Abstract
We investigated the prognostic effects of hyperuricemia and high or low body mass index (BMI) in peripheral artery disease (PAD) after endovascular therapy (EVT). Between July 2015–2016, 357 consecutive patients with PAD who underwent EVT were enrolled. Patients were divided into 2 groups: BMI < 25 kg/m2 (low BMI) and ≥ 25 kg/m2 (high BMI); they were also divided into 2 more groups based on the presence/absence of hyperuricemia. The primary and secondary endpoints were major adverse cardiovascular and limb events (MACLE), and all-cause death at 3 years post-EVT. Patients with hyperuricemia had significantly lower freedom from MACLE than patients without hyperuricemia at 3 years (57.0 vs 71.9%, p = .0068). The overall survival of patients with hyperuricemia was significantly lower than that of patients without hyperuricemia (63.9 vs 81.7%, p = .0012). Patients with hyperuricemia who had low BMI experienced significantly lower freedom from MACLE than those without hyperuricemia who had low BMI (48.2 vs 69.9%, p = .002). The overall survival of patients with hyperuricemia who had low BMI was significantly lower than that of patients without hyperuricemia who had low BMI (55.2 vs 77.1%, p = .003). Patients with hyperuricemia had significantly more MACLE and a lower survival at 3 years than patients without hyperuricemia, even if they had a low BMI.
Introduction
The association between hyperuricemia and atherosclerosis was difficult to assess because it was thought to be a consequence of the patient’s characteristics (e.g., obesity, chronic kidney disease (CKD)) and not to be an independent cause of atherosclerosis.1,2 Many patients with hyperuricemia (serum uric acid >7.0 mg/dL) have one or more lifestyle-related diseases, such as hypertension, obesity, diabetes, and dyslipidemia. 3
Several studies have reported a relationship between uric acid levels, metabolic syndrome, and obesity. 4 In Japan, a positive correlation has been found between visceral fat and uric acid levels.5,6 It has also been reported that uric acid levels decrease with weight loss.7–9 However, in recent years, hyperuricemia has been identified as an independent risk factor for atherosclerosis, cerebrovascular disease, and coronary artery disease.10–22 Hyperuricemia has been reported to also impair vascular endothelial function (e.g., reduction of flow-mediated dilation, FMD) in patients without metabolic syndrome.23,24 Therefore, it has been suggested that hyperuricemia is a risk factor for cardiovascular events, independent of obesity or metabolic syndrome.
Although hyperuricemia has been reported to be a prognostic factor in patients with coronary artery disease,17–22 little data on hyperuricemia have been reported in patients without obesity. In addition, there are few reports on the prognostic effects of hyperuricemia in patients with peripheral artery disease (PAD) after endovascular therapy (EVT). Therefore, we investigated the effects of hyperuricemia on post-EVT PAD patients and non-obese (low body mass index [BMI]) post-EVT PAD patients.
Materials and Methods
This study comprised a sub-analysis of the I-PAD NAGANO registry (improving prognosis of PAD patients undergoing endovascular treatment around NAGANO). The I-PAD NAGANO registry was a prospective, multicenter, observational registry in which 337 consecutive patients undergoing EVTs for any PAD of the lower extremities between August 2015 and August 2016 from 10 institutions were enrolled in Nagano prefecture, Japan. This registry had no exclusion criteria and was an all-comer registry. The patients provided their informed consent. The present study was approved by each hospital’s ethics committee and was conducted in accordance with the Declaration of Helsinki. The I-PAD NAGANO registry was registered with the University Hospital Medical Information Network Clinical Trials Registry, as accepted by the International Committee of Medical Journal Editors (UMIN-ID; 000018297). Enrolled PAD patients had intermittent claudication, resting pain, or leg ulcers and underwent standard EVT within the preceding period. PAD was diagnosed using angiography or duplex ultrasonography. The average ankle brachial index (ABI) of the affected limb before surgery was 0.76 ± 0.26. The median follow-up period was 3.3 years.
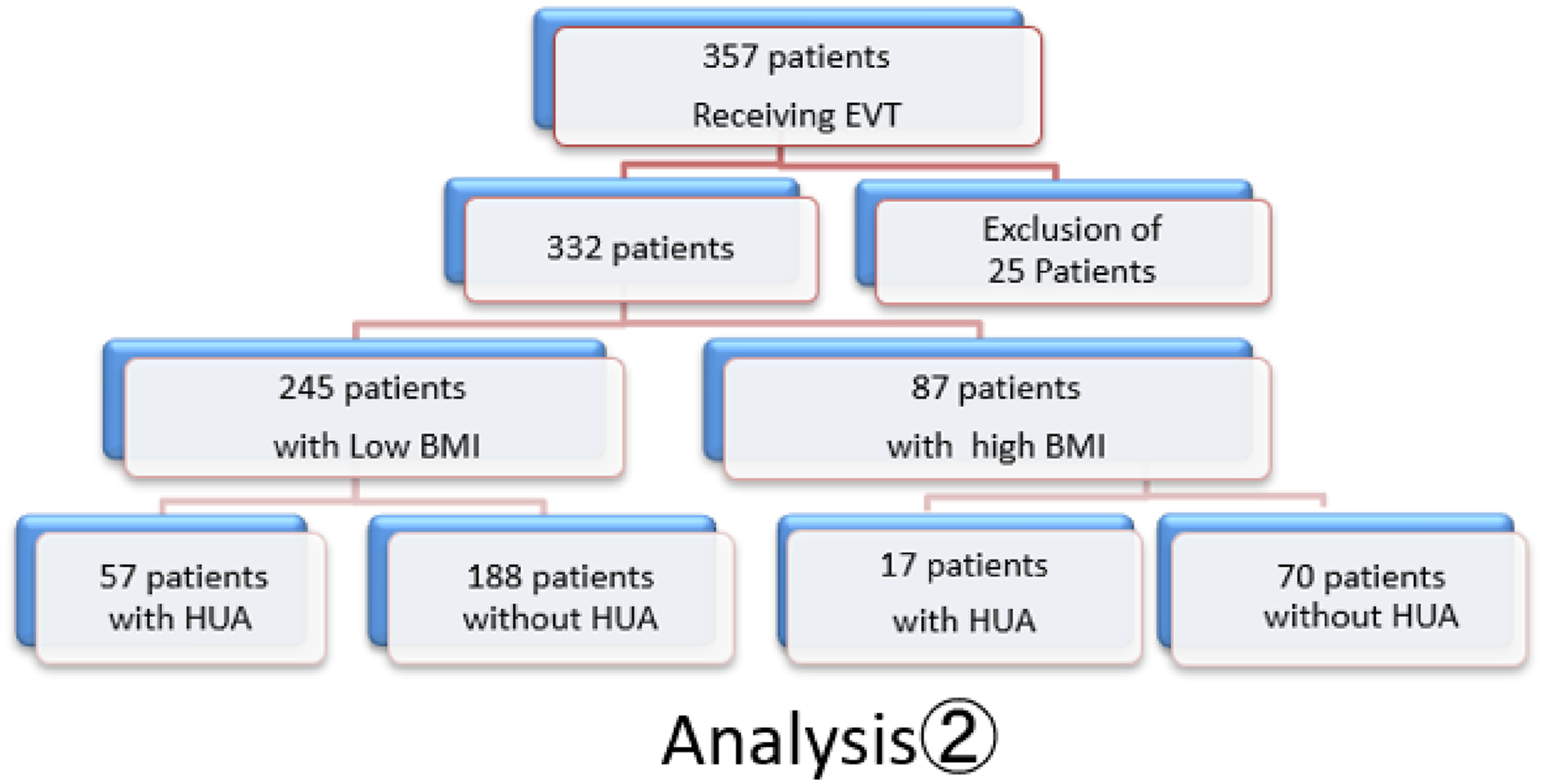
Patients were divided into 2 groups based on the presence or absence of hyperuricemia, which was defined as a serum uric acid level >7.0 mg/dL. The hyperuricemia and non-hyperuricemia groups comprised 74 and 258 patients, respectively (Analysis 1). All patients were also divided into 2 more groups: patients with a BMI <25 kg/m2 and those with a BMI ≥25 kg/m2 (245 patients and 87 patients, respectively). They were further divided into 2 groups based on the presence or absence of hyperuricemia. The groups consisted of 57 patients with low BMI and hyperuricemia, 188 patients with low BMI and no hyperuricemia, 17 patients with high BMI and hyperuricemia, and 70 patients with high BMI and no hyperuricemia (Analysis 2). The prognosis of each group after EVT was investigated. The study flow charts are shown in Figures 1 and 2. The uric acid and BMI values are at the time of admission. Study profile. Abbreviations: EVT: endovascular therapy; HUA: hyper uremic acid; BMI: body mass index. Study profile. Abbreviations: EVT: endovascular therapy; HUA: hyper uremic acid; BMI: body mass index.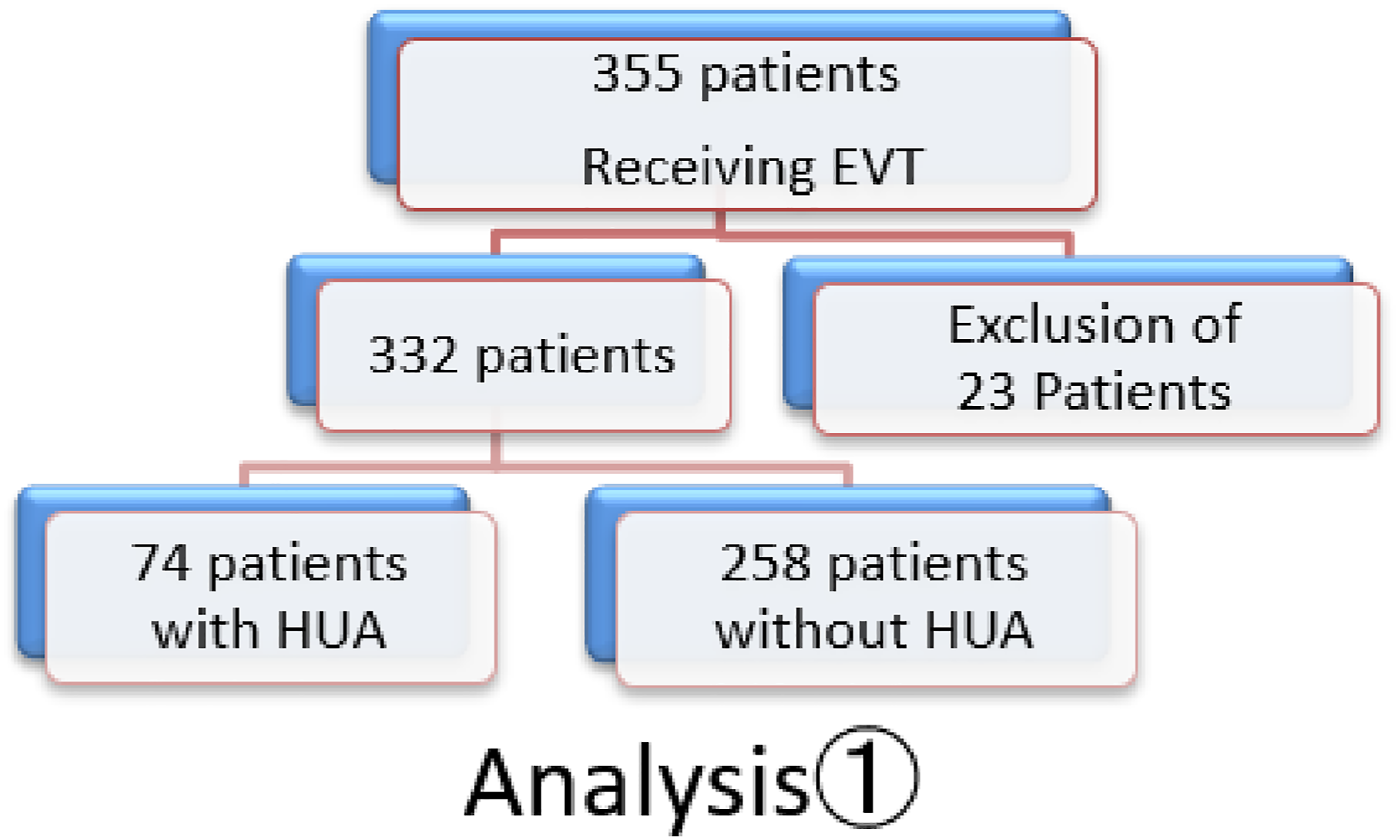
Exclusion Criteria
The following patients were excluded in this study: (1) patients lacking data, (2) patients with malignant tumors, and (3) patients with a BMI <18.5 kg/m2.
Based on 2016 Guidelines of the Japan Society for the Study of Obesity, patients with a BMI < 18.5 kg/m2were defined as underweight and patients with a BMI of ≥25 kg/m2 were defined as high BMI. 25 We defined a BMI <25 as low BMI. Underweight patients were excluded from this study because emaciation may be associated with prognosis. 26
Study End Points
The primary endpoint of this study was major adverse cardiovascular and limb events (MACLE), including cardiac death, nonfatal myocardial infarction, stroke, transient ischemic attack (TIA), major amputation, and limb revascularization. The secondary endpoint was all-cause death within 3 years of EVT.
Definitions
Myocardial infarction was defined as previously described.27,28 Ischemic stroke was defined as the presence of a new neurological deficit lasting ≥24 h. Brain magnetic resonance imaging or computed tomography was required to confirm ischemic stroke. TIA was defined as the presence of a new focal neurological deficit lasting <24 h. Limb revascularization was performed in cases with worsening symptoms, progression, or restenosis of the lesion. Restenosis after revascularization was defined as a decrease in the ABI and 50% stenosis on angiography or duplex ultrasonography.27,29 Major amputation was defined as amputation above the ankle.
Statistical Analyses
Continuous data are presented as mean ± standard deviation, and categorical data are presented as frequencies (percentages). Continuous variables were compared using the unpaired t-test, and categorical variables were compared using the chi-square test. Patency and event-free survival were estimated using the Kaplan–Meier method and are presented as the mean ± standard deviation. The curves were compared using log-rank tests. Data are presented as hazard ratios (HRs) with 95% confidence intervals (CIs). The threshold for statistical significance was set at p < .05 in two-sided test. All statistical analyses were performed using JMP version 10.1 (SAS Institute Inc., Cary, NC, USA).
Results
Characteristics and Comorbidities of Patients.
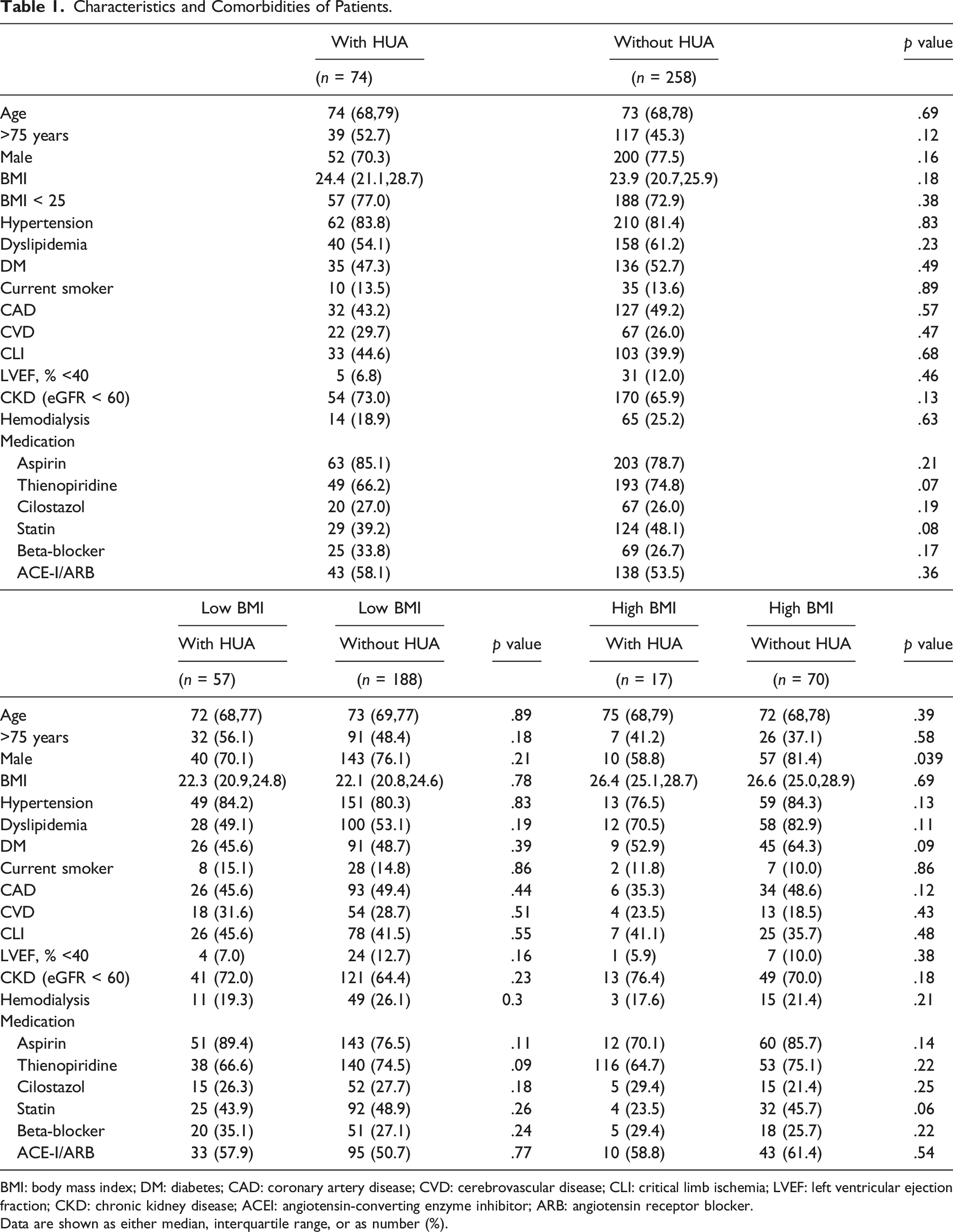
BMI: body mass index; DM: diabetes; CAD: coronary artery disease; CVD: cerebrovascular disease; CLI: critical limb ischemia; LVEF: left ventricular ejection fraction; CKD: chronic kidney disease; ACEI: angiotensin-converting enzyme inhibitor; ARB: angiotensin receptor blocker.
Data are shown as either median, interquartile range, or as number (%).
Each 2 groups were relatively matched in terms of comorbidities and oral medications. The presence or absence of a history of gout was not recorded.
Adverse Events at 3 Years Post-EVT.
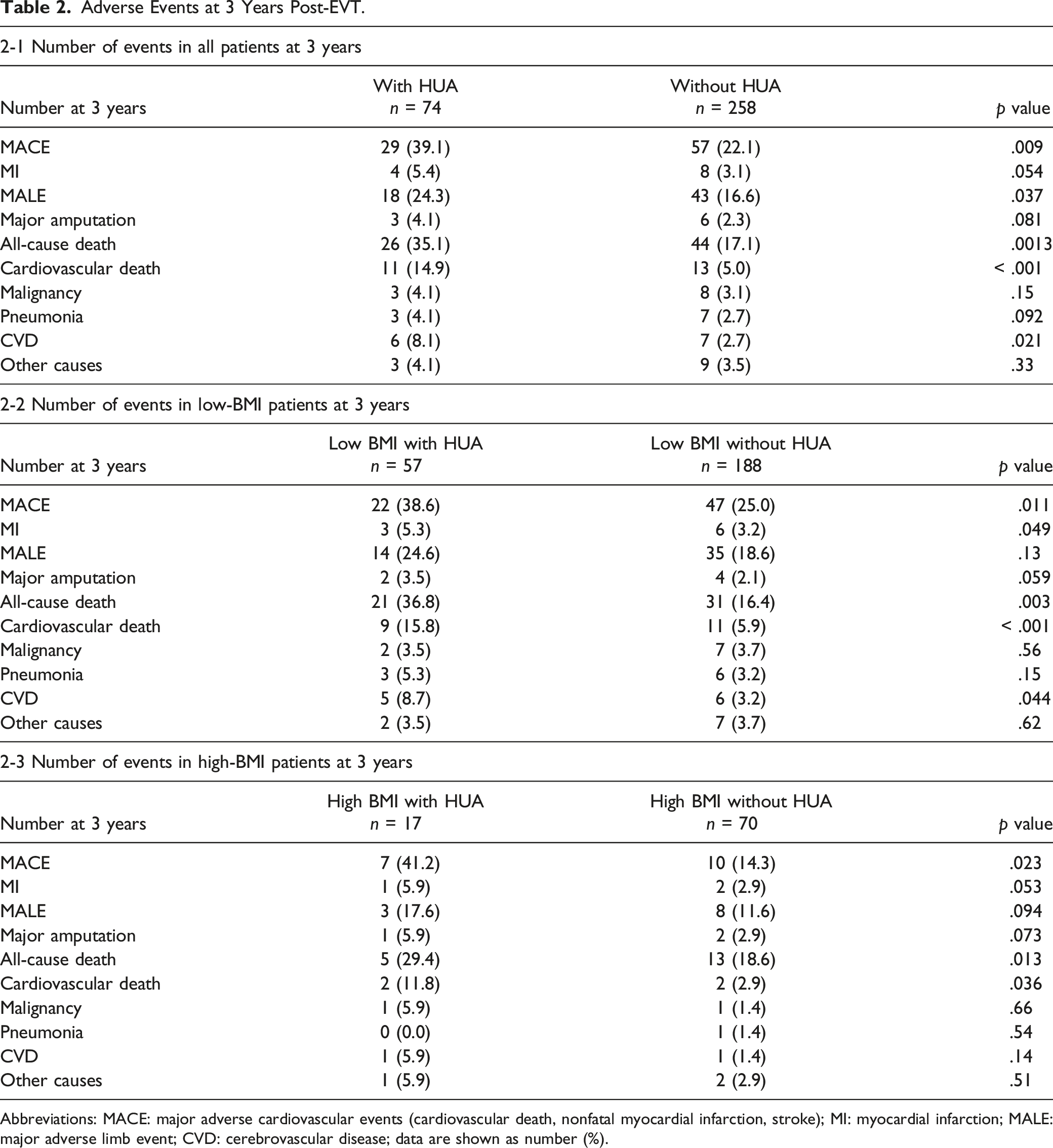
Abbreviations: MACE: major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction, stroke); MI: myocardial infarction; MALE: major adverse limb event; CVD: cerebrovascular disease; data are shown as number (%).
Patients with hyperuricemia had a significantly lower freedom from MACLE than patients without hyperuricemia at 3 years after EVT (57.0 vs 71.9%, p = .0068) (Figure 3). The 3-year overall survival of patients with hyperuricemia was significantly lower than that of patients without hyperuricemia (63.9 vs 81.7%, p = .0012) (Figure 4). Multivariate analysis, including BMI, revealed that hyperuricemia (HR: 1.2, 95% CI: 1.1–2.2, p = .036), age (HR: 1.3, 95% CI: 1.1–1.8, p = .027), critical limb ischemia (CLI) (HR: 1.5, 95% CI: 1.1–1.9, p = .021), and chronic kidney disease (CKD) (HR: 1.8, 95% CI: 1.1–2.3, p < .001) were independent predictors of MACLE (Table 3-1). Hyperuricemia (HR: 1.4, 95% CI: 1.1–1.9, p = .042), age (HR: 1.3, 95% CI: 1.1–1.8, p = .035), diabetes mellitus (DM) (HR: 2.0, 95% CI: 1.4–2.7, p < .001), CKD (HR: 1.6, 95% CI: 1.2–2.0, p < .001), and CLI (HR: 1.4, 95% CI: 1.1–5.2, p < .001) were independent predictors for overall survival (Table 3-2). Patients with hyperuricemia who had a low BMI had significantly lower freedom from MACLE than those without hyperuricemia who had a low BMI at 3 years post-EVT (48.2 vs 69.9%, p = .002) (Figure 5). The 3-year overall survival of patients with hyperuricemia who had a low BMI was significantly lower than that of patients without hyperuricemia who had a low BMI (55.2 vs 77.1%, p = .003) (Figure 6). Multivariate analysis revealed that hyperuricemia (HR: 1.4, 95% CI: 1.1–1.9, p = .016), DM (HR: 2.0, 95% CI: 1.4–2.7, p < .001), age (HR: 1.3, 95% CI: 1.1–1.8, p = .030), CKD (HR: 1.7, 95% CI: 1.2–2.4, p < .001), and CLI (HR: 1.7, 95% CI: 1.1–3.8, p < .001) were independent predictors of MACLE (Table 3-3). Hyperuricemia (HR: 1.3, 95% CI: 1.1–1.8, p = .037), age (HR: 1.3, 95% CI: 1.1–1.8, p = .013), CKD (HR: 1.7, 95% CI: 1.2–2.7, p < .001), and CLI (HR: 1.5, 95% CI: 1.1–1.8, p < .001) were independent predictors for overall survival (Tables 3-4).
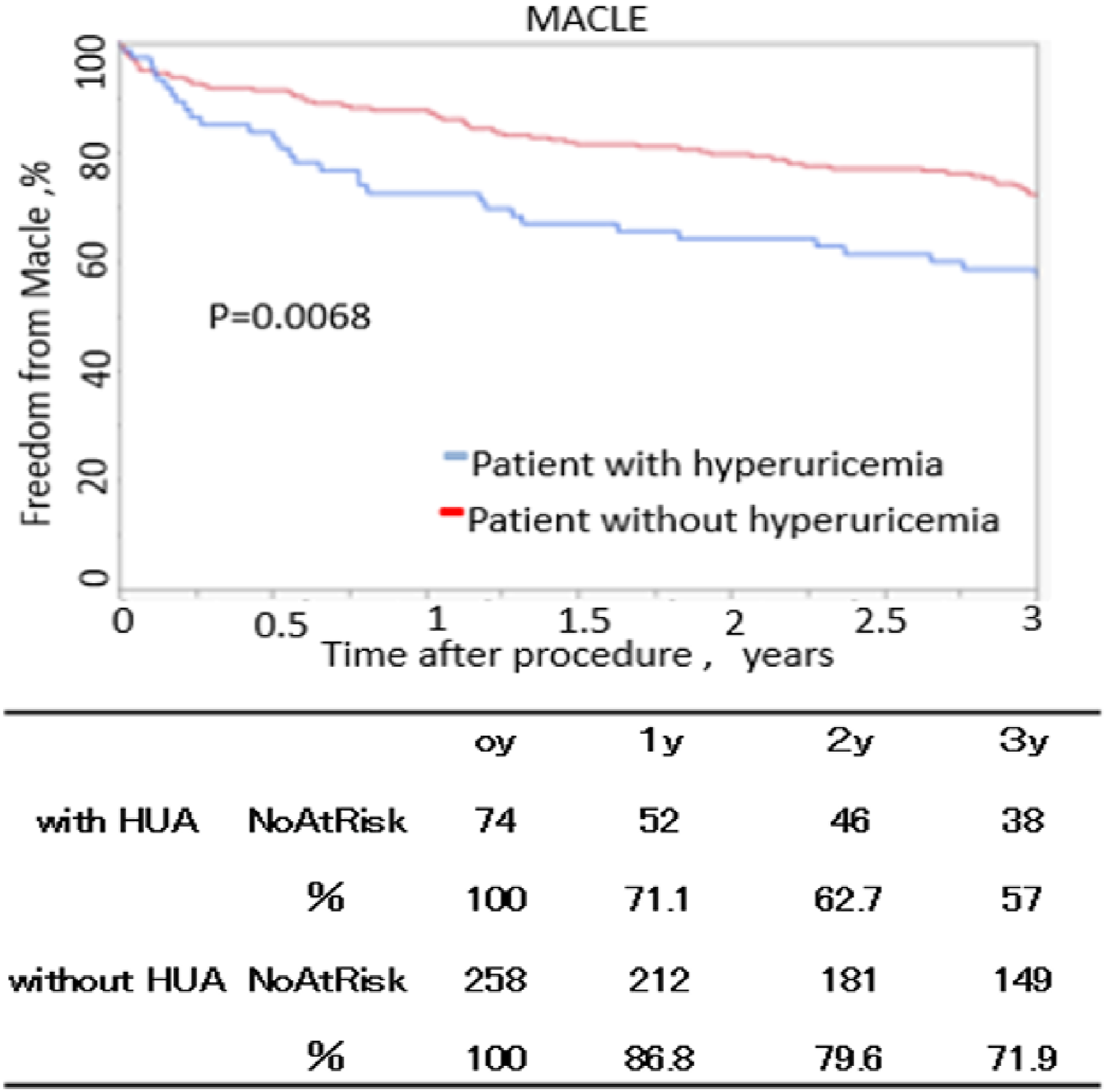
Freedom from major adverse cardiovascular and limb events. MACLE: major adverse cardiovascular and limb events; HD: hemodialysis. The standard errors did not exceed 10% at 5 years.
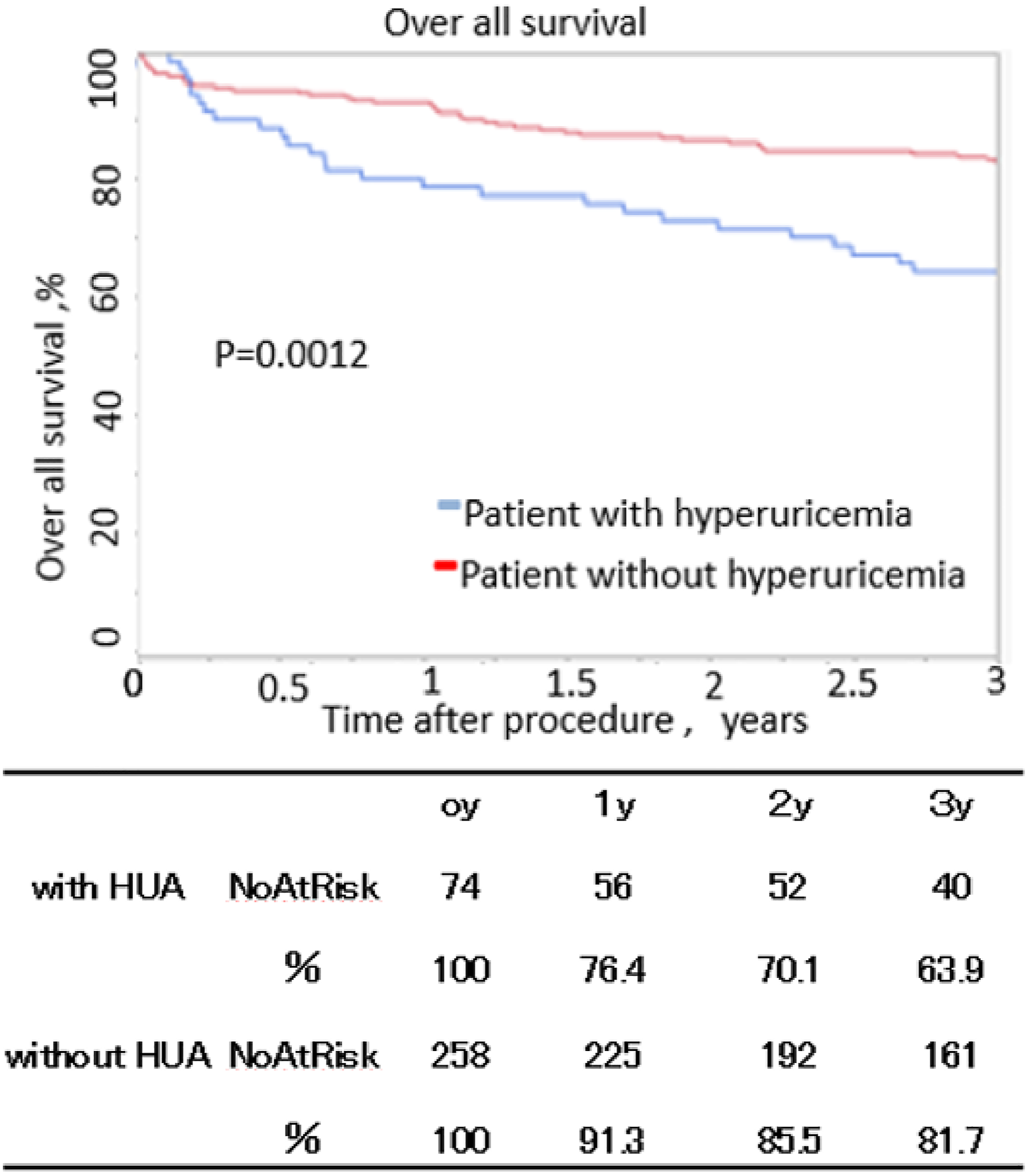
Overall 3-year survival. The standard errors did not exceed 10% at 5 years.
Risk Factors for Adverse Cardiovascular and Limb Events and All-Cause Death.
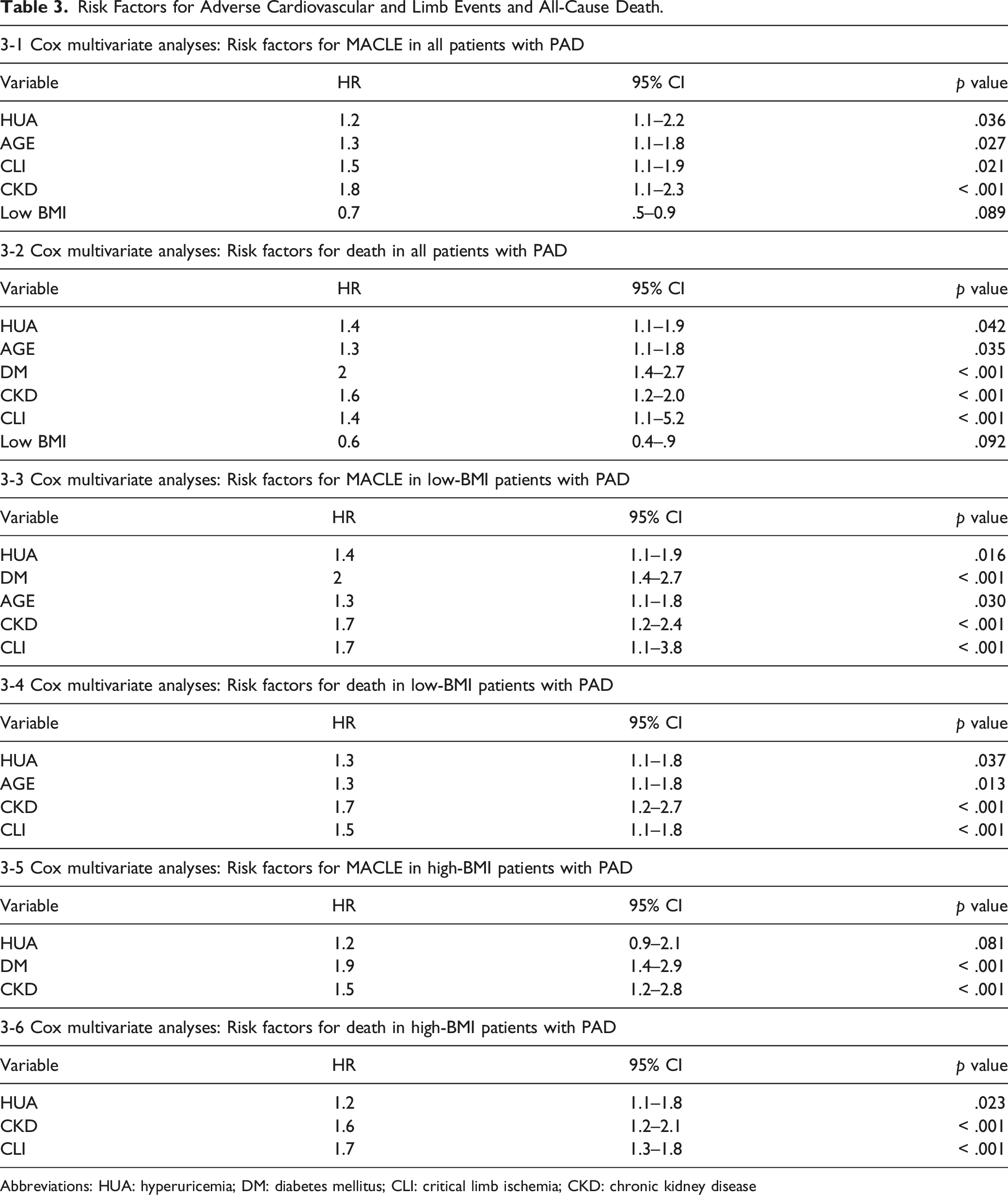
Abbreviations: HUA: hyperuricemia; DM: diabetes mellitus; CLI: critical limb ischemia; CKD: chronic kidney disease
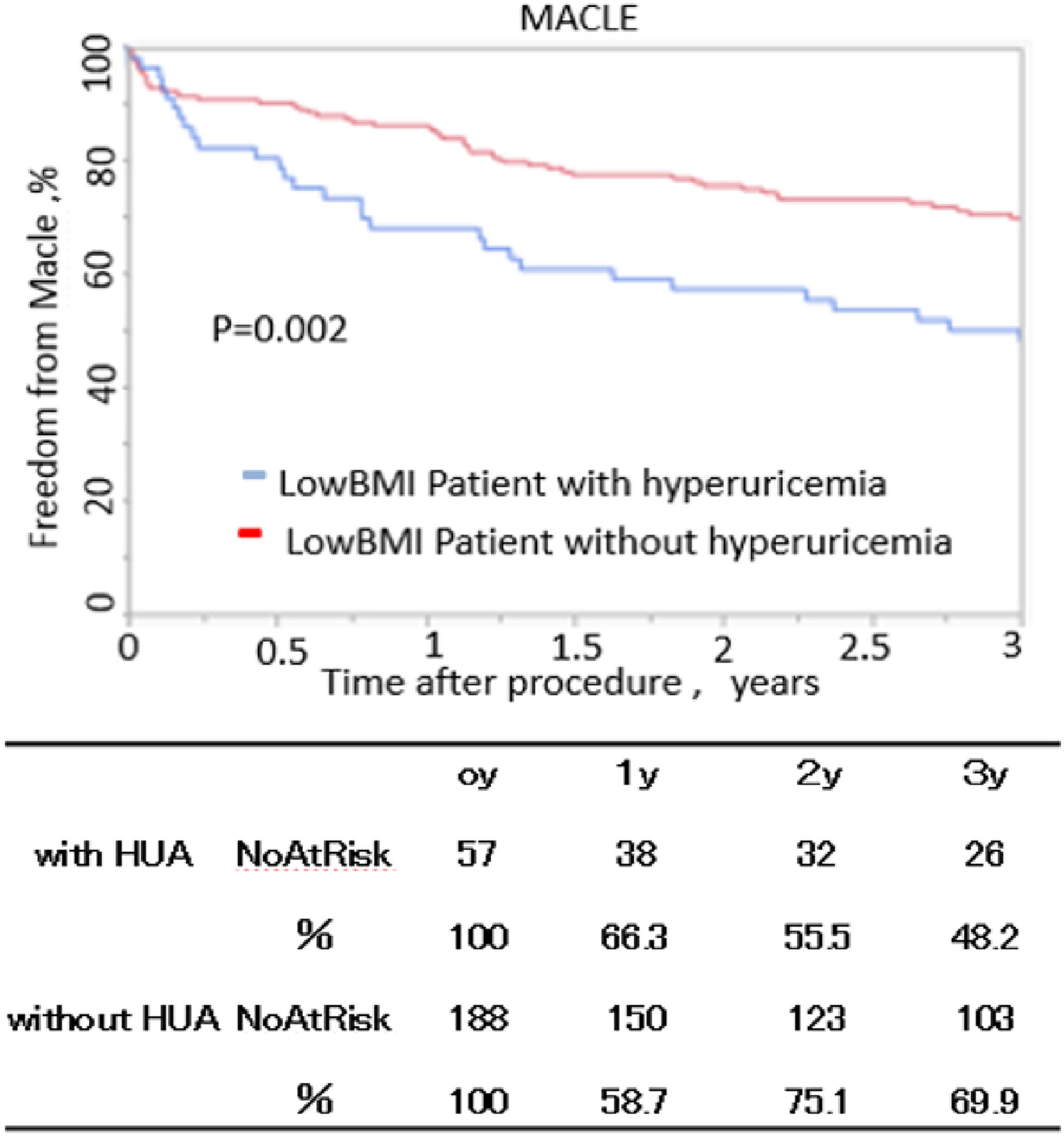
Freedom from major adverse cardiovascular and limb events in patients with low BMI. The standard errors did not exceed 10% at 5 years.
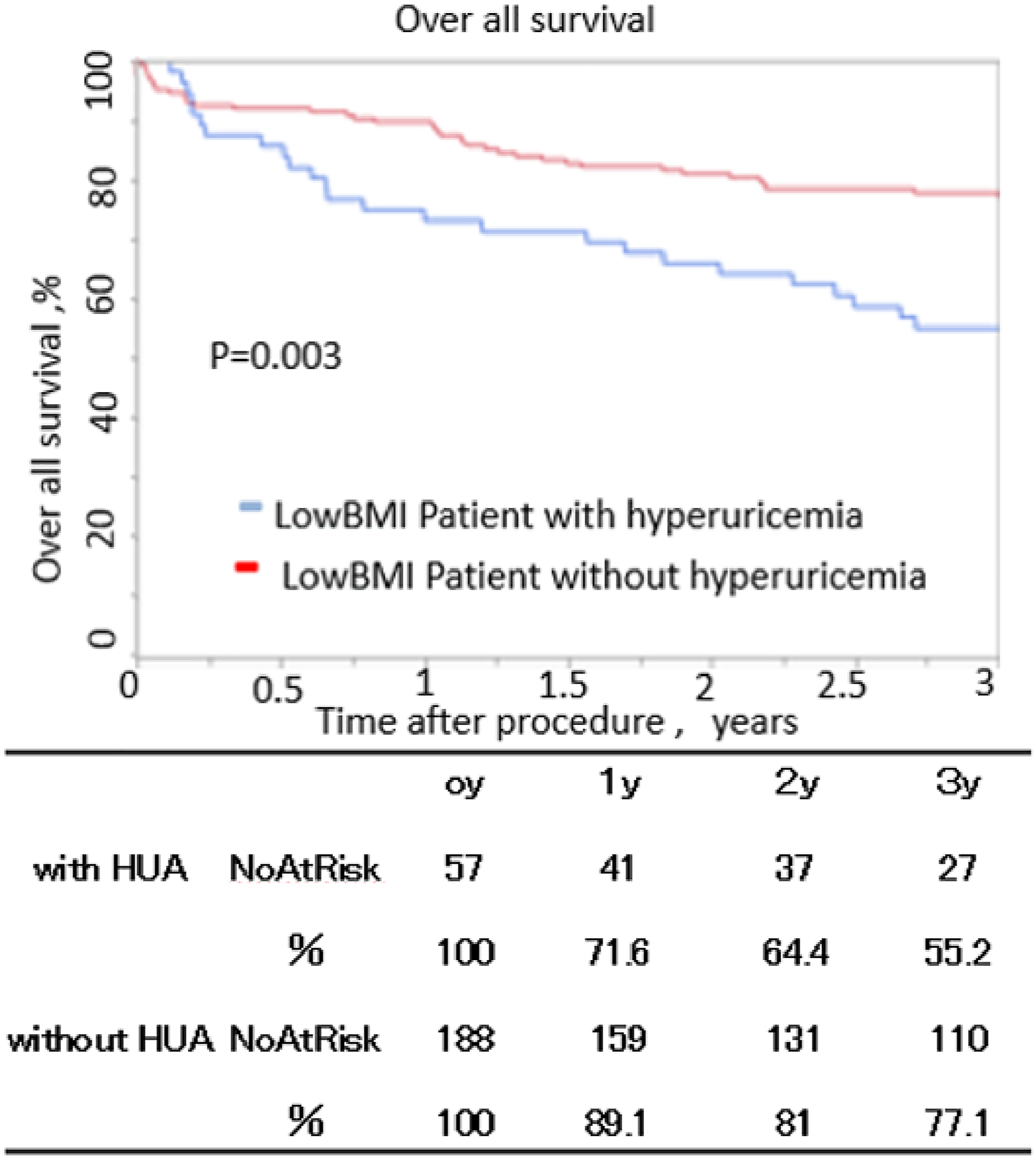
Overall 3-year survival in patients with low BMI. The standard errors did not exceed 10% at 5 years.
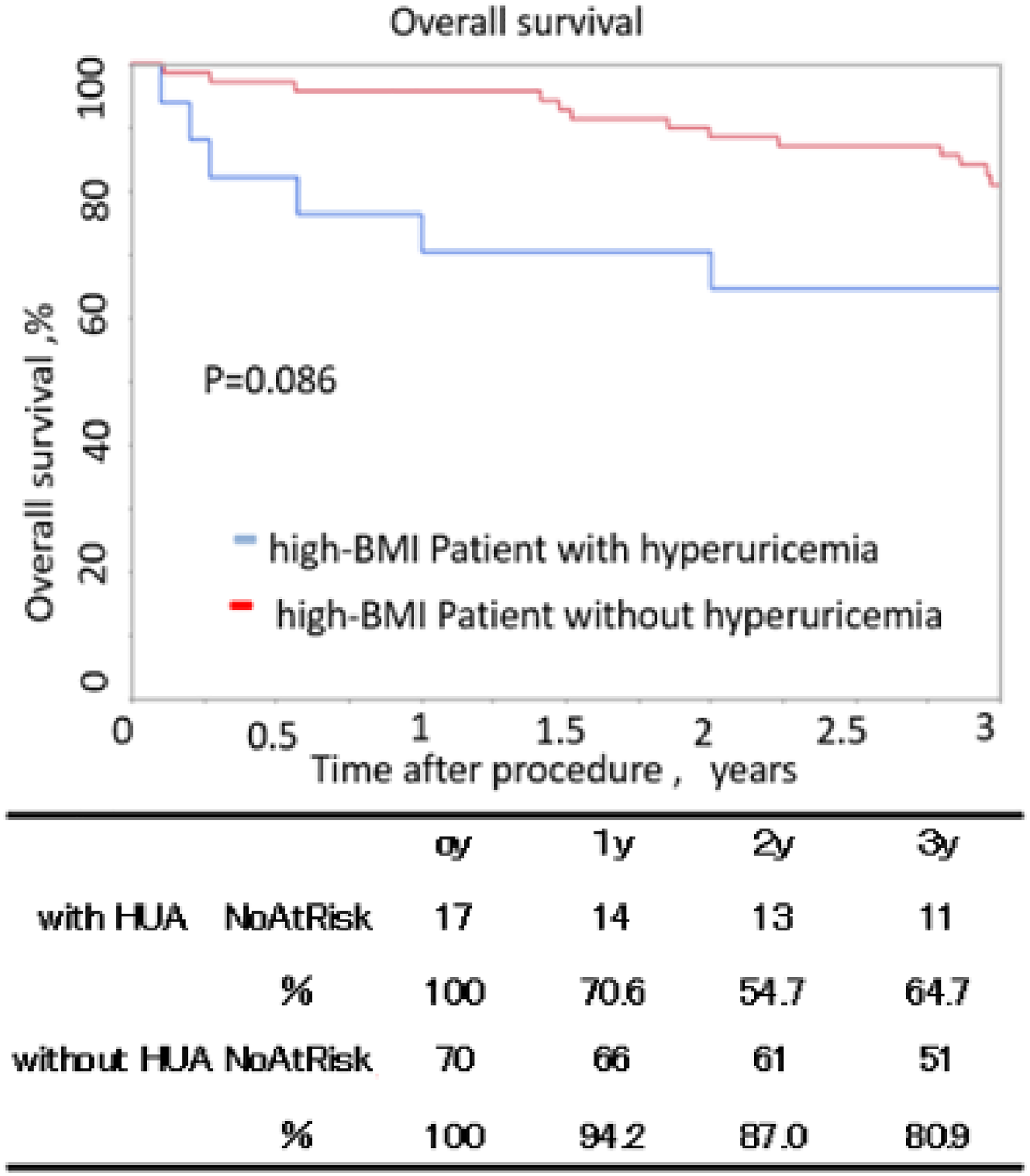
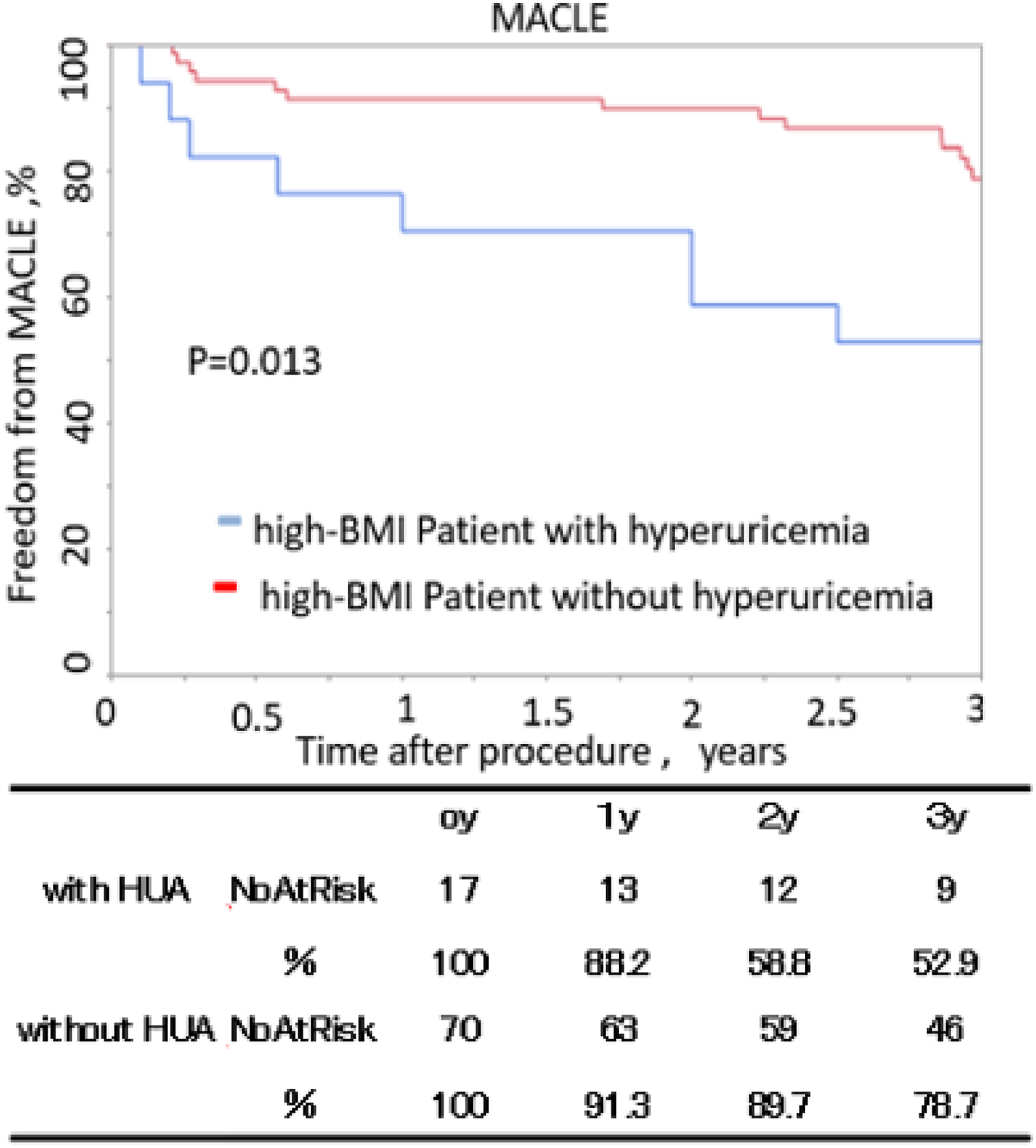
Patients with hyperuricemia who had a high BMI tended to lower freedom from MACLE than those without hyperuricemia, although there was no significant difference (64.7 vs 80.9%, p = .086) (Figure 7). The 3-year overall survival of patients with hyperuricemia who had a high BMI was significantly lower than that of patients without hyperuricemia (52.9 vs 78.7%, p = .013) (Figure 8). A multivariate analysis revealed that DM (HR: 1.9, 95% CI: 1.4–2.9, p < .001), CKD (HR: 1.5, 95% CI: 1.2–2.8, p < .001) were independent predictors for MACLE. Hyperuricemia (HR: 1.2, 95% CI: 0.9–2.1, p = .081) was not independent predictors for MACLE (Tables 3-5). Hyperuricemia (HR: 1.2, 95% CI: 1.1–1.8, p = .023), CKD (HR: 1.6, 95% CI: 1.2–2.1, p < .001), and CLI (HR: 1.7, 95% CI: 1.3–1.8, p < .001) were independent predictors for overall survival (Tables 3-6).
Freedom from major adverse cardiovascular and limb events in patients with high BMI. The standard errors did not exceed 10% at 5 years.
Overall 3-year survival in patients with high BMI. The standard errors did not exceed 10% at 5 years.
Discussion
We found that hyperuricemia is a risk factor for MACLE in all patients, including the low-BMI patient group. In addition, for overall mortality, hyperuricemia may be a risk factor for all patients, regardless of BMI. For MACLE in the high-BMI group, the number of patients was small (n = 87), which may be the reason why no significant difference was seen. Our results indicate that hyperuricemia may be an independent prognostic factor in PAD patients after EVT, regardless of BMI. It is of interest that this study suggests that the presence of hyperuricemia is a residual risk factor for atherosclerosis, even in patients with a low BMI (without obesity). Based on the above, this study suggests active intervention for hyperuricemia in patients with PAD to improve prognosis.
There have been many reports on the effects of hyperuricemia associated with the development of coronary artery disease, cerebrovascular disease, and hypertension.2–14 However, there are few reports on the prognostic effects of hyperuricemia in patients with PAD after EVT. Previous studies have indicated that the prognostic factors in patients with coronary artery disease and PAD are similar.30,31 In the present study, there was a significant difference in the occurrence of MACLE and the overall 3-year survival after EVT in patients with and without hyperuricemia. As shown in Table 2, as a sub-analysis, there was a significant difference in MACE and cardiovascular death at 3 years. This suggests that hyperuricemia may be a prognostic factor in patients with PAD, as in patients with coronary artery disease.
BMI has long been used as an indicator of obesity. 32 It has been reported that BMI is highly correlated with waist circumference (WC), which is an index of accumulation of visceral fat, subcutaneous fat, and visceral fat area (VFA), and that it is convenient and meaningful to use BMI instead of WC as a diagnostic criterion for metabolic syndrome. 33 Based on the above, in this study, analysis was performed using BMI as an index of subcutaneous fat and visceral fat and as an index of obesity. The debate about the obesity paradox is still ongoing. Even in recent reports, a high BMI did not improve the prognosis in acute coronary syndrome (ACS) patients. 34 It has been reported that obese patients develop the disease at an early age, and thus, the prognosis seems good because of the long follow-up period due to their youth; moreover, obesity is considered to be a risk factor of the disease. 35
Obesity leads to insulin resistance, which causes hyperinsulinemia and insulin resistance is associated with increased subcutaneous and visceral fat. 36 Through various metabolic pathways, hyperinsulinemia increases serum uric acid levels by promoting purine synthesis in the liver and uric acid reabsorption in the kidney.37–39 It has been reported that uric acid affects atherogenesis via the oxidative stress action of xanthine oxidase, intracellular uric acid crystals, inflammasomes, and uric acid transporters.40–43 Crystallization of uric acid due to a rapid increase in serum uric acid levels causes hypercoagulation, inflammatory reactions, increased active oxygen levels, and vascular disorders, and the incorporation of uric acid into macrophages promotes the formation of inflammasomes, resulting in vascular injury via inflammatory cytokines. 40 Active oxygen is generated when uric acid is produced by the action of xanthine oxidase, and oxidative stress is associated with atherosclerosis. 41 It has also been proposed that uric acid may be taken into cells via transporters expressed in various cells other than in the kidney and may act as a risk factor for vascular disorders.42,43
It has also been pointed out that monosodium urate (MSU) may deposit in coronary plaques and promote atherosclerosis and cardiovascular MSU deposits were detected by dual energy computed tomography significantly more often in patients with gout compared with controls and were associated with higher coronary calcium score. 44
As mentioned above, various mechanisms that may explain the association between uric acid and arteriosclerosis have been proposed. However, factors other than the oxidative stress action related to the uric acid production pathway are not directly related to the degree of obesity, and the existence of hyperuricemia itself is considered to be a risk factor for atherosclerosis. These findings support the theory that hyperuricemia may be a prognostic factor, regardless of BMI, as in the present study.
A strong point of our study is that regardless of the statistical power, hyperuricemia was associated with incidence of 3-year death and MACLE independently of the BMI, except in the high-BMI group, possibly due to the small number of participants. From these results, hyperuricemia might be a prognostic variable of patient with PAD. To establish hyperuricemia as a novel predictor, well-designed randomized controlled trials would be required (e.g., to assess the effect of drugs that lower serum uric acid levels).
In this study, we did not consider the role of therapeutic interventions. A case-control study reported that the incidence of myocardial infarction was lower in patients treated with allopurinol. 45 Studies have also reported that high-dose allopurinol (600 mg/day) in patients with stable angina prolongs exercise time before ischemia. 46 Goiciechea et al reported that cardiovascular events were significantly lower in the group undergoing uric acid–lowering therapy in a randomized study in which 100 mg/day of allopurinol was administered to patients with renal failure. 47 As described above, there are some reports that uric acid–lowering therapy reduces ischemic events. Uric acid–lowering drugs include febuxostat, a novel non-purine selective inhibitor of xanthine oxidase (XO) and drugs such as losartan and fenofibrate are also known to have having a uric acid–lowering effect.48,49 Febuxostat did not suppress the progression of carotid atherosclerosis in patients with asymptomatic hyperuricemia. 50 Therefore, it is unclear whether it suppresses arteriosclerosis like other uric acid lowering drugs. As mentioned above, the evidence of therapeutic interventions for hyperuricemia is insufficient.
Uric acid production inhibitors were used in these studies, and it is unclear whether the effects on cardiovascular disease are related to the decrease in uric acid or the suppression of uric acid production. Therefore, an examination of the therapeutic effect of drugs on patients without a high BMI or metabolic syndrome, as in this study, is necessary.
There are many factors that need to be clarified, such as the effects of drug treatment as described above and the therapeutic goals for uric acid levels. Further research is needed to overcome any uric acid–related residual risk.
Limitations
This study had some limitations. First, it was a retrospective study and was at risk of bias. Second, the patient population included in this study was relatively small. This might have influenced the subgroup analysis. Third, this study did not consider the presence or absence of therapeutic interventions for hyperuricemia. The presence or absence of oral medication was not investigated. Finally, this study also does not include diuretic data. Diuretics can affect renal function and uric acid levels. 51 Hence, the absence of these data is a limitation of this study.
Conclusion
In the present study, the post-EVT prognosis of PAD patients with hyperuricemia was found to be worse than that of patients without hyperuricemia, regardless of BMI. Hyperuricemia may be a residual risk for patients with PAD, as for patients with coronary artery disease.
Footnotes
Acknowledgments
We thank all the patients, investigators, and staff who participated in this study.
Author contribution
All authors contributed to: (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data, (2) drafting the article or revising it critically for important intellectual content, and (3) final approval of the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the Shinshu University Ethical Committee. Written informed consent was obtained from all subjects for publication of this study. A copy of the written consent is available for review upon request. The deidentified participant data will not be shared.
