Abstract
Stent under-expansion is a predictor of restenosis and stent thrombosis. It remains uncertain whether enhanced stent imaging (ESI) (CLEARstent) guidance can improve stent under-expansion. Our aim was to assess the effect of using ESI on stent under-expansion, after percutaneous coronary intervention (PCI) in a single center, cross-sectional observational study. Participants attending our cardiology clinic with stable angina or acute coronary syndrome, from March to September 2020 were recruited. A total of 164 patients who underwent post-PCI ESI (CLEARstent) were compared with 77 age- and sex-matched control patients. Post-procedural minimal lumen diameter (MLD) was calculated. The patients in the ESI-guided PCI group, had a median age of 61 (54–69 IQR 25-75) years and 76.8% (n = 126) were males. The patients in ESI-guided PCI group had a greater minimal lumen diameter compared with the X-ray guided PCI group (βeta coefficient:2.88 (95% CI:2.58–2.99) vs βeta coefficient 2.55 (95% CI 2.34–2.63), P < .001). Our finding supports the use of the ESI system to optimize stent placement as expressed by the MLD.
Introduction
Coronary angiography (CAG) and percutaneous coronary intervention (PCI) have been pivotal diagnostic and therapeutic tools for patients with coronary artery disease (CAD). Coronary stent under-expansion is not only important preventable factor of in-stent restenosis (ISR), but is also associated with stent thrombosis (ST)1-3 ST is a serious PCI complication, which has a substantial adverse impact on survival with a mortality rate of 23% and other complications (e.g., stroke, major bleeding, and emergency PCI). 4 One of the strongest predictors of ST is stent under-expansion, which increases the relative risk by approximately 13 times. 5 The contemporary rate of ISR for PCI is approximately 10% and under-expansion of the coronary stent is still an independent determinant of ISR causing ischemia.6,7 Therefore, appropriate coronary stent deployment during PCI is crucial. It has been noted that, intravascular ultrasound (IVUS) guided PCI is associated with lower rates of major adverse cardiovascular events than angiography-guided stent implantation. 8
Enhanced stent imaging (ESI) is a novel software based on angiographic imaging that improves coronary stent visualization and provides advantages regarding quantitative analysis of stent dimensions. 9 Although IVUS is the gold standard technique for the assessment of adequate stent expansion, correlation of minimum stent diameter is higher between IVUS and ESI when compared with IVUS and quantitative coronary angiography (QCA) or ESI and QCA. For this reason, ESI is a potential alternative imaging tool for the assessment of stent deployment in clinical practice with some advantages such as easy application and cost-effectiveness as well as requiring less training and no additional equipment.10,11
As ESI has favorable practical features, we aimed to find out whether it achieves better procedural success in terms of minimal lumen diameter (MLD) calculated by QCA compared with conventional PCI.
Patients and Methods
Study Population
We conducted a cross-sectional study. All patients who were hospitalized from March to September 2020 with stable angina or acute coronary syndrome were included in the study. Patients younger than 18 years old and/or who were not hospitalized were excluded from the study. Exclusion criteria included treatment with thrombolytic drugs in the previous 24 h, contraindications for dual antiplatelet therapy (active gastrointestinal bleeding, recent hemorrhagic stroke, and allergy to aspirin), cardiogenic shock and previous coronary artery bypass graft (CABG). A total of 164 patients who underwent post-PCI ESI were compared with 77 age-sex-matched control patients.
The local ethics committee of Dr Siyami Ersek Thoracic and Cardiovascular Research and Training Hospital approved the study. All enrolled patients signed written informed consents.
Data Collection
All data were obtained from hospital digital health record systems, including the “Health Management System module” specific data (symptoms, biomarkers, medication, and clinical outcomes during index hospitalization).
Procedure
Conventional stent implantations were performed with angiograms in 2 orthogonal projections. Whether to post-dilate the stent based on the post-stent angiogram was a decision made by the operator. ESI images and documenting the operator’s decision whether the ESI images changed the decision to post-dilatation the stent. This decision was made by the operator discretion. An example of an ESI is shown in Figure 1. The decision for the post-dilatation was made by the 9 experienced interventional cardiologist who were working together in the same clinic for a long time. All patients undergoing angioplasty received a loading dose of aspirin 300 mg and either clopidogrel 600 mg or ticagrelor 180 mg or 60 mg prasugrel prior to the procedure. All patients then received regular 75 mg aspirin and 75 mg clopidogrel or 10 mg prasugrel or 180 mg ticagrelor daily maintenance therapy. Drug eluting stents were implanted for all patients. A/B: Good apposition appearance after the stent implantation in the standard PCI, C: We changed our decision after Enhanced Stent Imaging (ESI) and performed non-compliant balloon dilatation in this patient.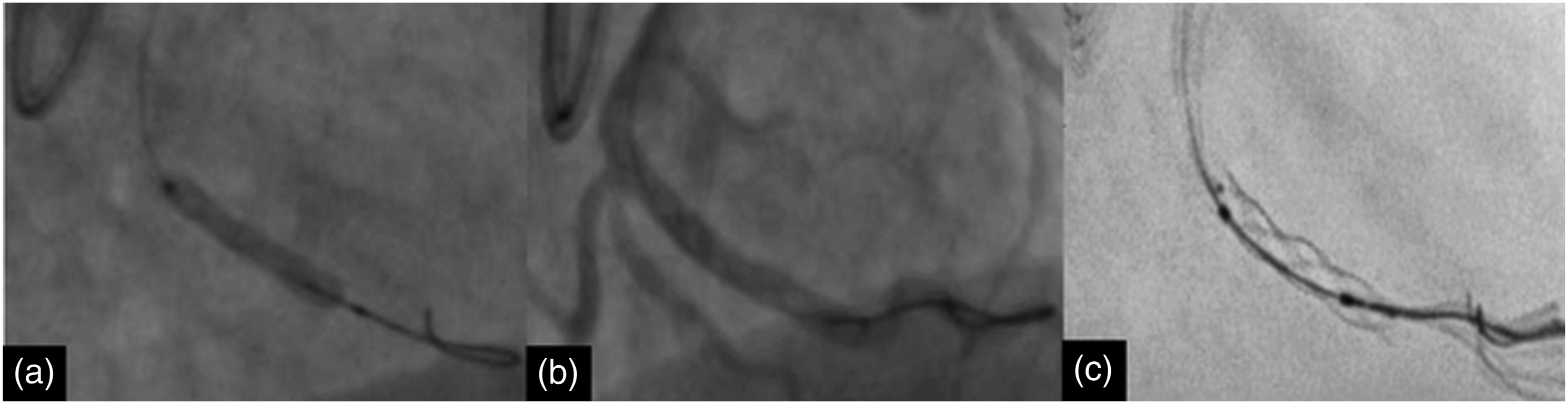
Enhanced Stent Imaging Procedure
CLEARstent is an image processing software system (Siemens Healthcare, Munich, Germany) designed as an add-on to conventional X-ray angiographic systems. After stent deployment and balloon deflation, an ESI is produced from a minimum of 20 cine frames over 3 sec using the radiopaque markers of the delivery balloon as an anchor to align the stent across all frames. The CLEARstent system automatically acquires the cine images to create a still image of the stent with enhanced edges and the associated region of interest. Stent visibility was assessed using both standard angiography and ESI. Following the procedure, ESI diameter measurements were obtained independent of and blinded to the QCA measurements.
Outcome Variables Measurements
Post-procedural minimal lumen diameter (MLD) was calculated using a quantitative coronary angiography module. QCA analysis was performed using a validated semi-automated QCA software (Siemens Healthcare, Munich, Germany) Frames for QCA analysis were selected from fully opacified angiograms that provided optimal visualization of the lesion/treated segment with the least degree of foreshortening. Calibration was performed with the use of the contrast-filled guiding catheter as the reference. Post-PCI diameters of the stented segments as well as the diameters at the proximal and distal stent edges were obtained. QCA measurements were obtained independent of and blinded to the ESI measurements.
Statistics
Continuous variables distribution assessed by histogram and Shapiro–Wilks’s test. Numerical variables were represented as median and interquartile ranges (25–75 IQR) or mean and standard deviation; for comparisons, the t-test or Mann–Whitney-U test were used. Categorical variables were expressed as percentages and number and for comparison the chi-square test was used.
Main Candidate Predictor and Adjustment Variables
The predictors for the primary outcome, which the intervention of interest and other variables in regression analysis should plausibly have an effect and have optimally with preliminary evidence of an effect.12,13 We considered all candidate predictors that we included in the model under these principles. The main candidate predictor, ESI usage was included in the model. Adjustment variables were determined as age, pre-dilation, classification, post-dilation, lesion type, stent diameter, stent balloon atmosphere (ATM), and post-clear stent extra-balloon usage.
Statistical Modeling
To examine the relationship between MLD and the CLEARstent system, we used the ordinary least square regression model. The associations between candidate predictors and MLD were quantified by the adjusted Beta coefficient with a 95% confidence interval (CI). We did not remove any of these predictors based on statistical significance. To visualize the unique effect of predictors while accounting for other variable in regression, a partial effect plot is presented by plotting on the horizontal axis, against the residuals of MLD on the vertical axis.
The variables having missing value >10% were not included in the model while for that <10%, with the assumption of missing at random, multiple imputations were used either to minimize bias or to avoid exclusions of participants. Multiple imputations were applied for missing values using aregImpute function (rms R package). For analyses, R-software version 4.01 (Vienna, Austria) with “rms” and “desctool” packages were used.
Results
Baseline Clinic Laboratory and Procedure-Related Factors Analysis Between X-Ray Guided PCI and ESI-Guided PCI Group.
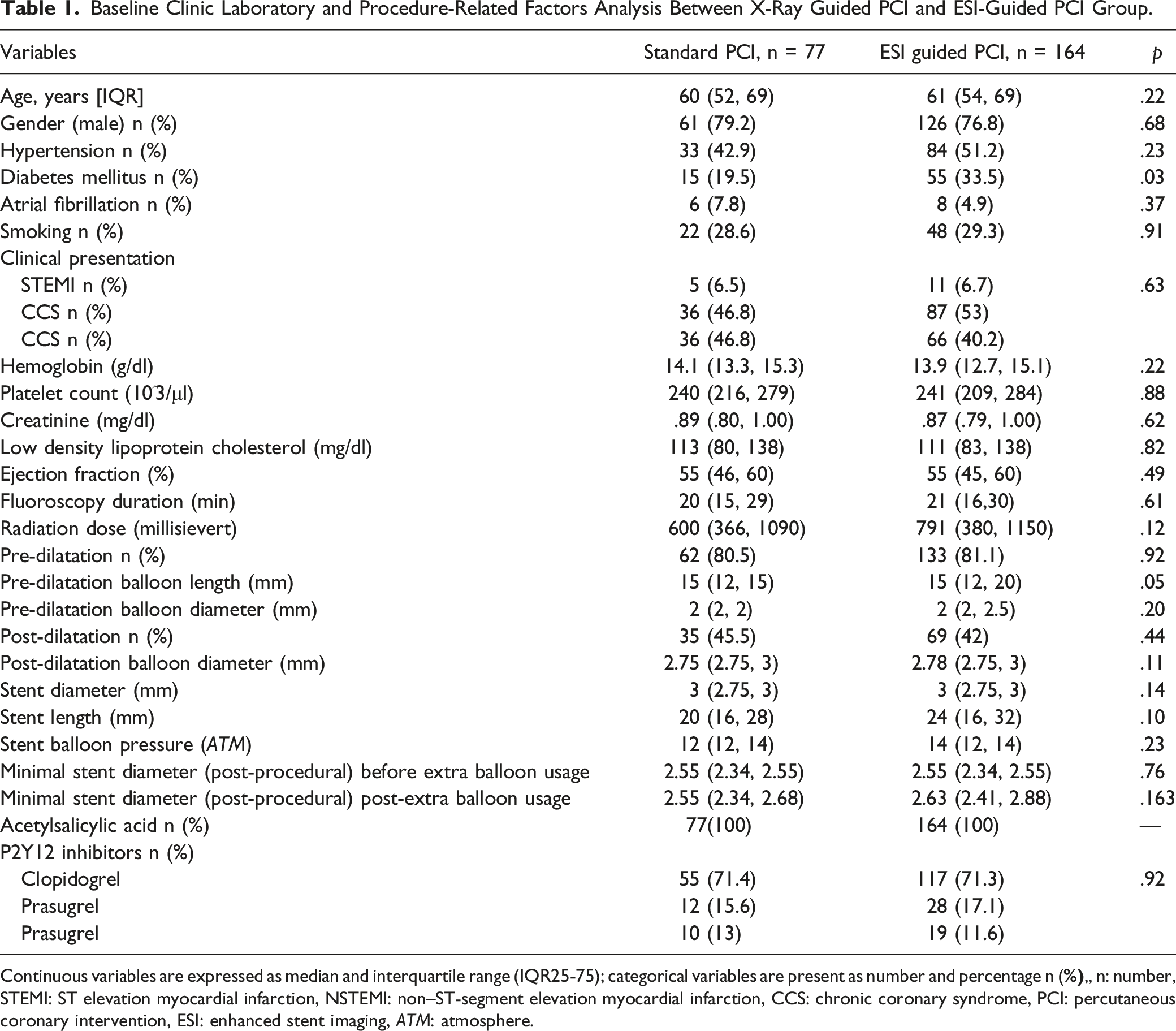
Continuous variables are expressed as median and interquartile range (IQR25-75); categorical variables are present as number and percentage n (
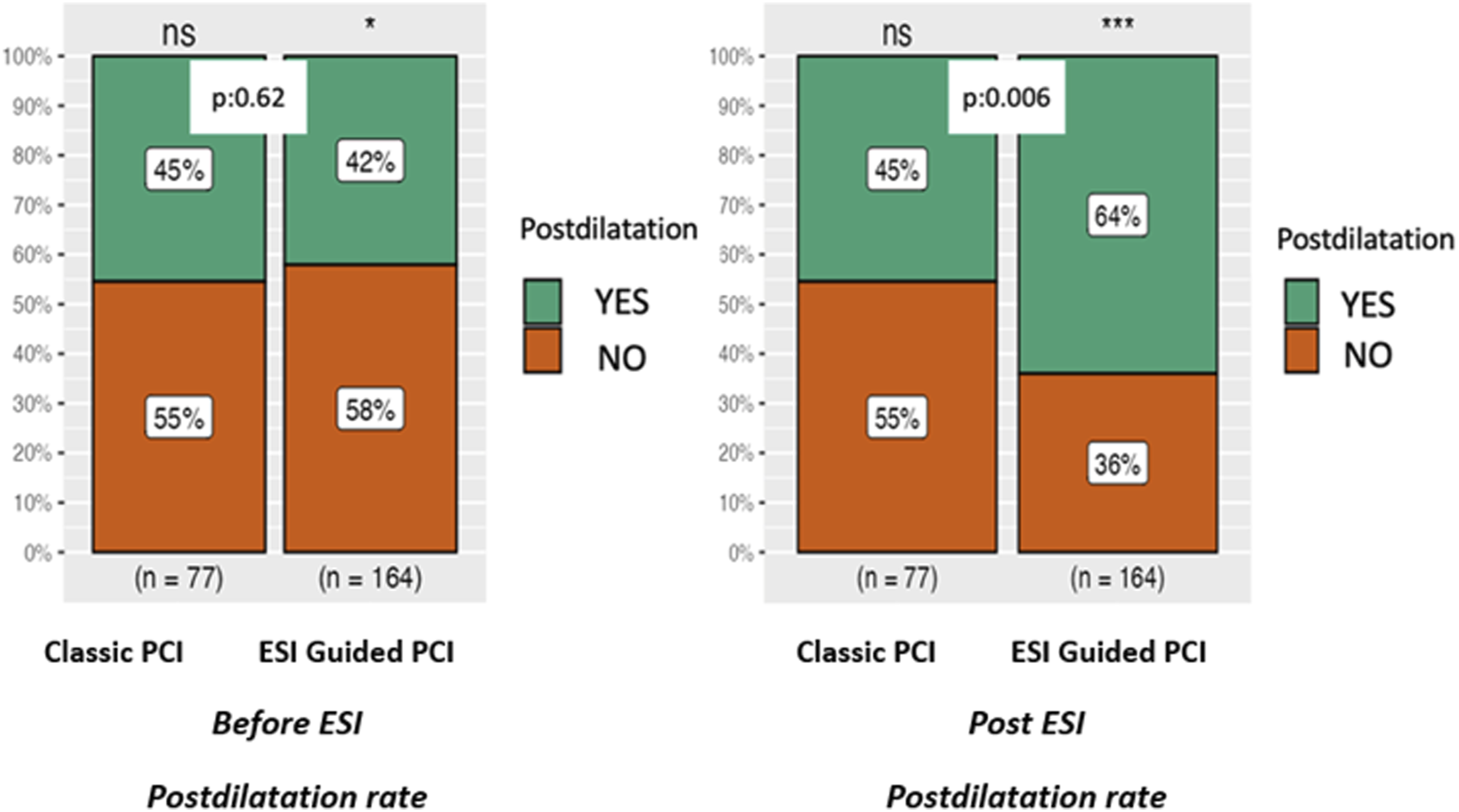
Post-dilatation rate before and after ESI comparison, between X-ray guided and ESI-guided PCI.
Procedure-Related Variables Between ESI vs X-ray Guided PCI.
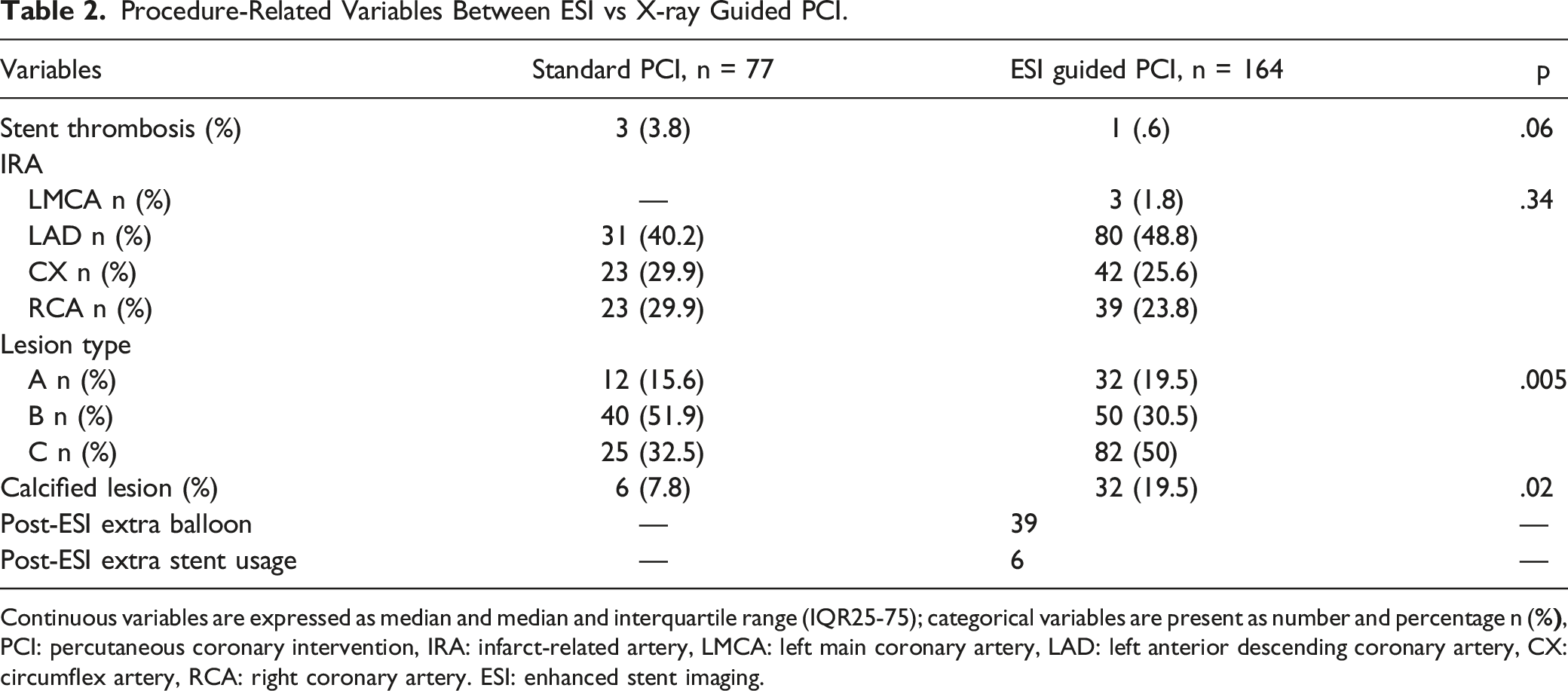
Continuous variables are expressed as median and median and interquartile range (IQR25-75); categorical variables are present as number and percentage n (
Comparison of Patients with and Without Post-Dilatation in Group ESI.
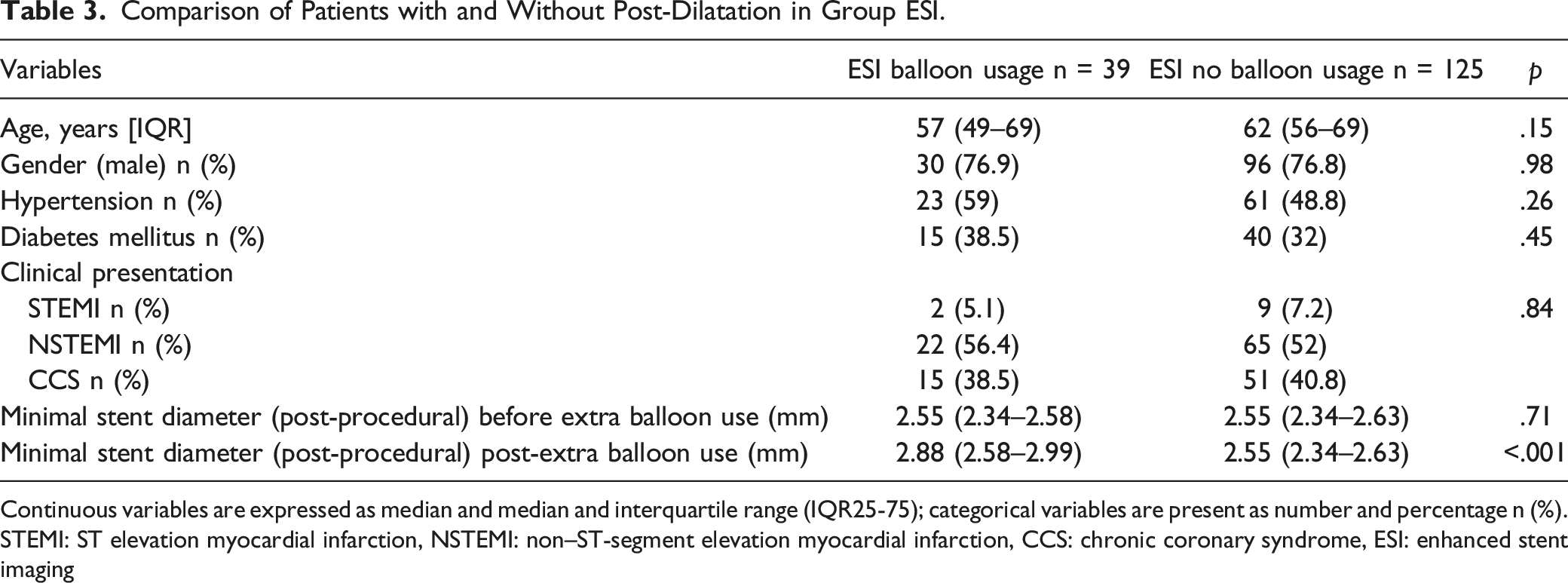
Continuous variables are expressed as median and median and interquartile range (IQR25-75); categorical variables are present as number and percentage n (%). STEMI: ST elevation myocardial infarction, NSTEMI: non–ST-segment elevation myocardial infarction, CCS: chronic coronary syndrome, ESI: enhanced stent imaging
Associated Variables With Final Minimal Lumen Diameter According to Multivariable Ordinary Least Square Regression.
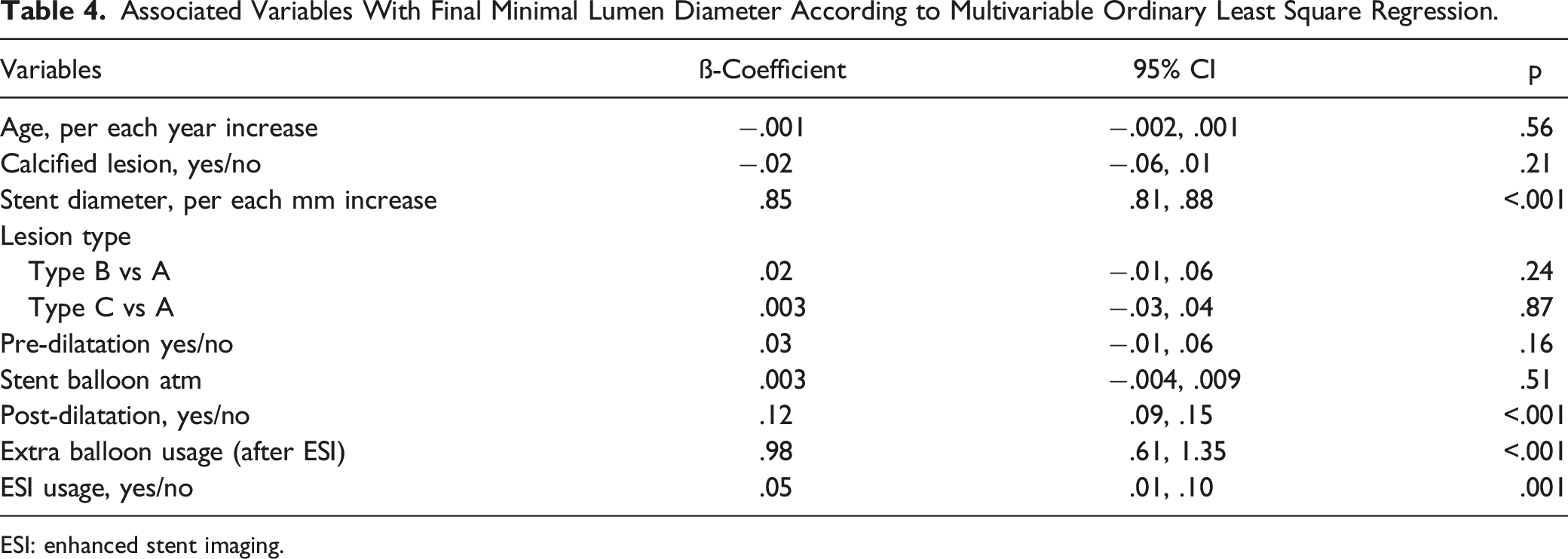
ESI: enhanced stent imaging.
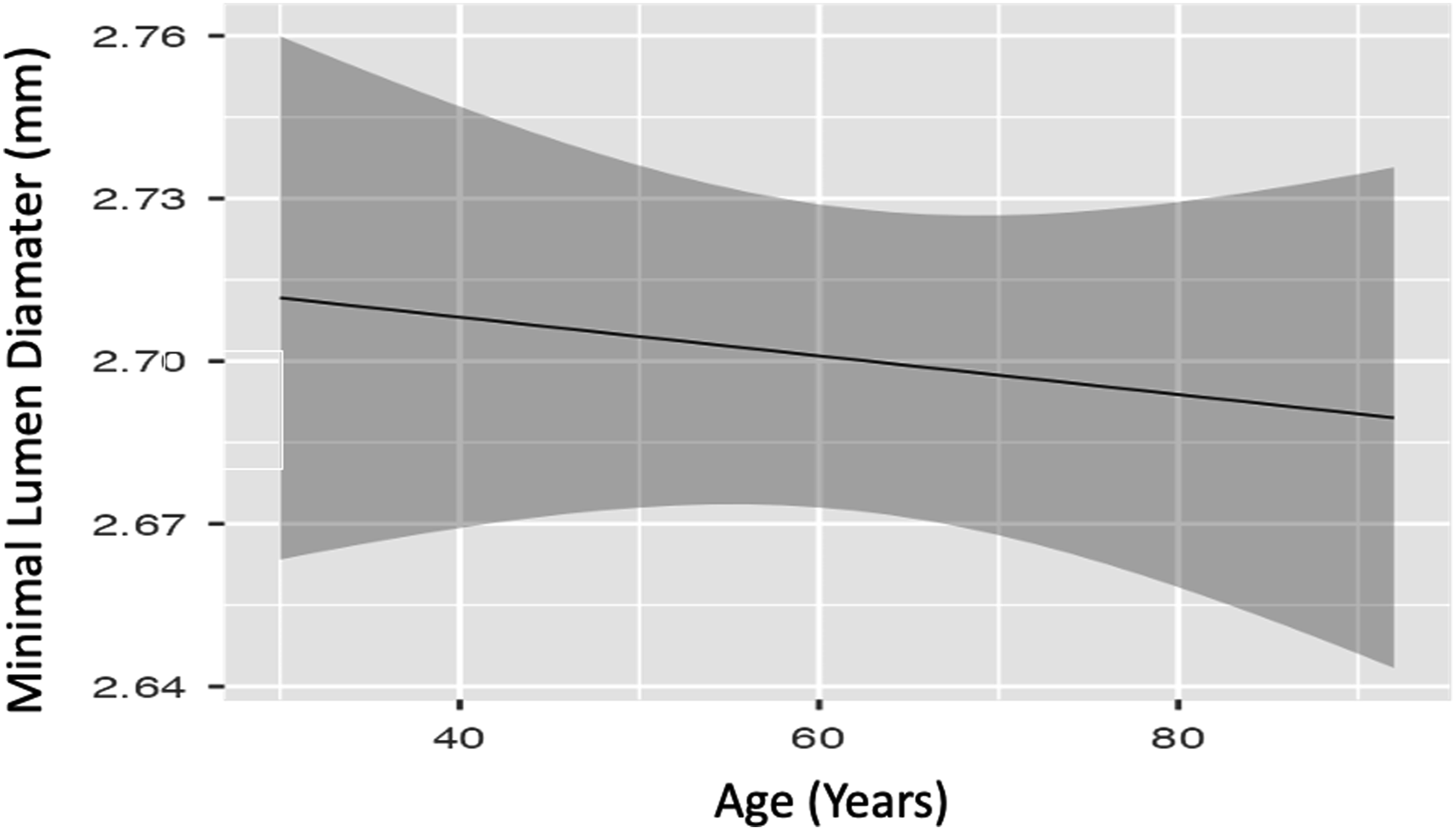
Partial effect plot for age with adjusted all variables according to regression model.
Discussion
This study found that stent diameter, post-dilation, extra balloon usage after “ClearStent Live” system and additional stent deployment by the help of ClearStent implementation was associated with better stent expansion as quantified by the MLD.
As conventional angiography-guided PCI relies on two-dimensional projections, assessing the appropriateness of stent placement can be difficult using this method alone. Therefore, there is a compelling requirement for more complex and practical imaging modalities for PCI guidance.
In early 1990s, Tobis et al 14 first showed that IVUS, which acquires cross-sectional images of the coronary arterial wall and provides information about plaque composition, could be used in vivo. After IVUS started to be used widely in clinical practice, compared with angiographic guidance, IVUS-guided stent implantation was associated with favorable outcomes in terms of major adverse cardiac events15,16 Optical coherence tomography (OCT) is another adjunctive imaging method which provides valuable information about coronary wall cross-sectional structure and atherosclerotic plaque architecture. 17 Studies have showed that, similar to IVUS, OCT guided coronary intervention may lead better results than PCI guided by angiography alone.18,19 Although these are favorable clinical results for IVUS and OCT, both imaging techniques have some disadvantages. Resolution or penetration challenges, limited tissue characterization, additional contrast media requirement, interpretation difficulties, and cost constitute of some restrictions of these modalities regarding routine clinical use. 20
Enhanced stent imaging techniques such as ClearStent (Siemens Healthcare, Munich, Germany) or StentBoost (Philips Medical Systems Nederland BV, Best, The Netherlands) are novel, software based, motion-compassed imaging techniques that provide enhanced visibility of deployed stents. This innovative technology promises a practical and cost-effective clinical use. Emerging clinical studies investigating the impact of ESI in cardiology practice revealed that, ESI-guided PCI is associated with lower rates of stent restenosis, target lesion revascularization, and major adverse cardiovascular events compared with no use of ESI.21,22 Our study demonstrated that enhanced visualization of deployed coronary stent was associated with better post-interventional result measured as MLD. It can be deduced that ESI-guided PCI provides better clinical outcome by means of better visualization of stent characteristics such as deployment status, which help needing for further post-dilation and stent strut fracture assessment.9,22 Sanitas et al demonstrated that ESI associated with increased of stent expansion and extra balloon needing similar to our study, also, McBeath et al showed that ESI associated with better medium-term angiographic and clinical outcomes.9,22
Besides its positive contributions, ESI-related fluoroscopy time and radiation exposure can be expected to be higher than standard PCI, but the fluoroscopy time and radiation exposure of ESI-guided coronary procedures are comparable with that of conventional angiography-guided PCI.22,23 In our study, fluoroscopy time and radiation exposure dose were compatible with the studies in literature.22,23 Applying an ESI procedure may reduce future complications such as stent thrombosis; therefore, it might reduce radiation as it will reduce new procedures in the long term. 22
Different commercial ESI systems such as StentViz (GE Healthcare, Milwaukee, WI, USA), StentBoost, ClearStent, and StentOptimizer (Paieon medical, Rosh Haayin, Israel) are currently available. By virtue of its improved imaging quality, ESI system can be used in different procedural scenarios such as assessing stent under-expansion, stent strut disruption, implantation a second stent in overlap, ostial lesion stenting, bifurcation stenting, and side branch preservation.24-27
Considering the emerging technology, ESI systems promise to be auxiliary tools that can guide the operator in both simple and complex PCI procedures, with features such as cost-effectiveness, availability, and procedural and clinically satisfactory results.
Our study had some limitations. The sample size was relatively small, and the study design was observational. Another limitation was the endpoint, the MLD, which was quantified with QCA, rather than compared with IVUS. Although we included all variables in the regression model, some unmeasured confounders may remain. On the other hand, as IVUS and OCT provide structural information such as plaque characteristic or vascular wall anatomy, ESI should not be considered as a substitute for those modalities. It should be noted that this is a cross-sectional study, and that additional longitudinal cohort studies evaluating both cardiovascular events and mortality are needed.
Conclusion
Our study addresses that use of ESI system with intend to optimize stent placement results in better procedural outcome expressed by MLD. In the era of complex intracoronary imaging modalities such as IVUS and OCT, ESI systems seem to be a good alternative. Although there are publications that show favorable results, it should be stated that this topic needs further research in randomized, controlled studies.
Footnotes
Author contribution
All authors contributed to: (1) substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
