Abstract
Introduction:
As the demand for otolaryngologists increases, there is a need to address workforce and training challenges. Technological advancements, such as high-fidelity 3-dimensional (3D) simulations, may help train the future workforce—particularly for conditions with limited exposure, such as sinonasal and skull base cancers.
Aims:
This study aimed to develop and validate a high-fidelity head and neck model, created using proprietary 3D printing technology, to simulate sinonasal and skull base surgery.
Methodology:
The sinonasal and skull base models were designed and produced using proprietary 3D printing technology (Fusetec). 3D printed models were incorporated into a 2-day dissection course. Surveys were completed by delegates and faculty on the course, with delegates having their surgical performance assessed using the Objective Structures Assessment of Technical Skills (OSATS).
Results:
A total of 21 delegates and 10 faculty had utilized the 3D printed models for sinonasal and skull base surgery training. Overall, 71.4% of delegates (n = 15) and 70.0% (n = 7) of faculty rated the realism of the models as very good/excellent. The mean pre course OSATS score was 22.57/40 (SD ±6.88) and the post course OSATS score was seen to be 25.7/40 (SD ±4.70; P < .0001). The total confidence reported by delegates prior to the course was 25.8/45 (SD ±7.00) and following the course the self-reported confidence was 33.6/45 (SD ±3.62; P < .001).
Conclusion:
This study developed and validated a novel 3D model for simulating sinonasal and skull base surgery, which improved surgical performance amongst participants. This offers a potential training solution to address workforce challenges in Otolaryngology.
Introduction
Surgical training enables surgeons to practice and refine their skills to achieve the best possible outcomes for patients.1 -3 The traditional “see one, do one, teach one” approach is now considered insufficient and unacceptable for ensuring patient safety.2,3 With advancements in technology, surgical training and education are evolving. Simulation plays a crucial role in expediting training and ensuring that surgeons become safe practitioners.2,4,5
Proficiency in endoscopic sinonasal and skull base surgery requires methodical practice, repetition, and feedback, which are often difficult for trainees to obtain due to limited exposure. 6 Endoscopic sinus surgery presents significant visuospatial challenges, as it requires working in a 3-dimensional space while relying only on a 2-dimensional image on the screen. This makes it particularly difficult for trainees to master. This anatomical region is fraught with the risk of life-altering complications due to its proximity to critical structures.6,7 While real patient experience remains a cornerstone of surgical training, ensuring trainees have appropriate foundational knowledge and supervised, graduated involvement based on their competency level is essential for maintaining patient safety.1 -3
Anatomically accurate models serve as excellent tools for both novice and experienced surgeons, especially in procedures with limited exposure. Simulation training enhances surgeon confidence, skills, and clinical outcomes.8,9 This is particularly valuable in sinonasal and skull base surgery due to the complex anatomy, challenging ergonomics, and high-risk complications.6,7 Moreover, due to the rarity of sinonasal and skull base cancers, trainees and fellows may have limited exposure to these conditions and, as a result, may not develop the necessary skills to manage such cases by the end of training program. This presents a significant challenge for the future Otolaryngology workforce, as a lack of adequate training could leave them ill-prepared to handle these complex cases competently. Therefore, it is essential that training programs evolve to ensure that Otolaryngology residents/fellows gain sufficient hands-on experience to be well rounded by the time they become attending surgeons. Simulation models, such as 3-dimensional (3D) printed models, could help bridge this gap by providing trainees with opportunities to practice and refine their skills in a controlled, risk-free environment, ensuring they are better equipped to manage these cases effectively in clinical practice.1,6,7 While the use of 3D models has been suggested as a potential method for providing hands-on training in endoscopic sinus surgery and improving the learning curve, 7 no previous study has developed and validated their use in endoscopic sinonasal and skull base cancer resection.
This study aimed to develop and validate a high-fidelity head and neck model, created using proprietary 3D printing technology for simulating sinonasal and skull base surgery for Otolaryngologists and Otolaryngology trainees/interns.
Methodology
3D-Printed Simulation Model Design
The 3D models were designed and produced by Fusetec (Adelaide, Australia) using a combination of techniques. New cassettes and tumors were created to surround a previously made off the shelf models: Nose 6 (Figure 1A) and the pituitary model (PT). 10 Although it was a finished product, Nose 6 was modified to include frontal sinus tumors (Figure 1B) and maxillary tumors (Figure 1C).
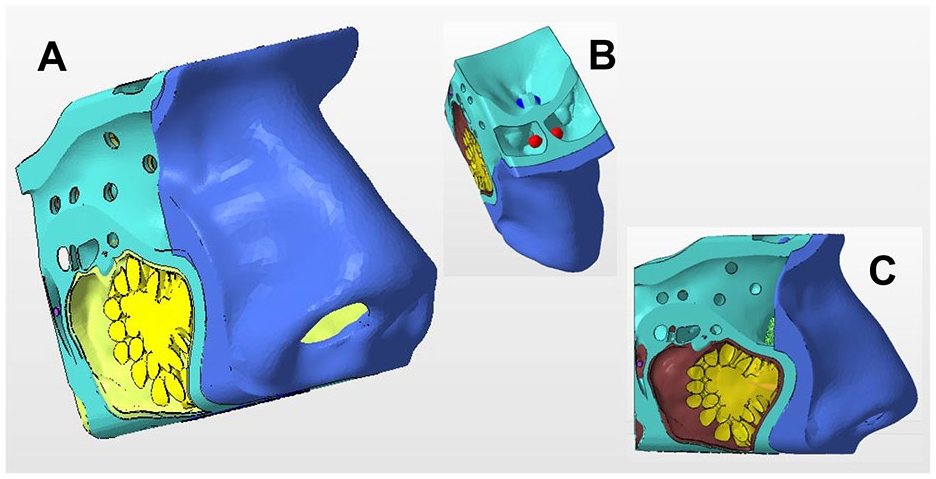
(A) Edited Nose-6 model. (B) Bilateral frontal sinus tumors (red). (C) Maxillary sinus tumor (yellow).
The head base (Figure 2A) and nasopharyngeal cassette (Figure 2B) were developed using the sculpting software ZBrush (Pixologic Inc, USA). Actual images and consultations with surgeons were utilized to ensure that the tumors were accurate in shape, size, and location. A surgical navigation system was connected to the models, enabling participants to better understand the anatomy.
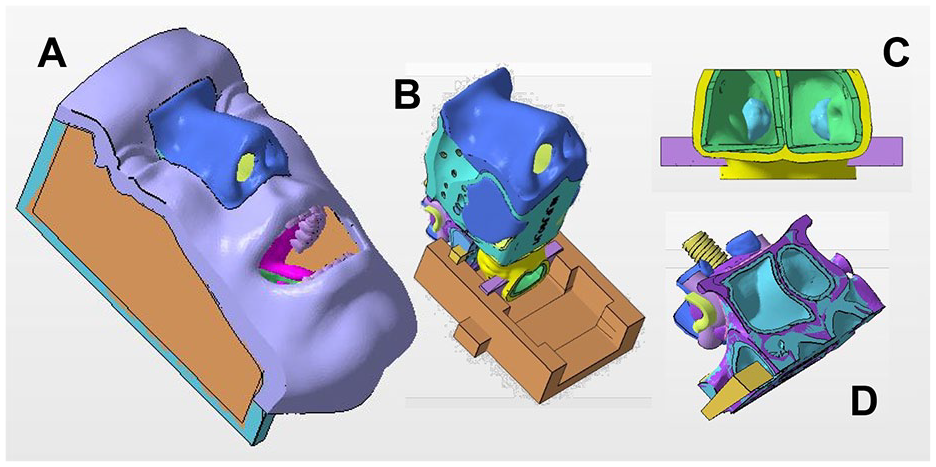
Internal model components. (A) Head base. (B) Edited nose-6. (C) Nasopharyngeal tumor. (D) Pituitary tumor.
The model components were then assembled and further edited on Netfabb (Autodesk, USA) for manufacturing. Materials were selected based upon surgeon feedback of haptic response. Models were printed using the Stratasys 3D Printer (Stratasys Ltd, USA; Figure3).
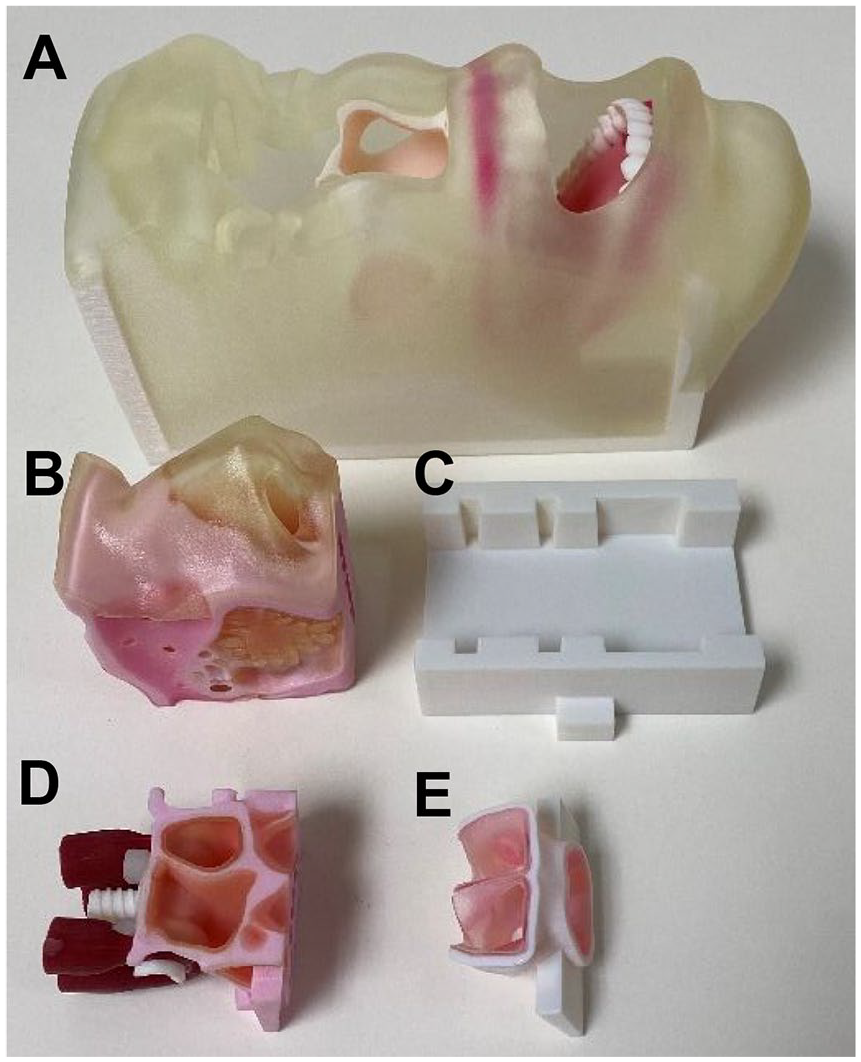
Printed model and components separated. (A) Head base. (B) Edited nose-6. (C) Base frame. (D) Pituitary tumor. (E) Nasopharyngeal tumor.
Sinonasal and Skull Base Training Course
The models were validated in a 2-day in person training course. The course included lectures, practical demonstrations, and hands-on skills sessions using 3D-printed head and neck models. Delegates completed a total of 4 sessions over the 2 days, covering maxillary sinus access and excision of a maxillary tumor, pituitary access and excision of pituitary tumor, nasopharyngectomy, and frontal sinus access and excision of a frontal sinus tumor, all transferable skills in endoscopic sinus surgery. The faculty consisted of Otolaryngology consultants/attendings, maintaining a faculty-to-delegate ratio of approximately 1:2 to assist with procedural steps and teaching.
The 3D model simulation training was conducted after delegates received content tutorials, followed by practical demonstrations of the surgical procedures on the 3D models performed by the faculty (surgical setup seen in Supplemental Figure A.1). Upon completion of the course, both delegates and faculty were asked to complete paper-based surveys, which included a combination of yes/no items and Likert scales to evaluate their experience with the 3D models.
Validation
The validation of the 3D models involved multiple methods to confirm their realism, educational value, and effectiveness. First, face validity was assessed by having delegates and faculty evaluate the realism of the models for performing sinonasal and skull base procedures. This included the overall accuracy in mimicking sinonasal/skull base surgery, haptic/tactile feedback, tumor visualization/haptic feedback, and anatomical accuracy. Second, content validity was ensured by mapping the included surgical procedures to the postgraduate Otolaryngology curriculum. Additionally, content validity was further assessed by having faculty and delegates rate the usefulness of the models for learning sinonasal and skull base cancer surgical skills.
Lastly, criterion validity was determined by having 2 independent examiners rate the surgical performance in the model dissection using the objective structured assessment of technical skills (OSATS) score at the start of the course (Day 1, Session 1) and at the end of the course (Day 2, Session 4). 1 The OSATS scoring system evaluates participant performance on specific aspects of endonasal surgery (understanding of anatomy, indications and objectives of surgery, use of endoscope, knowledge of instruments, instrument handling, respect for tissues, knowledge of procedure, time, and motion, and flow of operation) on a scale from 1 (unable to perform) to 5 (performs easily with good flow).1,5 An overall score was then calculated (maximum 40), averaging the scores from both examiners. Any faculty member involved in the design of the models did not complete the validation survey to eliminate any potential bias and were not involved in examining surgical performance.
Construct validity was assessed by having delegates complete self-reported confidence surveys on various sinonasal and skull base cancer surgical procedures before and after using the 3D models.
Statistical Analysis
The collected data were transferred and analyzed using GraphPad Prism 7 (GraphPad Software, San Diego, USA). Data were expressed as mean and standard deviation. A Shapiro-Wilk test was conducted to assess the normal distribution of the data. Data that followed a normal distribution (tumor visualization, pre/post-course surgical performance) were analyzed using parametric testing (paired/unpaired t-tests). Data that did not follow a normal distribution (anatomy rating, instrument haptic feedback, self-reported confidence, usefulness of models for learning, trainees’ access to sinonasal and skull base cancer surgery, mimicking real-life surgery, tissue texture, and tumor haptic feedback) were analyzed using a Mann-Whitney U test. Statistical significance was set at P < .05.
Ethical Statement
Delegates who participated in the course and completed the survey gave implied consent to partake in the study and have their data used for research purposes by voluntarily completing the survey. All delegates had the option to complete the survey and be assessed as part of the process, with no personal information collected. They were informed that their responses and feedback would be used for research and publication purposes.
In line with the ethical guidelines in the United Kingdom (UK), a thorough ethical assessment was carried out using the UK Research and Innovation (UKRI) Research Ethics Tool before the commencement of our study. According to the UKRI Research Ethics Tool, ethical approval was not required.
Results
Baseline Characteristics
A total of 21 delegates had utilized the 3D printed models for sinonasal and skull base cancer surgery training and responded to the survey. Among them, 28.6% (n = 6) were senior Otolaryngology Residents/Registrars, 23.8% (n = 5) were junior Otolaryngology Residents/Registrars, 14.3% (n = 3) were Associate Specialists in Otolaryngology, 9.5% (n = 2) were Rhinology and Skull Base Fellows, 14.3% (n = 3) were Otolaryngology attendings/consultants, and 9.5% (n = 2) were foundation doctors/interns. Nearly half of the delegates had 6 to 12 months’ experience in rhinology and skull base surgery (n = 10, 47.6%). This was followed by: 13 to 24 months (n = 4, 19.0%), 37 to 48 months (n = 2, 9.5%), >48 months (n = 2, 9.5%), 0 months (n = 2, 9.5%), and 25 to 36 months (n = 1, 4.8%). Prior to the course delegates had attended other endoscopic sinus surgery and skull base courses with 19.0% (n = 4) attending 2 courses, 19.0% (n = 4) attending 3 courses, 19.0% (n = 4) attending 5 or more courses, and 14.3% (n = 3) attending 1 or more courses. A 19.0% (n = 4) had attended 0 courses prior. Ten faculty members experienced in sinonasal and/or skull base surgery were surveyed. The group consisted of 9 Otolaryngology consultants/attendings and 1 Neurosurgery consultant/attending.
Training Opportunities in Sinonasal and Skull Base Surgery
Delegates and faculty were asked “how much do you agree with the following statement: it is difficult for registrars/residents to gain exposure to sinonasal and skull base cancer surgery.” With 38.1% (n = 8) strongly agreeing, 52.4% (n = 11) agreeing, 4.8% (n = 1) being neutral and 4.8% (n = 1) disagreeing with the statement. With 40.0% (n = 4) of faculty strongly agreeing with the same statement and 60.0% (n = 6) agreeing with the statement. Faculty were asked “how much do you agree with the following statement: there is a need to increase training opportunities in sinonasal and skull base cancer surgery.” To which 70.0% (n = 7) strongly agreed, 20.0% (n = 2) agreed, and 10.0% (n = 1) remained neutral.
Face Validity and Content Validity
Face validity was assessed by having the delegates rate how closely the model mimicked real life sinonasal and skull base surgery. With 71.4% (n = 15) rating this as very good or excellent and 28.6% (n = 6) rating this as average. Similarly, 70.0% (n = 7) of faculty rated the same measure as very good or excellent and 30.0% (n = 3) rated this as average. With no significant difference seen in the rating between delegates and faculty (P = .1936). With regards to the tissue texture of the 3D models in comparison to real/cadaveric tissue 61.9% (n = 13) of delegates rated this as having excellent or very good resemblance, 28.6% (n = 6) rating this as average resemblance and 9.5% (n = 2) rating this as below average resemblance. Similarly, faculty rated the tissue resemblance with 60.0% (n = 6) rating this as excellent or very good and 40.0% rating this as average. No statistically significant differences were found between delegates and faculty ratings of tissue resemblance (P = .3539).
The visual and haptic realism of the 3D printed tumors was then rated by delegates and faculty this is displayed in Table 1. The tactile feedback (delegates and faculty) from the surgical instruments on the 3D model tissue are seen in Figure 4A and 4B. No statistically significant differences were seen in realism ratings of the instruments on the models between delegates and faculty (P = .7621). The realism of the important anatomical structures relating to sinonasal, and skull base surgery was assessed using a Likert Scale (1-5) this is displayed in Figure 5A and B. There was a statistically significant difference in the overall anatomical ratings of realism between faculty and delegates, with faculty recording higher anatomical realism ratings versus delegates (P = .0009).
Tumor Visual and Haptic/Tactile Feedback and Realism.
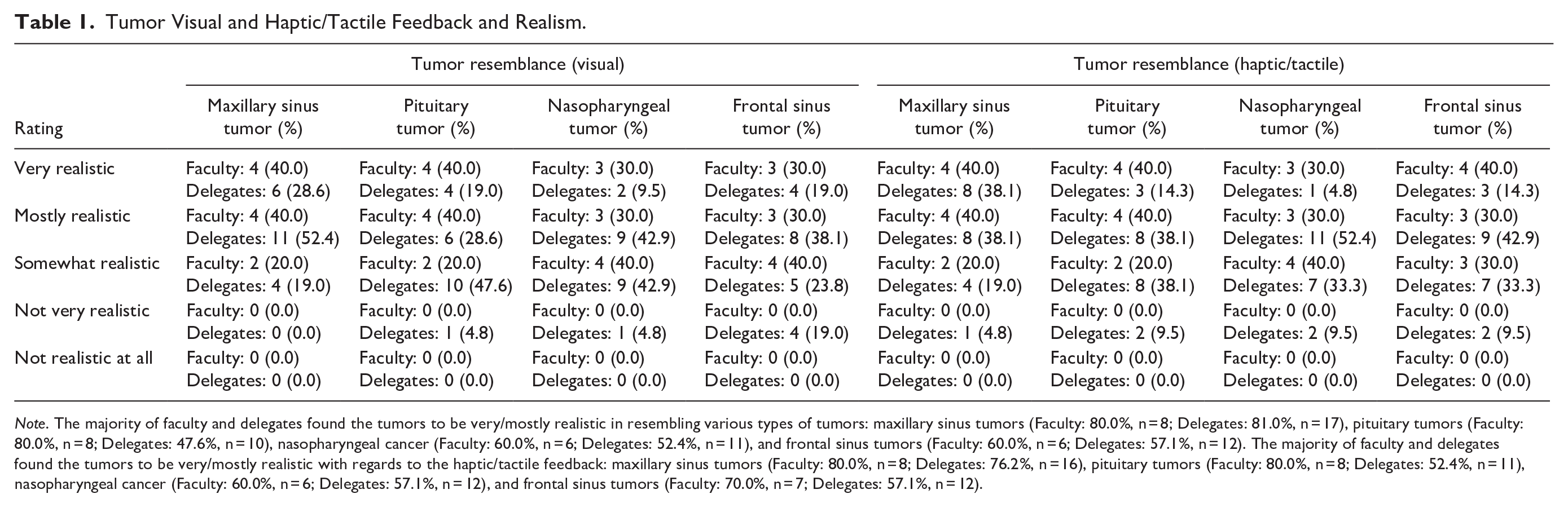
Note. The majority of faculty and delegates found the tumors to be very/mostly realistic in resembling various types of tumors: maxillary sinus tumors (Faculty: 80.0%, n = 8; Delegates: 81.0%, n = 17), pituitary tumors (Faculty: 80.0%, n = 8; Delegates: 47.6%, n = 10), nasopharyngeal cancer (Faculty: 60.0%, n = 6; Delegates: 52.4%, n = 11), and frontal sinus tumors (Faculty: 60.0%, n = 6; Delegates: 57.1%, n = 12). The majority of faculty and delegates found the tumors to be very/mostly realistic with regards to the haptic/tactile feedback: maxillary sinus tumors (Faculty: 80.0%, n = 8; Delegates: 76.2%, n = 16), pituitary tumors (Faculty: 80.0%, n = 8; Delegates: 52.4%, n = 11), nasopharyngeal cancer (Faculty: 60.0%, n = 6; Delegates: 57.1%, n = 12), and frontal sinus tumors (Faculty: 70.0%, n = 7; Delegates: 57.1%, n = 12).
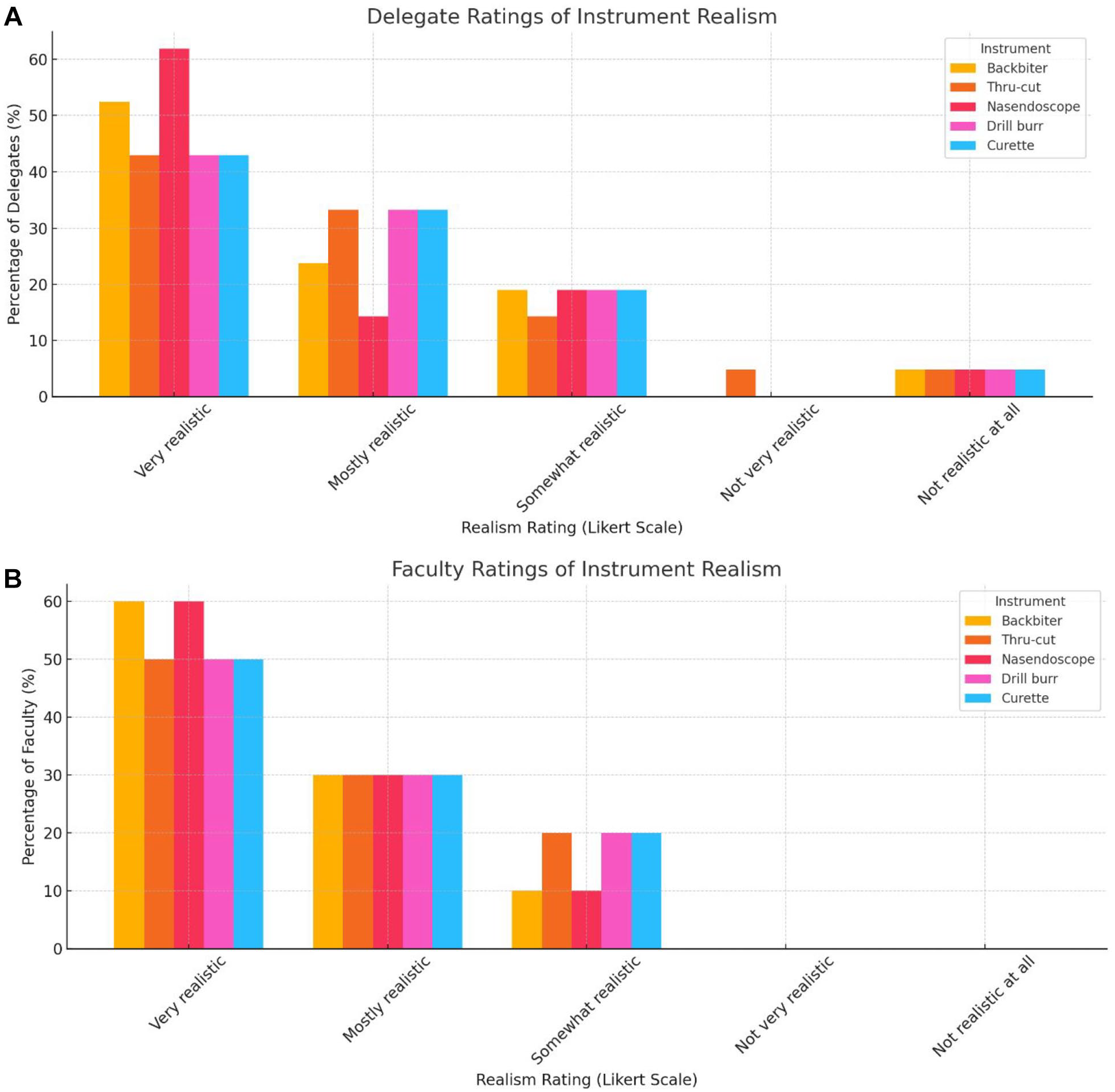
Realism and haptic/tactile feedback of surgical instruments on the 3D model. (A) Delegate ratings. (B) Faculty ratings. The majority of faculty and delegates found the haptic feedback from various instruments to be very/mostly realistic backbiter (Faculty: 90.0%, n = 9; Delegates: 67.7%, n = 14), thru-cut (Faculty: 80.0%, n = 8; Delegates: 76.2%, n = 16), nasendoscopy (Faculty: 90.0%, n = 9; Delegates: 76.2%, n = 16), drill-burr (Faculty: 80.0%, n = 8; Delegates: 76.2%, n = 16), and curette (Faculty: 80.0%, n = 8; Delegates: 76.2%, n = 16).
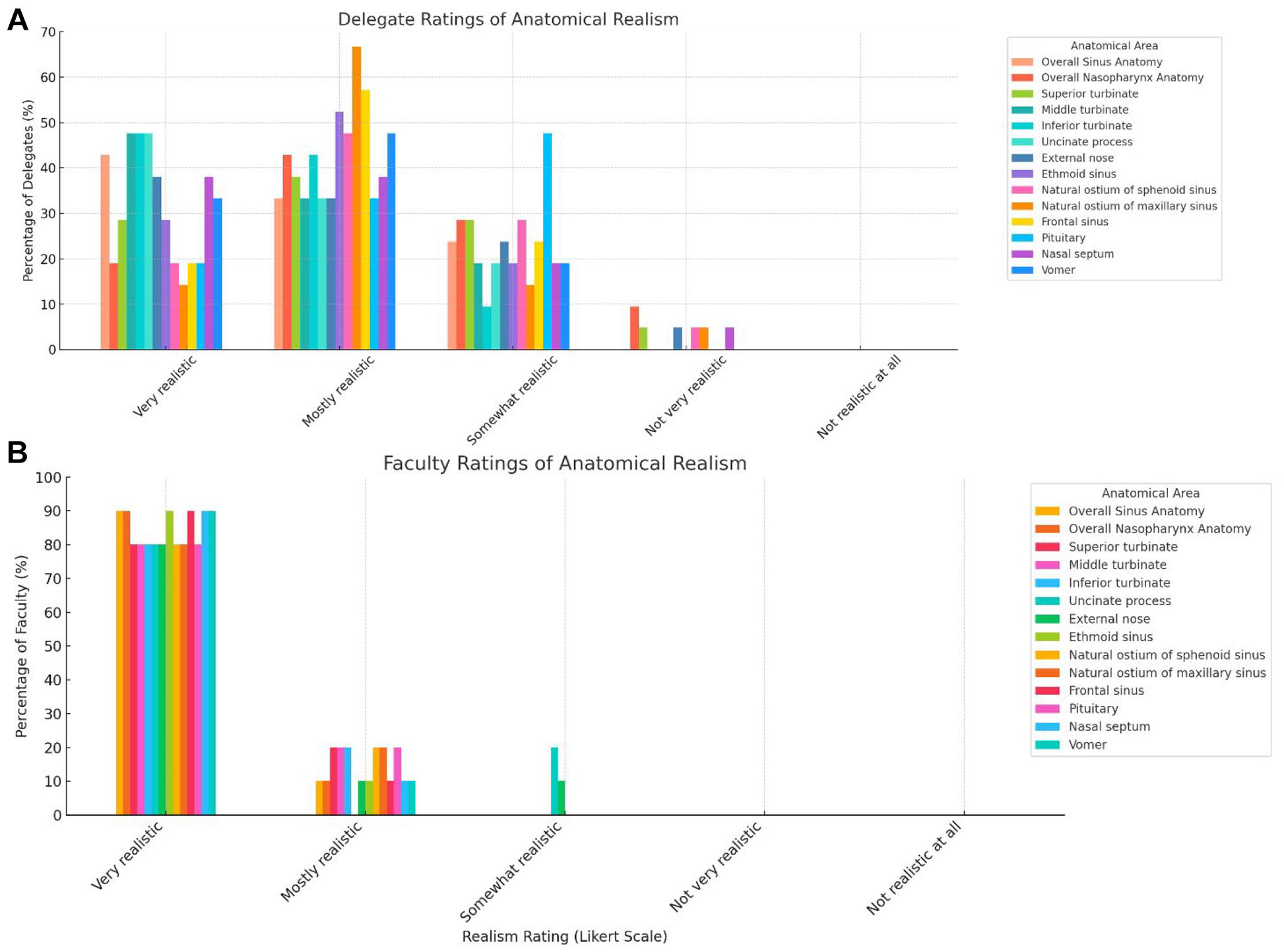
Anatomical realism of 3D models. (A) Delegate rating. (B) Faculty rating. The majority of faculty and delegates reported the anatomy of the models to be very/mostly realistic with regards to overall sinus anatomy (Faculty: 100.0%, n = 10; Delegates: 76.2%, n = 16), nasopharynx anatomy (Faculty: 100.0%, n = 10; Delegates: 61.9%, n = 13), frontal sinus anatomy (Faculty: 100.0%, n = 10; Delegates: 76.2%, n = 16), and pituitary anatomy (Faculty: 100.0%, n = 10; Delegates: 61.9%, n = 13).
Content validity was ensured by mapping the content of the course to the Otolaryngology Curriculum outlined by the Intercollegiate Surgical Curriculum Program. The faculty were asked “are there any anatomical details or procedural steps that are missing or inadequate for learning?” All faculty (n = 10, 100%) responded with no steps/details missing. Delegates and faculty were also asked to rate the usefulness of the 3D models for learning sinonasal and skull base cancer surgery. Overall, 57.1% (n = 12) of delegates found the models to be very or extremely helpful in enhancing their learning, 28.6% (n = 6) found them to be moderately helpful, and 14.3% (n = 3) found them to be slightly helpful. A 100% (n = 10) of faculty found the models to be extremely or very helpful for enhancing surgical trainees learning during the course. No statistically significant differences were seen between delegates and faculty (P = .0819).
Criterion and Construct Validity
Criterion validity was assessed by faculty rating delegates surgical performance using the OSATS score in the first session of the course and then rating them again in the final session at the end of the 2 days. The mean pre course OSATS score was 22.57/40 (SD ±6.88) and the post course OSATS score was seen to be 25.7/40 (SD ±4.70; P < .0001; Supplemental Figure A.2).
The mean improvements were then compared based upon grade of the delegate. Mean improvement by surgical experience was: surgical attendings/consultants (1 ± 0.81), surgical registrars/residents (4.2 ± 2.3), associate specialists/fellows (0.6 ± 0.8), and interns/foundation doctors (7 ± 1). On post hoc analysis only significant differences were seen in mean improvement in surgical performance scores between interns/foundation doctors and fellows/associate specialists (P = .044; Supplemental Figure A.3).
To assess construct validity delegates were asked to self-report their confidence before and after the course using a Likert Scale (1-5) and an overall score out of 45 calculated. Participants provided a score from 1 (no confidence) to 5 (complete confidence) to rate their confidence for each of the following procedural steps before and after the course: ethmoidectomy, sphenoidotomy, endoscopic nasopharyngectomy, maxillectomy, resection of maxillary tumor, endoscopic access to frontal sinus, endoscopic resection of frontal sinus tumor, endoscopic resection of pituitary tumor, and endoscopic access to pituitary gland. The total confidence reported by delegates prior to the course was 25.8/45 (SD ± 7.00) and following the course the self-reported confidence was 33.6/45 (SD ± 3.62; P < .001; Supplemental Figure A.4).
Future Directions
Faculty were asked on future courses what their preferred method of practicing endoscopic sinonasal and skull base surgery skill would be. Overall, 80.0% (n = 8) stated using both cadavers and 3D models whilst 20.0% (n = 2) stated 3D models alone. Similarly, delegates also stated they would prefer to have 3D models and cadavers in future courses (n = 16, 76.2%), followed by 3D models only (n = 2, 9.5%) and cadavers only (n = 2, 9.5%). A 100% (n = 10) of faculty recommended trainees use 3D models to learn endoscopic sinonasal and skull base cancer surgical skills. Similarly, 100% (n = 21) of delegates also would recommend the use of 3D models to their colleagues. Furthermore, 100% (n = 10) of faculty stated that 3D models could be used to complete Procedure Based Assessments/ Direct Observation of Procedural Skills to assess trainee surgical performance.
Discussion
To our knowledge, this is the first study that has developed and validated the use of a high-fidelity 3D model for simulating endoscopic sinonasal and skull base cancer resection. The study demonstrates the benefits of simulating sinonasal and skull base cancer resection using advanced 3D models. Models developed using proprietary technology demonstrated strong face, content, construct, and criterion validity. Training with 3D models significantly enhanced both subjective self-reported confidence and objective surgical performance scores. The most notable improvements were seen among foundation doctors/interns and residents/registrars, though gains were observed across all participant levels. Hence, 3D models may provide a potential method for evolving surgical training and ensuring the future Otolaryngology workforce have the skills necessary to manage a variety of pathology including rarer sinonasal and skull base cancers.
As the number of endoscopic surgical cases has expanded over the last few decades, simulation models for advanced endoscopic sinonasal and skull base surgery training have grown in popularity. 6 Previous studies have highlighted the limited exposure to advanced sinonasal and skull base surgery during otolaryngology training. 11 In our cohort, 100% of faculty and 90.5% of delegates reported difficulty in gaining exposure to sinonasal and skull base cancer surgery during training. Therefore, addressing this gap in exposure is crucial for training the future Otolaryngology workforce, with 3D models presenting a potential solution. High-fidelity 3D models offer a means for surgeons to gain procedural competency and optimize their performance in the operating room, making them a promising tool for training the future workforce. Similarly, previous studies have demonstrated that effective surgical simulation models enhance surgeon confidence, skills, and clinical outcomes, and may also reduce healthcare costs.1,6
High-fidelity surgical simulations are crucial in most surgical disciplines, particularly in sinonasal and skull base cancer surgery, where real-life exposure for trainees is limited.6,11 The complexity of the anatomy, challenging ergonomics, and high risk of potential complications make it difficult for trainees to learn through the traditional apprenticeship model and may result in gaps in surgical skills. 6 While real-life hands-on training through this model offers an ideal opportunity for observation, it is a high-stakes environment that can jeopardize patient safety.1,6 Therefore, providing additional surgical education through high-fidelity 3D models allows trainees to enhance, develop, and refine their surgical skills without compromising patient safety, ensuring that future surgeons are equipped with the skillset to competently manage a broader scope of disease.1,6,7
Within our cohort, faculty members found the anatomical resemblance of the models to be higher than what the delegates perceived. This difference may be attributed to the faculty’s greater experience and familiarity with the anatomy of the nose and sinuses compared to the delegates, who may be less familiar. However, there were no significant differences between the faculty and delegates regarding the tissue texture and tactile feedback from surgical instruments. Although all faculty and delegates found the models helpful for learning, a non-significant difference was observed in the proportion rating them as very or extremely helpful. This may reflect differences in experience and expectations. Trainees, who are often more familiar with cadaveric-based courses, may be more inclined toward or expect traditional training methods. In contrast, faculty—with broader clinical and teaching experience—may better appreciate the evolving role of 3D models in surgical education. These perceptions may shift as simulation becomes more integrated into training.
The majority of both faculty and delegates felt that the models should be incorporated into future courses alongside cadaveric dissection. Cadaveric models have been used to simulate endoscopic neurosurgical tumor resections, though they often utilize low-fidelity tumor mimics made from injectable polymer.6,12 -15 Previously used models in endoscopic sinus and skull base surgery offer insight into basic skills and dissection techniques but are often limited by inadequate haptic feedback—rated highly in the current model.16 -18 Moreover, prior models have not been developed to simulate training for endoscopic sinonasal cancer surgery but are more limited in their scope. Additionally, models in the literature generally focus on specific skills (eg, septoplasty and maxillary antrostomy), whereas the current model provides delegates the opportunity to practice both fundamental skills and advanced cancer access and resection techniques.16 -18 In contrast, our study found that experienced faculty rated the haptic/tactile feedback and visual realism of the 3D tumor models as very or mostly realistic across all 4 tumor subtypes. This allowed participants, from interns to attending surgeons, to practice a range of skills—from basic endoscopic sinus surgery to advanced tumor access and resection.
Our cohort showed similar improvements in final OSATS scores were similar to those reported by Suzuki et al. (2022), who found that OSATS scores improved more after repeated dissection sessions than after a single repetition. 1 Therefore, 3D models are valuable for extending and developing skills beyond the course, allowing trainees to practice either alone or with experienced surgeons, both of which have been shown to improve surgical skills in previous literature. Trainees can perform procedure steps and complete work-based assessments to demonstrate competency in specific steps, which can then be integrated into clinical practice under senior supervision. Moreover, the models can be utilized to develop skills in endoscopic sinus surgery for all indications. This was highlighted in our study, where 100% of faculty reported that the models could be used for procedure-based assessments/direct observation of procedural skills. Additionally, 3D models allow for creating pathology on demand, enabling training without the risk of harmful pathogens and without the need for personal protective equipment in non-wet lab conditions. Moreover, there is a potential for 3D personalized tumor models in difficult cases, enabling even experienced surgeons to better prepare for complex surgery.
Limitations
This study had several limitations. Firstly, respondents had varied experience in sinonasal and skull base cancer surgery, with participants ranging from interns to consultants/attendings although prior research focussing on 3D models in endoscopic sinus surgery have utilized delegates with varying surgical experiencing.19 -21 Secondly, the sample size of delegates and faculty was small, but comparable to prior research,19 -21 making comparisons of OSATS scores based on surgical experience unreliable.
Thirdly, the models were used in a non-validated training program. Thus, the goal was not to ensure that delegates were signed off as competent to perform the procedures; rather, these 3D models were part of a course and are still in the evaluation stage. Fourthly, no comparator or control group was used to assess differences in surgical performance between 3D models and cadavers. Additionally, the OSATS used was validated for functional endoscopic sinus surgery rather than for sinonasal and skull base cancer excisions; however, it was employed to assess general endoscopic sinus surgery skills performance rather than procedure-specific performance. The examiners were not blinded during the assessment, which is a limitation. Future research could address this by using videos for assessment, ensuring that all examiners are blinded.
Conclusion
The development of this novel 3D model for simulating sinonasal and skull base surgery demonstrated significant benefits in surgical training and could provide a valuable modality for training the future Otolaryngology workforce. These models were shown to enhance surgeon confidence and improve objective surgical performance, making them highly relevant for training in sinonasal and skull base cancer surgery. They exhibit strong face, content, construct, and criterion validity in this context. Future efforts aim to integrate these models into surgical training programs to support ongoing skill development beyond formal courses.
Supplemental Material
sj-docx-1-aor-10.1177_00034894251340879 – Supplemental material for The Development and Validation of Novel 3-Dimensional Models for Simulation Training in Sinonasal and Skull Base Surgery
Supplemental material, sj-docx-1-aor-10.1177_00034894251340879 for The Development and Validation of Novel 3-Dimensional Models for Simulation Training in Sinonasal and Skull Base Surgery by Umar Rehman, Saleh Okhovat, Robbie Woods, Nyall R. London, Benjamin Verillaud, Christian von Buchwald, Gary L. Gallia, Sacit Bulent Omay, Warren Bennett, Kumar Abhinav, Garret Choby, Jayakar V. Nayak, Samuel C. Leong, Erich Vyskocil and Matt Lechner in Annals of Otology, Rhinology & Laryngology
Footnotes
Acknowledgements
We would like to acknowledge the support by Mr. John Budgen and Mr. Mark Roe from Fusetec Inc. (Adelaide, Australia), who supported the development of the technology and provided the materials for the testing of the models and the validation study.
Author Contributions
Umar Rehman: conceptualization, validation, formal analysis, Writing original draft, visualization, and project administration. Saleh Okhovat: conceptualization, validation, formal analysis, Writing review and editing, visualization, and project administration. Robbie Woods: conceptualization, validation, formal analysis, Writing original draft, visualization, and project administration. Nyall R. London, Jr.: Writing review and editing, project administration, and conceptualization. Benjamin Verillaud: Writing review and editing, project administration, and conceptualization. Christian von Buchwald: Writing review and editing, project administration, and conceptualization. Gary L. Gallia: Writing review and editing, project administration, and conceptualization. Sacit Bulent Omay: Writing review and editing, project administration, and conceptualization. Warren Bennett: Writing review and editing, project administration, and conceptualization. Kumar Abhinav: Writing review and editing, project administration, and conceptualization. Garret Choby: Writing review and editing, project administration, and conceptualization. Jayakar Nayak: Writing review and editing, project administration, and conceptualization. Samuel C. Leong: conceptualization, validation, formal analysis, and project administration. Erich Vyskocil: conceptualization, validation, formal analysis, writing review and editing, visualization, and project administration. Matt Lechner: conceptualization, validation, formal analysis, Writing original draft, writing review and editing, visualization, project administration, and supervision.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ML is on the advisory board for the company Fusetec Inc. which provided the technology and Director of Innocrates Ltd.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Head and Neck Cancer Research Trust provided funding for the surgical equipment used for the study as part of the course. Fusetec Inc. supplied the 3D models for the validation process.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
