Abstract
Objectives:
Chronic rhinosinusitis with nasal polyps (CRSwNP), asthma, and non-steroidal anti-inflammatory drug-exacerbated respiratory disease (NSAID-ERD) are frequent coexisting conditions and share type 2 inflammatory pathophysiology, with interleukin (IL)-4 and IL-13 as key cytokines. Dupilumab is a monoclonal antibody that blocks the shared receptor for IL-4 and IL-13. The objective of this analysis was to evaluate dupilumab’s effect on type 2 inflammation biomarkers in patients with CRSwNP with/without coexisting asthma or NSAID-ERD from the SINUS-52 (NCT02898454) study.
Methods:
Patients received treatment with dupilumab or placebo for 52 weeks. Blood and urinary biomarkers were evaluated through 52 weeks, and nasal secretions and mucosa brushings through 24 weeks.
Results:
Of 447 patients, 60% had coexisting asthma and 27% had coexisting NSAID-ERD. At baseline, blood eotaxin-3, eosinophils, and periostin, nasal secretion eotaxin-3, and urinary leukotriene E4 were significantly higher in patients with coexisting NSAID-ERD than without. Dupilumab reduced eotaxin-3, thymus and activation-regulated chemokine, periostin, and total immunoglobulin E in blood, eotaxin-3, periostin, IL-5, and eosinophil cationic protein in nasal secretions, and leukotriene E4 in urine. Reductions were generally similar or greater in the subgroups with asthma and NSAID-ERD than without. Dupilumab also reduced MUC5AC and mast cell counts in nasal mucosa brushings.
Conclusion:
Dupilumab reduced local and systemic type 2 inflammatory biomarkers in patients with CRSwNP, including mast cells in nasal mucosa and cysteinyl leukotrienes in urine. These findings provide insight into the processes driving CRSwNP and the mechanisms of dupilumab’s therapeutic effects.
Clinical Trial Registry Name:
SINUS-52 https://www.clinicaltrials.gov/ct2/show/NCT02898454
ClinicalTrials.gov Identifier:
NCT02898454
Introduction
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a chronic inflammatory disease of the upper airways associated with high symptom burden and poor health-related quality of life (HRQoL). 1 Coexisting asthma is common, resulting in greater disease severity and higher risk of recurrence after surgery.1,2 Some patients with CRSwNP and asthma display hypersensitivity to aspirin and other non-steroidal anti-inflammatory drugs (NSAIDs), a disease syndrome termed NSAID-exacerbated respiratory disease (NSAID-ERD),3 -6 which increases disease burden.3,7,8
CRSwNP, asthma, and NSAID-ERD share a common type 2 inflammatory pathophysiology, with interleukin (IL)-4, IL-13, and IL-5 as key cytokines and tissue infiltration by eosinophils, lymphocytes, basophils, and mast cells.9,10 IL-4 and IL-13 act as central drivers of type 2 inflammation with unique and overlapping actions (Figure 1).11 -15 IL-5 is upregulated in nasal polyp (NP) tissue where it mediates a delay in eosinophil apoptosis.16,17
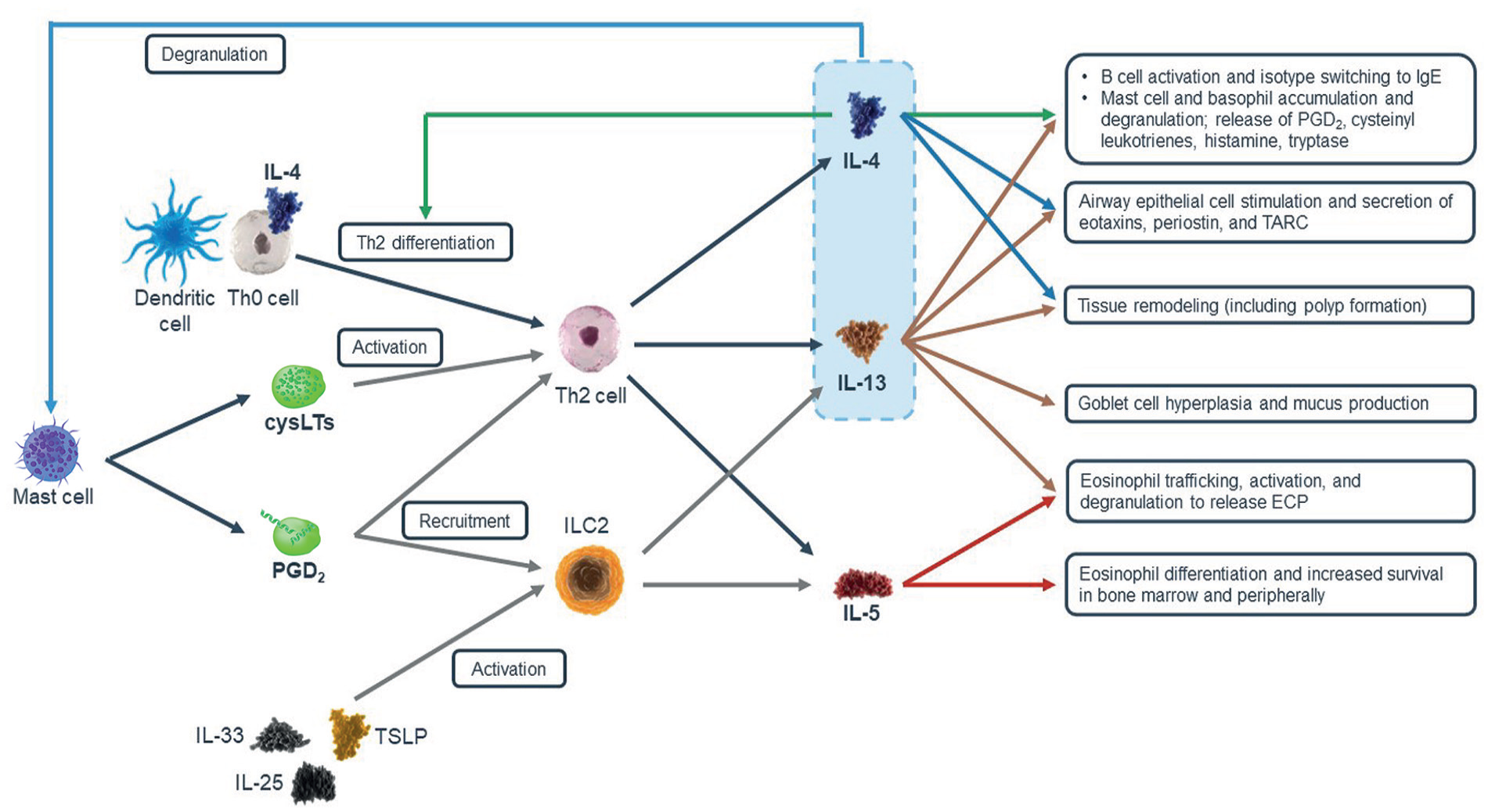
Type 2 inflammation involves both the innate and adaptive immune system.11 -13 Type 2 biomarkers relevant to the pathophysiology of CRSwNP and regulated by IL-4 and/or IL-13 include: total immunoglobulin E (IgE), an antibody class associated with allergic responses and found in elevated tissue concentrations in type 2 inflammation 19 -21; periostin, an extracellular matrix protein involved in eosinophil recruitment and airway remodeling; thymus and activation-regulated chemokine (TARC), a T helper 2 cell chemoattractant; and eotaxin-3, an eosinophil chemoattractant.9,10,13
Dupilumab is a fully human VelocImmune®-derived monoclonal antibody that blocks IL-4Rα, the shared IL-4 and IL-13 receptor component.11,22 In the SINUS-52 study (NCT02898454), dupilumab reduced NP size, sinus opacification, symptom severity, and improved HRQoL in patients with severe CRSwNP who were receiving mometasone furoate nasal spray (MFNS), and demonstrated safety consistent with the known dupilumab safety profile. 23 Improvements were also observed in patients with coexisting asthma or NSAID-ERD. 23
In a phase 2 study in patients with CRSwNP (NCT01920893; n = 60), dupilumab decreased the levels of type 2 biomarkers at Week 16 versus placebo in nasal secretions and polyp tissues, and serum total IgE, plasma eotaxin-3, and serum TARC.24,25 In SINUS-52, serum concentrations of total IgE, periostin, TARC, and plasma eotaxin-3, and nasal secretion concentrations of eosinophil cationic protein (ECP), total IgE, eotaxin-3, and IL-5 were reduced in dupilumab-treated patients. 23
Here we report the effects of dupilumab on mast cells and mucus production in nasal brushings in patients with CRSwNP. We also extend the analysis of local and systemic type 2 inflammatory biomarkers to subgroups of patients with CRSwNP with and without coexisting asthma or NSAID-ERD.
Methods
Study Design
SINUS-52 was a multicenter, randomized, placebo-controlled, double-blind, parallel-group, phase 3 study. 23 Patients were randomized 1:1:1 to receive dupilumab 300 mg subcutaneously every 2 weeks (q2w) for 52 weeks, 300 mg q2w for 24 weeks followed by every 4 weeks (q4w) to 52 weeks, or placebo throughout. Randomization was stratified by asthma/NSAID-ERD status, prior surgery, and country. All patients received MFNS 100 µg in each nostril twice daily from 4 weeks prior to randomization to study end. The study was conducted in accordance with the principles of the Declaration of Helsinki and Good Clinical Practice, and all patients provided written informed consent.
Patients
Eligible patients were aged ≥18 years with severe CRSwNP and had received systemic corticosteroids in the preceding 2 years or undergone sinonasal surgery. Patients had bilateral NP with nasal polyps score (NPS) ≥5 out of 8 (≥2 per nostril) and ≥2 rhinosinusitis symptoms (nasal congestion/obstruction and either anterior/posterior rhinorrhea or reduction/loss of smell). Key exclusion criteria included: monoclonal antibody or immunosuppressant treatment within 2 months or anti-IgE therapy within 130 days of screening; sinus surgery (including polypectomy) within 6 months before screening or sinonasal surgery changing the lateral wall structure of the nose and making the evaluation of NPS impossible; or forced expiratory volume in 1 second ≤50% of predicted normal. Asthma/NSAID-ERD was ascertained by patient-reported medical history. Patients with asthma were not required to alter their ongoing asthma medication regimen.
Biomarker Assessments
Levels of serum total IgE, TARC, periostin, and plasma eotaxin-3 were evaluated at 0, 24, and 52 weeks; blood eosinophil counts and urine leukotriene E4 (LTE4) and prostaglandin D2 metabolite (PGDM) levels at 0, 16, 24, and 52 weeks; and biomarkers in nasal secretions (total IgE, periostin, ECP, eotaxin-3, and IL-5), obtained by inserting nasal packings bilaterally into the nasal cavity for 5 minutes, at 0, 16, and 24 weeks. Cell counts (eosinophil, mast cell), mucus in cells, and total mucus were evaluated in mucosal brushings taken at baseline and Week 24 in a subset of patients (n = 65 interpretable). Nasal mucosa cells were collected bilaterally in the proximity of the lateral wall of the middle meatus (eg, surface of the middle turbinate) using a microbrush after completion of other endoscopic procedures (eg, polyp scoring).
Quantification of blood, urine, and nasal secretion protein biomarkers was carried out centrally, with samples shipped on dry ice and stored below −20°C. Serum total IgE was measured using the ImmunoCAP method (Phadia, Uppsala, Sweden); TARC using a validated enzyme-linked immunosorbent assay (ELISA) (R&D Systems, Minneapolis, MN, USA); and serum periostin using an ELISA (Shino-test, Kanagawa, Japan). For ELISA assays, any value under the lower limit of quantification (LLOQ) was set to half the LLOQ. Mast cells and eosinophils were quantified using a multiplex immunohistochemistry algorithm with dedicated settings for each marker (tryptase, eosinophil peroxidase, and epithelial cell adhesion molecule antibodies, respectively); hematoxylin and eosin stain was used to perform the quality control to exclude poor cellular samples. Cell and area quantification were performed on multiplex immunohistochemistry digitized slides using a machine-learning algorithm trained by a pathologist using HALO software (Indica Labs®, Albuquerque, NM, USA). Mucus was stained using anti-mucin 5AC antibodies (MUC5AC, Abcam, Cambridge, UK) and immunoreactivity quantified using an area quantification algorithm. Total mucus was the area occupied by mucus reported to the total area of the sample. Mucus in cells was the area within the mucus-secreting (goblet) cells of the respiratory epithelium corresponding to mucus vesicles. Using HALO software, a classifier was trained by a pathologist to distinguish total mucus from mucus within cells. All histologic analyses were blinded to treatment allocation.
Statistical Analyses
Biomarkers were analyzed in the overall population, and in asthma and NSAID-ERD subgroups. The dupilumab 300 mg q2w and dupilumab 300 mg q2w−q4w arms were pooled for analyses of nasal secretion biomarkers to Week 24 (both arms received dupilumab 300 mg q2w to Week 24). Differences between comorbidity subgroups at baseline were tested using the t-test for continuous variables, chi-square test for categorical variables in demographic characteristics, and Wilcoxon rank test for continuous variables in the biomarkers. Changes from baseline were assessed in the exposed population using only on-treatment post-baseline data, and were summarized by descriptive statistics. Nasal secretion biomarkers were normalized to total protein, and urine biomarkers to total creatinine. Nasal brushing biomarkers were normalized to total cells (for eosinophils and mast cells) or total mucus (for MUC5AC).
Results
Baseline Patient Characteristics
Of 447 patients in the exposed population, 267 (60%) had coexisting asthma and 120 (27%) had coexisting NSAID-ERD. Patients with coexisting asthma/NSAID-ERD were more likely to be female and had more severe CRSwNP (based on prior NP surgeries and Lund–Mackay computed tomography score) than those without (Table 1).
Baseline Characteristics of Patients With CRSwNP With or Without Asthma and NSAID-ERD.
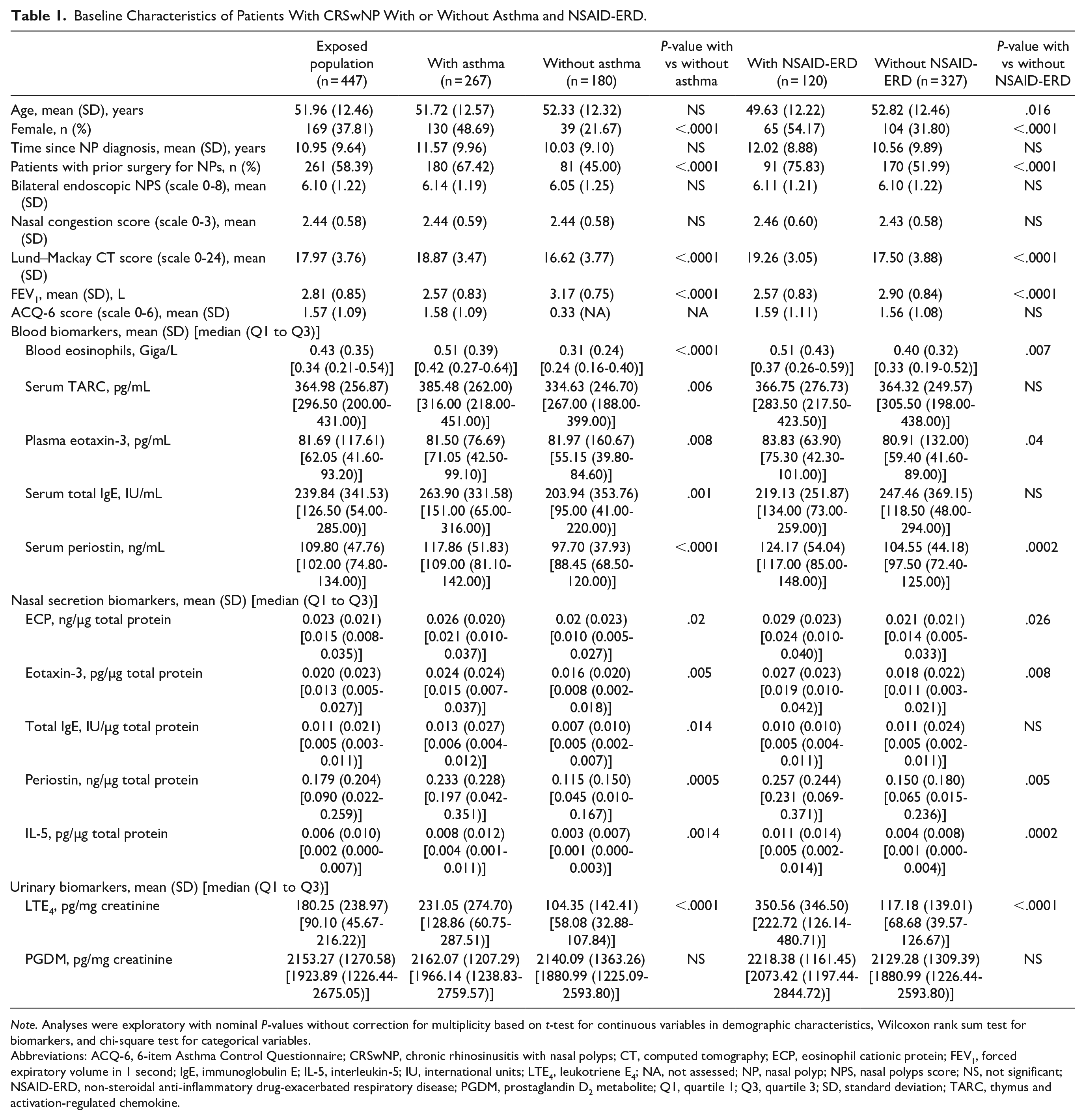
Note. Analyses were exploratory with nominal P-values without correction for multiplicity based on t-test for continuous variables in demographic characteristics, Wilcoxon rank sum test for biomarkers, and chi-square test for categorical variables.
Abbreviations: ACQ-6, 6-item Asthma Control Questionnaire; CRSwNP, chronic rhinosinusitis with nasal polyps; CT, computed tomography; ECP, eosinophil cationic protein; FEV1, forced expiratory volume in 1 second; IgE, immunoglobulin E; IL-5, interleukin-5; IU, international units; LTE4, leukotriene E4; NA, not assessed; NP, nasal polyp; NPS, nasal polyps score; NS, not significant; NSAID-ERD, non-steroidal anti-inflammatory drug-exacerbated respiratory disease; PGDM, prostaglandin D2 metabolite; Q1, quartile 1; Q3, quartile 3; SD, standard deviation; TARC, thymus and activation-regulated chemokine.
Baseline levels of blood eosinophils, eotaxin-3, and periostin, nasal secretion periostin, ECP, eotaxin-3, and IL-5, and urinary LTE4 were higher in patients with coexisting asthma/NSAID-ERD than without (Table 1). Baseline serum TARC and total IgE levels were higher in patients with asthma than without, but not different in those with and without NSAID-ERD.
Change in Blood Biomarkers
Dupilumab 300 mg q2w reduced serum total IgE, plasma eotaxin-3, serum periostin, and serum TARC with mean changes from baseline of −129.0 international units (IU)/mL, −37.4 pg/mL, −37.9 ng/mL, and −146.1 pg/mL, respectively, at Week 24, and −162.1 IU/mL, −40.8 pg/mL, −46.7 ng/mL, and −163.4 pg/mL, respectively, at Week 52. With placebo, mean levels remained essentially unchanged through Week 52 (Figure 2). The equivalent median data (Figure S1 and Table S1) showed similar results. Although there was little variation in median eosinophil count, a transient increase in mean count was apparent with dupilumab at Week 16, with a return toward baseline by Week 52, driven by a subset of patients who experienced eosinophil counts increase (Figure S2). 26
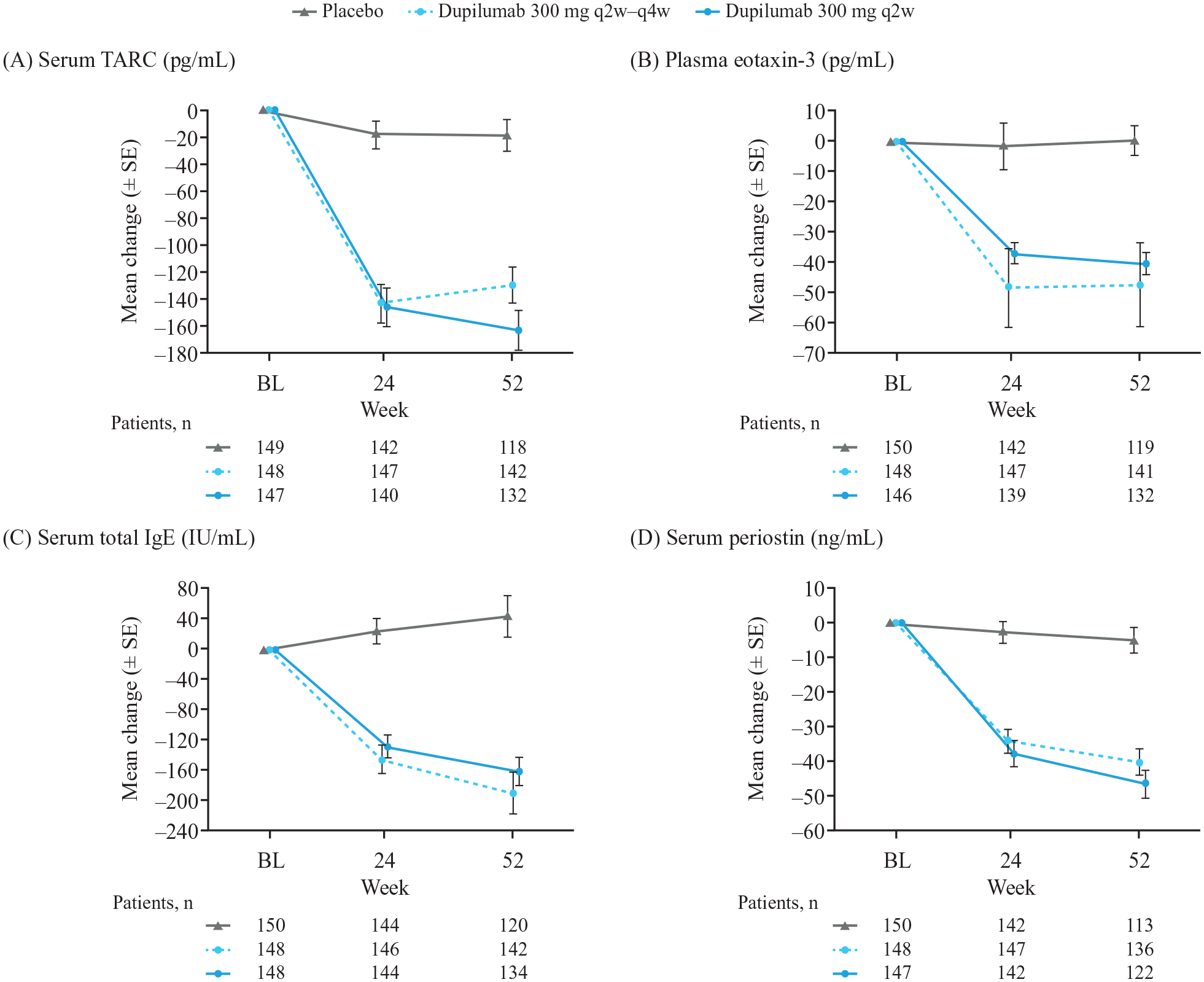
Mean change (±SE) from baseline to Week 52 in blood biomarkers. (A) TARC. (B) Eotaxin-3. (C) Total IgE. (D) Periostin.
Changes in Nasal Secretion and Nasal Mucosa Brushing Biomarkers
Dupilumab 300 mg q2w reduced eotaxin-3, total IgE, periostin, IL-5, and ECP in nasal secretions (mean change from baseline to Week 24: −0.021 pg/μg total protein, −0.005 IU/μg total protein, −0.151 ng/μg total protein, −0.003 pg/μg total protein, and −0.006 ng/μg total protein, respectively; Figure 3). Placebo mean changes were +0.005 pg/μg total protein (eotaxin-3), +0.001 IU/μg total protein (total IgE), and −0.074 ng/μg total protein (periostin), with no changes observed for IL-5 and ECP.
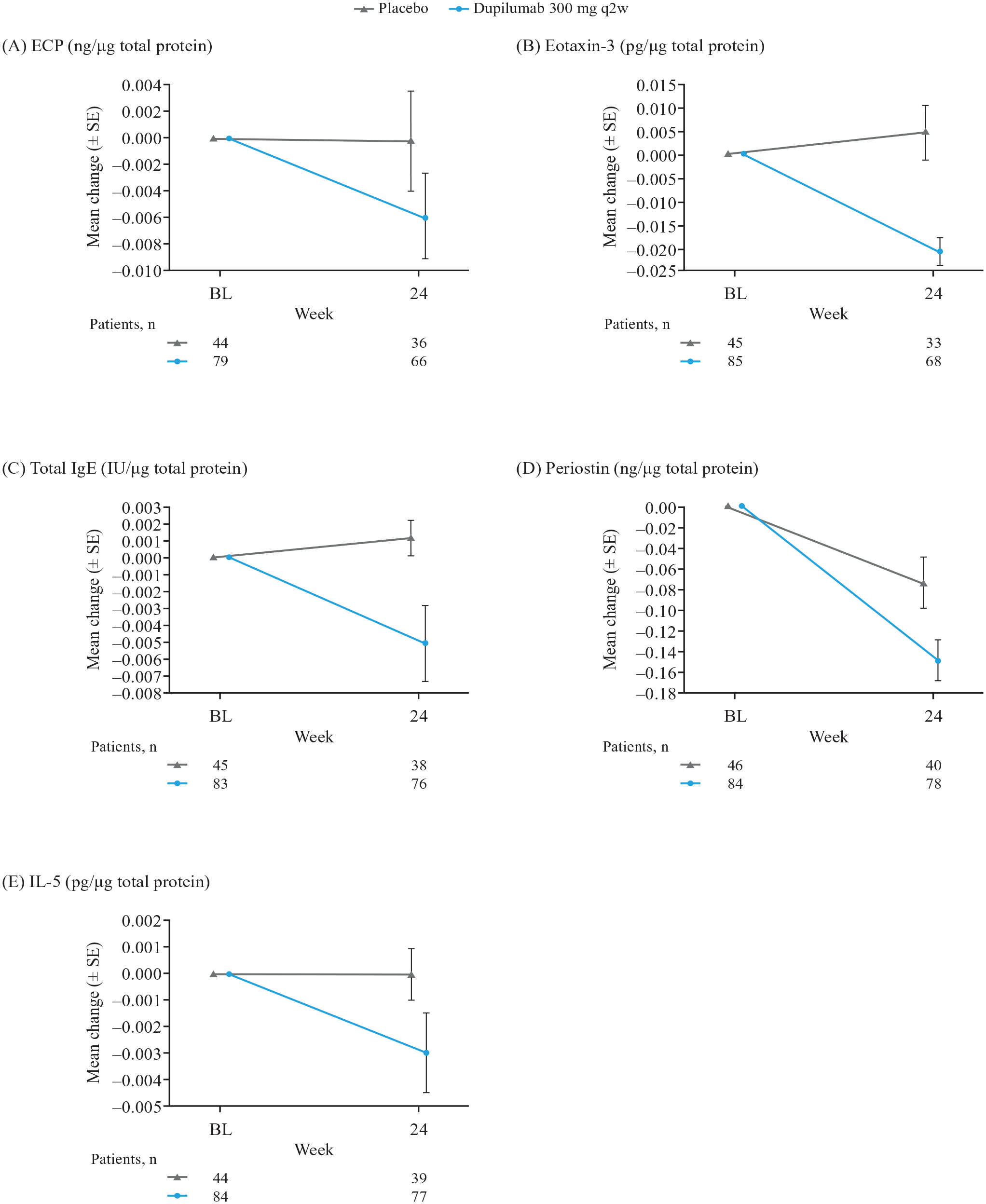
Mean change (±SE) from baseline to Week 24 in nasal secretion biomarkers. (A) ECP. (B) Eotaxin-3. (C) Total IgE. (D) Periostin. (E) IL-5.
In nasal mucosa brushings, dupilumab 300 mg q2w reduced mean total MUC5AC/mucus, mucus in goblet cells, and mast cell count at Week 24 (Figure 4). There was little difference in mean change in eosinophil count in nasal mucosa brushings at Week 24 between the dupilumab and placebo groups (Figure 4). Median data are presented in Figure S3, Figure S4, and Table S2.
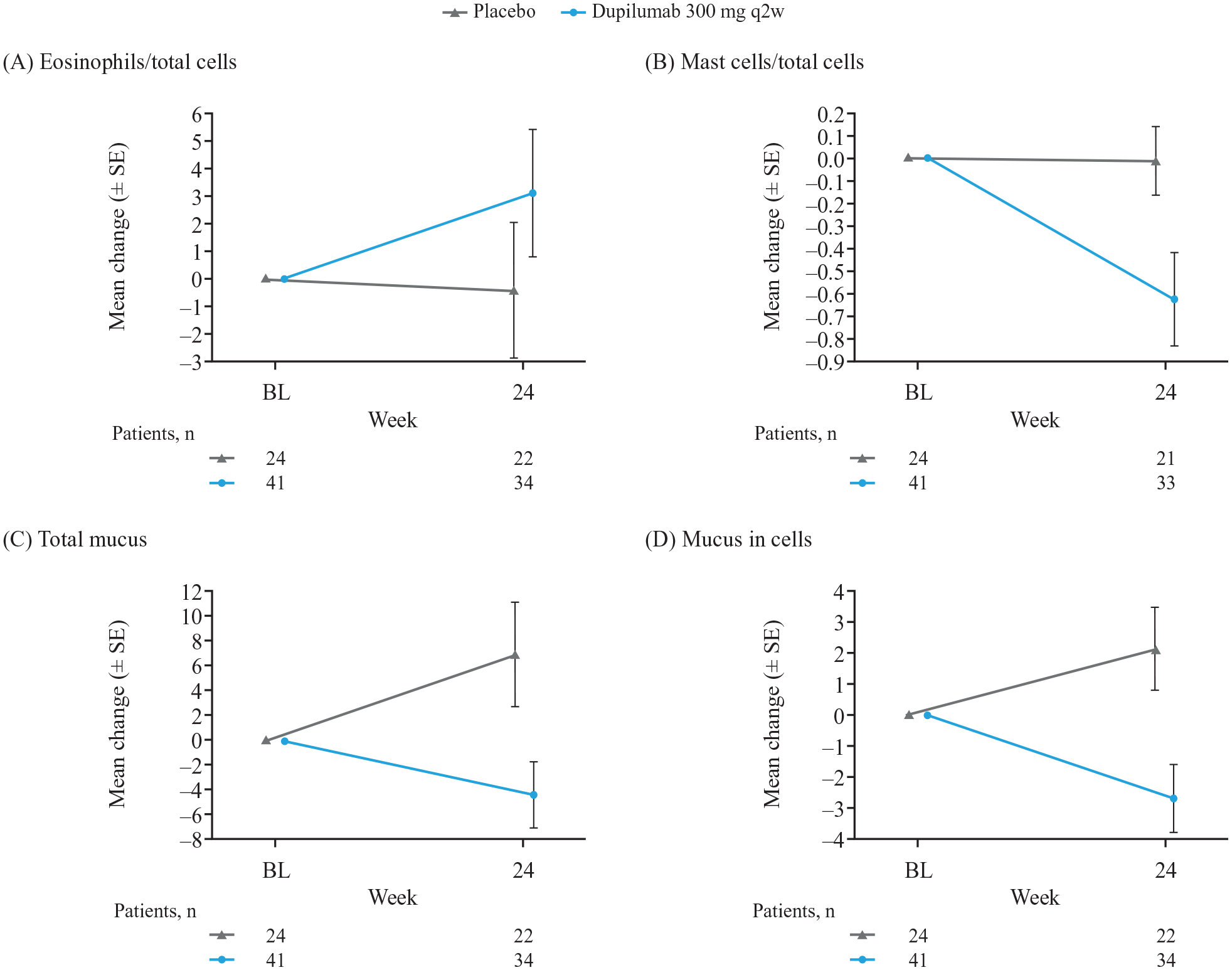
Mean change (±SE) from baseline to Week 24 in nasal mucosa brushing biomarkers. (A) Eosinophils. (B) Mast cells. (C) Total MUC5AC/mucus. (D) Mucus in cells.
Changes in Urinary Biomarkers
Dupilumab 300 mg q2w reduced urinary LTE4 concentrations at Weeks 24 and 52 (mean changes: −112.3 and −131.2 pg/mg creatinine, respectively; Figure 5); placebo changes were +0.01 and −22.4 pg/mg creatinine. PGDM concentrations were reduced with dupilumab 300 mg q2w (mean change from baseline to Weeks 24 and 52: −530.0 and −434.4 pg/mg creatinine, respectively; Figure 5); placebo changes were −290.6 and −41.1 pg/mg creatinine. Median data are presented in Figure S5 and Table S3.
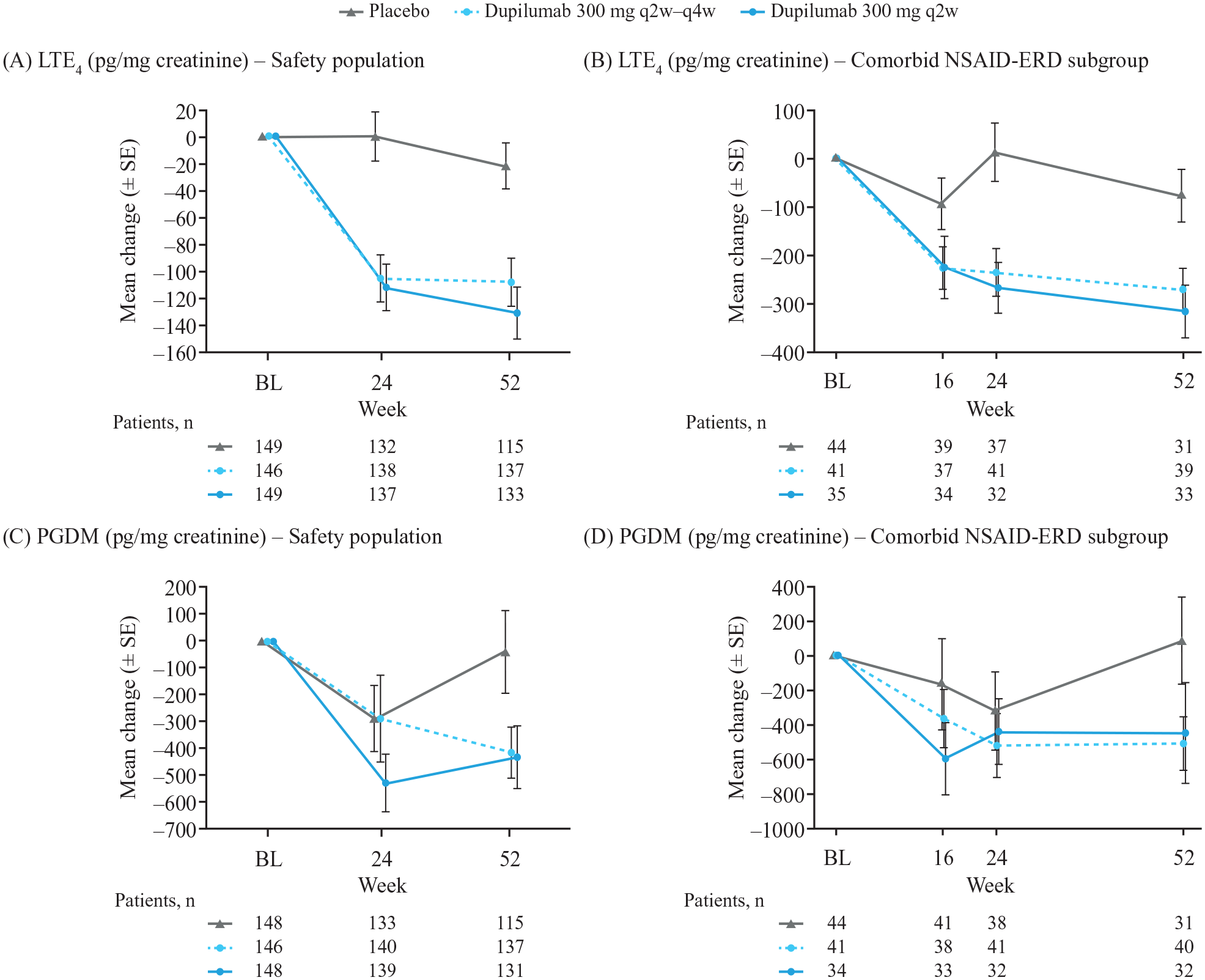
Mean change (±SE) from baseline to Week 52 in urinary biomarkers. (A) LTE4 (safety population). (B) LTE4 (NSAID-ERD subgroup). (C) PGDM (safety population). (D) PGDM (NSAID-ERD subgroup).
Change in Biomarkers in Patients With and Without Asthma
Dupilumab’s effect on type 2 biomarkers was generally similar or greater in patients with coexisting asthma than in patients without (Table S4). There were greater reductions with dupilumab versus placebo, regardless of coexisting asthma, in serum TARC, serum total IgE, plasma eotaxin-3, and serum periostin; eotaxin-3, periostin, and IL-5 in nasal secretions; and in urinary LTE4 (Table S4).
Change in Biomarkers in Patients With and Without NSAID-ERD
Dupilumab’s effect on type 2 biomarkers was generally similar or greater in patients with NSAID-ERD as in patients without (Table S5). There were greater reductions with dupilumab versus placebo, irrespective of coexisting NSAID-ERD status, in serum TARC, serum total IgE, plasma eotaxin-3, and serum periostin; eotaxin-3, periostin, and IL-5 in nasal secretions; and in urinary LTE4 (Figure 5, Figure S5, Table S5).
Discussion
This study confirms and expands on recent advances in the understanding of type 2 inflammation in CRSwNP and dupilumab’s mechanism of action in treating it. Our analysis of nasal brushings provides a signal for the first time that dupilumab reduces mast cell count and mucus in cells in nasal mucosa of patients with CRSwNP.
We did not observe a decrease in eosinophil counts in nasal mucosa brushings at 24 weeks despite a decrease in IL-5, eotaxin-3, and ECP in nasal secretions. A transient increase in mean blood eosinophil counts was seen with dupilumab in a subset of patients at Week 16, which returned to baseline levels by Week 52. 26 This transient increase may be explained by dupilumab blocking the trafficking of eosinophils from blood into tissue by inhibiting IL-4- and IL-13-induced eotaxin-3 production, but not eosinophil production or egress from the bone marrow, consistent with previous dupilumab clinical studies.23,24 Given the observed clinical improvements on dupilumab, these data suggest that decreases in tissue and peripheral eosinophil counts are not essential for robust suppression of symptoms and polyp burden in CRSwNP. This is supported by the observation that dexpramipexole treatment, which demonstrated a notable decrease in tissue eosinophils and increase in mast cells in patients with CRSwNP, was not associated with either a reduction in polyp size or an improvement in clinical symptoms at 6 months. 27 In contrast, a marked decrease in the number of mast cells but not eosinophils with dupilumab coupled with the observed improvement in CRSwNP symptoms and polyp burden may suggest that mast cells are key drivers in NP development. 28
In the current analysis, dupilumab decreased the density of mast cells in nasal brushings and, consistent with this, reduced urinary LTE4 levels, a cysteinyl leukotriene secreted by mast cells. As dupilumab has demonstrated significant improvements in CRSwNP symptoms, 23 these findings suggest that decreases in mast cells and cysteinyl leukotrienes, coupled with restoration of the anti-inflammatory effects of PGE2, are essential for robust suppression of symptoms and polyp burden in CRSwNP.23,28,29 Changes in urinary PGDM showed markedly less separation from placebo than LTE4, especially at Week 24, a timepoint at which a robust clinical improvement was observed with dupilumab. This observation is relevant given that recent studies with chemoattractant receptor-homologous molecule expressed on T-helper type 2 cells (CRTH2) and prostaglandin D2 receptor 2 antagonists in patients with CRSwNP failed to demonstrate polyp size reductions or symptom improvement.30,31 The apparent difference in relative effects on LTE4 and PDGM suggests that effects on PDGM may be a less important component/marker of dupilumab’s therapeutic effect and that PGD2 is not a key driver of the disease.
The current study adds to findings from a recent study of patients with aspirin-exacerbated respiratory disease who received dupilumab for severe asthma and/or CRSwNP. 28 As well as showing similar results to this analysis, dupilumab was shown to reverse the increase in proinflammatory cysteinyl leukotrienes and prostaglandin D2.25,32
In peripheral blood, dupilumab reduced serum TARC, periostin, and plasma eotaxin-3 levels—all of which are induced by IL-4 and IL-13 and secreted from respiratory epithelium and likely other cell types.33,34 Serum total IgE, a function of IL-4-polarized immunoglobulin switching in B cells and plasma cells, 35 was reduced by dupilumab. While systemic atopy is not strongly associated with CRSwNP, local IgE overproduction in respiratory tissue and secretions is a characteristic of the disease. 36 Although no decrease in the number of eosinophils was observed in nasal brushings, a decrease in eosinophil activation and migration was evident in dupilumab-treated patients, with reductions in eotaxin-3, ECP, and IL-5 in nasal secretions. In addition, CRSwNP severity is associated with coexisting asthma, eicosanoid dysmetabolism, and aspirin intolerance; thus, elevation in urinary LTE4 is characteristic of CRSwNP and is associated with coexisting asthma.37 -42 Interestingly, surgical removal of polyps has been reported to improve coexisting asthma and decrease urinary LTE4.43,44 These observations suggest that NPs and surrounding mucosa in the upper airway may promote asthmatic inflammation, possibly involving overproduction of cysteinyl leukotrienes and prostaglandin D2, a dysmetabolism that may be modulated by dupilumab treatment. 29
At baseline, subgroups with asthma and NSAID-ERD had higher levels of blood eosinophils, serum periostin, and nasal eotaxin-3 than subgroups without asthma and NSAID-ERD. In contrast, while levels of serum TARC and total IgE were higher in the asthma than the without-asthma subgroup, there was little difference in these biomarkers between the NSAID-ERD and without-NSAID-ERD subgroups. For IgE, this finding may reflect that NSAID-ERD is not primarily driven by an atopic mechanism. For TARC, this study population had only modest elevation relative to healthy subjects, 45 markedly lower than levels seen in atopic dermatitis for example, 32 which may limit the clinical significance of observed differences. Ascertainment of asthma and NSAID-ERD was based on patient-reported medical history, which may be subject to error, and further study is warranted for confirmation.
Dupilumab’s effect on type 2 biomarkers in CRSwNP was generally similar or greater in patients with coexisting asthma and NSAID-ERD as in patients without, which is consistent with clinical findings that dupilumab improved upper and lower airway outcomes in patients with coexisting asthma, 46 and improved objective and subjective measures to a greater extent in patients with coexisting NSAID-ERD than without. 47
A diminution in local secretion of activators of eosinophil degranulation may explain a decreased secretion of ECP and contribute to a clinical response. Dual suppression of IL-5 and eotaxin-3 secretion by dupilumab is relevant because IL-5 priming of eosinophils synergistically accentuates eotaxin-induced migration. 48 Expression of IgE receptors on eosinophils is upregulated by IL-4, facilitating their sensitization and degranulation by surface IgE–antigen interactions 49 ; in principle, dupilumab may downregulate IgE receptors on eosinophils in synergy with the diminished local IgE secretion shown in our study.
Assessment of biomarkers in nasal secretions and nasal mucosa brushings was limited to Week 24 collection, so changes beyond it and correlations with Week 52 blood biomarkers cannot be determined. However, most of the Week 52 improvements in symptoms were evident by Week 24, 23 and decreases in IL-13-regulated blood biomarkers (eg, eotaxin-3), have been shown to be near maximal by 4 weeks of dupilumab treatment in patients with CRSwNP and/or asthma. 24
In a separate analysis, we explored the correlation between changes in clinical measures of CRSwNP and changes in type 2 inflammation biomarkers. 50 Correlations were found between NPS improvement and decreased levels of IL-5, eotaxin-3, and periostin in nasal secretions. However, no single biomarker strongly correlated with clinical response, suggesting that, within the framework of type 2 inflammation, there are differences across patients in the relative importance of specific disease mediators. This is supported by variations in efficacy observed in the treatment of CRSwNP by antagonism of IL-5 (mepolizumab), IgE (omalizumab), leukotriene receptor (montelukast), prostaglandin D2 receptor 2 (fevipiprant), and CRTH2 (AZD1981).30,31,51,52 Dual IL-4 and IL-13 antagonism (dupilumab) demonstrated robust efficacy in CRSwNP. 23 While dupilumab has not been directly compared with other biologics, recent meta-analyses suggest that dupilumab may achieve greater clinical efficacy in CRSwNP than omalizumab or mepolizumab. 53 It is possible that greater efficacy with dupilumab may be a consequence of a broader effect on multiple mediators of type 2 inflammation; further studies are required to confirm.
Conclusion
Consistent with its mechanism of action, dupilumab reduced type 2 inflammatory biomarkers systemically and locally in patients with CRSwNP. Furthermore, this is the first study to demonstrate the impact of dupilumab on mast cells and mucus secreting cells, suggesting a key role of these effector cells in CRSwNP, and their importance as a target for the robust suppression of symptoms and polyp burden. These data add to the available evidence for the benefit of add-on dupilumab in patients with CRSwNP, including those with coexisting type 2 inflammatory diseases, who represent difficult-to-treat populations.
Supplemental Material
sj-docx-1-aor-10.1177_00034894231176334 – Supplemental material for Effect of Dupilumab on Type 2 Biomarkers in Chronic Rhinosinusitis With Nasal Polyps: SINUS-52 Study Results
Supplemental material, sj-docx-1-aor-10.1177_00034894231176334 for Effect of Dupilumab on Type 2 Biomarkers in Chronic Rhinosinusitis With Nasal Polyps: SINUS-52 Study Results by Claus Bachert, Tanya M. Laidlaw, Seong H. Cho, Joaquim Mullol, Brian N. Swanson, Souad Naimi, Marion Classe, Sivan Harel, Alexandre Jagerschmidt, Elizabeth Laws, Marcella Ruddy, Amy Praestgaard, Nikhil Amin and Leda P. Mannent in Annals of Otology, Rhinology & Laryngology
Footnotes
Acknowledgements
Medical writing/editorial assistance was provided by Peter Tran, PhD, of Adelphi Group, Macclesfield, UK, funded by Sanofi and Regeneron Pharmaceuticals Inc. according to the Good Publication Practice guideline.
Data Availability Statement
Qualified researchers may request access to data and related study documents. Patient-level data will be anonymized and study documents will be redacted to protect the privacy of trial patients. Further details on Sanofi’s data sharing criteria, eligible studies, and process for requesting access can be found at: ![]() .
.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C Bachert reports advisory board fees from ALK, ASIT Biotech, AstraZeneca, GlaxoSmithKline, Intrexon Actobiotics, Novartis, Sanofi, and Stallergenes Greer. TM Laidlaw reports advisory board fees from GlaxoSmithKline, Novartis, Regeneron Pharmaceuticals Inc., and Sanofi. SH Cho reports research grants from National Institutes of Health, Regeneron Pharmaceuticals Inc., and Sanofi. J Mullol has received speaker fees from GlaxoSmithKline, Menarini, Merck Sharp & Dohme, Mylan-Meda Pharmaceuticals (Viatris), Novartis, Regeneron Pharmaceuticals Inc., Sanofi, UCB Pharma, and Uriach; clinical trial funding from Genentech-Roche, Regeneron Pharmaceuticals Inc., and Sanofi; research grants from Mylan-Meda Pharmaceuticals (Viatris) and Uriach; and advisory board fees from Genentech-Roche, Mylan-Meda Pharmaceuticals (Viatris), Novartis, Regeneron Pharmaceuticals Inc., Sanofi, and Uriach. S Naimi, A Jagerschmidt, E Laws, A Praestgaard, and LP Mannent are employees and may hold stock and/or stock options in Sanofi. BN Swanson and M Classe are former employees and may hold stock and/or stock options in Sanofi. S Harel, M Ruddy, and N Amin are former employees and may hold stock and/or stock options in Regeneron Pharmaceuticals Inc.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was sponsored by Sanofi and Regeneron Pharmaceuticals Inc. ClinicalTrials.gov Identifier: NCT02898454. The funder of the study approved the study design and was involved in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
