Abstract
Peripheral Artery Disease (PAD) with Chronic Limb Threatening Ischemia (CLTI) is a severe condition at risk of amputation, and often conventional surgical and endovascular procedures and/or medical therapy are insufficient. The present review considers current knowledge regarding autologous cell therapy and regenerative medicine for PAD with CLTI. We evaluated cell-based therapies in the recent literature, that are pivotal in treating vascular disorders by promoting angiogenesis, vascular restoration, and tissue regeneration; furthermore, we provided an overview of the relevant main clinical studies. Clinical conditions and risk factors of patients correlate with the performance of autologous cell-based therapies and the probability of success. Nevertheless, the current evidence indicates that these therapies are promising, necessitating further research. Autologous cell therapy and regenerative medicine can offer additional support for treating PAD with CLTI. These advanced techniques are becoming more tailored and substantial for their application in clinical practice. Personalized Medicine represents a contemporary paradigm that calls for consideration of individual patient’s clinical conditions, risk factors, and biomarkers.
Introduction
Peripheral Artery Disease (PAD) encompasses a range of occlusive arterial conditions, from asymptomatic obstruction to severe cases of critical limb ischemia necessitating amputation. The severity and symptoms escalate as the condition disrupts the balance between distal perfusion pressure and the tissues’ metabolic needs. 1
Atherosclerotic PAD can lead to Chronic Limb Threatening Ischemia (CLTI). CLTI shares numerous risk factors with atherosclerotic conditions affecting other blood vessel regions.2,3
The risk factors include hypertension, hypercholesterolemia, cigarette smoking, and diabetes mellitus. 4
Patients with CLTI experience a swift decline in functionality, which negatively impacts on their quality of life and may result in a depressed mental state.5,6
CLTI is defined as the combination of evidence of ischemia together with any conditions of rest pain, nonhealing ulcers or gangrene in the lower extremities. CLTI leads to an amputation rate of 14% to 20% and a death rate of 25% within the first year and 50% within 5 years. 7
Treatment methods—medical, endovascular, and surgical—focus on restoring peripheral blood flow and managing risk factors to reduce pain, heal ulcers, and preserve the limbs.
A significant proportion of patients with CLTI are unable to undergo revascularization procedures due to vascular anatomical constraints, or experience procedural failure resulting from graft complications, stent thrombosis, or restenosis. 3 No medical treatment currently exists to improve blood flow beyond the occluded vessels. Research is exploring therapeutic angiogenesis in the affected limb distal to the occlusion. Regenerative medicine is a promising approach to enhance vascular surgery techniques. In the present review, we aim to provide a current update on regenerative medicine and the journey toward personalized medicine for CLTI.
Literature Highlights
Cell-Based Therapies for Vascular Diseases
Cell-based therapies are pivotal in treating vascular disorders by promoting angiogenesis, vascular restoration, and tissue regeneration. Stem cell vascular medicine involves angiogenesis, arteriogenesis, and vasculogenesis, which are related to distinct processes in blood vessel development and modification. Vasculogenesis, the formation of blood vessels in situ, involves the differentiation of angioblasts into blood vessels. This process is essential during embryonic development and in adults, where it aids in progenitor cell repair for ischemic conditions. Vasculogenesis is distinct from angiogenesis and arteriogenesis. 8
Angiogenesis, which is the formation of new blood vessels from existing vascular structures, is vital for embryonic development, wound healing, and menstrual cycle regulation. 9 It can occur through mechanisms such as branching from existing vessels, division of current vessels, and incorporation of endothelial progenitor cells, which are essential for maintaining healthy bodily functions and for supporting tissue growth and repair. 9
Arteriogenesis, the enlargement and remodeling of small arteries into larger ones, is vital for restoring the blood supply to regions impacted by narrowed or blocked arteries in conditions such as ischemic heart disease or peripheral artery disease. This process starts when endothelial cells in small arteries undergo increased fluid shear stress, initiating signaling cascades and attracting cells critical for vascular restructuring. 10
Neoangiogenesis, the formation of new blood vessels in mature tissues, occurs under both normal and abnormal conditions, such as tissue repair, muscle growth from exercise, tumor growth, and age-related eye disorders. 11 This process requires a balance between the factors that promote and inhibit blood vessel formation, along with the activation and proliferation of endothelial cells, supportive cells, and bone marrow-derived stem cells that contribute to vessel development. While both angiogenesis and vasculogenesis create new blood vessels, arteriogenesis specifically involves the modification and expansion of existing small arteries. These processes are essential for maintaining blood supply, oxygen delivery, and nutrient distribution. Disruptions can lead to various pathological conditions. 11
Neoangiogenesis and endothelial vascular repair processes are compromised in arteriosclerotic vascular disease, a condition frequently linked to hypertension, diabetes, and chronic kidney disease. 12
Notwithstanding the increasing significance of the Gut-Cardiovascular Axis is assuming an increasing importance in the cardiovascular field. 13 Alterations in the microbiota and, consequently, in the circulating microbiome impair the function of endothelial cells and endothelial progenitor cells (EPCs). 14
For these reasons, the journey toward the use of regenerative medicine in general and in vascular diseases is going toward a personalized approach.15,16
Adult stem cells, such as mesenchymal stem cells (MSCs) and EPCs, can be sourced from the bone marrow, adipose tissue, and peripheral blood. According to Asahara et al, a subset of bone marrow-derived cells, specifically CD34+ cells, contribute to the formation of new blood vessels. 17 These cells, known as EPCs, play a crucial role in the physiological processes that result in the development of new blood vessels.18 -20
The CD34 surface marker is a transmembrane protein classified as sialomucin, which consists of the hematopoietic progenitor cell antigen (HPCa) podocalyxin and endoglycan. CD34 serves as an identifier for vascular endothelial cells (ECs), EPCs, and hematopoietic progenitor cells (HPCs). 21
CD34+cell populations possess unique regenerative capabilities, that have attracted considerable attention in the field of vascular medicine.22 -24
Following Asahara’s pioneering work, 25 numerous studies have focused on understanding progenitor cell populations, particularly hematopoietic stem cells (HPS) and EPC.
Prominin-1, also known as CD133, is a transmembrane glycoprotein typically present in undifferentiated cells, such as EPC, HPC, fetal brainstem cells, and prostate epithelial cells. 26
The process of angiogenesis also encompasses several other important cellular groups, including endothelial colony forming cells (ECFC) expressing CD34+ and PROCR high, C-Kit cells, very small embryonic like cells (VSELc), and MSC.27 -29
Adult tissue contains a rare very small population of stem cells (3-5 µm) with characteristics resembling embryonic cells defined very small embryonic like cell (VSElc) and expressing CD34+/CD133+/CXCR4+/Lin− CD45− Oct-4, Nanog, and stage-specific embryonic antigen-430,31 that seems to play a supportive role in angiogenesis. 32
Within the vascular intima resides a population of cells designated as vascular endothelial progenitor cells (VSPCs), characterized by the expression of CD157 with homeostatic and regenerative properties. 33 The significance of ECFCs in angiogenesis is underscored by their putative origin from the vascular endothelial stem/progenitor cells (VESCs) situated within the vascular intima. These VESCs have been demonstrated to play a vital role in the processes of vascular regeneration. 34
It is crucial to acknowledge the importance of the interaction between endothelial colony-forming cells with high proliferative potential (ECFCs) and VESCs.35,36
Various cell types, including EPCs, ECFCs, C-kit, VSELc, and MSC, generate growth factors such as vascular endothelial growth factor A (VEGF-A), fibroblast growth factor (FGF), insulin growth factor 1 (IGF-1), platelet derived growth factor (PDGF), and transforming growth factor ( TGF), which promote the formation of new blood vessels.37 -40
The International Society for Cell and Gene Therapy (ISCT) defines MSCs as a heterogeneous cell population with distinct properties, including plastic adherence, multilineage differentiation potential (into osteoblasts, chondrocytes, and adipocytes), and secretion of various factors that modulate immune responses and promote tissue repair. 41 MSCs express markers 75, 90, and 105. 41
MSCs are notable for their paracrine mechanisms, which support angiogenesis and tissue repair. The MSC secretome, which includes growth factors, cytokines, and extracellular vesicles (EVs), plays a critical role in intercellular communication and modulating recipient cell behavior. 42
EVs facilitate the transfer of bioactive molecules like microRNAs, which regulate gene expression and enhance angiogenesis. 43 MSC-derived EVs deliver angiogenic factors such as VEGF, Von Willebrand faactor (vWF), and TGF-β1 to endothelial cells. Studies have shown that MSCs from adipose tissue, bone marrow, and cord blood have similar angiogenic effects, especially under hypoxic conditions. 44
The angiogenic efficacy of MSC-derived EVs may vary depending on their tissue of origin. For example, the secretome of AT-MSCs (adipose tissue-derived mesenchymal stem cells) exhibits greater tubulogenic efficiency compared with BM-MSCs (bone marrow-derived), attributable to a greater expression of angiogenic factors such as IGF-1, VEGF-D, and interleukin 8 (IL-8). 44
Hypoxia enhances the angiogenic potential of MSC-derived EVs. Studies have demonstrated that EVs derived from hypoxia-stimulated AT-MSCs, BM-MSCs, and CB-MSCs (cord blood-derived) promote angiogenesis more effectively.45,46 Hypoxia not only induces VEGF expression in MSCs and their EVs but also increases VEGF expression and activates the protein kinase A (PKA) signaling pathway in endothelial cells. 47 In addition to growth factors, EVs also transport miRNAs that can regulate angiogenesis. For example, miR-132, miR-125a, and miR-210 have been identified in MSC-derived EVs and have been demonstrated to promote angiogenesis in vitro and in vivo.48 -50
Platelets and monocytes are fundamental players in angiogenesis as well as monocytes.
Platelets contain various pro- and anti-angiogenic proteins in their α-granules, that are released upon activation to significantly affect endothelial cell behavior and stimulate blood vessel formation, crucial for wound healing and tumor growth.51 -53 Lopatina et al 44 demonstrated that MSCs stimulated by platelet-derived growth factor (PDGF) showed an increased ability to promote angiogenesis. Compared with EVs from unstimulated cells, those from PDGF-stimulated MSCs contained angiogenic factors like c-kit, stem cell factor (SCF), and matrix metalloproteinases (MMPs). 54
BMCs and peripheral blood mononuclear cells (PBMNc) differ in their cytokine expression. VEGF is mainly expressed by BMCs. 55
EPCs interact directly with MSC and ECFC cells present in the vessel wall and tissue. 56
EPCs, ECFCs, C-kit, VSELc, and MSCs contribute to paracrine actions that promote vasculogenesis and angiogenic gene induction by producing cytokines and miRNAs. EPCs lose CD133+ markers and express CD31+ and vascular endothelial cadherin during differentiation. 56
Ischemic events cause a complex pathophysiological process that aims to repair the damage. Cytokines released during ischemia interact with the cell niches of the bone marrow, resulting in the migration of endothelial progenitor cells into the ischemic tissue through processes of adhesion, proliferation, differentiation, and the release of cytokines. 57 Of note, in chronic limb ischemia the expression of genes of vascular endothelial growth factor (VEGF) and stromal derived factor 1α (SDF-1α) is reduced in the tissue. 57
Others Stem Cell Therapy
Arango-Rodríguez et al 48 compared the therapeutic effects of auto-BM-MNC and allo-Wharton Jelly-MSCs in diabetic patients with CLTI (Rutherford stage 3-5) aged 51 to 85 years in a randomized, double-blind, controlled study. They evaluated the safety and efficacy of arterial wall injections of these cells. Both cell types showed potential therapeutic benefits, suggesting that stem cell therapy could be a promising treatment alternative for CLTI in diabetic patients. 58
Adipose-Derived Stem Cells
Human adipose-derived stem cells (ASCs) display mesenchymal stem cell markers such as CD29, CD44, CD146, CD166, CD90, CD105, and the GPI-anchored enzyme CD73, with minimal (<2%) expression of the hematopoietic antigens CD11b, CD13, CD14, CD19, and CD45. 59
It is imperative that they lack the expression of the endothelial markers CD31 and human leucocyte antigen (HLA-DR). 59
Adipose tissue, especially subcutaneous white adipose tissue from the abdomen, thighs, hips, and/or buttocks, is the primary source for stem cell isolation. 60 Mechanical or enzymatic digestion, typically with collagenase, breaks down the tissue to produce the stromal vascular fraction (SVF), excluding adipocytes. Mechanical isolation is preferred for obtaining SVF owing to its enzyme-free approach and preservation of extracellular matrix (ECM) components, which are vital for ASCs. Techniques, such as emulsification, vibration, and mincing, enhance cell yield and viability, although excessive mechanical force may reduce cell viability. Research indicates that mechanical methods may yield more peripheral blood mononuclear cells and retain progenitor cells within the vascular structures. 61 Further studies are needed to identify the most effective mechanical techniques for optimal SVF in regenerative therapies. 50 The SVF is abundant in ASCs, a relatively uniform group of spindle-shaped, fibroblast-like cells. 60
ASCs have generated significant interest in the field of regenerative medicine. The complexity of adipose tissue and its cellular activities highlight the potential of ASCs in regenerative therapies for PAD, particularly in neovascularization.62,63 ASCs have demonstrated efficacy in improving muscle function, increasing perfusion, and reducing inflammatory infiltrates in a mouse model of PAD.64,65 Concerns have been raised regarding about ASCs, in pathologies such as diabetes, arteriosclerosis, obesity, and smoking that cause inflammation and impair their angiogenic potential.66 -68
Diabetes negatively affects ASCs by altering niche conditions and depleting crucial cellular subpopulations necessary for angiogenesis, thereby impairing ASCs’ angiogenic potential and their ability to promote neovascularization and support tissue healing. 66
Marędziak et al evaluated aging’s impact on human ASCs adipose-derived mesenchymal stem cells’ regenerative capabilities. 69 They assessed oxidative stress, senescence, and gene expression profiles to determine functional changes with age. Results showed a decline in regenerative potential, evidenced by increased oxidative stress, senescence, and altered gene expression profile. 69 Gender differences are particularly important regarding ASCs and should be considered. 70
Gentile and Garcovich have suggested a possible interaction with the tumor microenvironment and pro-oncogenic effects. 71
When used associated with ozone ASCs switch toward adipogenesis. 72
The scientific community underscores the importance of understanding the functionality and safety of ASCs in clinical environments. Although initial research is promising, further studies are needed to thoroughly investigate the therapeutic benefits of ASCs for PAD and to assess any potential risks (Table 1).
Cell Therapies for CLTI.

Table 1 summarizes the cell population described.
Clinical Studies
Rigato et al 73 conducted a meta-analysis of 19 randomized clinical trials (n = 837) investigating autologous cell therapy for PAD and CLTI. The studies used bone marrow (BM) mononuclear cells (BM-MNCs, n = 8), BM concentrate (n = 2), BM mesenchymal stem cells (BM-MSCs, n = 3), or mobilized peripheral blood (PB)-MNCs. Several elements of the data compiled in the meta-analysis merit discussion. The meta-analysis indicates that the requisite sample size for achieving significant pooled effects in randomized controlled trials (RCTs) was not attained. This suggests that some significant findings may be false positives, arising from insufficient statistical power. There was substantial intrinsic heterogeneity among the studies, as they differed in setting, underlying disease, type and dose of cells, route of administration, and follow-up duration. Significant heterogeneity was observed for key outcomes such as amputation-free survival, ankle brachial index (ABI), transcutaneous oxygen tension (TcO2), rest pain score, and pain free walking distance (PFWD). This heterogeneity can obscure the true effect of cell therapy and complicate the interpretation of results. This analysis suggests that cell therapy may improve amputation-free survival, and limb perfusion, and functional capacity endpoints, although the evidence varies. The authors recommend high-quality trials to further evaluate cell therapy’s benefits in PAD/CLTI. 73
Gao et al 74 analyzed 27 randomized trials with 1186 PAD patients, discovering that autologous stem cell therapy improved ulcer healing compared with standard treatments, but limb salvage outcomes were similar. Also, in this meta-analysis some studies were characterized by small sample sizes, which may limit statistical power of the findings and contribute to result instability, particularly concerning outcomes such as major amputation rate. There was considerable variation among the included randomized controlled trials (RCTs) regarding patient characteristics, types of stem cells utilized, transplantation methods, control group treatments, and follow-up durations. These differences resulted in significant heterogeneity in the meta-analysis, especially for outcomes such as ABI, TcO2, and rest pain score (Table 2).
Key clinical trial findings.
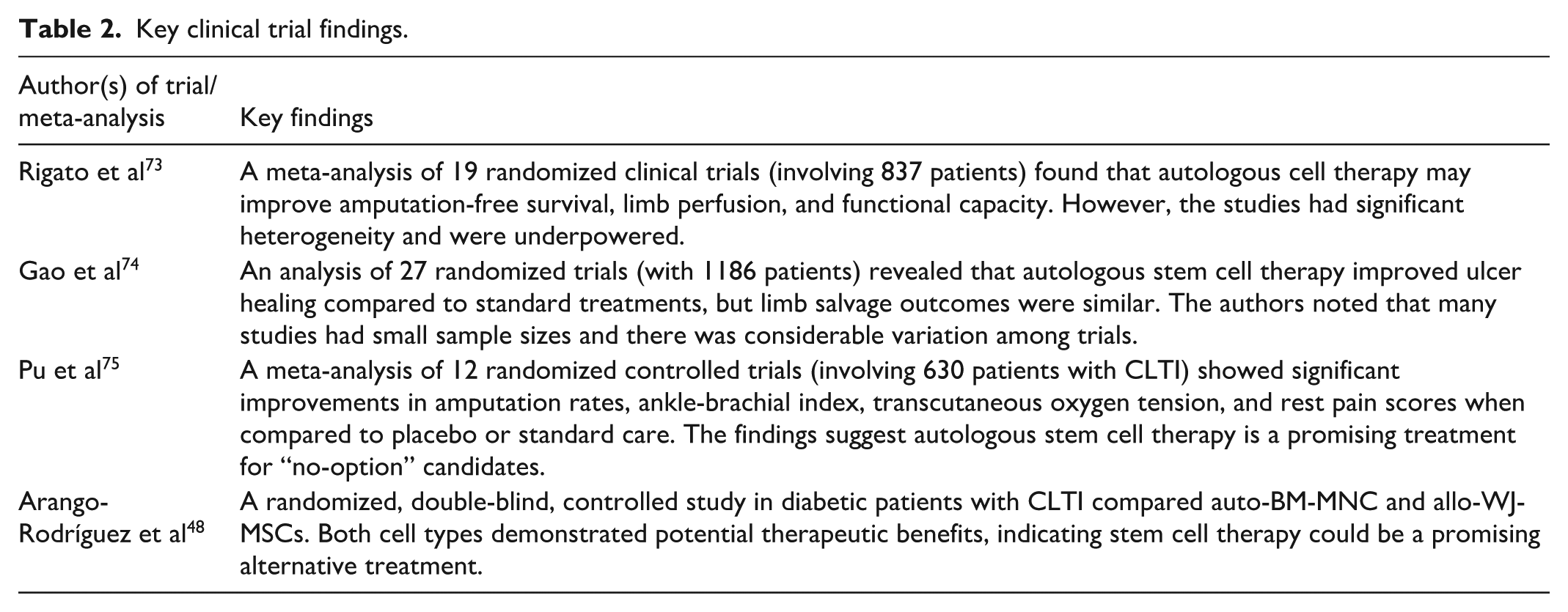
This highlights the necessity for more rigorous research and the standardization of transplantation techniques, stem cell types, and dosages to enhance safety and effectiveness. Despite its potential, autologous stem cell therapy demands further well-structured studies to verify its safety and efficacy
Pu et al 75 conducted a meta-analysis of 12 randomized controlled trials with 630 CLTI patients, examining the safety and efficacy of autologous stem cell therapy from bone marrow and granulocyte colony stimulating factor (G-CSF)-mobilized peripheral blood. Findings revealed major enhancements in amputation rates, ABI, transcutaneous oxygen tension, and rest pain scores over placebo or standard care. 75
These meta-analyses significantly impact future therapeutic options for CLTI patients, especially “no-option” candidates for conventional treatments. The findings suggest that autologous stem cell therapy is a promising treatment option for these patients.
Considerations
The patient’s overall condition is crucial for autologous regenerative therapies, such as BMCs or PBMNC. 76 Furthermore, in bone marrow aspirate (BMA) and PBMNC acquisition, erythrocytes and leukocytes are the primary cellular populations. 77
Aging diminishes stem cell regenerative potential, notably the angiogenic capacity required for tissue repair. This decline involves cellular senescence, reduced stem cell numbers, and self-renewal, and changes in systemic regulatory mechanisms. 78
Comorbidities are prevalent in this patient population. When managing severe PAD, it is essential to consider all relevant factors before applying regenerative treatments. Arteriosclerosis reduces the regenerative capacity of circulating EPCs. The decline in EPC quantity and function in atherosclerotic conditions, along with their link to osteogenic phenotypes in coronary atherosclerosis, contributes to this impairment.79,80
Further investigation is warranted to elucidate the mechanisms underlying this impairment and to develop strategies for enhancing the regenerative potential of EPCs in arteriosclerosis. The relationship between heart failure (HF) and PAD is well-documented. 81 Ischemic cardiomyopathy is linked to a specific impairment in the function and quantity of progenitor cells in bone marrow and peripheral blood, marked by depletion of the bone marrow niche. 82
The bone marrow microenvironments of diabetic patients show changes that impair hematopoietic stem cell (HSC) mobilization in response to granulocyte colony-stimulating factor (G-CSF). 83
These changes include increased quiescent and cycling HSCs, fewer osteoblasts, more sympathetic neurons, and elevated levels of β3-adrenergic receptors. 84 Diabetes disrupts bone marrow niche dynamics, affecting HSC mobilization through both HSC-independent and microenvironment-dependent mechanisms. 83 Clinical trials using mobilized peripheral blood mononuclear cells (PBMNCs) and granulocyte G-CSF raise concerns about off-label G-CSF use and the associated risks in critically ill patients.84,85
Diabetes negatively affects the number and functionality of EPCs, contributing to the progression of vascular complications.86 -89 Hyperglycaemia is a key factor in microvascular complications, but other elements also heighten cardiovascular risk in diabetes by affecting EPC biology. 90 Elevated high-sensitivity C-reactive protein (hs-CRP) levels, indicative of inflammation, are linked to reduced circulating EPCs in diabetic patients, suggesting that inflammation contributes to EPC depletion in diabetic vascular disease. 91 Moreover, peripheral mononuclear cells and monocytes are reduced in the blood of diabetic patients. A comprehensive description of the complex world of macrophages and monocytes is beyond the scope of this review, and we refer readers to Ferrante and Leibovich for further information. 92
Monocytes are crucial to the immune system and exhibit heterogeneity, with subsets having specific functions. The most prevalent subset, Classical Monocytes (CD14++CD16-), are termed “inflammatory” monocytes. 93 Non-Classical Monocytes (CD14+CD16++) engage in immune surveillance, tissue repair, and vascular integrity. Intermediate Monocytes (CD14++CD16+) are a transitional subset with roles in both inflammation and patrolling.94,95 Aged Monocytes express an increased number of pro-inflammatory genes. 96
Monocytes are crucial in arteriosclerosis pathogenesis, being recruited to arterial walls, interacting with growth factors, and engaging in inflammatory processes. Atherogenic risk factors activate monocytes, causing persistent inflammation and epigenetic changes linked to chronic low-grade inflammation in arteriosclerotic cardiovascular disease. These stimuli influence monocyte epigenetic regulation, altering gene expression without changing the DNA sequence. This epigenetic reprogramming results in enduring changes in monocyte function and inflammatory response.97,98
Diabetes is a condition characterized by a shift in the monocyte population toward a proinflammatory phenotype, which plays a significant role in the altered immune response that exacerbates atherosclerosis and contributes to increased tissue resistance to insulin.99,100
The connection between monocytes, arteriosclerosis, and diabetes highlights the importance of these cells in both conditions. The presence of monocytes and macrophages in conditions associated with diabetes disrupts lipid uptake and efflux pathways, leading to the accumulation of lipids. 101
Patients with chronic kidney disease (CKD) exhibit diminished levels of proangiogenic mediators, including the circulating VEGF ligand and endostatin, as well as circulating endothelial cells in their peripheral blood.102,103 CKD is also associated with reduced angiogenic potential in EPCs. Patients with hypertension have significantly lower levels of circulating EPCs compared with normotensive individuals.14,104
The gut microbiota is gaining significance in the cardiovascular system. It produces trimethylamine (TMA) from sources such as choline, carnitine, lecithin, gamma-butyrobetaine, and phosphatidylcholine, primarily derived from diets rich in choline, L-carnitine, and phosphatidylcholine. 105 TMA is transformed into trimethylamine-N-oxide (TMAO) through the action of specific bacterial enzymes. In the liver, TMA is primarily oxidized to TMAO by flavin monooxygenase 3 (FMO3), which demonstrates 5-fold greater activity compared with flavin monooxygenase 1 (FMO1). FMO1 is predominantly expressed in the kidneys and intestines. 106
A key effect of TMAO above physiological range, is to facilitate the onset of atherosclerosis through several complex mechanisms, and it is a known marker of arteriosclerotic vascular disease. 107 Plasma TMAO predicts the risk of adverse events in patients with PAD 108 and inhibits angiogenesis and perfusion in patients with PAD.108,109 TMAO levels are abnormally high in diabetic patients as well as during continuous statins treatment, which may contribute to the development and progression of atherosclerosis. 110 TMAO increase platelet activation and the risk of thrombosis. 111 Elevated levels of TMAO in the plasma were linked to decreased numbers of circulating EPCs, impaired endothelial function, and a higher incidence of adverse cardiovascular events. 112 TMAO serves as a biomarker in the field of cardiovascular disease, particularly in the context of applying autologous regenerative therapies.
Statins increase EPCs in both quantity and quality via the phosphoinositide-3-Kinase / protein kinase B (PI3K/Akt) signaling pathway and targeted progenitor cell differentiation. 113 These mechanisms likely contribute to statins’ therapeutic effects in preventing and treating vascular disorders, beyond merely lowering cholesterol levels.113,114 Pravastatin has the potential to enhance the angiogenic capacity of EPCs, whereas simvastatin can inhibit the expression of proinflammatory cytokines in monocytes.115,116 A recent study found that administering atorvastatin before bone marrow cell (BMC) transplantation significantly improved transcutaneous oxygen pressure (TcPO2) and reduced pain compared with the non-atorvastatin group. 117 Atorvastatin recipients had a higher number of BM-MNCs in the transplanted bone marrow, potentially contributing to the positive results of cell therapy.
Patients who received renin-angiotensin-acting agents (RAS-acting agents) experienced decreased pain and improved TcPO2. 118 These findings suggest that atorvastatin and RAS-acting agents may enhance the therapeutic effects of BMC treatment in patients with CLTI, potentially conferring benefits for limb salvage and wound healing in this high-risk population. 117
Various medications utilized in the management of diabetes have demonstrated an ability to increase the number of circulating stem cells, particularly endothelial progenitor cells. Metformin has been observed to enhance both the quantity and functionality of endothelial progenitor cells. 119 Thiazolidinediones (TZDs), like pioglitazone and rosiglitazone, as well as Glucagon Peptide Like-1 (GLP-1) receptor agonists, such as exenatide and liraglutide, have also demonstrated the capacity to enhance the function and numbers of circulating endothelial progenitor cells.120 -122 Furthermore, dipeptidyl peptidase-4 (DPP-4) inhibitors such as sitagliptin and saxagliptin, as well as sodium-glucose cotransporter-2 (SGLT2) inhibitors including empagliflozin and canagliflozin, have demonstrated efficacy in enhancing the quantity of endothelial progenitor cells.123,124
To effectively address the complex interactions of regenerative therapies and improve outcomes, it is essential to focus on the patients’ preparation and cell harvesting process. Harvesting EPCs from bone marrow has been demonstrated to impact both the quality and clinical outcomes.77,125
Extracting bone marrow from the iliac crest without using centrifugation seems to be more effective in terms of cell yield and shows less contamination from peripheral blood. The presence of contaminating red blood cells negatively impacts the functionality of the isolated bone marrow cells. 126 Preliminary results suggest improved outcomes in critical limb ischemia patients.77,125
BMC aspirate contains megakaryocytes, platelets, and cytokines. 127
Recent transcriptomic advancements reveal the complexity and heterogeneity of megakaryocytes, highlighting their multifaceted roles beyond platelet production, including significant contributions to immune functions and inflammatory responses. 128
In a recent study, Nazzal et al 129 investigated the angiogenic properties of megakaryocytes (MKs) and their conditioned media (CM) on bone marrow endothelial cells (BMECs), with a focus on the differences between young and aged, male and female MKs. The findings revealed that MK CM from both young and aged female mice significantly enhanced BMEC proliferation (over 65%) and vessel formation (>115%). 129 Additionally, young male MK CM notably increased vessel formation by 160%. 129
Aged male MK CM significantly boosts vascular node and mesh formation (>150%) while reducing vessel quantity by 62% compared with young male MK CM. 129 Aged female MK CM greatly increases cellular migration by >250% but is less effective in wound closure. 129
Of note, MK present in the blood are different from the ones of bone marrow and in the cord blood. 129 MK could be involved in angiogenesis, immune responses, inflammation. 129
As per the findings of Dregalla et al, it has been established that platelets derived from bone marrow exhibit a unique secretome profile, which differs significantly from those derived from peripheral blood. This distinct profile is characterized by a high concentration of anti-inflammatory cytokines. 130
As previously reported, the quantity and functionality of PBMNCs are diminished in CLTI patients. 85
The process of peripheral blood filtration is characterized by a high concentration of erythrocytes that can induce eryptosis.126,131,132 Eryptosis is the programmed cell death of red blood cells (RBC). 131 The filtration process and the surrounding tissue environment contribute to cellular damage, stress, and energy depletion, ultimately leading to eryptosis. The breakdown of red blood cells (RBCs) results in the release of pro-inflammatory cytokines, such as macrophage migration inhibitory factor. Furthermore, the inflammatory response is exacerbated by the presence of free hemoglobin (Hb) and its derivatives, oxyhemoglobin (oxyHb) and ferry hemoglobin (ferryHb). The intricate process of eryptosis induces oxidative stress, tissue damage, and the activation of inflammatory pathways.
The clinical outcomes reported in the Italian experience of laboratory and observational studies with a focus on diabetic foot and ulcer healing, are likely attributable to the presence of an elevated platelet count.133 -135
As chronic and inflammatory disease, diabetes and arteriosclerosis cause a profound alteration in innate and adaptive immune mechanisms and cellular component.136,137
The result of filtration could be classified as leucocyte platelet rich plasma with one time increase of platelets toward basal level and relevant presence of red blood cells (Red L- PRP IA3).131,138
In the process of angiogenesis, platelets play a vital role by aiding endothelial cell growth, movement, and the development of new blood vessels.139 -144 The association of BMNC with an appropriately formulated platelet rich plasma may potentially enhance the efficacy of the treatment approach. 145
Laboratory medicine is essential for advancing stem cell therapies for vascular regeneration, providing methods to characterize stem cells, understand differentiation, and evaluate therapeutic potential. However, laboratory results emphasize the need for extensive clinical validation to ensure the safety and efficacy of these therapies in humans.
It is recommended to conduct a preoperative count of circulating EPCs (at least CD34+, KDR) to achieve optimal results.
Laboratory Medicine and Regenerative Medicine
Laboratory Medicine aids Regenerative Medicine by:
Quantifying analytes and biomarkers integral to human biological regeneration.
Identifying, separating, and preserving cell derivatives or lines for regenerative therapy.
Identifying and monitoring biomarkers to predict disease risk in individuals, utilizing artificial intelligence to generate personalized risk indices based on clinical and family history.
Laboratory Medicine Techniques and Regenerative Medicine
Flow Cytometry
The first flow cytometer, based on impedance measurement and Coulter’s principle, was developed by Wallace H. Coulter and Mack Fulwyler in 1963. 146 Flow cytometry employs lasers to generate scatter and fluorescence signals, captured by photodiodes or photomultiplier tubes and converted into electronic data, stored as standardized (.fcs) files. This method allows for the analysis or purification of cell populations based on their light scattering or fluorescence characteristics. Flowcytometry utilizes a range of fluorescent reagents, including conjugated antibodies and various dyes, making it indispensable in fields like immunology, molecular biology, and cancer research. Over the last 3 decades, it has significantly advanced the study of the immune system and cell biology, notably facilitating the identification, counting, and separation of viable cell lines for Cell-Based Therapies. 147
Conclusions
Vascular Surgery has exhibited a dynamic and adaptable nature since its onset. In the final decade of the 20th century, endovascular surgery emerged as a significant addition to the therapeutic armamentarium, subsequently followed by the development of hybrid surgical techniques and enhanced recovery after surgery protocols.
Surgical intervention, even when accompanied by appropriate pharmacotherapy, is not always sufficient to address the complex issues of patients suffering from pathologies, particularly those affecting the lower limbs, complicated by critical comorbidities. Unfortunately, PAD and CLTI are increasing in the population. Many patients are not candidates for revascularization, or the procedure is only partially successful. The potential benefit provided by regenerative medicine is associated with the neovascularization process, involving some or all aspects of angiogenesis, arteriogenesis, and vasculogenesis.
Cell-based therapies have emerged as a promising avenue for treating various vascular diseases, offering novel methods to stimulate angiogenesis, repair blood vessels, and regenerate tissue. Extensive preclinical research has been conducted to assess the safety and effectiveness of stem cell treatments for vascular conditions. 148 Researchers have employed animal models of ischemic heart diseases, PAD, and other vascular disorders to explore the potential of stem cells in promoting blood vessel growth, decreasing inflammation, and enhancing tissue blood flow. For PAD patients with CLTI, gene therapy primarily aims to induce therapeutic angiogenesis.149,150
Biomaterials represent a potentially revolutionary approach to CLTI treatment, providing a means to improve cell-based therapies and enhance clinical outcomes. To effectively treat CLTI, bioengineering strategies are crucial for improving the viability and functionality of transplanted cells.
The field of Vascular Surgery is continuously evolving, capitalizing on the advancements in modern biomedical technology to combat diseases that predominantly affect the elderly population, whose average lifespan has increased. This specialty employs cutting-edge and appropriate methods to address these conditions. Moreover, younger patients, whether due to multiple health issues or the need for effective reintegration into society and the workforce, can increasingly benefit from the innovative approaches offered by autologous cell therapy and regenerative medicine.
In conclusion, PAD with CLTI represents a severe and critical condition. Despite optimal medical therapy, vascular and endovascular surgery interventions may prove insufficient or ineffective, potentially resulting in amputation. Autologous cell therapy and regenerative medicine can provide support as complementary or alternative options to conventional surgical and/or medical therapies.
These advanced techniques are becoming more refined and substantive in their application in clinical practice. Further research, clinical studies, and new evidence are necessary.
Personalized medicine represents the contemporary paradigm that calls for consideration of the individual patient’s clinical conditions, risk factors, and biomarkers.
Footnotes
Author Contributions
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and, (3) final approval of the version to be published.
Funding
The authors disclosed receipt of the following financial support forthe research, authorship, and/or publication of this article: This work has been supported by Ministry of Health – Ricerca Corrente to IRCCS MultiMedica.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Not applicable.
