Abstract
Objectives/Hypothesis:
Systemic glucocorticoids (GC)s are employed to treat various voice disorders. However, GCs have varying pharmacodynamic properties with adverse effects ranging from changes in epithelial integrity, skeletal muscle catabolism, and altered body weight. We sought to characterize the acute temporal effects of systemic dexamethasone and methylprednisolone on vocal fold (VF) epithelial glucocorticoid receptor (GR) nuclear translocation, epithelial tight junction (ZO-1) expression, thyroarytenoid (TA) muscle fiber morphology, and body weight using an established pre-clinical model. We hypothesized dexamethasone and methylprednisolone will elicit changes in VF epithelial GR nuclear translocation, epithelial ZO-1 expression, TA muscle morphology, and body weight compared to placebo-treated controls.
Methods:
Forty-five New Zealand white rabbits received intramuscular injections of methylprednisolone (4.5 mg; n = 15), dexamethasone (450 µg; n = 15), or volume matched saline (n = 15) into the iliocostalis/longissimus muscle for 6 consecutive days. Vocal folds from 5 rabbits from each treatment group were harvested at 1-, 3-, or 7 days following the final injection and subjected to immunohistochemistry for ZO-1 and GR as well as TA muscle fiber cross-sectional area (CSA) measures.
Results:
Dexamethasone increased epithelial GR nuclear translocation and ZO-1 expression 1-day following injections compared to methylprednisolone (P = .024; P = .012). Dexamethasone and methylprednisolone increased TA CSA 1-day following injections (P = .011). Methylprednisolone decreased body weight 7 days following injections compared to controls (P = .004).
Conclusions:
Systemic dexamethasone may more efficiently activate GR in the VF epithelium with a lower risk of body weight loss, suggesting a role for more refined approaches to GC selection for laryngeal pathology.
Keywords
Introduction
Glucocorticoids (GCs) are commonly used to treat a wide variety of voice disorders due to their potent anti-inflammatory properties and hypothesized anti-fibrotic effects.1-3 Systemic GCs, including oral, intravenous, and/or intramuscular injection (IM), are associated with variable efficacy to treat laryngeal indications.1,4-11 Oral GCs are most commonly prescribed to treat rheumatologic laryngeal lesions and acute laryngitis. 1 Intravenous GCs are commonly employed to manage vocal fold (VF) and airway angioedema and laryngotracheobronchitis.1,12 Intramuscular GCs have demonstrated efficacy for laryngotracheobronchitis.4,5 Systemic GCs have also been reported to manage laryngeal sarcoidosis.9-11 Dexamethasone and methylprednisolone are the most frequently prescribed GCs in otolaryngology, 1 and drug bioavailability between oral and IM administration of these GCs is comparable.13-15 The mechanism of action of GCs is well-established in other tissues and disorders, and known to be initiated via intracellular binding of GC to the glucocorticoid receptor (GR).16-19 The GC-GR complex dissociates from chaperone proteins to regulate gene expression via 3 mechanisms: (1) direct binding to GC responsive elements on genomic DNA to promote anti-inflammatory protein production16,20; (2) indirectly through interactions between proteins and other transcription factors, mitigating inflammatory protein production17,20; and (3) via non-genomic pathways in the cytoplasm and at cell and organelle membranes.18,20-22
Although these immunomodulatory properties make GCs an attractive therapeutic option for treating laryngeal pathologies, it remains unclear how GCs induce physiologic changes within vocal fold (VF) tissue. Glucocorticoids vary in pharmacodynamic properties, including differences in drug potency, half-life, and mineralocorticoid activity. Glucocorticoids are also associated with adverse effects ranging from hypertension, hyperglycemia, 20 behavioral/cognitive changes,2,23 reduced therapeutic effect, 24 changes in tight junction/epithelial barrier function,25-28 and skeletal muscle catabolism.29,30 Glucocorticoid receptor nuclear translocation is an indicator of genomic steroid activation associated with therapeutic efficacy.31-34 Tight junction proteins, like ZO-1, contribute to epithelial cell barrier35,36 and protect the vocal folds from environmental pathogens 37 as well as phonotraumatic injury. 38 GC-induced skeletal muscle catabolism is also well documented in non-laryngeal skeletal muscle.30,39,40 Notably, reports of GC-induced VF atrophy are increasing. Mechanistically, GC injection into the VFs decreased hyaluronic acid synthesis in the lamina propria 41 and thyroarytenoid (TA) muscle fiber size. 42 Clinically, a single triamcinolone injection into the VFs was associated with VF atrophy in some patients. 43 Similarly, 5 to 6-weekly subepithelial dexamethasone injections into the VF were associated with VF atrophy in a retrospective case report. 44 Vocal fold atrophy can have profound detrimental effects on voice production.44-46
Despite known diversity of drug activity and associated adverse effects across GCs, clinical judgment was reported to drive GC selection rather than evidence of differential efficacy. 1 Characterization of GC-induced tissue changes in the VFs, and in particular, differential effects between GCs, is necessary to better understand the utility of GCs and to accurately customize drug selection for patient-specific needs. In the current study, we sought to characterize the temporal effects of acute systemic GC treatment on healthy VF tissue. Specifically, we investigated epithelial GR activation, tight junction expression, TA muscle morphology, and body weight. We hypothesized methylprednisolone and dexamethasone, 2 of the most prescribed GCs in otolaryngology, 1 will elicit changes in these tissue-level events. Information regarding the effects of GCs on healthy VF tissue will provide foundational insight regarding the utility of these compounds to improve clinical outcomes and mitigate potential adverse effects.
Methods
Preclinical GC Injections
This study was approved by the Vanderbilt Institutional Animal Care and Use Committee. Forty-five New Zealand white rabbits (Oryctolagus cuniculus) were weighed, and then 0.225 mL of 4.5 mg methylprednisolone (n = 15), 450 µg dexamethasone (n = 15), or volume matched saline (n = 15) was injected into the iliocostalis/longissimus muscle. This protocol was repeated for 6 consecutive days at consistent times. Doses were derived from the highest dosing ranges for GC IM injections: 1 to 9 mg for dexamethasone47-50,10 to 90 mg for methylprednisolone,51,52 assuming an average weight of 60 kg for humans and 3 kg for rabbits (eg, 4.5 mg methylprednisolone for 3 kg rabbit is analogous to 90 mg methylprednisolone for 60 kg human). Selected doses were based on clinical dosing ranges for IM dexamethasone and methylprednisolone and are not intended to be equivalent between the 2 GCs. Rabbits were randomized to treatment group.
After the 6 days injection protocol, 5 rabbits from each treatment group were weighed and euthanized at 1 day, 3 days, or 7 days. The larynges were harvested and bisected in the sagittal plane; the right half was formalin-fixed and paraffin processed, and the left half was fresh frozen in Optimal Cutting Temperature (OCT) medium. These timepoints were selected based on previous reports regarding GC-induced changes in tight junction expression26-28 and skeletal muscle morphology53-55 between 1 and 7 days following treatment.
Body Weight
Body weight was acquired immediately prior to the first injection and immediately prior to euthanasia at each timepoint using a Rice Lake VS-11 scale (Rice Lake, Wisconsin). Percent change from pre-injection weight and post-injection weight was calculated for each animal.
Immunofluorescence Labeling
Immunofluorescence labeling techniques were previously described by our group. 56 Briefly, OCT blocks were sectioned coronally at 12 µm using a Leica CM1900 cryostat and adhered to positively charged slides. Labeling was performed in triplicate on mid membranous VF sections. Samples were methanol fixed and permeabilized in 0.01% Triton X-100. Slides were antigen blocked in 10% goat serum (1 hour, 20°C) and incubated in mouse monoclonal primary antibodies against tight junction marker ZO-1 (Thermo Fisher Scientific; 1:250) and GR (Abcam; 1:250) in 1% goat serum overnight at 4°C. Secondary antibody detection was performed using Alexa Fluor® 594 Goat anti-Mouse IgG conjugate (Thermo Fisher Scientific) diluted 1:500 in 1% goat serum for 1 hour at room temperature in a darkened chamber. Samples were mounted with glass coverslips using Vectashield® Mounting Medium with DAPI (Vector Laboratories).
Hematoxylin and Eosin (H&E) Staining
Paraffin blocks were sectioned coronally at 10 µm using a Leica HistoCore BIOCUT microtome, floated in a Leica HI1210 heated water bath, and adhered to positively charged slides. Once dried, samples were deparaffinized using xylene and dehydrated in descending gradients of ethanol. Slides were stained with hematoxylin and counterstained with eosin performed in triplicate on midmembranous VF sections, followed by rehydration in an ascending gradient of ethanol. Following clarification with xylene, samples were mounted with glass coverslips and SecureMount™ low viscosity mounting medium.
Microscopy
All samples were imaged using a Nikon Eclipse 90i microscope. Immunofluorescence images were captured digitally using a Hamamatsu C10600 black and white camera and H&E images were captured digitally using a Nikon DS-Fi1 color camera. All images were captured at 10× magnification, to include the full depth of the VF including the epithelium, lamina propria (LP), and muscle fibers.
Image Analysis
ImageJ (National Institutes of Health) was employed to calculate epithelial GR nuclear translocation by a blinded rater. A region of interest (ROI) of the VF epithelium was acquired. “Otzu” thresholding, an algorithm to dichotomizes pixels into foreground and background, was used for DAPI (nuclei) labeled images. “Triangle” thresholding was also employed to differentiate foreground and background pixels in GR labeled images. A ratio was derived from positive GR pixels that overlapped with positive DAPI pixels, indicating GR nuclear translocation.
To calculate epithelial ZO-1 expression, images were magnified to 200% at a consistent, fixed region of the epithelium in all samples. A ROI of the epithelium was acquired, and “Moments” thresholding was used to identify ZO-1 positive pixels. This algorithm is better suited for automatic image segmentation based on ZO-1 expression. A ratio was calculated of positive ZO-1 pixels within the total epithelial ROI.
Thyroarytenoid muscle fiber cross-sectional area (CSA) is a standard technique used to assess skeletal muscle atrophy,39,40,54,57 and has been described previously by our group. 58 Briefly, a ROI outlining muscle fibers in H&E stained samples was acquired. “Default” thresholding was performed and the particle analysis tool was utilized to calculate total muscle fiber area in each sample. Individual muscle fibers were then manually counted using the multipoint tool and the average individual muscle fiber CSA for each image was calculated.
Statistical Analyses
All statistical analyses were calculated using GraphPad Prism V.7.0 (GraphPad Software, San Diego, California, USA) and employed non-parametric Kruskal-Wallis tests with Dunn’s multiple comparisons tests due to heterogeneity of variance between groups and/or non-normal distribution within conditions.
Results
Dexamethasone Increased Epithelial GR Nuclear Translocation
Dexamethasone significantly increased GR nuclear translocation compared to methylprednisolone 1-day following injections (H(2) = 7.46, P = .024; Figure 1). No differences were observed at the other time points. Intra-rater reliability was established using the correlation coefficient of 10 randomly selected and redundantly analyzed samples; excellent reliability was observed (r = .98; P < .01).
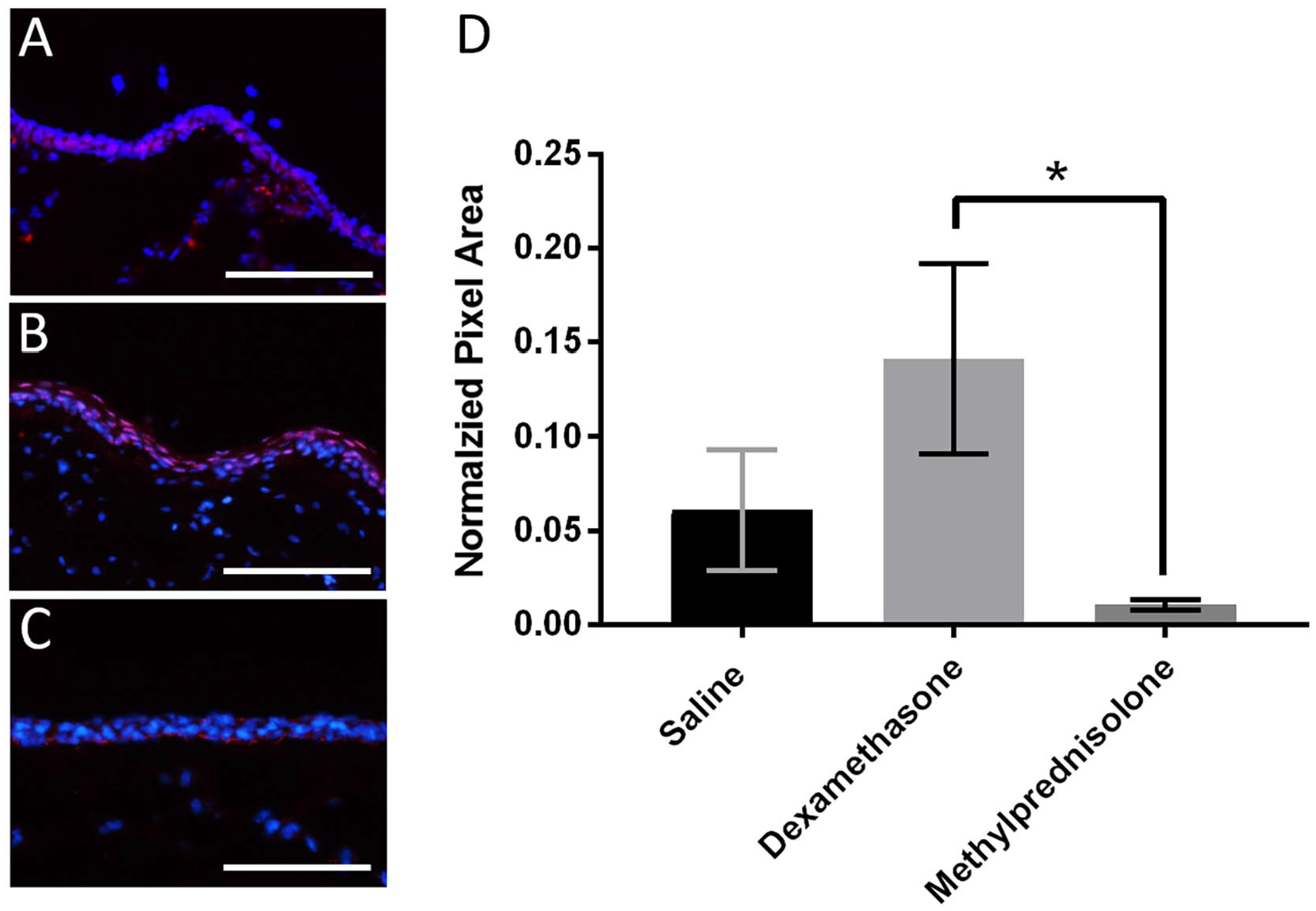
Epithelial GR nuclear translocation in (A) saline, (B) dexamethasone, and (C) methylprednisolone treated groups 1-day following injections. Graph (D) displays the mean and SEM across treatments.
Methylprednisolone Decreased Epithelial ZO-1 Expression
Methylprednisolone significantly decreased epithelial ZO-1 protein abundance compared to dexamethasone 1-day following injections (H(2) = 8.66, P = .012; Figure 2). No differences were observed at the other time points. Intra-rater reliability was established using the correlation coefficient of 10 randomly selected and redundantly analyzed samples; excellent reliability was observed (r = .97; P < .01).
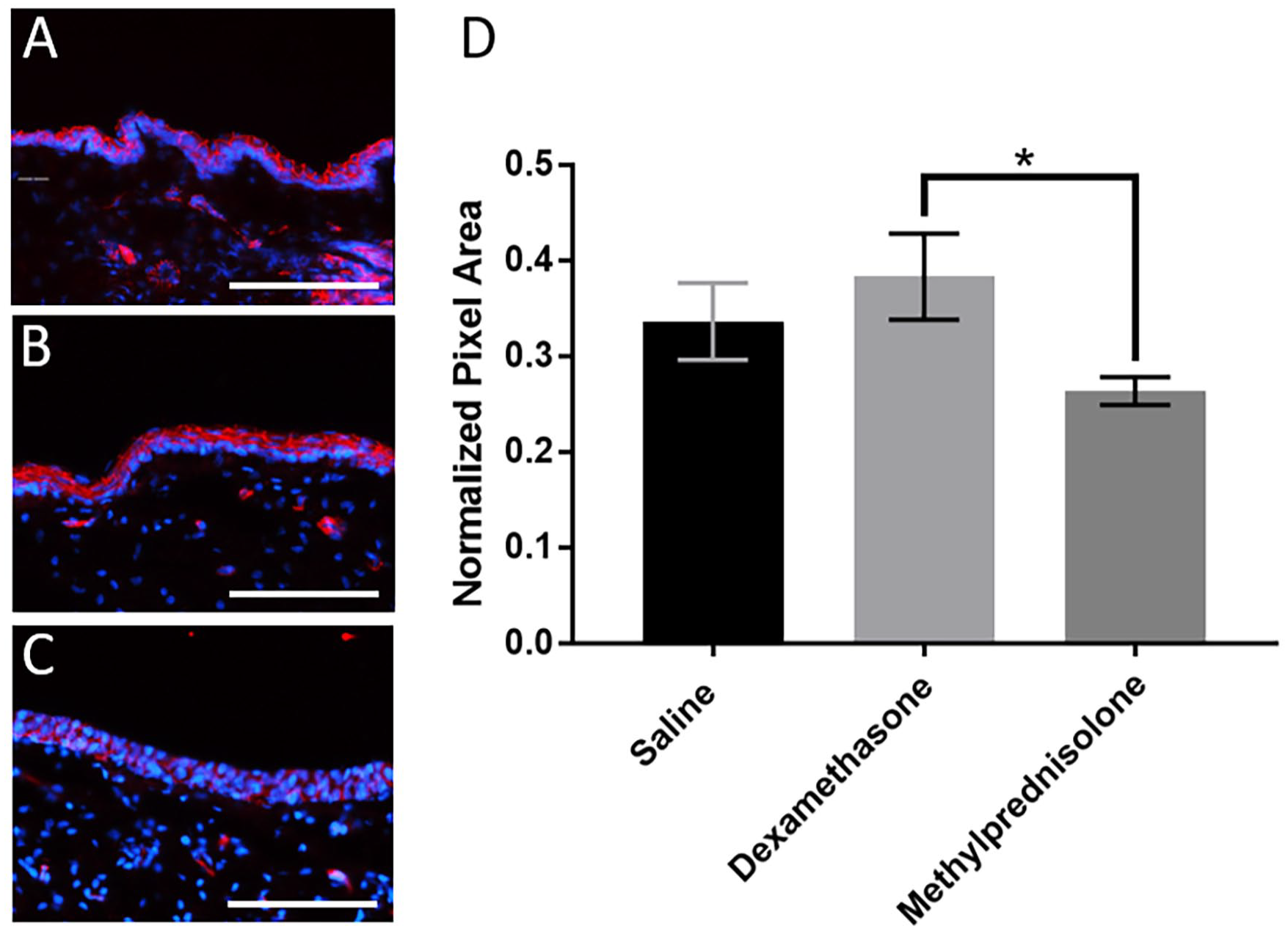
Epithelial ZO-1 fluorescence expression in (A) saline, (B) dexamethasone, and (C) methylprednisolone treated groups 1-day following injections. Graph (D) displays the mean and SEM across treatments.
Glucocorticoids Increased TA Muscle Fiber CSA
Dexamethasone and methylprednisolone significantly increased TA CSA compared to the controls 1-day following injections (H(2) = 8.91, P = .026; 0.029; Figure 3). No differences were observed between other time points or treatment conditions. Intra-rater reliability for quantifying muscle CSA was established using the correlation coefficient of 10 randomly selected and redundantly analyzed samples; excellent reliability (r = .96; P < .01) was observed.
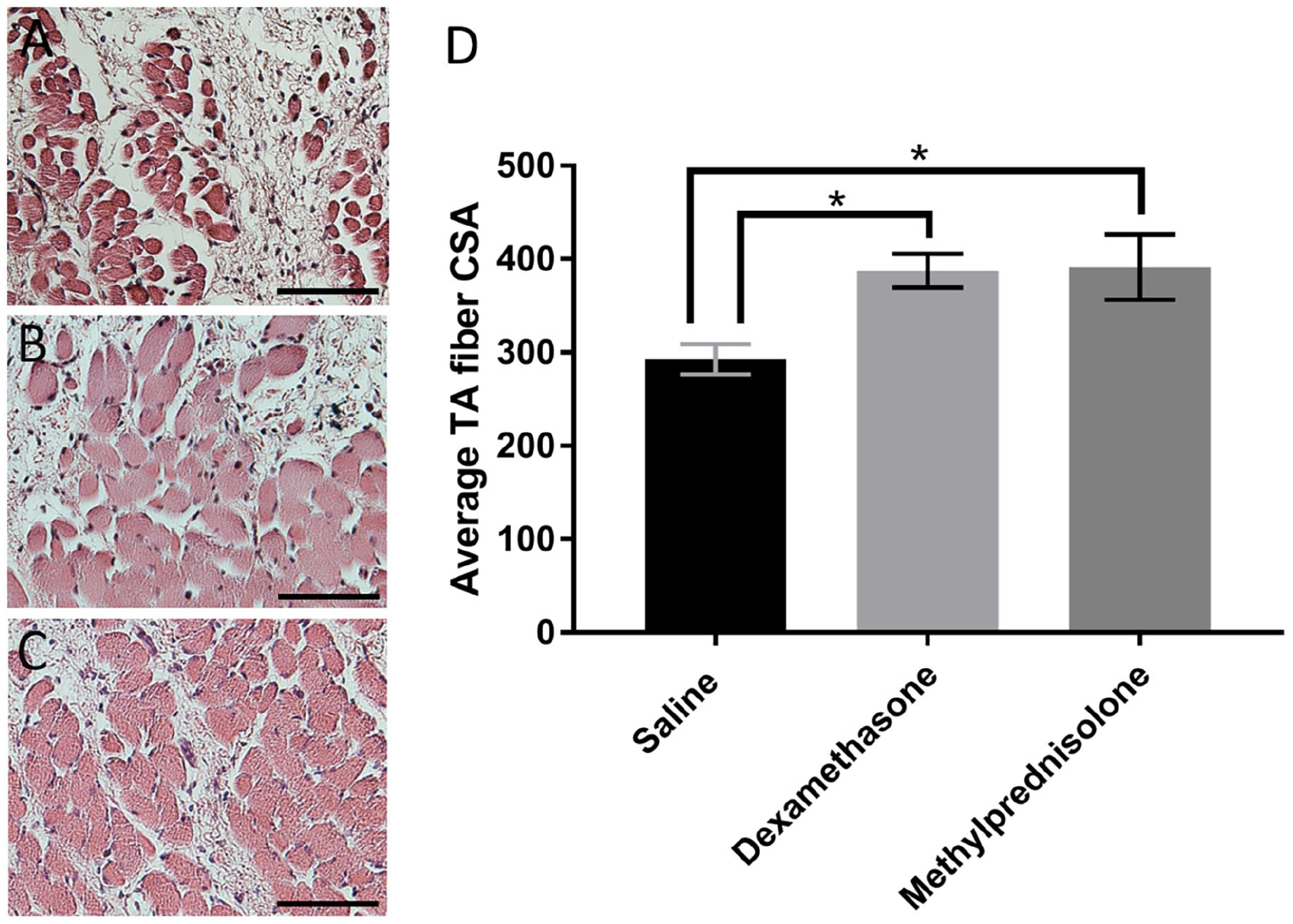
TA muscle fiber CSA in (A) saline, (B) dexamethasone, and (C) methylprednisolone treated groups 1-day following injections. Graph (D) displays the mean and SEM across treatments.
Methylprednisolone Decreased Body Weight
Methylprednisolone significantly decreased body weight 7 days post-injection compared to control (H(2) = 10.5, P = .004; Figure 4). No differences were observed at other time points.
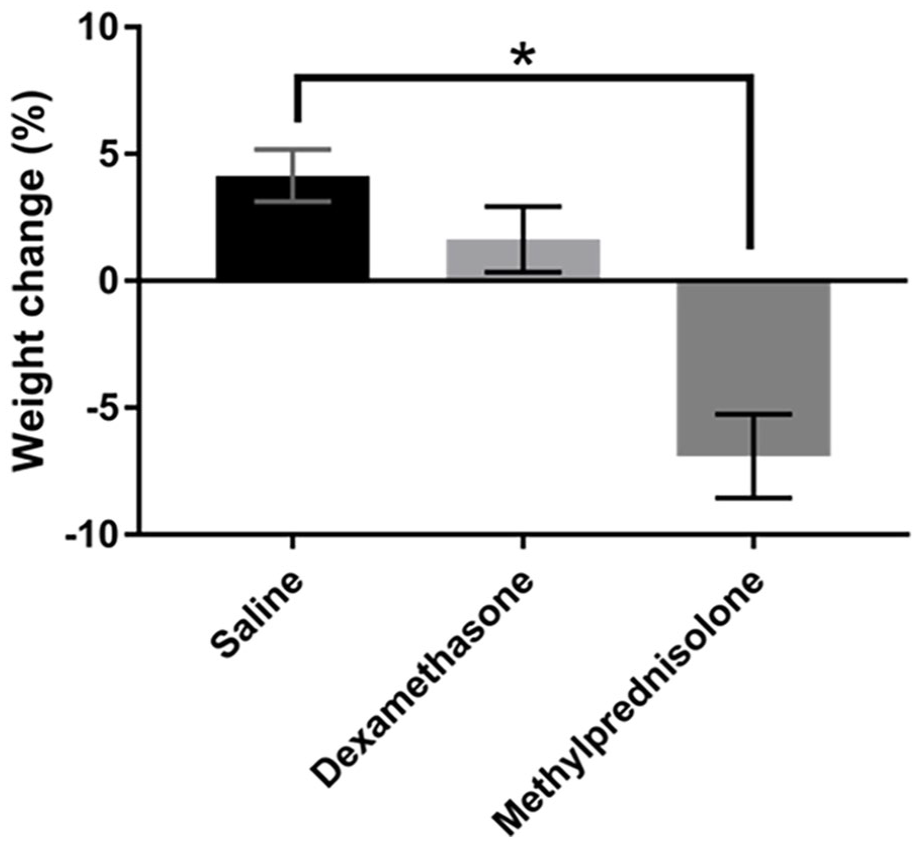
Changes in body weight in response to saline, dexamethasone, and methylprednisolone treated groups 7 days following injections. Graph displays the mean and SEM across treatments.
Discussion
Systemic GCs are commonly used to treat a wide variety of voice disorders. However, GCs have varying pharmacodynamic properties with adverse effects ranging from changes in epithelial tight junctions, skeletal muscle catabolism, and changes in body weight. It is critical to characterize the effects of GCs in healthy VF tissue to provide foundational insight regarding utility and optimal dosing of these compounds. In the current study, we sought to provide foundational data to, ultimately, improve clinical outcomes and mitigate potential adverse drug effects.
Glucocorticoid receptor nuclear translocation is associated with therapeutic response to GCs. 59 Dexamethasone significantly increased epithelial GR nuclear translocation compared to methylprednisolone 1-day following injections. However, GR nuclear translocation did not significantly differ between GCs and control group. No differences were observed between treatment conditions at the 3 and 7 days post-injection time points. Dexamethasone (36-72 hours) has a significantly longer biological half-life than methylprednisolone (12-36 hours). 60 This difference may, at least partially, explain these findings. Dexamethasone may have actively induced epithelial GR nuclear translocation 1 day following treatment, and this effect diminished by day 3. It is also unclear if methylprednisolone induced immediate GR nuclear translocation, but this induction diminished 1 day following injection. Repeated methylprednisolone injections may have decreased GR nuclear translocation due to tolerance and/or excessive stimulation of transcription factors interacting with GR,61,62 possibly related to the unique pharmacokinetic profile of methylprednisolone.
GR nuclear translocation in the VFs confirmed adequate concentration of systemic dexamethasone was delivered to the vocal folds to elicit a predictable response. Dexamethasone may more efficiently activate GR in the epithelium compared to methylprednisolone. Other extraneous factors such as sleep 63 and stress 64 have been shown to influence endogenous GC secretion. Although attempts were made to control for potential confounding variables, the inherent variability in the dexamethasone and saline conditions precludes meaningful statistical analyses. Further studies regarding GC effects on GR activation in in the context of laryngeal injury are required to interrogate the clinical efficiency of these drugs.
ZO-1, an epithelial tight junction protein, adheres adjacent cells to decrease paracellular permeability and maintain barrier integrity. 36 In the current study, GCs did not significantly alter ZO-1 expression compared to controls, and ZO-1 was distributed in the epithelial cell-cell junction across all treatment groups. Dexamethasone increased epithelial ZO-1 expression compared to methylprednisolone 1 day following injections. The translational value of this finding is unclear due to ZO-1 being present in the epithelium in all conditions, and no significant differences between GCs and controls. Dexamethasone has been reported to increase ZO-1 expression in other tissues.65-67 The effects of methylprednisolone on ZO-1 across tissue types is unknown, but other GCs have variable effects on epithelial permeability.25-28 Decreased ZO-1 expression is associated with increased endothelial and epithelial paracellular permeability.68,69 Increased ZO-1 may suggest systemic dexamethasone favorably impacted epithelial barrier function in the vocal folds, an under-investigated and favorable therapeutic effect of steroids given that barrier function is compromised in injury.70-72 Regardless, these data suggest systemic GCs do not acutely affect epithelial ZO-1 distribution.
Skeletal muscle atrophy an observed adverse effect of GCs. Many catabolic conditions result in muscle atrophy (eg, sepsis, cachexia, starvation, and metabolic acidosis) are associated with increased GC levels. 73 Additionally, GC induced skeletal muscle catabolism has been observed in vitro and in vivo.40,55,74 These catabolic effects are believed to be elicited, in part, via inhibitory effects on the IGF-I/PI3K/Akt/mTOR pathway and stimulatory effects on the Ubiquitin-Proteasome System (UPS).29,30 Surprisingly, dexamethasone and methylprednisolone increased TA muscle fiber CSA 1-day following systemic injection. Only a small subset of studies utilizing intracordal injections address GC induced atrophy in VF tissue, with little attention to the specific mechanism(s) involved, mucosa/muscle specificity, or dose and GC specificity.41,43,44 Increased TA muscle fiber CSA may be related to VF tissue specificity and/or influence of variables associated with systemic injections. However, these mechanistic hypotheses are speculative and require further investigation.
Methylprednisolone decreased body weight 7 days following injections. This finding may be due to the inhibitory effects of methylprednisolone on the endocrine system, specifically IGF-1.55,75 Dose-dependent, decreased body weight was reported in rats after repeated methylprednisolone administration, 76 which was postulated to result from decreased circulating IGF-I. Decreased body weight may also be related to reduced food intake, as GCs are reported to have mixed effects on appetite. Dose-dependent decrease in food intake and body weight were reported in rats77,78; contradictory data have also been reported.79-81 Rabbits in the current study had open access to food and associations between food intake, body weight, and GC treatment were not included in the current study.
Certainly, the uniqueness of rabbit VF structure and function may influence the generalizability of GC effects to human VFs. Although rabbits demonstrate similarities to humans in the context of VF tissue, microarchitectural properties, and extracellular matrix components, the rabbit only has 2 to 4 layers of epithelium and a 2-layered LP 82-84; humans have 5 to 10 epithelial layers and a tertiary intermediate LP layer.85,86 Additionally, although humans have both type I and II muscle fibers in the TA muscle, rabbits have been reported to only have type II muscle fibers, 87 which are believed to be less resistant to skeletal muscle atrophy. 88 Although no atrophy was observed in the current experiments, it is unclear whether different muscle fiber types have variable resistance to skeletal muscle atrophy induced by GCs. In that regard, “atrophy” in this study was defined as only a muscular finding, but atrophy is a multi-modal phenomenal observed to effect the VF lamina propria and epithelium.41,42 Additionally, GCs are typically administered for laryngeal indications that frequently involve an inflammatory response in the VFs or surrounding tissue. It is reasonable to believe that this inflammatory response, characterized by an influx of immune cells, the release of inflammatory mediators, and chemoattraction of other cell types, influences how GCs interact with the VF tissue. Uninjured VFs were employed in the current study as a starting point for mechanistic inquiry.
Conclusion
Repeated systemic GC treatment did not adversely affect VF epithelial tight junction ZO-1 or TA muscle morphology compared to control tissue. However, methylprednisolone decreased body weight. Additionally, VF epithelial GR nuclear translocation was less efficient in systemic methylprednisolone treatment than dexamethasone. These results suggest systemic dexamethasone may more efficiently activate GR in the VF epithelium with a lower risk of body weight loss. Although further investigation is needed, these data suggest differential systemic effects of GCs. This finding suggests a role for a refined approach to GC selection and administration for laryngeal disease. Future studies will interrogate similar outcomes in the context of VF injury and additional GCs, doses, duration, and timepoints to elucidate chronic steroid use and mimic clinical scenarios more closely.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by research supported by The University of Pittsburgh Clinical and Translational Science Institute (NIH/NCATS TL1-TR001858) and the NIH/NIDCD (R01DC015405 and R01DC017397).
