Abstract
Objective:
This study aimed to determine the prevalence, subtypes, and control status of chronic rhinosinusitis (CRS) among patients with severe asthma receiving biologic therapy in a tertiary allergy clinic.
Methods:
A single-center, cross-sectional observational study was conducted in adult patients with severe asthma receiving biologics for at least 6 months. CRS diagnosis and control were assessed based on European Position Paper on Rhinosinusitis and Nasal Polyps (EPOS) 2020 criteria. The Sinonasal Outcome Test-22 (SNOT-22) was also administered. Statistical analyses were performed using appropriate univariate and multivariate methods.
Results:
Among 148 patients with severe asthma receiving biologic therapy, 97 (65.5%) were diagnosed with comorbid CRS. Of these, 68 (45.9%) had CRS with nasal polyps (CRSwNP) and 29 (19.5%) had CRS without nasal polyps (CRSsNP). A total of 54 patients (55.6%) were treated with omalizumab and 43 (44.3%) with mepolizumab. Overall, 32.9% of patients had controlled CRS, 36.0% were partly controlled, and 30.9% were uncontrolled. In multivariable ordinal regression analysis, the presence of N-ERD and higher BMI were associated with poorer CRS control, whereas early-onset asthma and prior maintenance systemic corticosteroid use were associated with better control, suggesting that these factors may be related to CRS control status in this population.
Conclusions:
CRS is highly prevalent among patients with severe asthma receiving biologics, and approximately one-third remain uncontrolled despite treatment. These findings emphasize the importance of regular upper airway assessment and may inform future strategies for optimizing biologic selection and treatment outcomes in this population.
Introduction
Chronic rhinosinusitis is a prevalent upper respiratory tract condition characterized by persistent inflammation of the sinonasal cavities. 1 It is commonly classified into 2 subtypes as CRSwNP and CRSsNP. Patients with CRSwNP predominantly exhibit a type 2 inflammation pattern, marked by eosinophilia and elevated levels of immunoglobulin E (IgE), interleukin-4 (IL-4), IL-5, and IL-13. 2 Stratifying CRS based on endotype can facilitate personalized treatment approaches by targeting the specific pathophysiological mechanisms underlying the condition in each patient.1,3
The incidence of CRS is estimated to be between 6% and 15% in the general population. 4 Asthma is commonly associated with CRS, particularly in patients with CRSwNP, with prevalence rates ranging from 30% to 70%. The frequency of CRSwNP in asthma correlates with disease severity; while CRSwNP occurs in 10% to 30% of individuals with mild asthma, this rate increases to 70% to 90% in those with severe asthma.5,6 Recent advancements in biologics for severe asthma have also highlighted their potential in treating CRSwNP, supported by high-level evidence from recent studies. 5 However, accurately defining disease control and severity in CRS is crucial for both selecting appropriate treatments and for effective follow-up. The 2020 EPOS update redefined CRS control by incorporating the visual analog scale (VAS) to assess symptom severity in research settings and encouraged further investigation on this topic. 1
At our tertiary allergy clinic, many patients with severe asthma receiving biologics also have comorbid CRS, consistent with prior reports. This cross-sectional study aims to determine the prevalence and subtypes of CRS among patients with severe asthma receiving biologics at our center, to evaluate the level of CRS control, and to identify factors associated with CRS control status.
Materials and Methods
Results
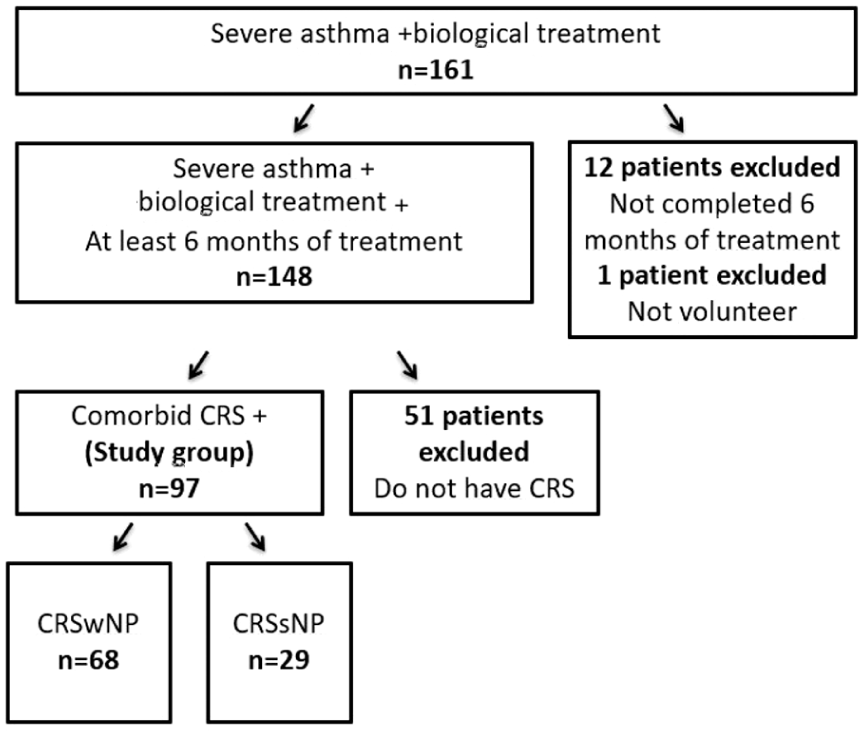
Flow chart of the study.
Fifty-four patients (55.6%) were on omalizumab, and 43 patients (44.3%) were on mepolizumab treatment. The mean ages of the patients in these groups were 52.9 ± 12.5 years and 43.5 ± 12.5 years, respectively (P = .001). The median follow-up period for biologics at the time of evaluation for all patients was 24 months (range: 6-131 months). This period was longer in patients receiving omalizumab (51 months [range: 6-131] vs 14 months [range: 6-36], [P < .001]). Atopy (P = .02) and perennial allergen sensitization (P = .001) were more common in the omalizumab group, while absolute eosinophil count before biologics was higher in mepolizumab group (P = .03). Patients’ BMI was significantly higher in the omalizumab group compared to the mepolizumab group (median BMI: 27.8 vs 25.7, respectively; P = .01). Demographic and clinical characteristics of the study population are presented in Table 1.
Demographic and Clinical Characteristics of the Study Population.
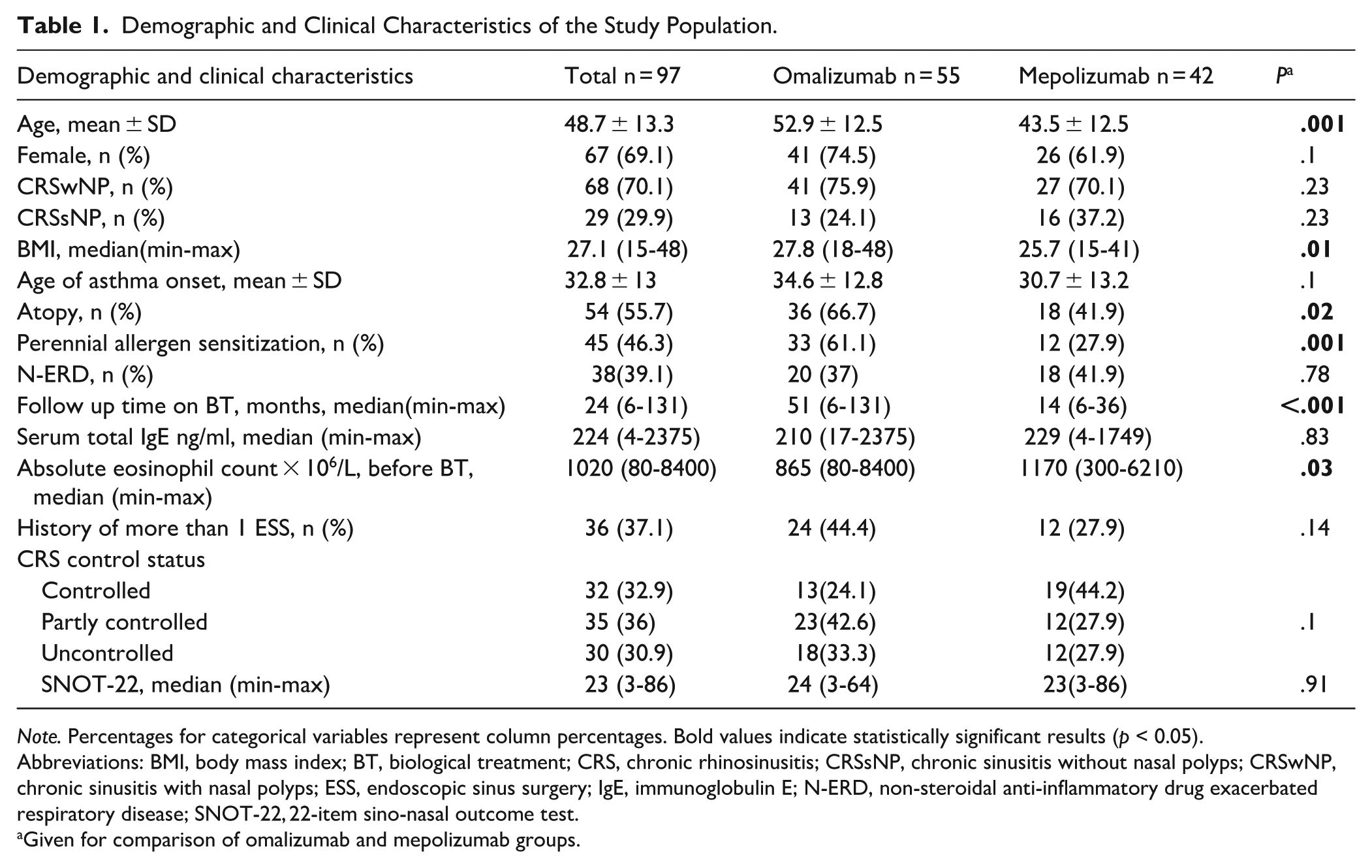
Note. Percentages for categorical variables represent column percentages. Bold values indicate statistically significant results (p < 0.05).
Abbreviations: BMI, body mass index; BT, biological treatment; CRS, chronic rhinosinusitis; CRSsNP, chronic sinusitis without nasal polyps; CRSwNP, chronic sinusitis with nasal polyps; ESS, endoscopic sinus surgery; IgE, immunoglobulin E; N-ERD, non-steroidal anti-inflammatory drug exacerbated respiratory disease; SNOT-22, 22-item sino-nasal outcome test.
Given for comparison of omalizumab and mepolizumab groups.
Relationship of Different Variables with EPOS CRS Control Status.
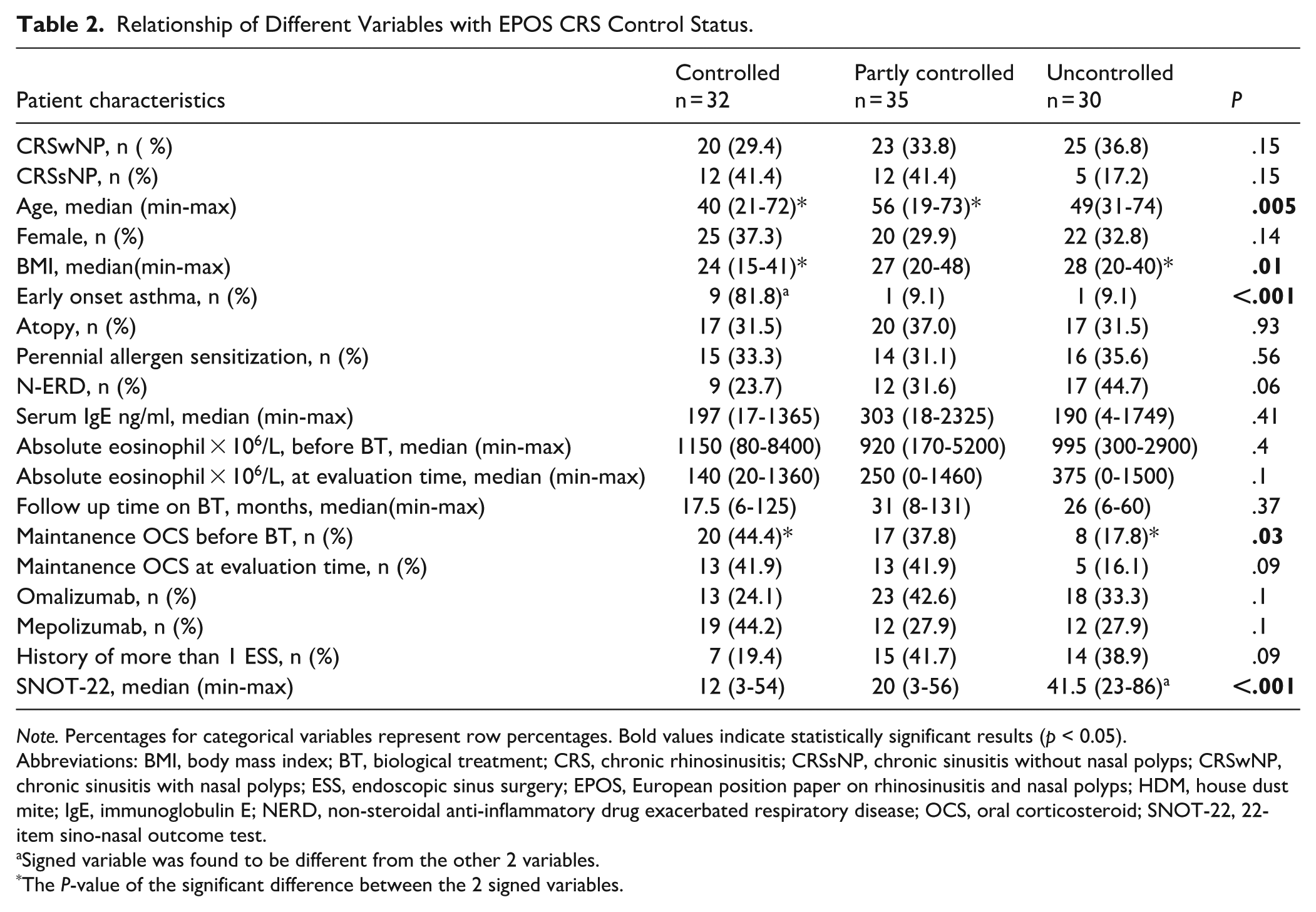
Note. Percentages for categorical variables represent row percentages. Bold values indicate statistically significant results (p < 0.05).
Abbreviations: BMI, body mass index; BT, biological treatment; CRS, chronic rhinosinusitis; CRSsNP, chronic sinusitis without nasal polyps; CRSwNP, chronic sinusitis with nasal polyps; ESS, endoscopic sinus surgery; EPOS, European position paper on rhinosinusitis and nasal polyps; HDM, house dust mite; IgE, immunoglobulin E; NERD, non-steroidal anti-inflammatory drug exacerbated respiratory disease; OCS, oral corticosteroid; SNOT-22, 22-item sino-nasal outcome test.
Signed variable was found to be different from the other 2 variables.
The P-value of the significant difference between the 2 signed variables.
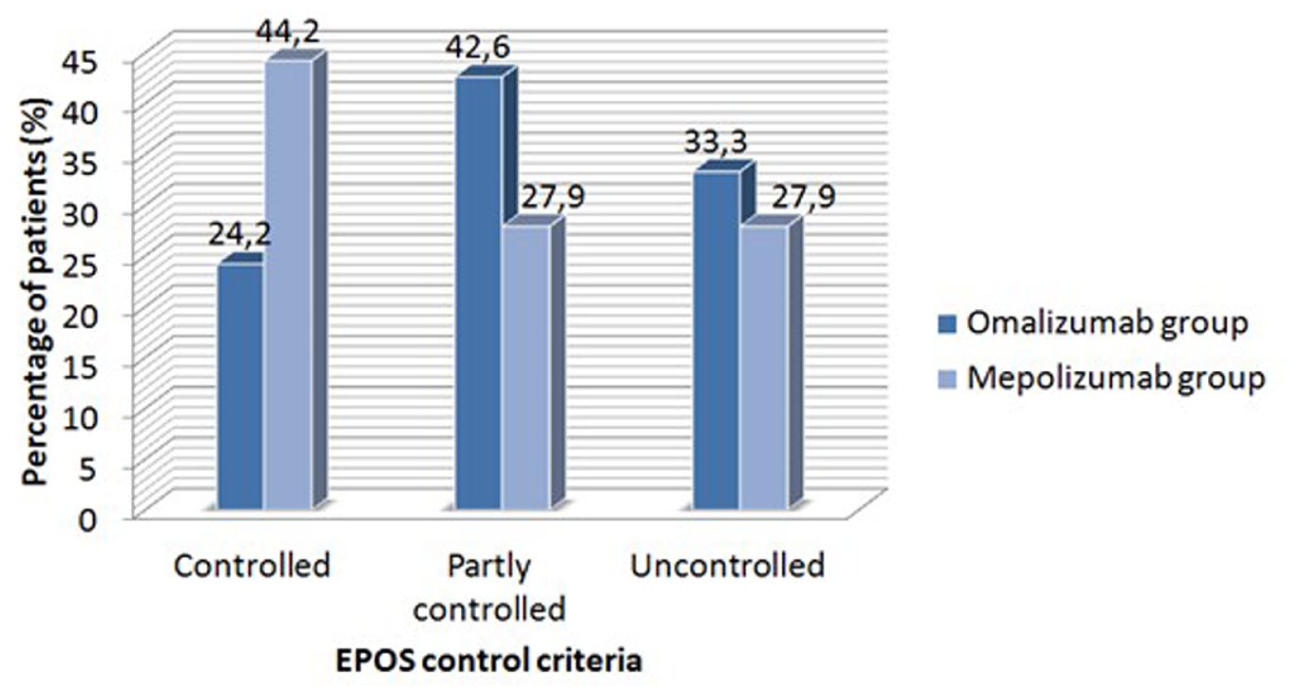
Chronic rhinosinusitis control status of patients on omalizumab and mepolizumab treatments.
In multivariable ordinal logistic regression, the presence of N-ERD (OR = 2.27, 95% CI: 1.02-5.26, P = .046) and higher body mass index (BMI; OR = 1.08 per unit increase, 95% CI: 1.01-1.16, P = .037) were independently associated with poorer CRS control, whereas early-onset asthma (OR = 0.09, 95% CI: 0.02-0.51, P = .006) and prior maintenance systemic corticosteroid use (OR = 0.41, 95% CI: 0.19-0.92, P = .029) were independently associated with better CRS control. Age and sex were not independently associated with CRS control. These results are summarized in Table 3 and illustrated in Figure 3.
Multivariable Ordinal Logistic Regression Analysis of Factors Associated with CRS Control in Severe Asthma Patients Receiving Biologic Therapy.
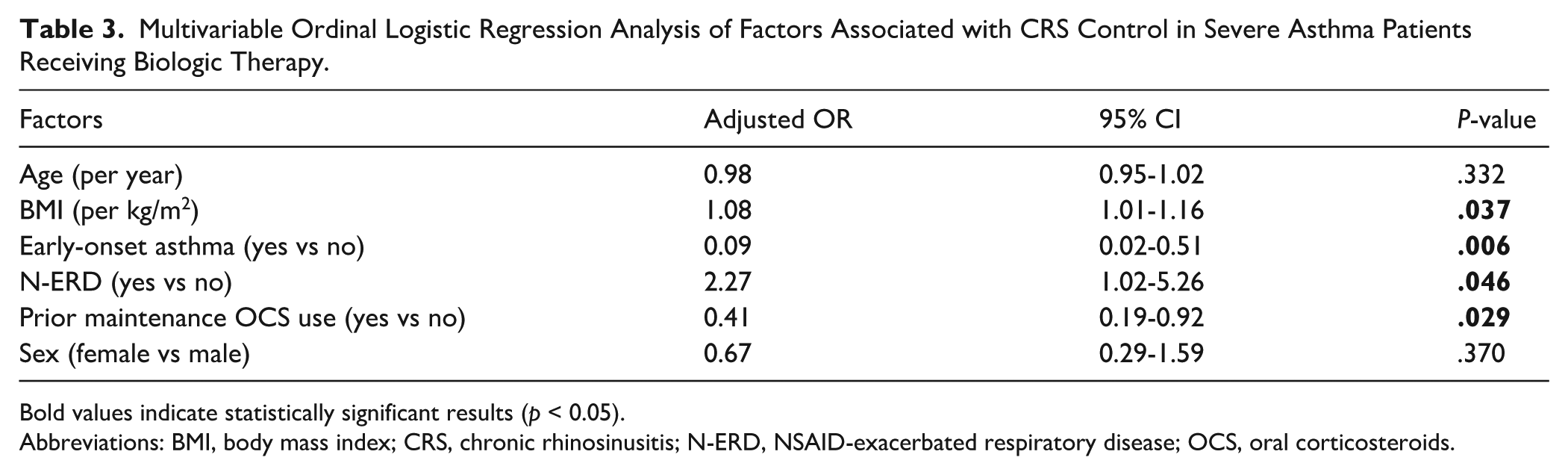
Bold values indicate statistically significant results (p < 0.05).
Abbreviations: BMI, body mass index; CRS, chronic rhinosinusitis; N-ERD, NSAID-exacerbated respiratory disease; OCS, oral corticosteroids.
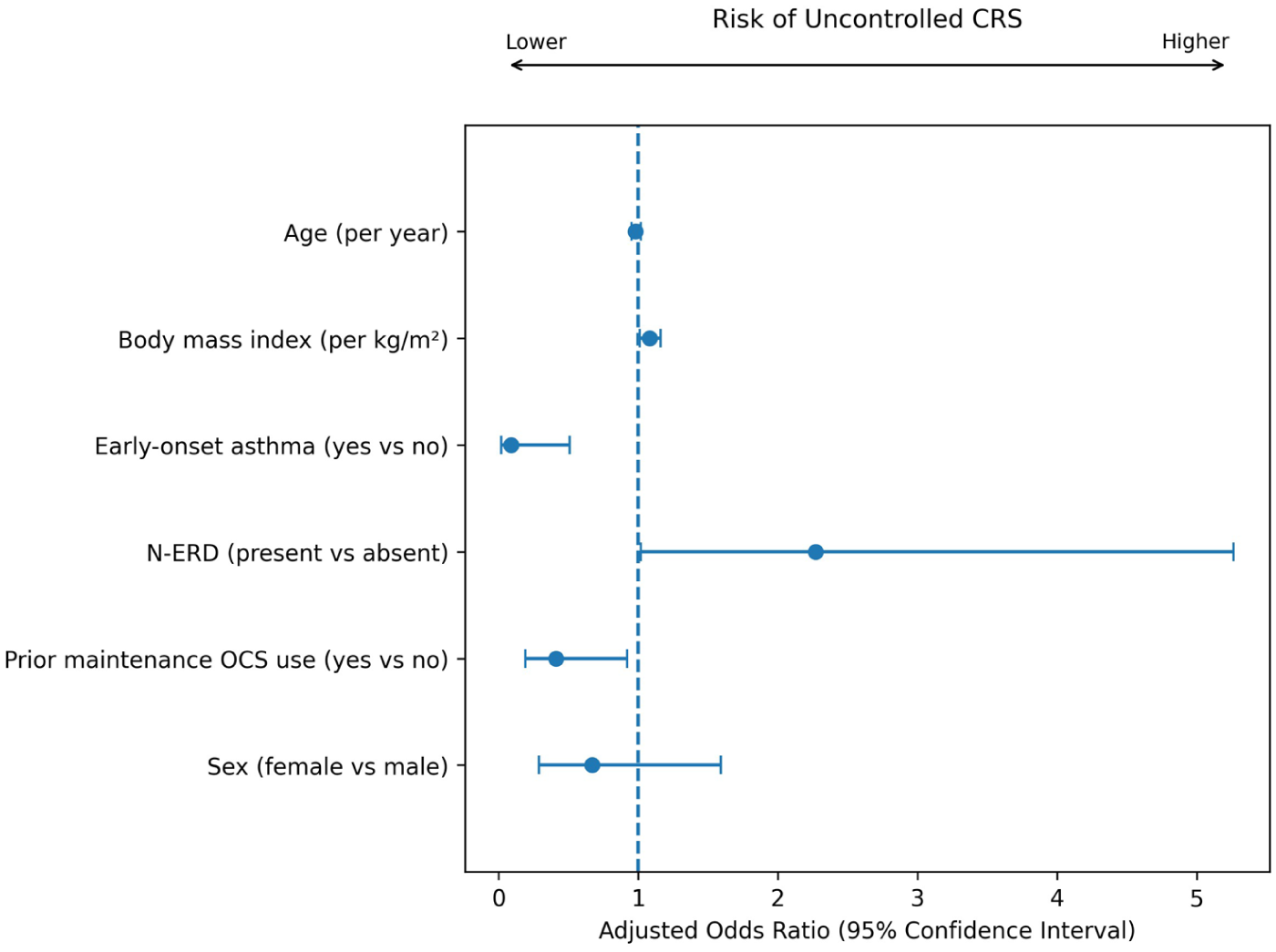
Forest plot showing factors associated with CRS control in severe asthma patients receiving biologic therapy based on multivariable ordinal logistic regression analysis.
In sensitivity analyses, time on biologic therapy was additionally included as a continuous covariate in the multivariable model, which did not materially change the direction or magnitude of the observed associations. Similarly, in an analysis restricted to patients treated within the overlapping exposure window of 6 to 36 months (n = 49), no significant differences in CRS control status were observed between biologic agents. In exploratory subgroup analyses, N-ERD was significantly associated with poorer CRS control (ordinal OR = 2.46, 95% CI: 1.14-5.32; P = .022). CRS subtype showed a non-significant trend toward poorer control in CRSwNP compared with CRSsNP (ordinal OR = 2.04, 95% CI: 0.90-4.61; P = .086).
Discussion
This single-center, cross-sectional study describes the distribution of CRS control per EPOS 2020 among patients with severe asthma receiving biologic therapy. CRS was present in 65.5% of the cohort; within the CRS subgroup, 32.9% were controlled, 36.0% partly controlled, and 30.9% uncontrolled. These findings provide a real-world snapshot of CRS control in this population.
Chronic rhinosinusitis, as a common comorbidity of asthma, is associated with more severe disease. 12 Previous studies report CRS prevalence in asthmatics ranging from 26.8% to 40.6%, depending on disease severity and population.13,14 Our finding of a 65.5% CRS prevalence in severe asthma patients aligns with this trend and underscores the importance of managing upper airway involvement in this group.
Our study was inspired by the cross-sectional design employed by van der Veen et al., although the patient populations, interventions, and follow-up durations differ substantially. Van der Veen et al. assessed CRS outcomes 3 to 5 years after sinus surgery and reported that approximately 19.5% of patients were well controlled, 36.8% were partly controlled, and 43.7% were uncontrolled. In our cohort of severe asthma patients receiving biologic therapy, 32.9% were controlled, 36.0% partly controlled, and 30.9% uncontrolled. While these results are not directly comparable due to methodological differences, both studies underscore the persistent challenge of achieving CRS control in clinical practice.
To date, omalizumab, mepolizumab and dupilumab have been approved by the FDA for the treatment of CRSwNP in patients unresponsive to standard therapies. All have been proven to improve disease-specific quality of life, dupilumab has also been shown to reduce disease severity. 15 Real-world studies have reported that omalizumab improved symptom scores and/or the nasal polyp score (NPS) at 6 to 12 months in patients with CRSwNP and severe allergic asthma.16,17 A recent randomized controlled trial also demonstrated that omalizumab reduces polyp tissue regeneration and decreases relapse frequency. 18 Mepolizumab has been shown to improve SNOT-22 scores after 12 months of treatment. 19 Real-life studies show that in patients with severe asthma and CRSwNP, biologic treatments (anti-IgE, anti-IL-5/R, anti-IL-4R) led to significant improvements in SNOT-22, VAS, and CT scores after 6 months.20-22 However, due to the cross-sectional design of our study and the lack of baseline CRS control data prior to biologic initiation, it is not possible to draw definitive conclusions regarding the direct impact of biologic therapy on CRS symptom improvement in our cohort. Nonetheless, the observation that approximately two-thirds of patients had either controlled or partly controlled CRS, along with the low rate of endoscopic sinus surgery (12.4%) during follow-up, might suggest a favorable trend in CRS management under biologic treatment in real-life clinical practice.
There are limited data on CRS control among patients with severe asthma receiving biologics. In a prospective study of 40 patients with severe asthma who received omalizumab, mepolizumab, or benralizumab for 52 weeks, the proportion of patients with uncontrolled CRS decreased from 82.5% at baseline to 37.5% after treatment. Following treatment, 15% of patients achieved controlled CRS, 47.5% were partly controlled, and 37.5% remained uncontrolled. 23 The proportion of patients with uncontrolled CRS on biologics was 30.9% in our study, closely aligning with the post-treatment outcomes reported in this study.
In multivariable analysis, early-onset asthma and prior maintenance oral corticosteroid use were independently associated with better CRS control, whereas N-ERD and higher BMI were independently associated with poorer CRS control (Table 3, Figure 3). Although age-related differences in inflammatory profiles and treatment responsiveness have been reported previously,24,25 age was not independently associated with CRS control in our cohort after multivariable adjustment, despite showing an association in univariate analyses. Early-onset asthma, typically associated with allergic sensitization and type 2 inflammation, is known to respond more favorably to corticosteroids and anti-IgE therapy.26,27 This may support the united airway hypothesis and the effectiveness of biologics targeting type 2 inflammation in this subgroup. The observation that patients with prior OCS use were more likely to have controlled CRS may also reflect a steroid-responsive disease profile. In addition, our findings are consistent with prior evidence linking higher BMI to worse CRS outcomes, possibly due to the pro-inflammatory effects of obesity and its association with non-type 2 inflammation. 28 Lastly, the presence of N-ERD was independently associated with poorer CRS control, which is consistent with previous evidence linking N-ERD to more severe and treatment refractory CRS. 29 Subgroup analyses also suggested higher symptom burden and poorer control among patients with N-ERD, consistent with these observations. While these findings are exploratory, they may inform future hypotheses regarding clinical and biological predictors of CRS control in patients with severe asthma receiving biologic therapy.
In patients with severe asthma, the selection of biologic therapies is guided by current international guidelines and limited to agents approved for use in our country. The higher prevalence of atopy and perennial allergen sensitization in the omalizumab group, along with the elevated eosinophil counts observed in the mepolizumab group, likely reflects an endotype-driven approach to biologic prescribing in our cohort. The longer duration of biologic use in the omalizumab group is presumably due to its earlier national approval and wider availability. Nevertheless, sensitivity analyses accounting for treatment duration yielded similar findings, suggesting that differences in biologic exposure time may not have affected the main results. Additionally, while most existing literature and therapeutic indications primarily focus on CRSwNP, this study also included patients with CRSsNP, who similarly exhibited features of type 2 inflammation. This highlights the need to recognize CRSsNP as part of the type 2–high airway disease spectrum and to ensure its inclusion in future clinical studies and treatment strategies. Future longitudinal studies with larger cohorts may benefit from applying propensity score–based or weighting approaches to better address confounding by indication in comparative analyses of biologic therapies.
This study has several limitations. First, its single-center, cross-sectional design permits only observational associations and does not allow for conclusions about causality. The absence of standardized data on CRS control prior to biologic initiation limits the ability to assess treatment-related changes over time. Additionally, CRS control was evaluated primarily through patient-reported outcomes, without objective radiologic or endoscopic confirmation, which may introduce subjective bias. Although information on rescue treatment was supported by medical record review whenever possible, some degree of recall bias related to patient-reported data cannot be completely excluded. Moreover, at the time of data collection, only omalizumab and mepolizumab were licensed and reimbursed in our country; therefore, the potential effects of other biologic agents, such as dupilumab or benralizumab, could not be assessed. Although sensitivity analyses accounted for treatment duration, differences in biologic exposure time between groups cannot be completely excluded as a potential source of residual confounding.
Nevertheless, this study offers a valuable real-world snapshot of CRS control among patients with severe asthma receiving biologic therapy in a considerable number of patients for this specific clinical population. As the concept of disease control gains prominence in the management of chronic airway diseases, routine assessment of CRS status becomes increasingly relevant in optimizing outcomes. Furthermore, the observation that CRS may remain uncontrolled despite well-controlled asthma highlights the need for independent evaluation of upper airway disease during follow-up. In selected cases, persistent CRS symptoms could prompt clinicians to reconsider the adequacy of current biologic therapy and potentially explore switching strategies.
In conclusion, this real-life observational study underscores the high prevalence of comorbid CRS among patients with severe asthma receiving biologics, and reveals that CRS control remains suboptimal in approximately one-third of patients. These findings highlight the importance of routinely evaluating upper airway disease alongside asthma management and suggest that future longitudinal and multicenter studies are warranted to identify predictive factors and optimize biologic selection for comprehensive airway control.
Supplemental Material
sj-docx-1-aor-10.1177_00034894261434347 – Supplemental material for Control Status of Chronic Rhinosinusitis in Severe Asthma Patients Receiving Biologics: A Real-World Cross-Sectional Study
Supplemental material, sj-docx-1-aor-10.1177_00034894261434347 for Control Status of Chronic Rhinosinusitis in Severe Asthma Patients Receiving Biologics: A Real-World Cross-Sectional Study by Sevgi Çolak, Zeynep Çelebi Sözener, Ömür Aydin, Mustafa Çolak, Sevim Bavbek, Betül Ayşe Sin, Yavuz Selim Demirel and Dilşad Mungan in Annals of Otology, Rhinology & Laryngology
Footnotes
Ethical Considerations
This study was approved by the Ankara University Faculty of Medicine Ethics Committee (approval number: İ3-162-20), and informed consent was obtained from all participants.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.*
Supplemental Material
Supplemental material for this article is available online.
