Abstract
Ethyl chloride (EC) spray is a vapocoolant which is used in interventional procedures as a topical local anesthetic. The aim of the present study was to investigate whether or not EC spray can facilitate radial coronary angiography. The study was conducted on a study group EC of 106 (age: 58.1 ± 12.4) patients and a control group (placebo) of 104 (age: 59.1 ± 10.8) patients. Visual analogue scale scores (VAS), radial cannulation failure (RCF), the rates of clinical and anatomical radial artery spasm (RAS), pain lasting >2 day and >1 week, and radial artery occlusion (RAO) at first week and first month were evaluated in both groups. VAS scores, the rates of RCF, clinical and anatomical RAS and pain lasting >1 week were found significantly lower in the study group than the control group (p < .001, p = .024, p=.009, p = .028 and p = .042, respectively). But the rates of RAO were similar in both groups. Topical spray including EC, which has low-cost and rapid efficacy, may be preferred in transradial coronary angiography as it reduces the risk of RCF and RAS, and increases patient comfort with its analgesic effect, without significant side-effects.
Keywords
Introduction
Vapocoolants are local anesthetics in liquid form that provide temporary desensitization in the pain receptors of the region by evaporating and creating a sudden temperature drop in the region where topically applied. 1 Ethyl chloride (EC) is widely used as a vapocoolant in minor surgical procedures, minor sports injuries, in anesthesia before injection, and in myofacial pain. 2 Conflicting results have been reported in previous studies that investigated the effect of EC on the pain during radial arterial puncture performed for arterial blood gas analysis. In two studies, no superiority was shown for EC spray over placebo or ice application,3,4 while another study reported that EC spray provided a significant decrease in pain compared with lidocaine. 5
In coronary angiography, the transradial approach is preferred to the transfemoral approach because of the advantages of lower vascular complication rates, shorter length of stay in hospital, and increased patient comfort. 6 However, radial artery spasm (RAS) seen at high rates such as 30%, continues to be the leading cause of failure of the procedure.7-10
In a previous study, a eutectic mixture of local anesthetic cream containing lidocaine and prilocaine (EMLA®; Laboratoire ASTRA, Manterre, France) reduced radial pain and the sympathetic response in radial angiography. 11 In the present study, we investigated whether or not EC spray, which acts more quickly and is applied more practically compared with this cream, reduces pain, RAS, and other complications in radial angiography.
Methods
This study was designed as a single-center, prospective, randomized, placebo-controlled, and double-blinded. Approval for the study was granted by the Local Ethics Committee. Patients who were planned to undergo diagnostic coronary angiography with transradial artery approach between January 2023 and April 2023 were screened. Patients were included if they had a diagnosis of chronic coronary syndromes due to symptoms or evidence of ischemia with a non-invasive test. Patients were excluded from the study if they had a known history of EC allergy, a history of cold intolerance (Raynaud phenomenon), used relaxant or sedative topical or oral analgesic drugs within the last 24 h, had any emergency coronary angiography indication (myocardial infarction, cardiac shock), had a history of anticoagulant drug use for any indication, had an abnormal Allen test for the wrist where the puncture was planned, required a procedure other than angiography, had undergone a coronary bypass, and were diagnosed with any cognitive disorder. After the implementation of these criteria, a total of 224 patients provided written informed consent and were included in the study (Figure 1). Study flowchart.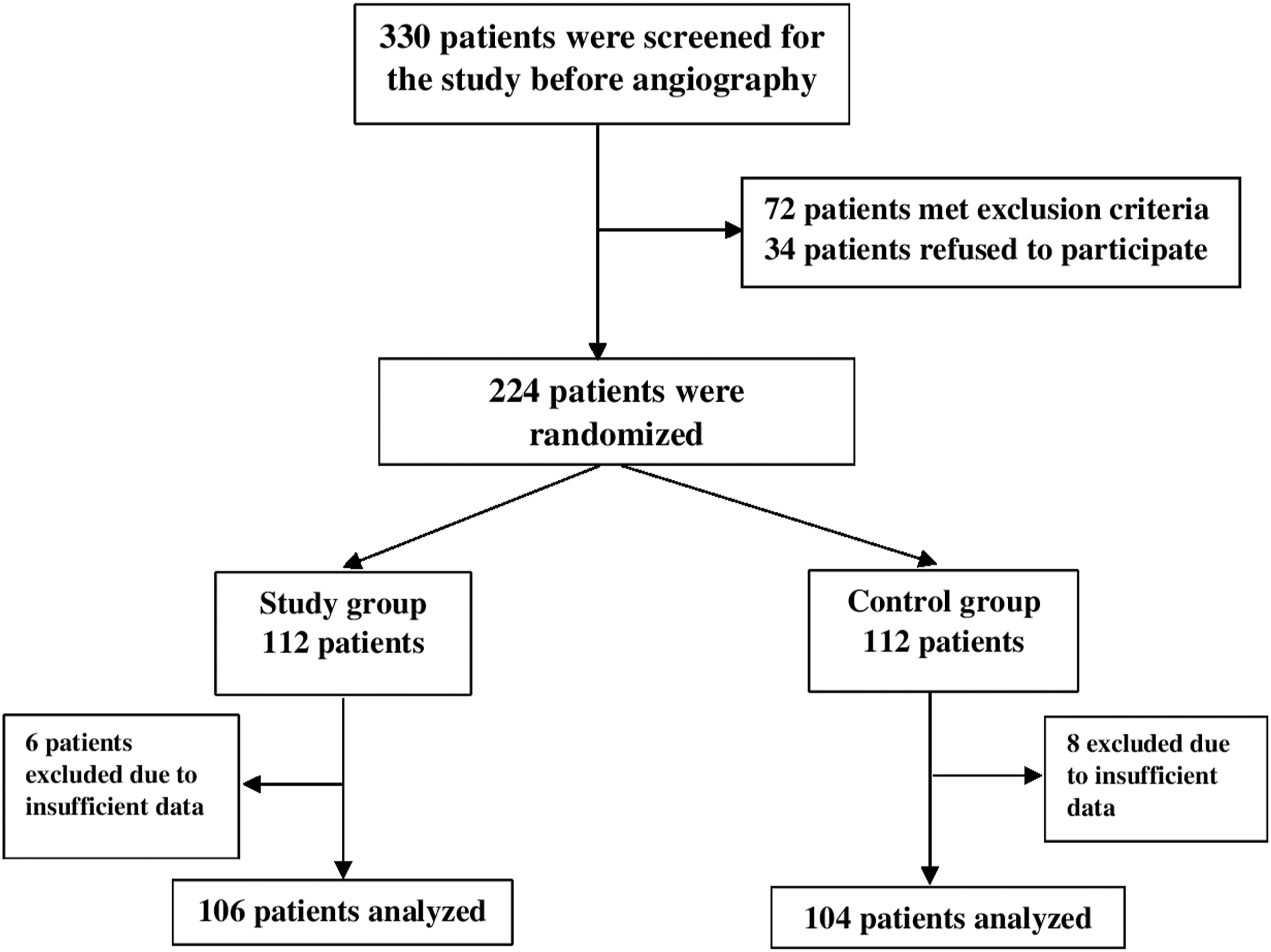
Radial artery cannulation (placement of the sheath through the radial artery) was performed in the radial artery of the arm that was preferred, by a single experienced operator. In supine position, the preferred arm was placed at the side of the patient’s body and the wrist was moved into hyperextension. The patient was told to turn their head to the opposite side of the arm then the patient was told that a spray would be applied to the arm to reduce procedure related pain. Taking the target as 2 cm above the styloid notch of the radial bone, the EC spray (Clorethyl spray®, EBT Health, Bursa, Turkey) was then applied for approximately 3 s from a height of 10 cm to the study group of patients and a placebo spray (0.9% isotonic sodium chloride solution at room temperature) was applied to the control group.
The spray bottles used for both groups were of similar size, unlabeled, and color-coded for content. Deciding to which group the patients would be assigned was made by an observer using computer program-generated random numbers. After application of the spray, the puncture area was wiped with 10% povidone iodine antiseptic solution. Before the radial artery cannulation, local anesthesia of 1 mL 2% lidocaine was administered to the puncture area subcutaneously with a 25-gauge needle (Beybi, Turkey) in both study and placebo groups. Any local anesthetic except for lidocaine and EC/placebo spray was not used. Immediately after the radial artery puncture performed with a 21-gauge needle (TERUMO, Tokyo, Japan), a 0.018-inch guidewire (TERUMO, Japan) was advanced through the needle. The needle was removed then a 10 cm long 6F hydrophilic sheath (TERUMO, Japan) was advanced over the wire. Immediately after placement of the sheath, 5000 units of heparin, 200 µg nitroglycerine, and 5 mL physiological saline were consecutively administered through the radial artery.
The level of pain was evaluated with a Visual Analogue Scale (VAS) just after the administration of heparin and nitroglycerine within the sheath. The scale consists of a 100 mm horizontal line, marked in 10 mm sections, where 0 mm represents “no pain” and 100 mm represents “intolerable pain.” The scale was held so that the patients could easily see it, then the patients were told that a nurse would move a pen slowly from 0 to 100 mm along the scale. The patients were instructed to stop the nurse when she reached a point corresponding to the level of pain they felt at that time. Values >30 mm are evaluated as moderate pain and values >54 mm as severe pain. 12
The number of punctures and puncture time were recorded for each patient. The puncture time was defined as the time interval from first puncture attempt to successful puncture. The total procedure time was defined as the time from the application of local anesthesia to completion of the angiography. Inability to perform the cannulation because of reduced or loss of pulse in the radial artery during the puncture intervention and therefore conversion to the transfemoral approach, was accepted as radial cannulation failure (RCF). After removal of the sheath, a compression device was placed on the radial artery in accordance with the manufacturer’s instructions, and was left in place for 2 h (Seal-One®, Hauts de France, France). At the 4th h after the procedure, the area of the procedure was evaluated in respect of pulsation, hematoma, redness, paleness, thrombophlebitis, and local skin reaction.
Radial Artery Spasm
Radial Artery Spasm (RAS) was evaluated both anatomically and clinically. Software was used for the anatomic evaluation of RAS (Automatic Reference Analysis, Siemens Axiom Artis zee, München, Germany). The radial arteriography image of a 40 mm radial artery segment from the distal tip of the sheath was obtained by injecting contrast agent through the radial sheath before the catheter was advanced. The mean radial artery diameter in the 40 mm segment was measured by the software and called as reference diameter (rd). The similar image was obtained after the coronary angiography procedure was completed. The mean radial artery diameter measured on that image was called as control diameter (cd). A narrowing of >50% was accepted as anatomical RAS. The amount of narrowing was calculated with the formula of “rd-cd/rd” (Figure 2). Determination of anatomical radial artery spasm, 2a: measurement of radial artery diameter before angiography, 2b: measurement of radial artery diameter after angiography.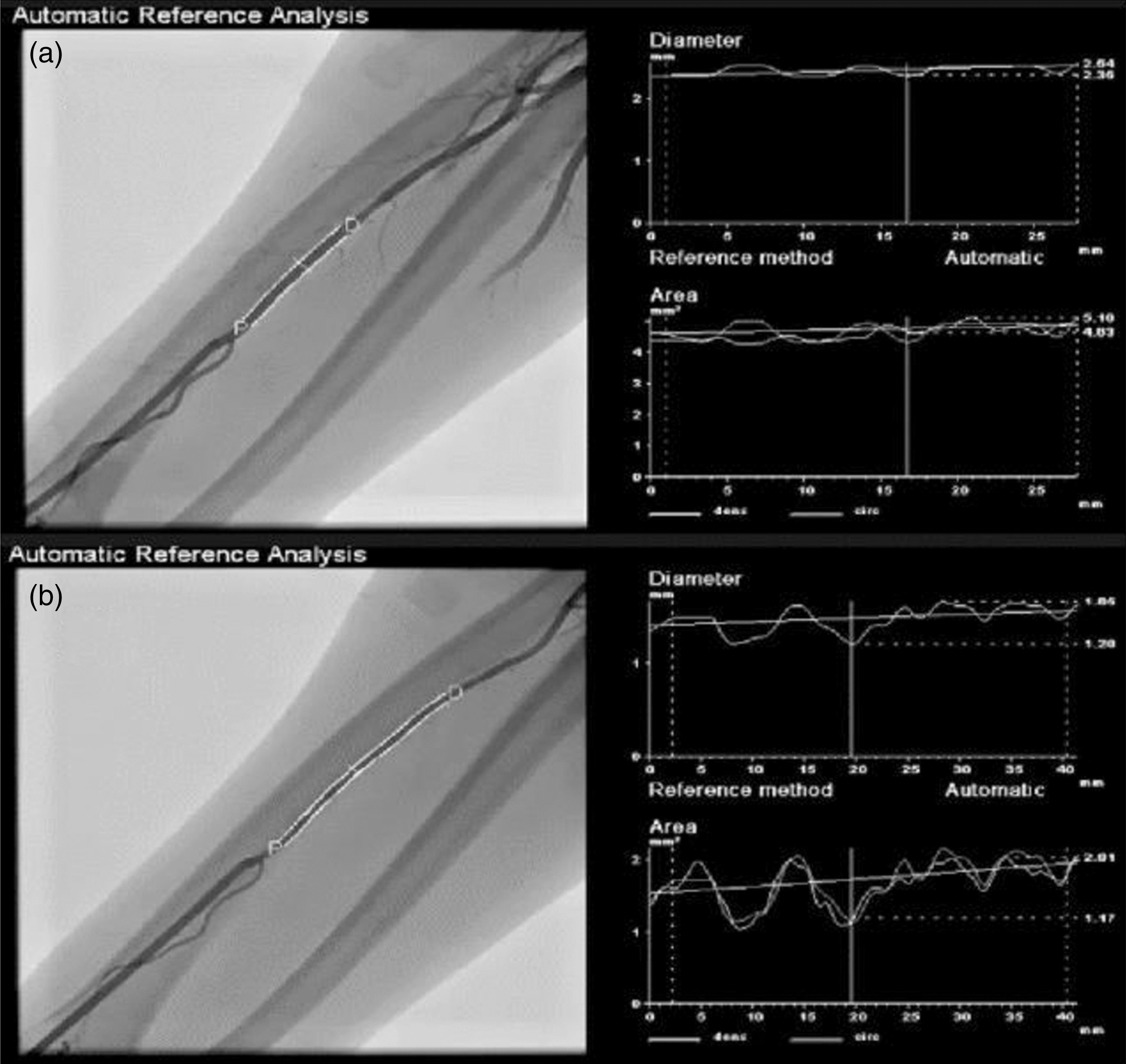
In the clinical examination, the presence of at least two of persistent forearm pain, a painful response to catheter manipulation, pain on withdrawal of the sheath, or difficult catheter manipulation, was accepted as clinical RAS. 11
Radial Artery Occlusion
The patients were called for follow-up examination at 1 week and 1 month after discharge. Ultrasonographic evaluation (7.5 MHz linear transducer, Toshiba USDI-790 A, Otawara, Japan) of the radial artery was made by an experienced operator blinded to the patient data at each follow-up visit. Radial artery occlusion (RAO) was evaluated as the obstruction of radial artery on two dimensional ultrasound imaging and the absence of Doppler flow signal at the cannulation site.
Statistical Analysis
All statistical analyses were performed using IBM SPSS Statistics, version 26.0 (IBM Corp, Armonk, NY, USA). According to the Kolmogorov–Smirnov test, continuous variables showing normal distribution were stated as mean ± standard deviation values, and those not showing normal distribution as median (25th–75th interquartile range) values. Categorical variables were stated as number (n) and percentage (%). Comparisons of demographic, clinical, and procedure-related variables between the study and control groups were made using the Independent Samples t-test for continuous variables showing normal distribution and with the Mann–Whitney U-test for continuous variables not showing normal distribution. The Chi-square test or Fisher’s Exact test were used for comparisons of categorical variables. A 2-sided p < .05 was accepted as statistically significant.
Results
Demographic, laboratory findings, and medications of the study and control groups.
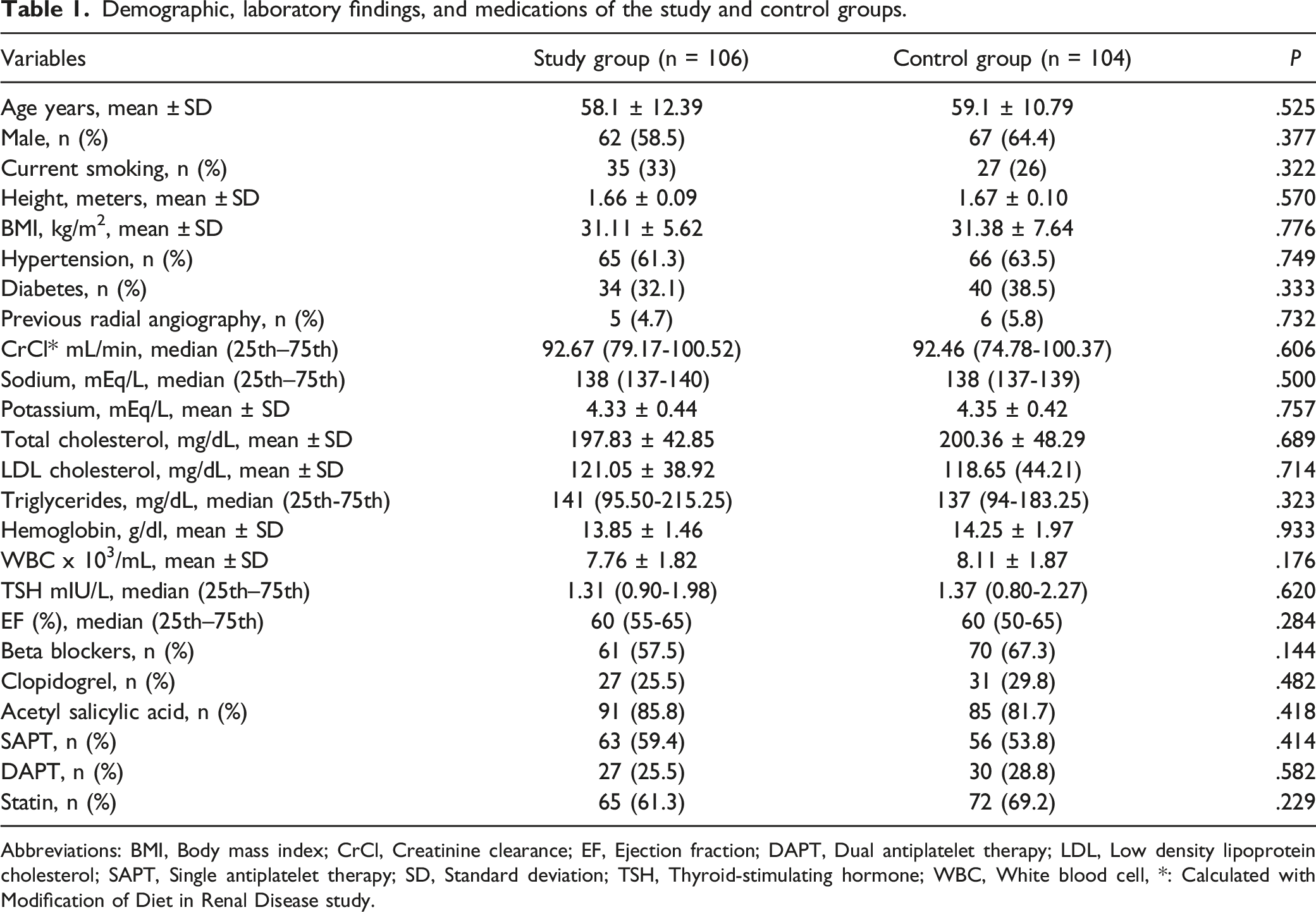
Abbreviations: BMI, Body mass index; CrCl, Creatinine clearance; EF, Ejection fraction; DAPT, Dual antiplatelet therapy; LDL, Low density lipoprotein cholesterol; SAPT, Single antiplatelet therapy; SD, Standard deviation; TSH, Thyroid-stimulating hormone; WBC, White blood cell, *: Calculated with Modification of Diet in Renal Disease study.
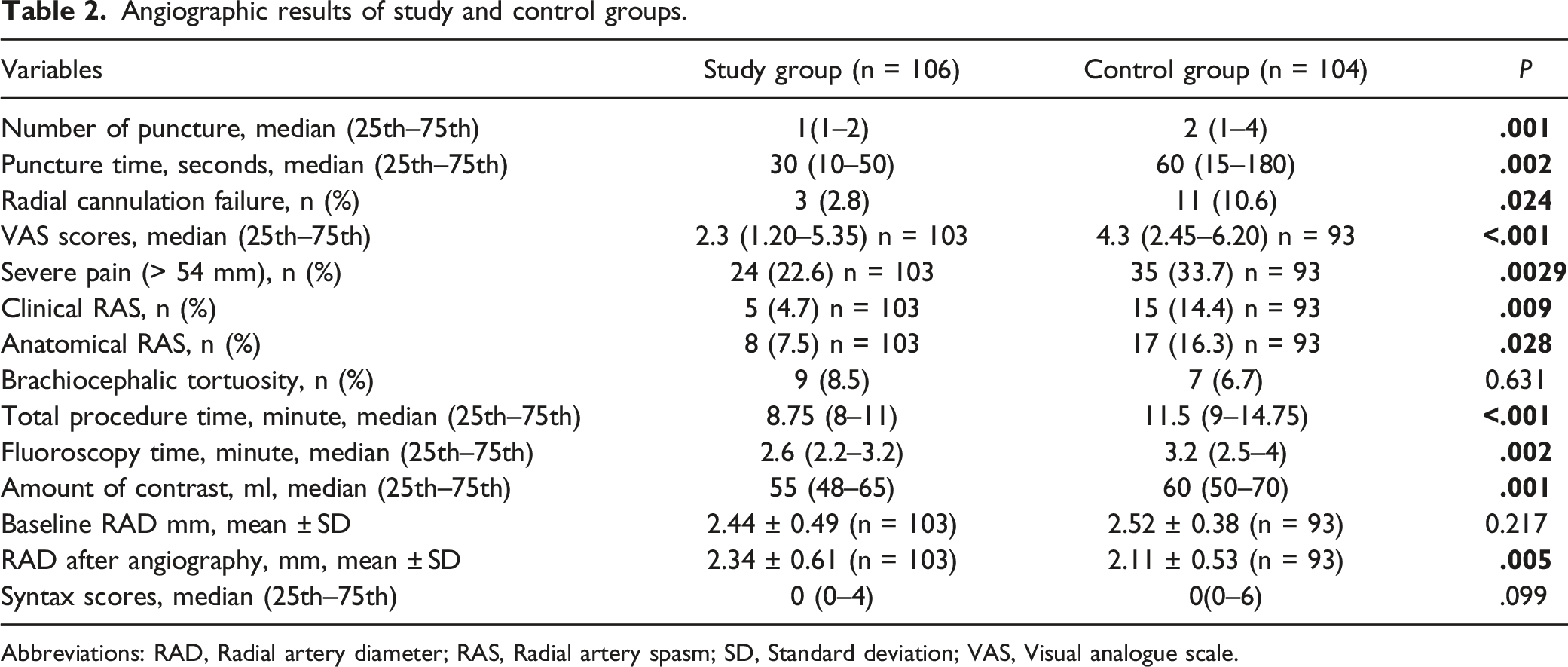
The median number of punctures, puncture time, VAS pain score (Figure 3), amount of contrast agent used, and total procedure time were found to be significantly lower in the study group than in the control group. Both clinical RAS and anatomical RAS were observed at a significantly lower rate in the study group than in the control group (p = .009, p = .028, respectively). The comparisons of the procedure-related variables are shown in Table 2. Visual analogue scale (VAS) scores of study and control groups. Angiographic results of study and control groups. Abbreviations: RAD, Radial artery diameter; RAS, Radial artery spasm; SD, Standard deviation; VAS, Visual analogue scale.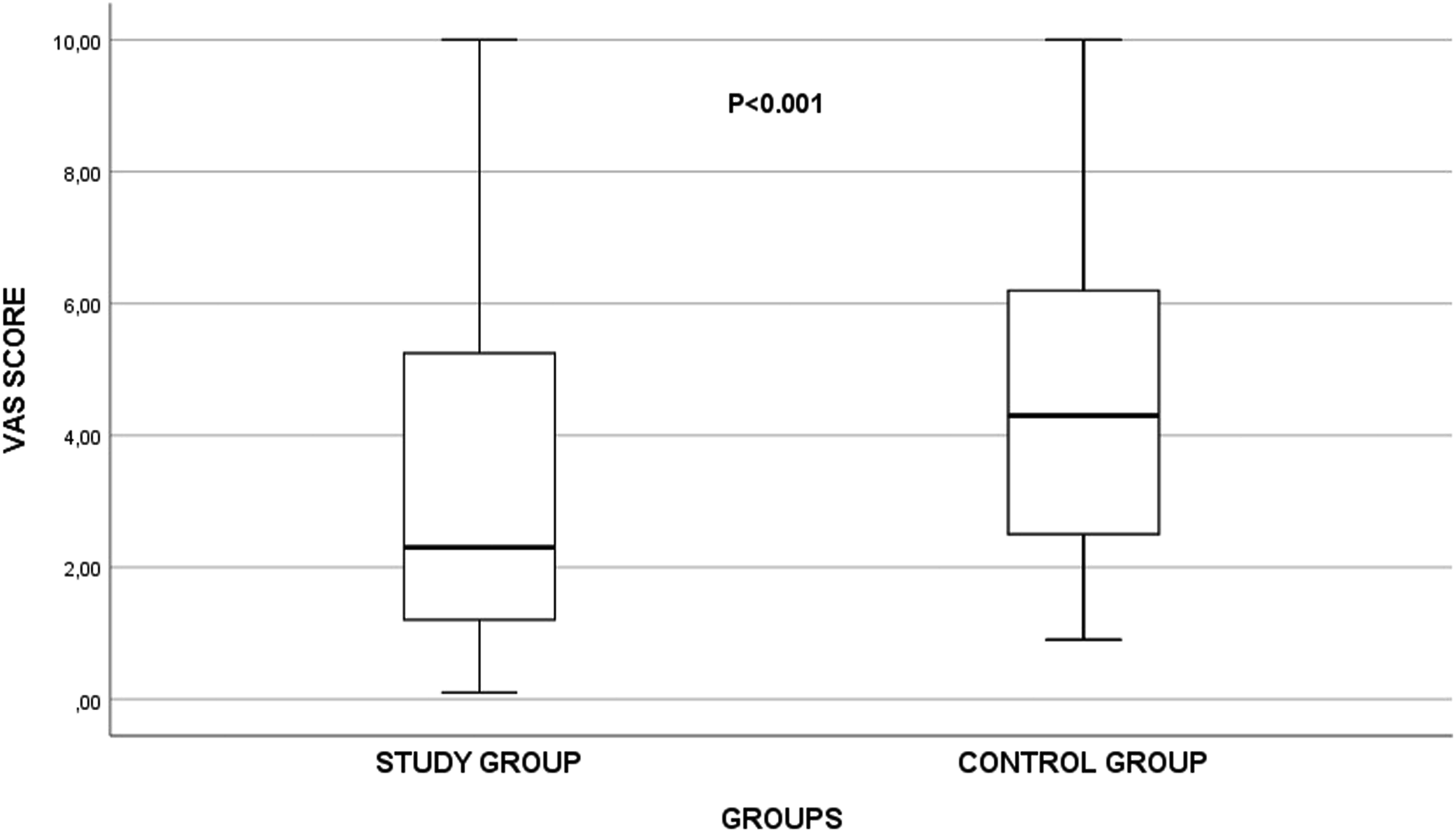
Follow up results of study and control groups.
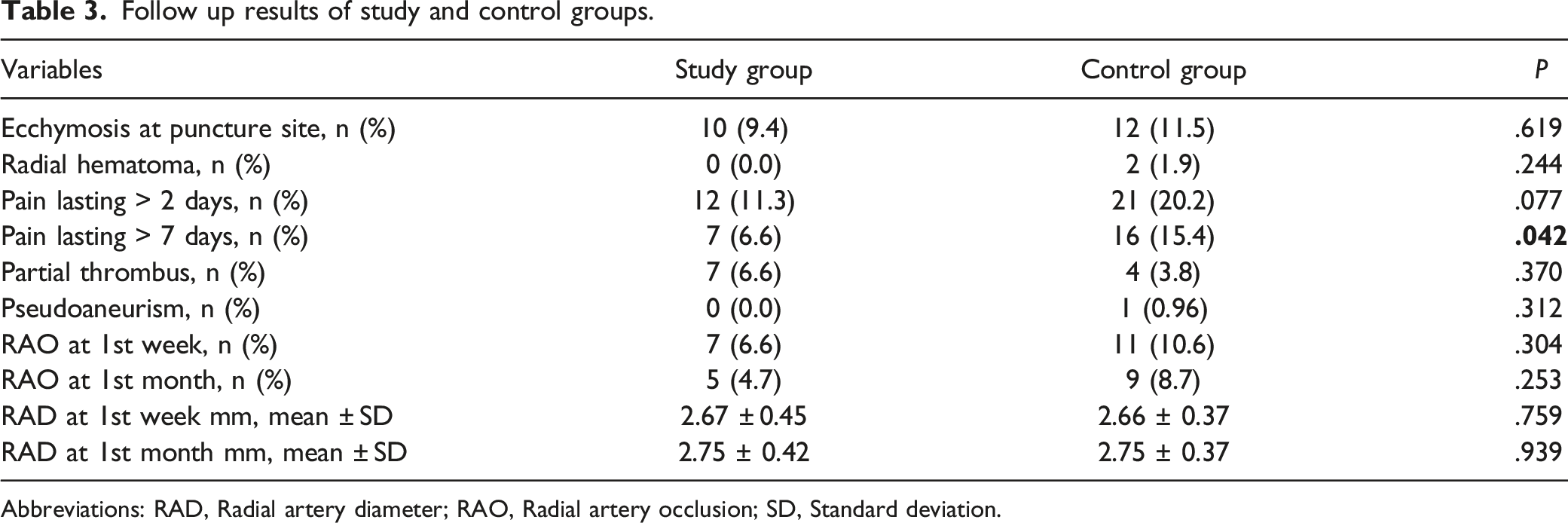
Abbreviations: RAD, Radial artery diameter; RAO, Radial artery occlusion; SD, Standard deviation.
Discussion
The aim of this study was to investigate whether a cooling spray with EC creating a local cryoanesthetic effect facilitated transradial coronary angiography and whether or not it reduced procedure-related complications. The results demonstrated that the rates of RCF, number of punctures, puncture time, total procedure time, peri-procedural pain, prolonged pain, clinical RAS, and anatomical RAS were lower in the study group. No increase in local or systemic reaction was observed due to EC, and no significant difference was found between the groups in respect of RAO rates.
Despite reports in literature of freezing, contact dermatitis, and even death associated with the use of EC, especially in conditions of prolonged exposure, such side-effects were not observed in any patient in the present study.13-15 The rate of itching (2.8%) observed in the area of the intervention was found to be lower than the 4.4% reported in the literature. 1
In previous radial angiography series, RAS rates have been observed to be between 5% and 30%7,9,16 whereas in the present study, RAS (clinical and anatomical) was observed in 12.9% of all the 196 patients (clinical RAS: 9.5%, anatomical RAS: 11.9%, clinical + anatomical RAS: 8.5%). In a systemic review of 3 studies that included a total of 697 patients undergoing transradial coronary intervention, 2 studies used a topical eutectic mixture of local anesthetic (EMLA) and 1 used topical lidocaine + nitroglycerine, and a significant decrease in the RAS rate was determined only in the patients where EMLA was used, compared with subcutaneous lidocaine infiltration. It was also shown that pain was significantly reduced in the EMLA group.11,17-19 In the present study, similar to EMLA, EC spray reduced procedure-related pain, the rates of RCF and RAS compared with placebo when applied before subcutaneous lidocaine infiltration. Compared with EMLA, the use of EC spray is more advantageous as it is low-cost and shows an effect much more quickly (in seconds).2,20
The radial artery is predisposed to spasm as it contains a thick smooth muscle layer and dense alpha-1 adrenergic receptors.21,22 Increased expression of catecholamines (adrenalin, noradrenalin) due to pain causing vasoconstriction mediated by alpha-1 receptors may be a factor increasing RAS.23,24 Thus, the decreased perception of pain due to the use of topical EC could explain the lower rate of RAS. It was thought that RCF due to decrease or loss in pulse in 14 patients could be associated with RAS developing due to repeated unsuccessful punctures. 25 The lower rate of RCF and lower number of unsuccessful punctures in the study group may be attributed to significant reduction in pain in that group.
Despite the short-lasting effect of EC, long lasting pain (≥ 7 days) was seen at a lower rate in the study group, and this may have been due to the lower number of unsuccessful punctures and the shorter procedure/cannulation time in that group.
This was a single-center study in which a limited number of patients included. Another limitation was that the sympathetic response developing after radial pain was not evaluated. In addition, the possibility that participants could distinguish between EC spray and placebo was another limitation. Finally, other factors, such as anxiety, affecting RCF, and RAS were not evaluated.
Conclusion
Topical spray including EC, which has low-cost and rapid efficacy, may be preferred in transradial coronary angiography as it reduces the risk of RCF and RAS, and increases patient comfort with its analgesic effect, without significant side-effects. There is a need for further studies to support these results.
Footnotes
Acknowledgements
We would like to thank Prof. Dr. Hasan Ari and Prof. Dr. Tahsin Bozat for their valuable contributions to our study.
Author Contribution
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
