Abstract
Objective:
To examine the relationship between conferred immunity after standard pneumococcal series and refractory otolaryngologic infections in pediatric patients using post-vaccination antibody titers, and to identify contributory underlying conditions revealed when vaccination/re-vaccination fails to confer protective immunity.
Study Design:
IRB-reviewed and “exempt” retrospective case series with chart review using the Epic® Electronic Medical Record system from 2013 to 2021.
Setting:
Dedicated tertiary referral children’s hospital.
Methods:
Pneumococcal antibody titer results were assessed for children ages 0 to 21 years and: (1) at least 1 of 7 otolaryngologic disease diagnoses and (2) having received the 4-dose schedule of pneumococcal conjugate vaccine (PCV 7 or 13).
Results:
A total of 241 subjects met inclusion criteria with 356 laboratory tests. Recurrent acute otitis media, chronic rhinitis, and chronic otitis media with effusion were the 3 most frequent diagnoses. At presentation, only 27.0% of subjects had titers conferring immunity from their prior vaccinations with PCV. About 85 subjects had been subsequently revaccinated with Pneumococcal Polysaccharide Vaccine (PPSV), and antibody responses conferring immunity reached 91.8%. Seven subjects never developed adequate responses; 5 of these had recurrent acute otitis media as the primary otolaryngologic diagnosis. Secondary “revealed” diagnoses included Juvenile Rheumatoid Arthritis (n = 1), unresolved specific antibody deficiency (n = 2), and Hypogammaglobulinemia (n = 1).
Conclusion:
In pediatric patients with recurrent infectious otolaryngologic disease refractory to traditional medical and surgical therapy, inadequate responses to pneumococcal vaccination may be revealed. This correlation represents a potential pathway for diagnosis and therapy.
Keywords
Introduction
Streptococcus pneumoniae is the most common bacterial cause of sinus and middle ear infections. 1 The use of pneumococcal vaccination in the pediatric population has been common since the advent of heptavalent conjugated vaccine, and widespread vaccination has reduced the national burden of pneumococcal disease in children.2,3 Pneumococcal Conjugate Vaccines (PCV) are designed for the infant immune system by conjugating the antibody target onto an immunogenic protein carrier 3 that can trigger an immune response from the immature immune system. These immunizations are routinely administered in infancy as a 4-injection series. The Pneumococcal Polysaccharide Vaccine (PPSV) is a traditional vaccine with best responses by the “mature” immune system and is considered a “one-time” vaccination recommended for certain populations. 4 In addition to recommended uses, it is used by many Allergy/Immunology specialists to test humoral immune-responsiveness and as a booster dose for patients lacking pneumococcus-specific protective antibodies.4,5 Literature exists on revaccination effectiveness for children with chronic diseases such as asplenia but not for those with chronic otolaryngologic disease.6,7
Immunologic responses to vaccination vary between individuals and with age or vaccination type. Generally, vaccination responsiveness is highly effective: the CDC reports protective response rates against invasive pneumococcal disease as high as 97% in pediatric subjects who received PCV4,8 and 60% to 70% for PPSV. 4 Low vaccination titers have been associated with higher incidence of respiratory tract infections and allergic conditions. 9 Laboratory assessments of antibody titers are not routine for all otolaryngology patients, but testing is recommended when disease is refractory to traditional medical and surgical management or when Allergy/Immunology consultation is considered for refractory disease. 10
The primary goal of this exploratory study was to characterize the relationship between the existence of refractory (chronic or recurrent) otolaryngologic infectious disease and pneumococcal vaccination responsiveness in pediatric patients, as tested by titer measurements. Secondarily, the goal was to determine if those patients in whom PCV vaccination and PPSV re-vaccination failed to confer protective immunity had any explanatory underlying conditions revealed.
Methods
This study was designed as an IRB-review and exempt retrospective chart review using the Epic® Electronic Medical Record system. A database was constructed including all records for patients meeting subject inclusion criteria at Children’s Hospital New Orleans between 2013 and 2021:
(1) at least 1 of 7 otolaryngologic disease diagnoses: chronic otitis media with effusion, recurrent acute otitis media, chronic mastoiditis, chronic sinusitis, recurrent sinusitis, chronic rhinitis, and recurrent rhinitis (ICD-10 H65.3 [chronic otitis media], H65.07 [recurrent acute otitis media], H70.10 [chronic mastoiditis], J32.9 [chronic sinusitis], J01.91 [recurrent acute sinusitis], J31.0 [chronic rhinitis], J30.9 [allergic rhinitis, unspecified]);
(2) 14-serotype pneumococcal antibody titer laboratory data (CPT 86317(x14)) available for review; and
(3) completion of the scheduled series of PCV (PCV-7 or PCV-13) before the subject’s first titer draw (considered “at presentation”).
Subjects were excluded if:
(1) previously unvaccinated, previously diagnosed with an immunodeficiency, or if they received a PCV vaccination series during the titer collection period;
(2) given PPSV23 before their first titer measurement; or
(3) revaccinated with PPSV23 despite a protective titer.
Diagnosis criteria for chronic and recurrent otolaryngologic disease is standardized in the literature. Chronic otitis media with effusion is defined as persistent drainage of greater than 6 weeks. 1 Chronic sinusitis is characterized by symptom duration greater than 12 weeks (3 months). Recurrent rhinitis is defined as a recurrence of acute rhinitis of at least 4 times in 12 months with absence of symptoms between episodes. 1
Study subject data points included basic demographics (date of birth, sex, and race), relevant diagnoses, vaccination status and dates of vaccination, and 14-serotype titer measurements for each date of sample collection. For this study, “revaccination” was considered vaccination with PPSV23 after the original PCV dose(s) in infancy. Similar 14-serotype pneumococcal antibody titer data were gathered for revaccinated subjects.
Responses were then classified as “protective” versus “non-protective.” A “protective” response was considered to confer immunity based on age and percentage of “protective” serotypes as follows:
- A “protective serotype” was classified as a value greater than 1.5mg/mL.
- For patients less than 6 years old, a titer measurement was considered a “protective response” if at least 50% of serotypes were greater than 1.5 mg/mL.
- For patients greater than or equal to 6 years old, a titer measurement was considered “a protective response” if at least 70% of serotypes were greater than 1.5 mg/mL.
Measurement cutoffs were based on a compilation of published expert opinion regarding protective antibody levels against Streptococcus pneumonia.4,5,11,12,13 Consensus guidelines from the American Academy of Allergy, Asthma, & Immunology describe adequate responses for these vaccines as related to tests of humoral immunity; these reference points use similar age-related percentages but with a cutoff of 1.3 mg/mL. 12 Other studies use higher cutoff points to capture more clinically relevant responses, between 2 and 3 mg/mL.13,14 Since no true consensus on clinically relevant cutoffs were found in the literature, we combine these guidelines in the current study and use a cutoff of 1.5 mg/mL.
Results
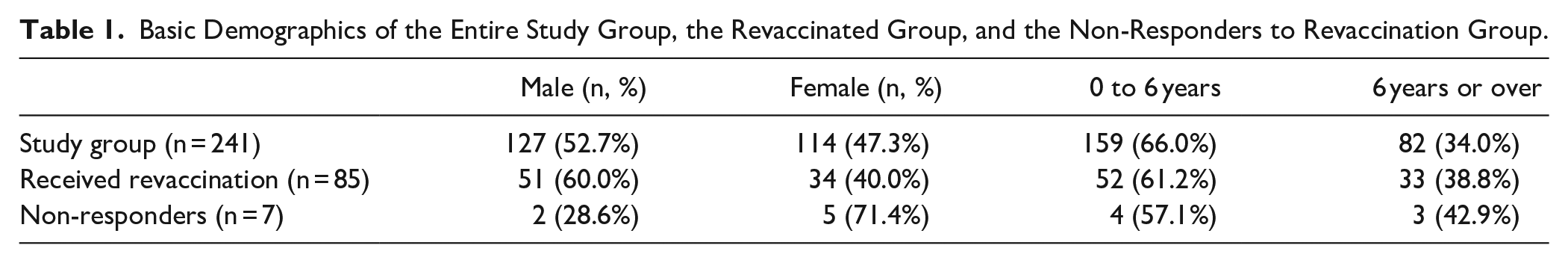
First pass identified 288 potential subjects with at least a single PCV vaccination, however 47 patients were excluded due to incomplete PCV vaccination (<4 doses). Final inclusion criteria were met by 241 subjects with 532 diagnoses, 356 laboratory tests, and 4871 individual serotype studies. A total of 169 subjects fell into more than 1 otolaryngologic diagnosis category (70.12%). Basic demographics are shown in Table 1. Age is categorized based on serotype level guidelines as described above. The overall mean number of PCV vaccinations received was 4.03 (SD 0.17). Recurrent acute otitis media was the most common diagnosis, representing 64.73% of subjects; chronic rhinitis (57.68%) was the second most common diagnosis (See Table 2).
Basic Demographics of the Entire Study Group, the Revaccinated Group, and the Non-Responders to Revaccination Group.
Distribution of the Otolaryngologic Diseases Amongst Study Subjects.
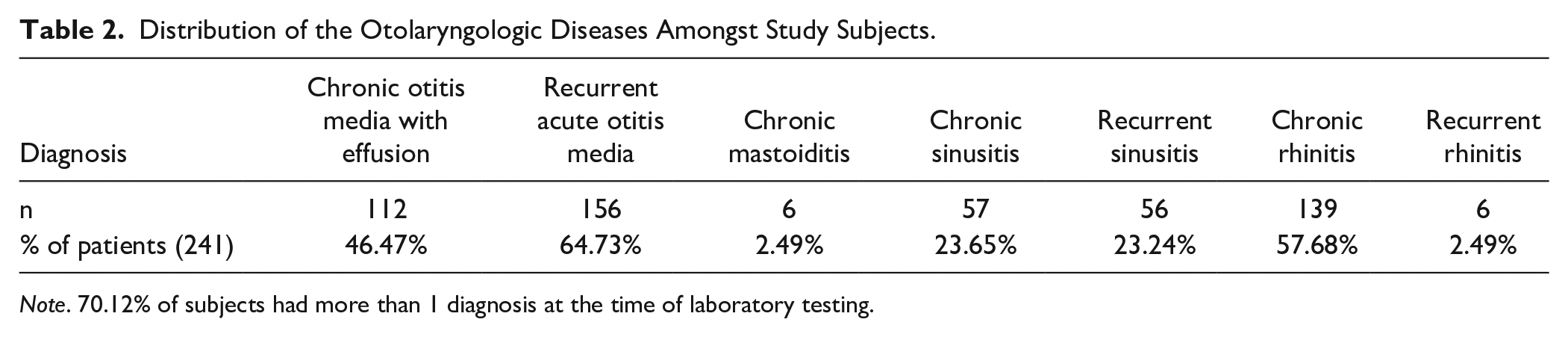
Note. 70.12% of subjects had more than 1 diagnosis at the time of laboratory testing.
At presentation, only 26.97% of all subjects were determined to have “protective responses,” conferring protective immunity, with the remaining 73.03% having “non-protective responses,” or titers that were considered inadequate to confer immunity.
Of the 241 original subjects, 85 had been revaccinated with PPSV23 (35.27%). Per exclusion criteria, all revaccinated subjects originally had titers consistent with “non-protective responses.” Titer measurements taken after revaccination with PPSV23 showed a “protective” response rate of 91.76% according to age-based criteria outlined above, with the other 8.24% of revaccinated subjects classified with “non-protective” responses.
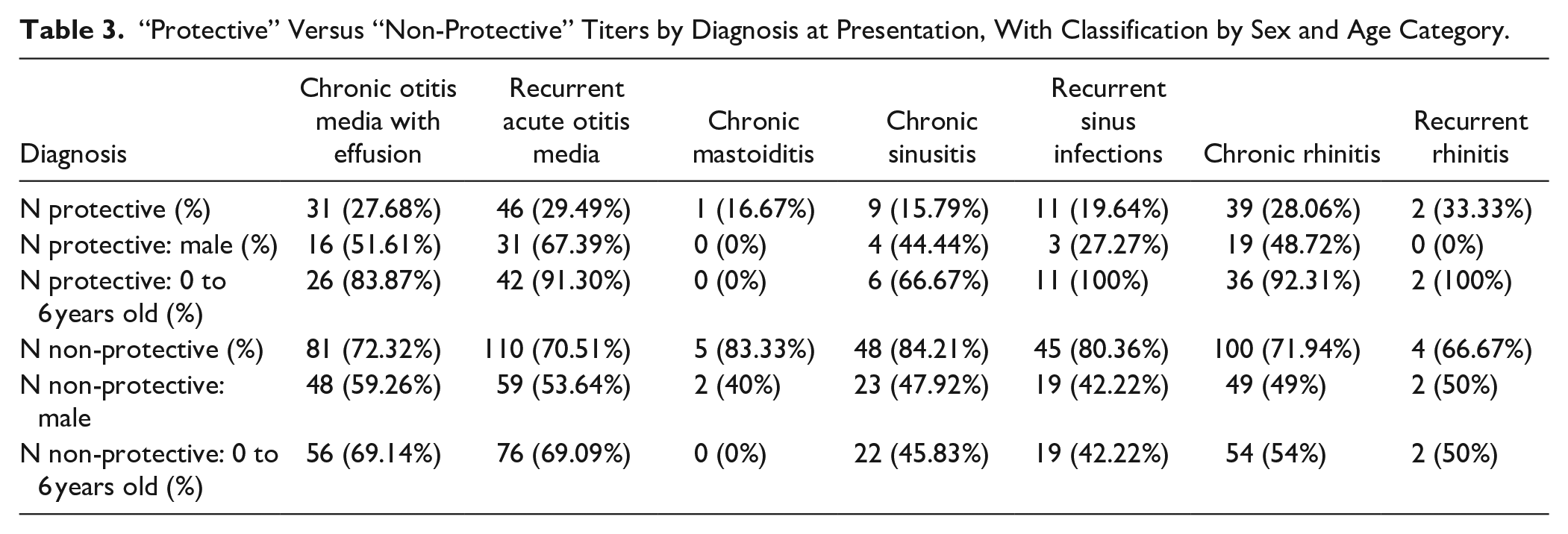
Pneumococcal antibody titer response was then analyzed by diagnosis. Responsiveness to primary vaccination, or antibody titer responses conferring immunity, for each disease category at presentation was measured. Percentages of subjects with “protective responses” conferring immunity are reported along with characterization by sex and age category. Chronic otitis media with effusion subjects demonstrated only 27.68% protective responses. Similarly, 29.49% for recurrent acute otitis media, 16.67% for chronic mastoiditis, 15.79% for chronic sinusitis, 19.64% for recurrent sinus infections, 28.06% for chronic rhinitis, and 33.33% for recurrent rhinitis (see Table 3).
“Protective” Versus “Non-Protective” Titers by Diagnosis at Presentation, With Classification by Sex and Age Category.
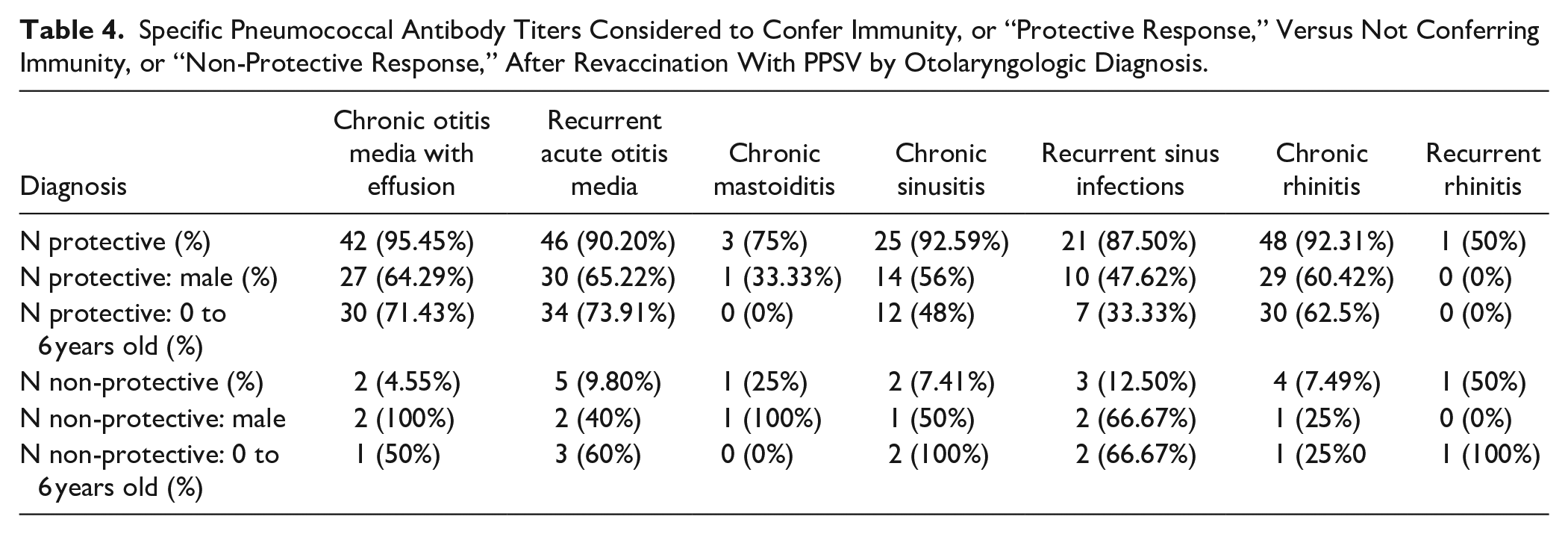
After revaccination with PPSV23, 95.45% of subjects with chronic otitis media with effusion had “protective responses;” similar observations were 90.20% for recurrent acute otitis media, 75% for chronic mastoiditis, 92.59% for chronic sinusitis, 87.5% for recurrent sinus infections, 92.31% for chronic rhinitis, and 50% for recurrent rhinitis. Data is again characterized by sex and age category (see Table 4).
Specific Pneumococcal Antibody Titers Considered to Confer Immunity, or “Protective Response,” Versus Not Conferring Immunity, or “Non-Protective Response,” After Revaccination With PPSV by Otolaryngologic Diagnosis.
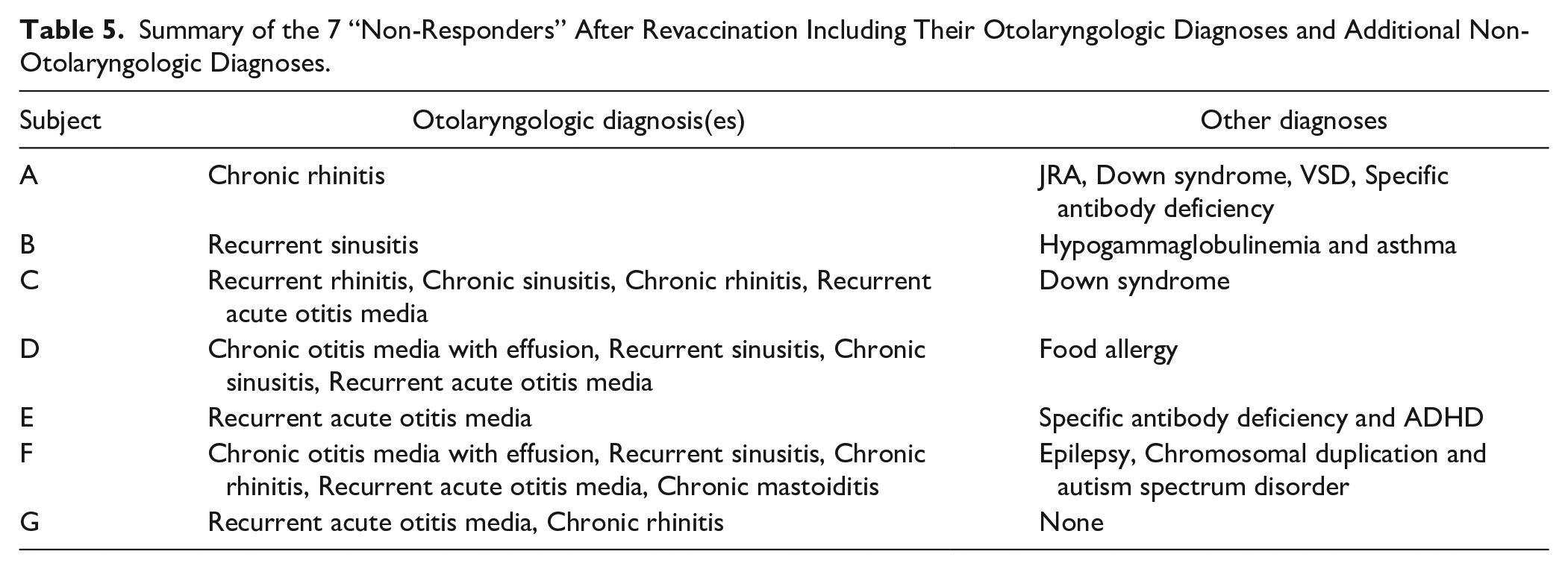
A total of 7 subjects were classified as “non-responders”, or those who did not develop a protective titer despite revaccination. Five of these subjects had a diagnosis of recurrent acute otitis media (71.43%). Four subjects had more than 1 disease diagnosis (57.14%). Six subjects had another non-otolaryngologic medical diagnosis (85.71%). Two subjects were male (28.57%) and 5 female (71.43%), 4 subjects were 0 to 6 years old (57.14%) and 3 were greater than 6 years old (42.86%), and all “non-responders” identified as White/Caucasian (100%). The otolaryngologic and non-otolaryngologic diagnoses of these subjects are shown in Table 5.
Summary of the 7 “Non-Responders” After Revaccination Including Their Otolaryngologic Diagnoses and Additional Non-Otolaryngologic Diagnoses.
Discussion
The current study was designed to examine the correlation between pediatric patients with 1 of 7 refractory infectious otolaryngologic disorders and pneumococcal vaccination responsiveness, defined as a response conferring immunity based on specific anti-pneumococcal antibody titer measurements at the time of “presentation” and in response to the standard 4-series PCV schedule. The 241 subjects included were not re-diagnosed, rather it was assumed that the specialty clinicians considered the disease to be refractory and beyond anticipated responsiveness to medical and surgical management. These results demonstrated that only 27% of symptomatic subjects had pneumococcal antibody titers conferring protective immunity at presentation. This percentage is low compared to the CDC-reported vaccine response rate for children receiving a conjugated pneumococcal vaccine of 97% efficacy against invasive disease. 4 After revaccination with PPSV, 92% of patients demonstrated pneumococcal antibody titers that correlated with conferred protective immunity. This represents a potentially clinically significant therapeutic response.
Clinical diagnosis at presentation did not appear to impact the potential to respond to revaccination. Lack of protective response was associated with underlying conditions for 3 of the 7 non-responders, yielding additional diagnoses potentially pertinent to clinical management.
Our data demonstrated the most common diagnoses to be recurrent acute otitis media followed by chronic rhinitis; these diagnoses were also the most common among subjects with “non-protective” responses. Chronic rhinitis does not have specific diagnostic criteria, in contrast to recurrent, subacute, and chronic sinusitis. It is also a difficult diagnosis to specifically link to a bacterial infectious etiology, as opposed to sinusitis.
Otitis, on the other hand, is known to be highly associated with S. pneumoniae infections and responsive to vaccination. Finnish studies demonstrated a 9% reduction in infections and a 25% reduction in requirements for tympanostomy tube placement after vaccination. 15 This association of refractory otitis after medical and surgical therapy with antibody titers too low to confer adequate immunity could represent a population of patients who could benefit from titer checks as part of medical management.
The dramatic responsiveness seen after PPSV23 vaccination similarly suggests that this is another tool for both diagnosis and therapy for this difficult population. This may hold true despite the concern for the rise of Haemophilus influenza in middle ear pathology.
Variations in response between diagnostic groups may be due to selection bias and small sample size, especially in certain diagnostic groups (ie, recurrent rhinitis = 6).
The current study was limited by its retrospective nature, including variability in medical management and diagnosis of individual patients as well as variability in chart records. Not all patients were revaccinated after a non-protective titer, which limited sample size. Additionally, no control groups were used for comparison purposes, as titers are currently not routinely ordered unless there is a specific immunologic concern.
For this preliminary exploratory study, data was not collected on clinical outcomes of patients after revaccination. Future studies will focus on age-matched controls and clinical outcomes. The minimum serum antibody concentration necessary for protection against invasive pneumococcal disease or against pneumococcal otitis media has not been determined for any serotype, as discussed above.13,14,15 The vaccination response rates published for FDA approval of the vaccinations are specifically for prevention of invasive disease but not perfectly comparable to our analysis. Future studies may investigate these items further.
Variabilities in age and waning titers due to age are also important possible confounding variables to consider for future studies. This study included patients with a specific set of vaccination criteria but did not account for time from most recent vaccination. Further studies could consider other vaccination combinations or inclusion/exclusion criteria.
The total prevalence of specimens positive for Streptococcus pneumoniae at this institution is not known. Additionally, the incidence of invasive disease such as pneumonia or meningitis in this subject population is not available secondary to chart review limitations.
By design, it was outside the scope of this study to determine whether vaccination response determined future outcomes for patients; rather the current goal was to only determine if any correlation existed between refractory otolaryngologic infectious disease and vaccine responsiveness. This remains an important area of investigation, particularly in the modern era wherein vaccination may be refused but highly concentrated unvaccinated children may increase risk of transmission of infectious disease, that is large daycare utilization and participation in public education with increasing frequencies of vaccination exemptions.
Conclusion
Pneumococcal antibody titer measurement in patients presenting with refractory otolaryngologic disease can reveal poor response to PCV, possibly contributing to the refractory nature of the disease. Revaccination with PPSV for patients with non-protective titers may be of diagnostic and therapeutic relevance, and further studies should explore this hypothesis further.
Footnotes
Acknowledgements
The authors would like to acknowledge the contributions of Dr. Joel Oubre and David Thompson for their contributions toward fundamental data collection.
Authors’ Note
The authors attest that this is original work, not under consideration for publication at any other journal. The work in overview was presented at the 49th Society for Ear, Nose and Throat Advances in Children (SENTAC) in Phoenix, Arizona December 4, 2021 and upon further review at the 12th International Symposium on Pneumococcus and Pneumococcal Diseases, in Toronto, Canada, June 23 to 26, 2022.
Author Contributions
Dr. Evans fundamentally designed this project, and Dr. Bonaventure performed the data analysis and re-analysis after review, to refine the project. The manuscript was developed by Drs. Bonaventure and Evans, and editing was accomplished by both.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
