Abstract
Objectives:
The present study was performed to determine whether the inhalation of carboxymethyl (CM)-chitosan can alleviate tracheal fibrosis in a rabbit model.
Methods:
We designed a rabbit model of tracheal stenosis involving electrocoagulation with a spherical electrode. Twenty New Zealand white rabbits were randomly divided into experimental and control groups (10 animals each). Tracheal damage was successfully established by electrocoagulation in all animals. The experimental group was given CM-chitosan (inhalation for 28 days), while the control group inhaled saline. The effects of CM-chitosan inhalation on tracheal fibrosis were analyzed. Laryngoscopy was performed to evaluate and grade tracheal granulation, while tracheal fibrosis was evaluated by histological examination. The effects of CM-chitosan inhalation on the tracheal mucosa were examined by scanning electron microscopy (SEM), and hydroxyproline content in tracheal scar tissue was determined by enzyme-linked immunosorbent assay (ELISA).
Results:
Laryngoscopy showed that the tracheal cross-sectional area was smaller in the experimental than control group. The amounts of loose connective tissue and damaged cartilage, as well as the severity of collagen and fibrosis, decreased following inhalation of CM-chitosan. According to the ELISA, the experimental group had low levels of hydroxyproline in the tracheal scar tissue.
Conclusion:
The findings presented here showed that inhalation of CM-chitosan mitigated posttraumatic tracheal fibrosis in a rabbit model, thus suggesting a potential new treatment for tracheal stenosis.
Introduction and Background
Both benign and malignant diseases of the tracheal airway are significant causes of morbidity and mortality associated with tracheal obstruction. Nonmalignant airway stenosis is mainly associated with iatrogenic complications. Such causes of tracheal trauma or disease, which may result in cartilage injury, mucous membrane rupture, or hematoma formation, followed by fibrous connective tissue and scar tissue repair, ultimately lead to the development of tracheal stenosis. 1 Treatment of tracheal stenosis is still very difficult. However, surgical treatment is now in a mature stage and drug treatment can prevent tracheal stenosis at an early stage. 2
Fibrosis is a frequently observed wound-healing reaction that promotes restoration of tissue integrity. However, it can induce tracheal stenosis, and may ultimately lead to organ dysfunction and death. Granulomas grow around the edges and fill the wound, and the fibroblasts produce collagen fibers for 5 to 6 days after injury. With the increase in collagen fibers, keloid is fully formed about 1 month after injury. Therefore, earlier suppression or slowing of these pathological phenomena represents an effective strategy to prevent or reduce fibrosis.
TGF-β1 stimulates the expression of fibronectin, collagen, and extracellular matrix proteins, which may be involved in many fibrotic processes. 3 There is accumulating evidence that overexpression of TGF-β1, as a master regulator of extracellular matrix and collagen accumulation, results in hypertrophic scar formation.4,5 In this study, we evaluated the expression of TGF-β1 in tracheal scar tissue following drug intervention to determine whether it plays a key role in the progression of fibrosis. Inhalation of CM-chitosan was associated with reduced TGF-β expression in tracheal scar tissue, suggesting that it may prevent tracheal fibrosis and scar formation.
In recent decades, chitosan derivatives have attracted a great deal of attention in medicine and biotechnology. Carboxymethyl chitosan (CM-chitosan) is a water-soluble polysaccharide derived from chitosan by carboxymethylation characterized by high biodegradability and histocompatibility; it is effective for hemostasis, pain relief, and the inhibition of microorganism growth. In addition, CM-chitosan can promote the growth of epithelial cells and inhibit fibroblast proliferation and migration, thus inhibiting scar tissue formation and accelerating wound healing.6-9 Moreover, CM-chitosan significantly inhibited the expression of type I collagen in keloid fibroblasts.7,9 To some extent, the effects of a drug depend on its mode of administration. Experimental evidence has shown that most of the inhaled drug remains in the laryngotracheal lumen, with only 10% to 20% reaching the lungs. 10 Here, we hypothesized that CM-chitosan inhalation may have value for the treatment of posttraumatic tracheal fibrosis. To test this hypothesis, we explored the therapeutic efficacy of CM-chitosan inhalation in an electrocoagulation-induced tracheal stenosis rabbit model.
Materials and Methods
Animal Experiments
Twenty New Zealand White rabbits of both sexes weighing 2.12 to 2.42 kg (mean weight, 2.27 kg) were allowed to acclimatize to the experimental animal center of Naval Medical University for at least 1 week, to aid manipulation and postoperative nursing care (ie, inhalation and checking of the incision). The study was approved by the Ethics Committee of Changhai Hospital and local governmental authorities (SCXK (Hu) 2012-0007), and was performed in accordance with the European Guidelines for Animal Experimentation (directive 2010/63). All rabbits were randomly divided into experimental and control groups (n = 10 per group) .Penicillin was administered intraperitoneally as prophylaxis against infection.
Rabbits were anesthetized with intraperitoneal chloral hydrate at a dose of 5 mg/ml. Anesthesia was maintained by inhalation of low-concentration isoflurane. The back and anterior neck area were shaved. The animals were placed in the supine position and held in place with bandages.
The larynx and trachea were electrocoagulated for 5 seconds using a 3-mm spherical electrode (20 W) after tracheotomy in the seventh to eighth tracheal rings (Figure 1A and B). Each animal was observed over 4 weeks, with nebulizer therapy performed for 15 minutes daily at the end of this period. The aerosols were passed into the oral-nasal inhalation system via a compression pump atomizer (NE-C801; Omron Ltd., Kyoto, Japan), which is used to separate liquid drugs into particles <5.5 μm (Figure 1C). Animals in the experimental group inhaled CM-chitosan aerosol at a concentration of 5 mg/ml [3 ml for 15 min once per day on postoperative days (PODs) 1-28]. Animals in the control group inhaled 3 ml of normal saline for 15 minutes once a day on PODs 1 to 28 (Figure 1C). To accurately determine the effects of CM-chitosan, the doses were physiologically and clinically relevant, based on regulatory guidelines. 11 All rabbits were euthanized as scheduled on POD 28 by an overdose of chloral hydrate.
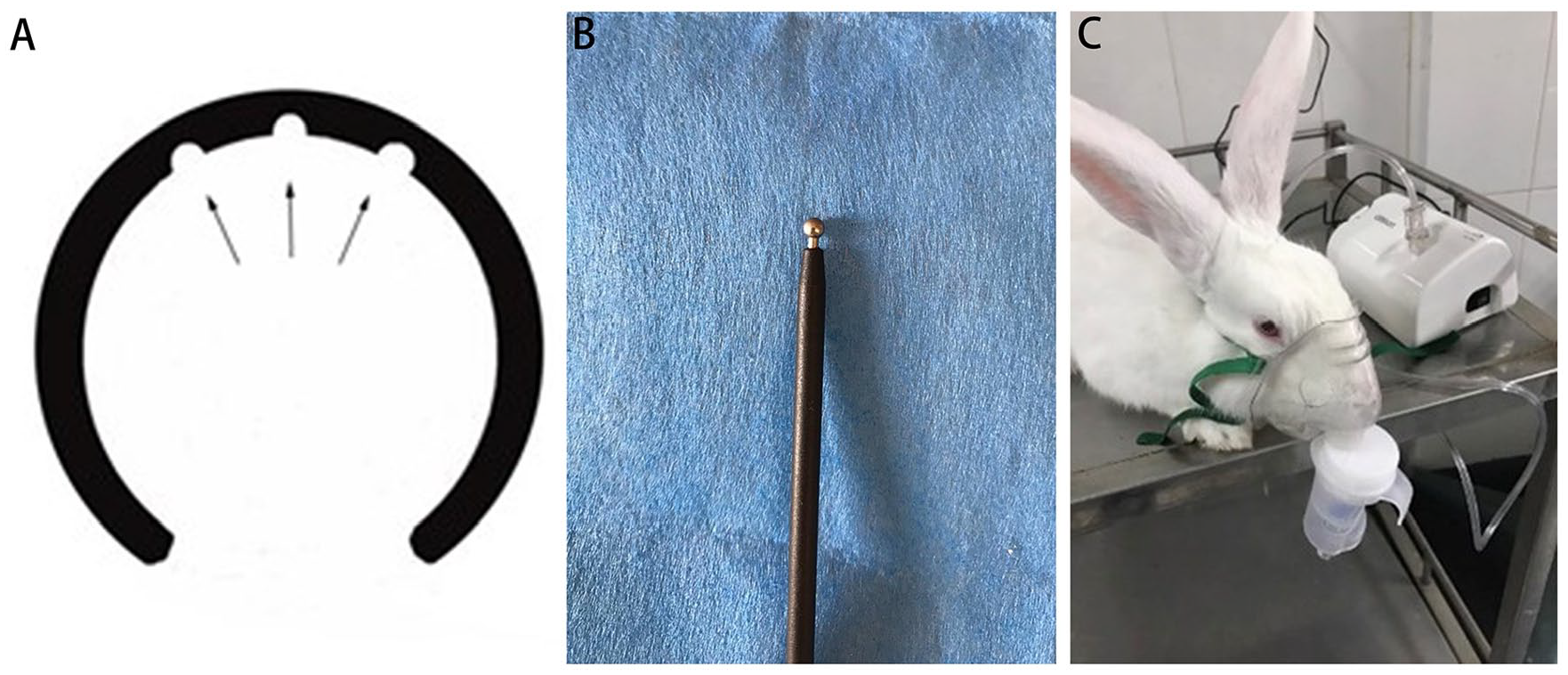
(A) Schematic diagram of the area of tracheal damage (2π × 32 mm) according to cauterization in the cervical trachea. (B) Electrocoagulation. (C) The nebulizer used in the experiments (NE-C801; Omron Ltd., Kyoto, Japan).
Fiberoptic Endoscopic Evaluation and CT
Fiberoptic endoscopic evaluation (EV-N; XION GmbH, Berlin, Germany) and computed tomography (CT; SSC-64X; Siemens, Erlangen, Germany) was used to measure tracheal mucosal granulation and the diameter of the tracheal lumen. The laryngoscope was placed on a “tracheal stenosis plane” created on the fifth tracheal ring by electrocoagulation to ensure accuracy of the collected images. The degree of stenosis was calculated in pixels as follows: (Original area − Tracheal stenosis plane)/Original area × 100%. The original tracheal cavity area and that during postoperative examination were calculated using histograms created in Photoshop (Adobe Systems Inc., San Jose, CA, USA). The CT scanning layer thickness was 1 mm.
Histological Examination
Tracheal tissues were embedded in paraffin, cut into 4-μm-thick sections, and stained with hematoxylin and eosin (H&E) and Masson’s trichrome. The sections were examined for morphological and structural alterations under an orthotopic light microscope (ECLIPSE Ci; Nikon, Tokyo, Japan).
Enzyme-Linked Immunosorbent assay (ELISA)
Tracheal specimens were weighed and hydrolyzed, and hydroxyproline content was determined by ELISA using commercial kits (A030-2; Nanjing Jincheng Bioengineering Institute, Nanjing, China; and SEA124Rb; USCN, Wuhan, China) according to the manufacturer protocols. Concentrations were calculated from a standard curve.
Scanning Electron Microscopy
For precise observation of ciliary beating, tracheal specimens were examined by scanning electron microscopy (SEM) (JEOL-6380LV; JEOL, Tokyo, Japan).
Immunofluorescence Staining
Paraffin-embedded tracheal tissue specimens were deparaffinized and rehydrated in xylene prior to antigen retrieval, blocked in 3% bovine serum albumin (BSA) at room temperature for 30 minutes, and then incubated with primary antibody (GB13179; Servicebio, Wuhan, China) at 4°C overnight. Specimens were then incubated with appropriate secondary antibody (GB25303; Servicebio) for 50 minutes at room temperature. The nucleus was counterstained with DAPI (G1012; Servicebio) in phosphate-buffered saline for 5 minutes at room temperature. Tissue sections were scanned with an inverted microscope (ECLIPSE Ti-SR; Nikon) and images were analyzed with the Nikon DS-U3 imaging system.
Statistical Analysis
The experimental results were subjected to analysis of variance (ANOVA) performed using SPSS for Windows software (ver. 22.0; SPSS Inc., Chicago, IL, USA). The results are presented as the mean ± SD. In all analyses, P < .05 was taken to indicate statistical significance.
Results
Animal Model Construction and Drug Intervention
The animals were sacrificed, laryngotracheal tissues and viscera were dissected out, and histomorphological examination was performed. Fourteen days after model construction, the median tracheal stenosis was 14.40% in the experimental group and 30.39% in the control group (Figure 2A). The rabbits in the experimental group showed less airway blockage compared with the control group (Figure 2A). In the experimental group, endoscopy revealed less granulation tissue on the luminal surface and reduced airway stenosis compared to the control group. Fiberoptic endoscopic examination on POD 28 showed more serious scar contracture of the luminal surface and a greater region of stenosis in the control group (Figure 2A). The median percentage of tracheal stenosis was 27.28% in the experimental group and 54.26% in the control group (Figure 2A). On POD 28, CT showed slight airway blockage in the experimental group (Figure 2C). The diameter of the tracheal lumen between the seventh and eighth cartilage rings was significantly greater in the experimental group (P < .05; Figure 2C). These results demonstrated the accuracy of the fiberoptic endoscopic technique.
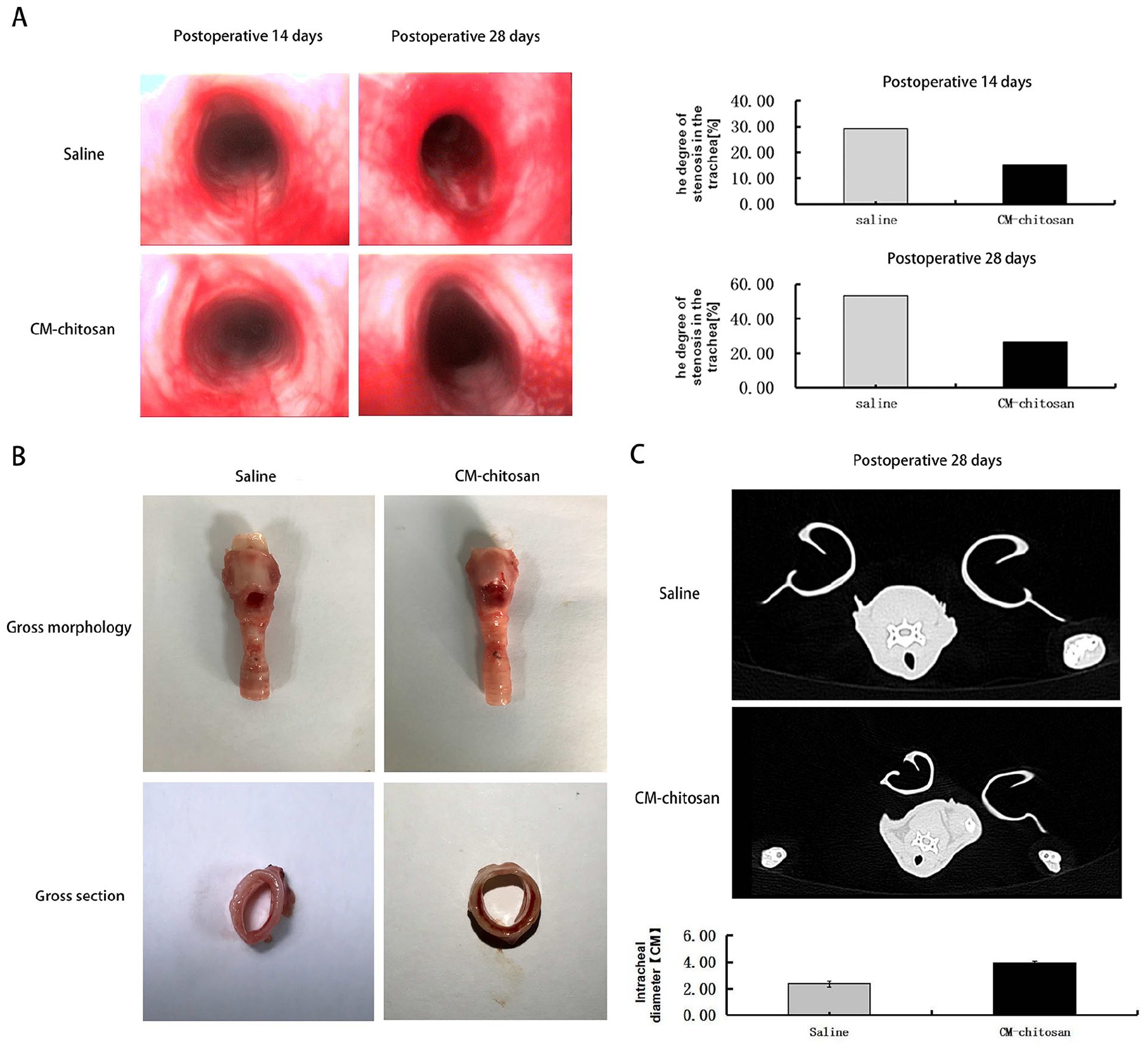
(A) Endoscopic findings of the tracheal mucosa and difference in tracheal stenosis between groups and time points. Bar graphs show the group differences in tracheal stenosis. The experimental group showed decreased tracheal stenosis compared to the control group. (B) Gross morphology and cross-sections of the trachea in both groups. (C) CT of the control group (first line) and experimental group (second line). Bar graphs show the group differences in tracheal lumen diameter. Data are presented as the mean ± SEM. P < .05 was taken to indicate statistical significance.
H&E Staining and Masson’s Trichrome Staining
The scar tissue of rabbits from the experimental and control groups were dissected out for histological examination with H&E and Masson’s trichrome staining. Mild inflammatory changes were seen in all specimens. In the control group, the scar tissue showed proliferation of fibroblasts and thickened collagen fibers, and the damaged cartilage was contracted by collagen fibers. The experimental group with CM-chitosan inhalation showed thinner collagen fibers and fewer fibroblasts in the cauterized lesions (Figure 3).
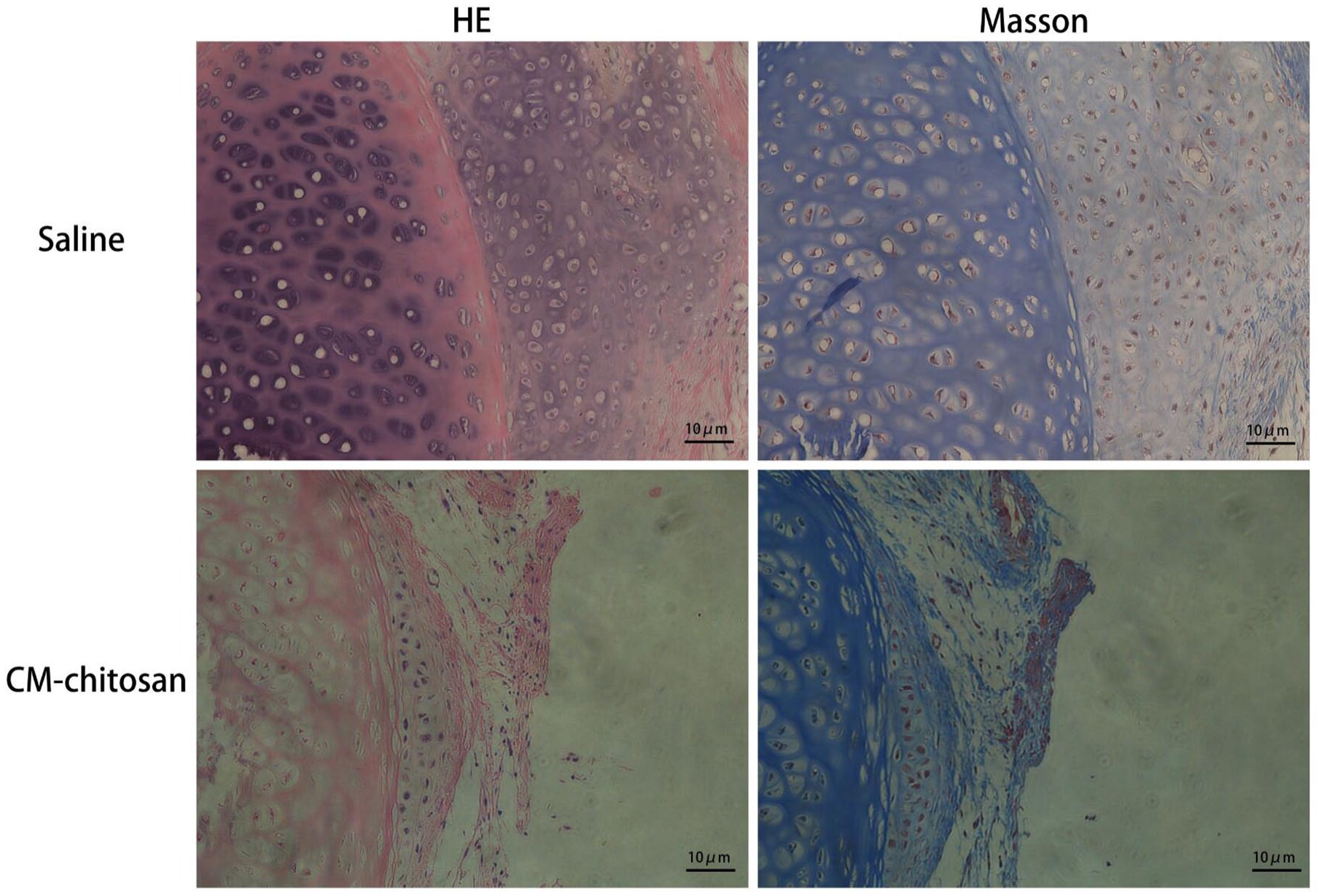
Photomicrographs of control and experimental group specimens with H&E and Masson’s trichrome staining (scale bars, 10 μm).
Hydroxyproline Determination
The experimental group showed significantly reduced hydroxyproline content in scar tissue on POD 28 compared with the control group (P < .05; Figure 4).
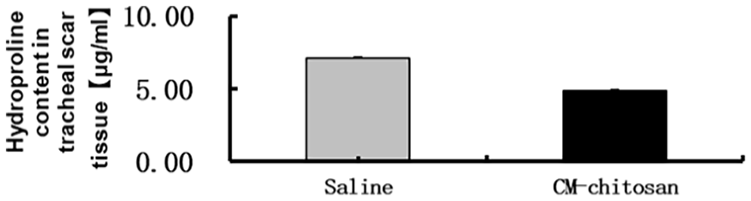
Group differences in hydroxyproline content. Data are shown as the mean ± SEM. P < .05 was taken to indicate statistical significance.
Scanning Electron Microscopy
On SEM examination of the tracheal rings on POD 28, ciliated and non-ciliated cells in the luminal surface were similar in appearance and number between the experimental group with CM-chitosan inhalation and saline-treated control group. Both groups showed a similar dense and orderly distribution of cilia on the whole mucosal surface (Figure 5).
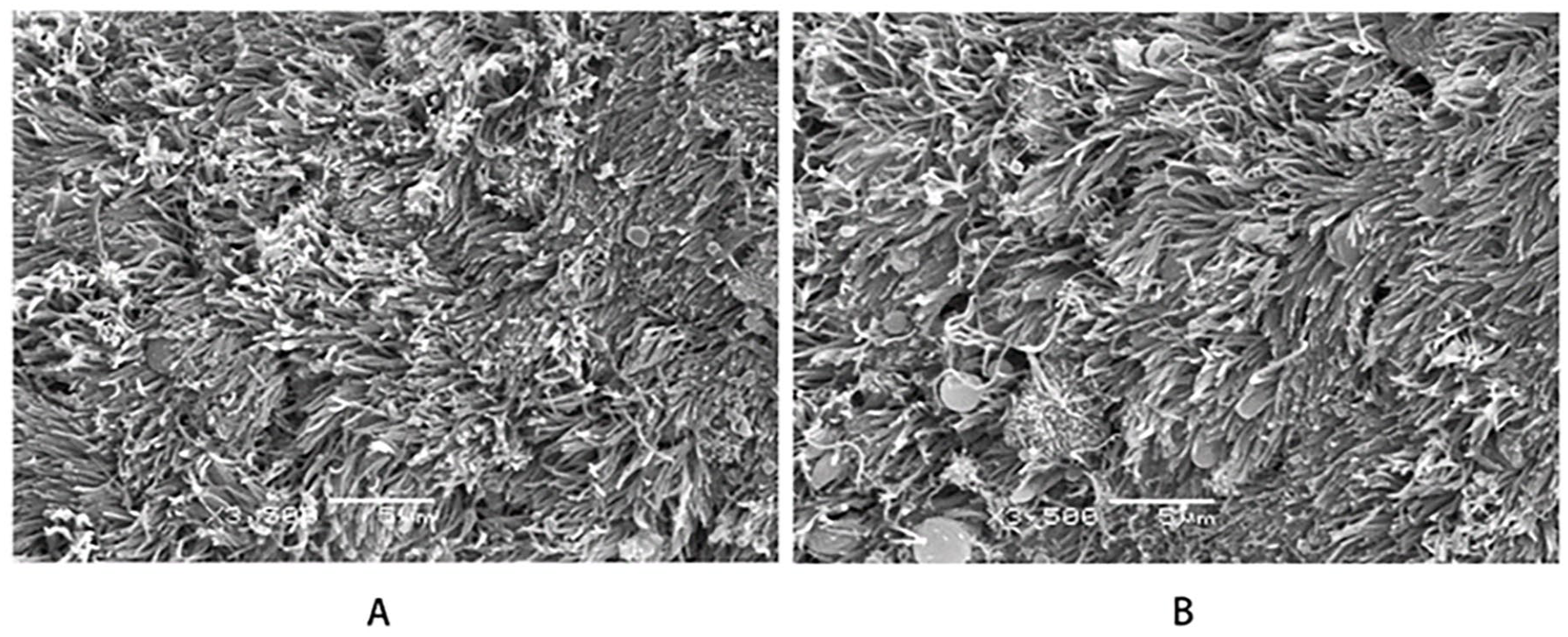
SEM examination of the tracheal epithelium. Micrograph of the tracheal epithelium on POD 28 with aspiration of saline (control group) or CM-chitosan (experimental group) (original magnification: ×3,500). (A) Saline. (B) CM-Chitosan.
TGF-β1 Expression
The level of TGF-β1 expression in the tracheal scar tissue was determined by ELISA in both groups. The TGF-β1 expression in the scar tissue was significantly decreased in the experimental group compared to the control group (P < .05; Figure 6B). Fluorescence staining showed that TGF-β1 was mainly present on the cell membrane and in the cytoplasm. The expression of TGF-β1 in the tracheal scar tissue was significantly suppressed in the experimental group treated with CM-chitosan compared with the control group (Figure 6A).
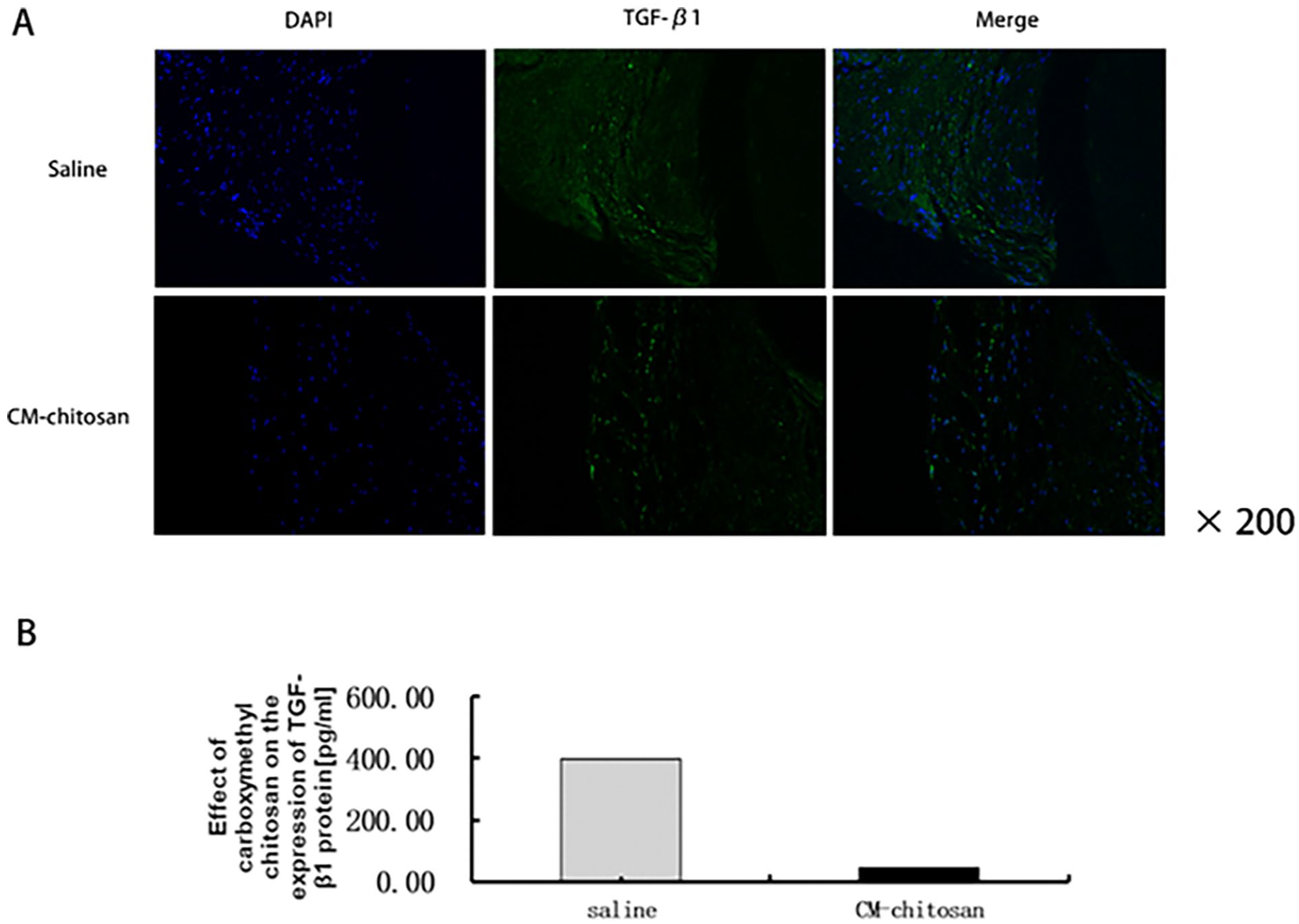
(A) Photomicrographs showing immunolocalization of TGF-β1 in the tracheal scar tissue in both groups (original magnification ×200, scale bar = 100 μm). (B) TGF-β1 expression in both groups. Data are shown as the mean ± SEM. P < 0.05 was taken to indicate statistical significance.
Discussion
Tracheal stenosis is a common symptom of a number of benign and malignant diseases that requires urgent attention. The benign diseases include iatrogenic complications and trauma. A number of studies have provided interesting perspectives regarding treatment strategies for iatrogenic complications after endotracheal intubation or tracheal tube insertion leading to airway obstruction.12-14 Wagner et al 15 demonstrated that the outcome of keloid following burns was different from that developing after mechanical trauma. However, there has been little research on traumatic complications related to thermal burns and blast injuries that result in severe tracheal stenosis. Thermal injury has been used to cause tracheal stenosis in animal models.16-18 Some studies have established tracheal stenosis models by thermal injury methods such as laser and radio frequency. Although it has the advantages of accuracy, quantification and repeatability, it also has many disadvantages, such as the complexity of the operation process, and the small area of burning may not be enough to form scar and granulation tissue proliferation. Through burning multiple times to form a burning point, the burning degree and range is not easy to control; too shallow a burn is not enough to damage the tracheal cartilage but too heavy a burn is easy to cause a tracheoesopahgeal fistula and mediastinal emphysema. In a previous study, we showed that in a rabbit model electrocoagulation can be used to establish tracheal stenosis. 19 In this study, electrocoagulation was used and the output power of 20w could cause a moderate burn point, which could cause damage and destruction of the tracheal mucosa, perichondrium and cartilage without repeated burning. In this study, a rabbit model of the 7th to 8th tracheal ring stenosis was established. After tracheotomy, the trachea was accurately positioned and destroyed by a spherical electrode tip, resulting in the destruction of the tracheal mucosa, perichondrium and cartilage, resulting in the formation of scar and granulation tissue in the trachea of the experimental rabbits on the 28th day after operation. At the same time, in this study, the specific power of spherical electrode electrocoagulation device was used to accurately locate the burn, avoid damage to the tracheal membrane, and prevent the occurrence of tracheal perforation and tracheoesophageal fistula. In addition, thermal damage to the trachea may cause damage to the tissues around the trachea. 20 There are abundant blood vessels and nerves on both sides of the neck of rabbits, so it is important to avoid damaging the recurrent nerve during burning. In this study, the complications such as asphyxia and hunger caused by direct or indirect thermal injury to the recurrent nerve were effectively avoided by burning the anterior wall of the trachea. Given the abovementioned factors, we used an animal model of tracheal damage induced by electrocoagulation to mimic the effects of thermal burns and blast injuries, and focused on the effects of CM-chitosan inhalation after traumatic tracheal injury.
However, no effective therapies for tracheal stenosis have been reported. At present, tracheal stenosis is treated surgically, including by tracheal resection and anastomosis, tracheal autograft tracheoplasty, stent implantation, and laser incision. Although surgical interventions can reconstruct the trachea, restenosis is likely to occur due to the interaction of multiple factors. In recent decades, the efficacy of drug treatment for tracheal stenosis has been studied extensively, particularly for early prevention and treatment. Drug therapy has a number of major advantages, including simplicity, minimal trauma, and rapid recovery. However, there is a lack of solid evidence for the efficacy of currently available medications, and it is therefore necessary to identify new pharmaceutical agents for the treatment of tracheal stenosis.
A number of drugs have been investigated for their potential ability to prevent tracheal fibrosis and scar formation. The use of topical mitomycin-C has attracted attention for use in laryngotracheal stenosis.21-23 However, recent studies showed that mitomycin-C does not prevent airway stenosis, and may lead to the deposition of fibrin resulting in airway obstruction.24,25 A great deal of attention has been paid to 5-fluorouracil (5-FU), alone or in combination with other drugs, in the treatment of keloid and hypertrophic scars.26,27 5-FU, alone and in combination with other drugs, ameliorated airway stenosis secondary to induced tracheal trauma in rabbit models.28,29 However, 5-FU may have toxic effects in chemotherapy. Recently, glutamine was suggested as an effective option to inhibit fibroblast proliferation in laryngotracheal stenosis. 30 Other adjuvant drug therapies, including tacrolimus, rapamycin, and vitamin A, have been investigated as a means to prevent scar formation in laryngotracheal stenosis.31-33
CM-chitosan is a water-soluble polysaccharide characterized by high biodegradability and histocompatibility; it is beneficial for hemostasis, pain relief, and inhibition of fibroblast proliferation and migration. This agent has been used in many studies to inhibit skin, liver, lung, and vocal cord fibrosis.7,34-36 CM-chitosan was shown to have greater efficacy than mitomycin-C in preventing anterior glottic stenosis after endoscopic surgery. 36 As these studies suggested that CM-chitosan is safe and effective, it could serve as a novel therapeutic strategy for inhibiting tracheal stenosis.
Accordingly, we evaluated the therapeutic efficacy of CM-chitosan inhalation for alleviating traumatic trachea injury and fibrosis in our rabbit model of spherical electrocoagulation-induced tracheal injury. To determine whether CM-chitosan plays a key role in preventing the pathogenesis of airway obstruction, we compared experimental and control groups following traumatic tracheal injury and found that CM-chitosan reduced the expression of TGF-β1. This study serves as a proof of concept that CM-chitosan inhalation may represent a novel therapeutic strategy for tracheal stenosis. Further studies are required to assess the effects of the CM-chitosan concentration and residue time in the tracheal lumen when administered using a nebulizer, and to determine the precise mechanism underlying the effects of CM-chitosan inhalation on tracheal fibrosis and scar formation.
Conclusion
This study suggested that the antifibrotic effects of CM-chitosan inhalation may help alleviate tracheal fibrosis and reduce the expression of TGF-β1-induced fibrosis. Based on the results of our rabbit model, the inhalation of CM-chitosan has potential as an alternative antifibrotic strategy for the treatment of tracheal fibrosis.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China (Nos. 81970868).
