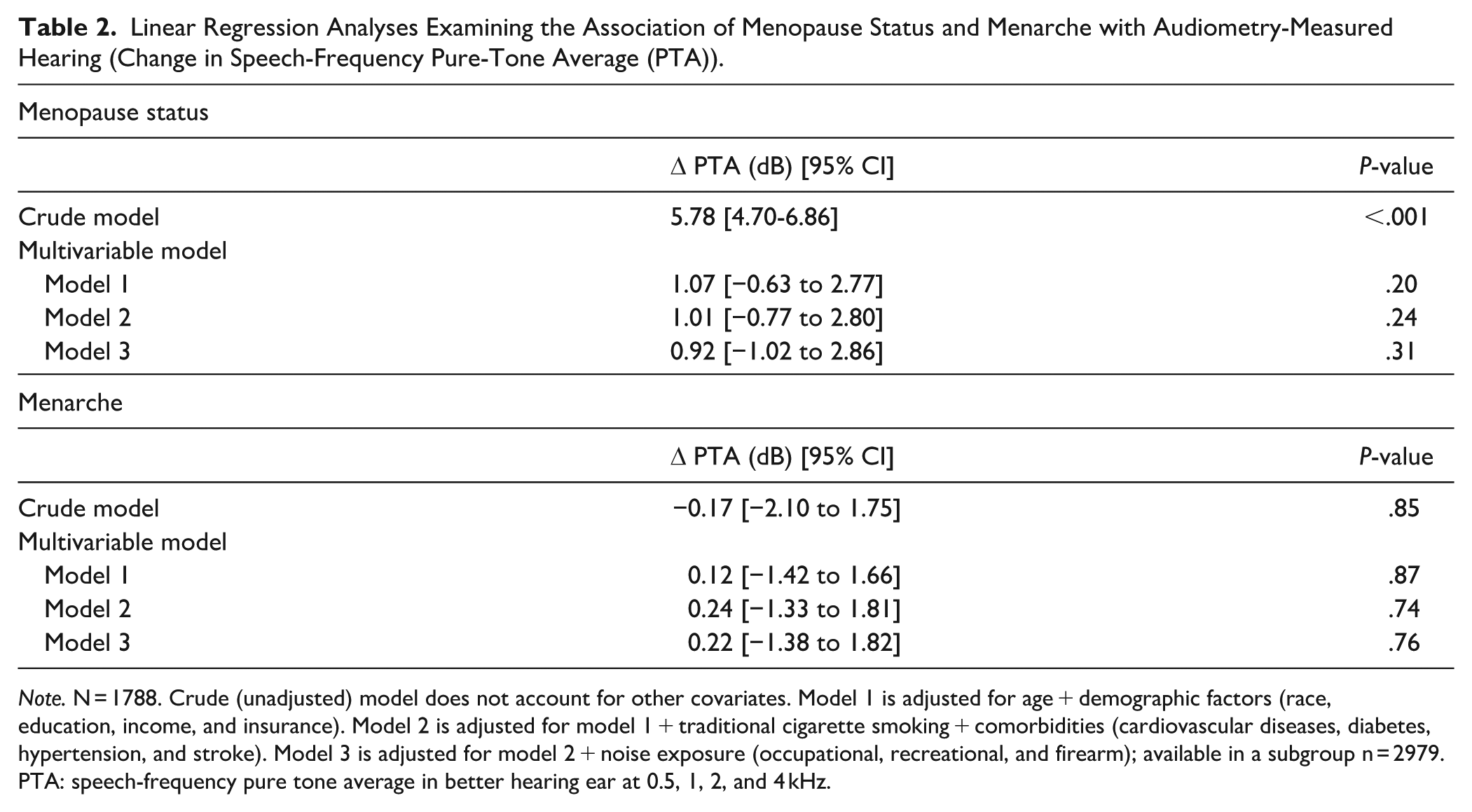
Note. N = 1788. Crude (unadjusted) model does not account for other covariates. Model 1 is adjusted for age + demographic factors (race, education, income, and insurance). Model 2 is adjusted for model 1 + traditional cigarette smoking + comorbidities (cardiovascular diseases, diabetes, hypertension, and stroke). Model 3 is adjusted for model 2 + noise exposure (occupational, recreational, and firearm); available in a subgroup n = 2979. PTA: speech-frequency pure tone average in better hearing ear at 0.5, 1, 2, and 4 kHz.
Table 3 summarizes the associations of age of menopause onset, reproductive lifespan, and HRT with audiometry-measured hearing in postmenopausal women. Similarly, sequential models incorporate additional adjustments for demographics, medical comorbidities, and noise exposure history. In the multivariate model adjusting for all the covariates, older age at menopause onset was significantly associated with better hearing (β: −0.16 dB per year, [95% CI: −0.30 to −0.006]). When compared to early age of menopause onset, individuals with normal and late onset of menopause had significantly better hearing (β: −3.21 dB, [95% CI: −5.39 to −1.03], and β: −4.60 dB, [95% CI: −8.42 to −0.79] respectively). Longer reproductive lifespan was significantly associated with better hearing (β: −0.16 dB per year, [95% CI: −0.32 to −0.002]. Individuals in the highest quartile had significantly better hearing than those in the lowest quartile (β: −4.86 dB, [95% CI: −8.28 to −1.44]). No significant association was found between HRT use and hearing loss (β: 0.45 dB, [95% CI: −1.69 to 2.58]). An additional supplemental model including heavy alcohol use as an additional covariate among those with complete data on alcohol use (n = 1366) demonstrated similar results (Supplemental Table 1).
Linear Regression Analyses Examining the Association of Age of Menopause Onset, Reproductive Lifespan, and Hormone Replacement Therapy (HRT) with audiometry-Measured Hearing (Change in Speech-Frequency Pure-Tone Average (PTA)) in Postmenopausal Women.
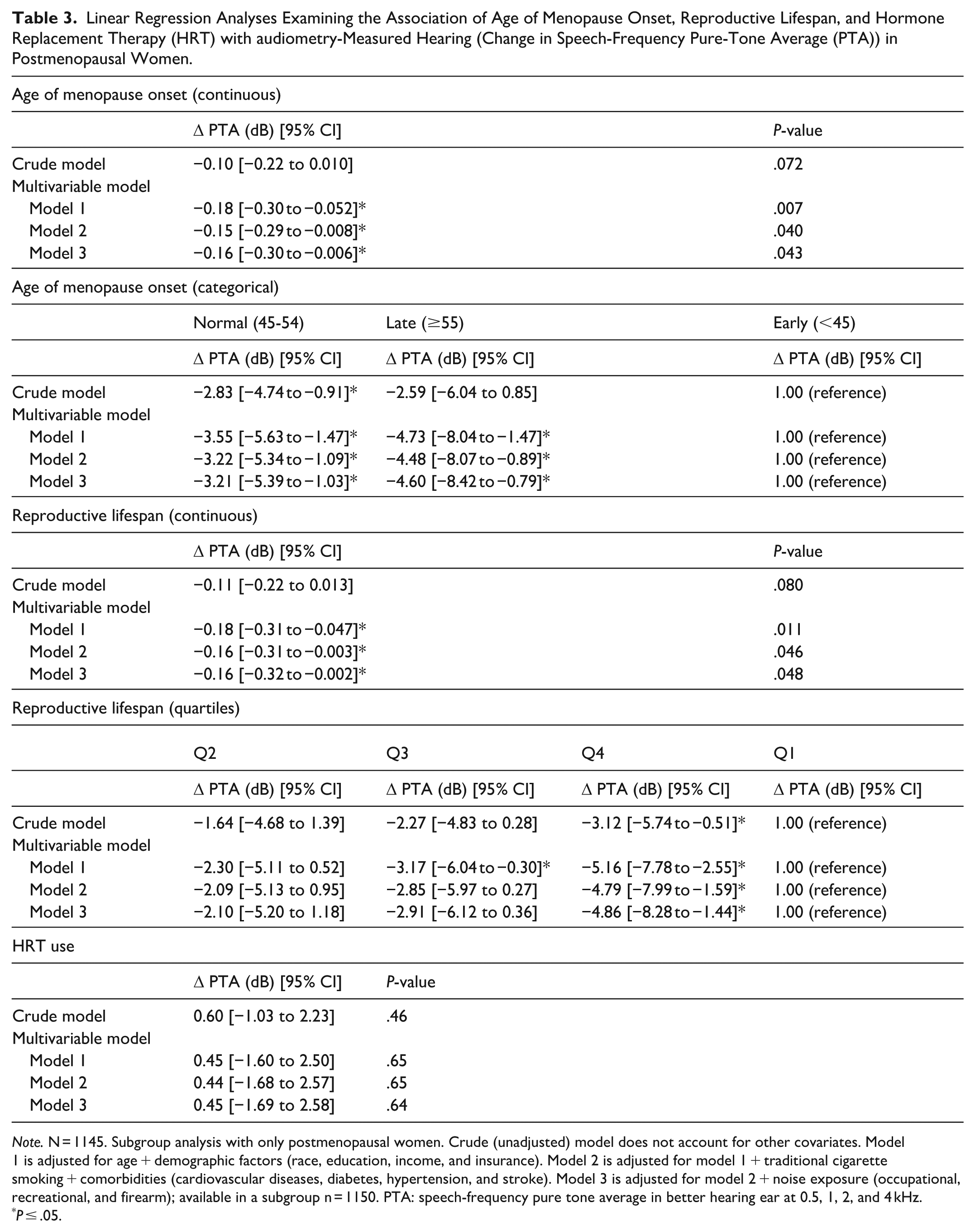
Note. N = 1145. Subgroup analysis with only postmenopausal women. Crude (unadjusted) model does not account for other covariates. Model 1 is adjusted for age + demographic factors (race, education, income, and insurance). Model 2 is adjusted for model 1 + traditional cigarette smoking + comorbidities (cardiovascular diseases, diabetes, hypertension, and stroke). Model 3 is adjusted for model 2 + noise exposure (occupational, recreational, and firearm); available in a subgroup n = 1150. PTA: speech-frequency pure tone average in better hearing ear at 0.5, 1, 2, and 4 kHz.
P ≤ .05.
Discussion
In this nationally representative sample of U.S. women, there was no significant association between menopausal status and hearing loss. Yet, amongst postmenopausal women, later onset of menopause and longer reproductive lifespans were significantly associated with better hearing. Use of HRT was not independently associated with hearing among postmenopausal women.
A few prior studies investigating the relationship between menopause and hearing loss reported mixed findings.22-24,30,31 For example, several population-based studies have suggested that postmenopausal status may accelerate hearing decline, aligning with our findings.22,23,30 In contrast, a large cross-sectional study reported that postmenopausal status was not an independent risk factor for self-reported hearing loss, though a later onset of menopause was associated with an increased risk of self-reported hearing loss. 24 Similarly, a smaller cross-sectional study conducted in Turkey found no significant differences in hearing thresholds based on menopausal status. 31 Our study, which utilized a large sample size and audiometry-measured hearing thresholds, demonstrated that menopausal status itself was not significantly associated with hearing loss.
Upon further consideration of the onset of menopause and reproductive lifespans among postmenopausal women, our study found that a later onset of menopause and longer reproductive lifespans were significantly associated with better hearing. These results suggest that the duration of physiological estrogen exposure may have a greater impact on hearing than the occurrence of menopause itself. This finding is consistent with a cross-sectional population-based study from Korea, which reported that longer reproductive lifespan and older age of menopause were associated with better hearing thresholds at speech frequencies (0.5, 1, 2, and 4 kHz). 32 However, given the relatively small effect sizes of these associations, the clinical significance of the findings remains uncertain.
Estrogen has been well established to play an integral role in auditory function. Cross-sectional correlation analyses between serum estradiol levels and hearing loss revealed that lower estradiol levels corresponded with declining hearing sensitivities, indicating that higher estradiol levels may help protect against hearing loss.16,17 Evidence shows that estrogen plays an active role in auditory function through its effects on the primary auditory cortex and inner ear structures via estrogen receptors ERα and ERβ.18,19 Experimental studies using ERβ−/− mice further support this protective role; these mice develop deafness by 12 months, whereas wild-type mice exhibit no hearing deficits. 33 Similarly, a study using a rat model to examine the effects of ovariectomy on auditory brainstem responses (ABR) found that estrogen deficiency due to the loss of ovaries impaired auditory processing at the brainstem level. However, estrogen replacement therapy following ovariectomy reversed some of these ABR deficits, underscoring estrogen’s critical role in maintaining auditory function. 34
The influence of sex hormones on auditory function is complex, and the exact mechanisms remain unclear. However, several effects have been proposed. Estrogen exhibits protective properties, particularly in the bone, cardiovascular system, and central nervous system, by mitigating oxidative stress.9,35-37 It has also been shown to counteract the ototoxic effects of aminoglycosides by inhibiting the JNK pro-apoptotic pathway in hair cells. 38 Additionally, estrogen demonstrates anti-inflammatory effects by inhibiting NLRP3 inflammasomes, thereby reducing neuroinflammation.10,11 Emerging research indicates that estrogen improves auditory function by increasing blood flow to the stria vascularis via upregulation of vascular endothelial growth factor through the PI3K/Akt pathway. 15 The impact of menopause on bone integrity has also been suggested as a potential mechanism influencing hearing health. Menopause is a major risk factor for osteoporosis due to estrogen’s direct role in regulating osteoclast and osteoblast activity. 39 A large-scale cross-sectional study of U.S. female nurses found that women with osteoporosis had a higher likelihood of experiencing hearing loss. 40 These findings suggest that bone density loss may underlie the connection between declining estrogen levels and hearing impairment. 41
In this study, there was no significant association between the use of HRT and hearing thresholds. While some studies also found no association between HRT use and hearing outcomes,42-44 others reported contrasting findings, with HRT use being linked to both improved hearing44-47 and worse hearing.24,48 For example, a large nationwide population study in Taiwan showed that the risk of developing sensorineural hearing loss after a 10 year time span did not differ across postmenopausal women that were on HRT compared to those that were not on HRT. 42 Alternatively, a study examining a cohort of 83 women of the same age found that postmenopausal women treated with tibolone had a significant lower hearing threshold in the right ear at 4000 to 6000 Hz relative to postmenopausal women that were not on any form of HRT. 44 Hederstierna et al 45 similarly implicates the potential protective role HRT has on hearing in postmenopausal women; postmenopausal women without HRT demonstrated worse hearing in selective frequencies, 2000, 3000, and 8000 Hz, relative to postmenopausal women on HRT and premenopausal women. Conversely, a large population study following over 80 000 women in the United States reported an association opposing HRT’s potential benefit on hearing. Among these postmenopausal women, longer duration of HRT use was associated with self-reported hearing loss. 24 The variability in these studies may have been influenced by factors such as the type of HRT administered, its dosage, onset, and the treatment regimen.49,50
This study has limitations. This is a cross-sectional study, and the temporal and causal relationships are unable to be established. Menopausal status and HRT use were determined based on self-reported data, which lacks objective measurements and may be prone to misreporting and recall bias. Furthermore, the data on HRT did not include information on the type, dosage, or duration, limiting the ability to comprehensively assess its effectiveness. The audiometry data were based on pure-tone air-conduction testing, which did not allow for the determination of the type of hearing loss. Additionally, residual confounders or potential mediators (eg, genetic predisposition, nutritional status, depression, etc) that were not available in this dataset could not be fully accounted for. Lastly, although our findings regarding hearing loss were statistically significant, their clinical relevance may be limited due to the small effect size. Future research should aim to evaluate the complex relationship between reproductive health and hearing health, as well as investigate the potential role of therapeutic options, such as HRT, and the underlying mechanisms involved.
Conclusion
In a nationally representative sample of U.S. women, menopausal status and HRT use were not significantly associated with hearing loss. However, later menopause and longer reproductive lifespan were significantly associated with better hearing among post-menopausal women. These results indicate a possible relationship between reproductive health and auditory function that requires further study to clarify clinical relevance and causal mechanism.
Supplemental Material
sj-docx-1-aor-10.1177_00034894261435259 – Supplemental material for Association of Menopause and Hormone Replacement Therapy with Hearing Loss
Supplemental material, sj-docx-1-aor-10.1177_00034894261435259 for Association of Menopause and Hormone Replacement Therapy with Hearing Loss by Jonathan Z. Hsu, Kaiqi Gu, Breana Nguyen and Janet S. Choi in Annals of Otology, Rhinology & Laryngology
Footnotes
Author Contributions
Jonathan Z. Hsu: Study conception, data collection, data analysis, and manuscript writing. Kaiqi Gu: Data collection, Data analysis, and manuscript revision. Breana Nguyen: Data collection and manuscript writing. Janet S. Choi: Study conception, data analysis, data interpretation, and critical revision of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
