Abstract
Contrast-induced nephropathy (CIN) is common. Risk factors include preexisting renal impairment, diabetes, elderly age, and dehydration. In a single-centre prospective study, we investigated which factors are implicated for CIN in patients with peripheral arterial disease due for angiography. Serum creatinine was measured before, 1, 2, and 7 days post-angiography. We also considered the chronic kidney disease stage of the patients at admission and 48 hours post-contrast. All patients received 500 mL normal saline pre- and post-angiography and a low-osmolality contrast medium. 6 of 94 patients developed CIN: 1 required dialysis and 1 died partly due to renal failure. Only 2 factors were associated with CIN: body mass index (BMI; P = .019) and kidney function (P = .001); 4 of 6 patients with CIN were obese (BMI ≥30) and only 2 were nonobese (P = .0092). Diabetes, contrast volume, and age were not significant risk factors. Our results confirm renal impairment raises the risk of CIN. To our knowledge, we report for the first time that obesity may be a risk factor for CIN. Pending confirmatory studies and given the rising prevalence of obesity, this finding could help identify at-risk patients and hence reduce the burden of CIN.
Introduction
Imaging techniques such as computed tomography scanning and angiography use radiographic contrast media (RCM), which can cause an acute decline in kidney function — known as contrast-induced nephropathy (CIN). 1,2 The exact mechanisms involved in the decline in renal function following RCM use are not known. However, studies based on animal models suggest that acute renal tubular injury or necrosis is involved, which is of a lesser degree compared to other forms of renal injury or from reduced function of the renal tubular epithelium. 1,2 Alterations in renal perfusion and oxygenation are considered the main mechanisms. 3
Contrast-induced nephropathy has been defined in several ways 4 ; we chose a rise of 25% or 0.5 mg/dL (∼45 μmol/L) in serum creatinine (SCr) at 48 hours post-RCM as it is the commonest definition 1,4 and hence allows comparison with other studies. Quoted rates of CIN vary from 0% to 90% depending on the presence or absence of risk factors such as preexisting renal insufficiency, hypovolemia, the dose and type of RCM used, diabetes mellitus, hypertension, advanced age, and concurrent intake of potentially nephrotoxic drugs. 5 –9 In a meta-analysis of 18 061 patients undergoing coronary angiography, the incidence of CIN was 3.8%. 10 Apart from periprocedural hydration, which seems to reduce but not completely prevent the risk of renal injury following RCM administration, 11 –13 the results of other agents such as dopamine, mannitol, N-acetylcysteine, and captopril are equivocal. 14 –20 Statin therapy has also been extensively investigated with some evidence of a protective effect, especially for high-dose statin therapy prior to coronary angiography—particularly in the context of acute coronary syndromes. 21,22 However, there is some uncertainty on the impact of statins on renal disease and kidney protection. 23 In summary, neither the joint British Renal Association, Cardiovascular Intervention Society, and Royal College of Radiology Guidance nor the American College of Radiology Committee have supported the use of any drugs to prevent CIN. 24,25
Patients with peripheral arterial disease (PAD) are known to have many risk factors that increase the risk of CIN, including diabetes mellitus, pre-existing renal disease, and renal artery stenosis. 4 We aimed to identify the risk of CIN in this group of patients who underwent angiography with prophylactic hydration and to identify the risk factors involved.
Methods
We invited all patients with PAD due for elective angiographic assessment or intervention to participate in this study. Approval was obtained from the ethics committee of the Royal Free Hampstead NHS Trust.
Structured History
A clinical history was obtained with specific reference to a diagnosis of diabetes mellitus, age, gender, and renal impairment. Patients with end-stage renal failure currently on dialysis were excluded from this study.
Body Mass Index
Height without footwear was measured to the nearest centimeter and weight without footwear and with the patient in a hospital gown measured to the nearest 0.5 kg. Body mass index (BMI) was calculated by dividing the weight (kg) by the height2 (m2).
All patients received 500 mL of normal saline over 4 to 6 hours and 6 to 12 hours prior to the angiographic procedure and again after it. Serum creatinine was measured prior to angiography and again at 1 and 2 days after angiography.
Outcome measures were change in SCr, incidence of CIN, and any significant morbidity and mortality. We also noted the chronic kidney disease (CKD) stage of the patient at admission and any change at 48 hours postprocedure.
We also made note of the type of procedure and volume of RCM used. All patients received Omnipaque 300 (300 mg of iodine/mL of solution) marketed by GE Healthcare AS.
Statistics
Associations were sought between each potential risk factor and CIN. Those risk factors found to be significantly related in univariate analysis were entered into a multiple logistic regression model (SPSS version 11.0) so that independent associations could be identified.
Results
A total of 94 consecutive patients were recruited. Six patients developed CIN: 1 required dialysis and 1 died partly due to renal failure.
Of the 94 patients, 45 had infrainguinal and 8 aortoiliac interventional procedures (angioplasty/stenting); there were 31 lower limb, 7 carotid, and 3 upper limb angiograms. Of the 6 patients with CIN, 3 had diagnostic lower limb angiograms and 3 lower limb angioplasties.
Table 1 shows that increasing SCr and BMI are associated with CIN. This is shown graphically in Figure 1. In this study, CIN was not affected by RCM dose, diabetic history, age, or gender. To convert from contrast dose in gram to volume of contrast in milliliter using Omnipaque 300 (GE Healthcare AS), we divided by 0.3, as each milliliter of Omnipaque 300 has 300 mg of iodine.
Potential Risk Factors and Their Impact on CIN.a,b
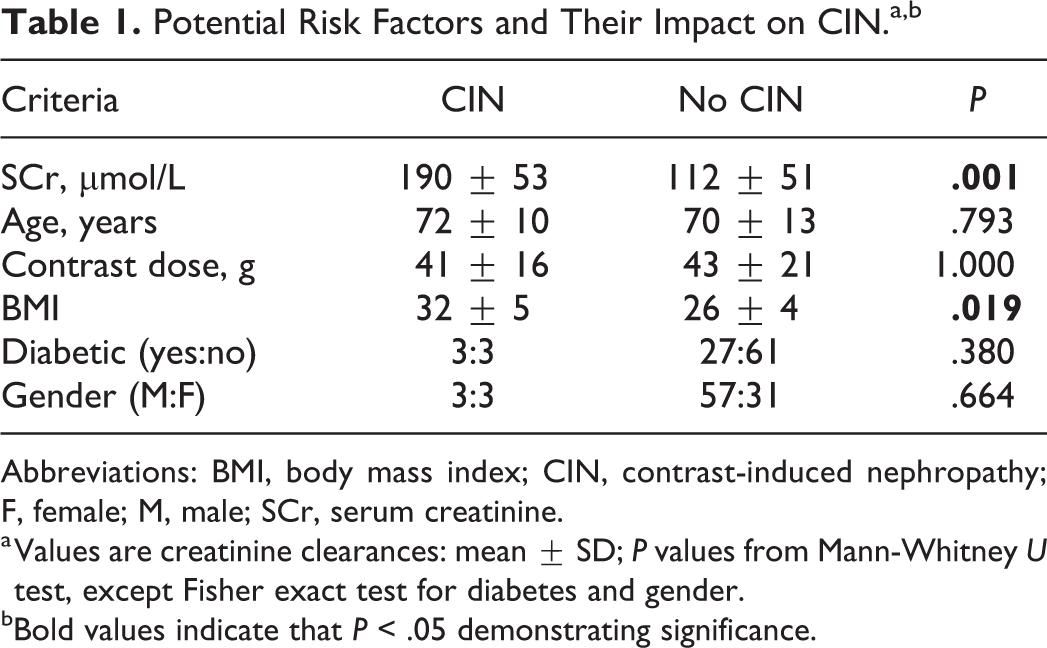
Abbreviations: BMI, body mass index; CIN, contrast-induced nephropathy; F, female; M, male; SCr, serum creatinine.
a Values are creatinine clearances: mean ± SD; P values from Mann-Whitney U test, except Fisher exact test for diabetes and gender.
bBold values indicate that P < .05 demonstrating significance.
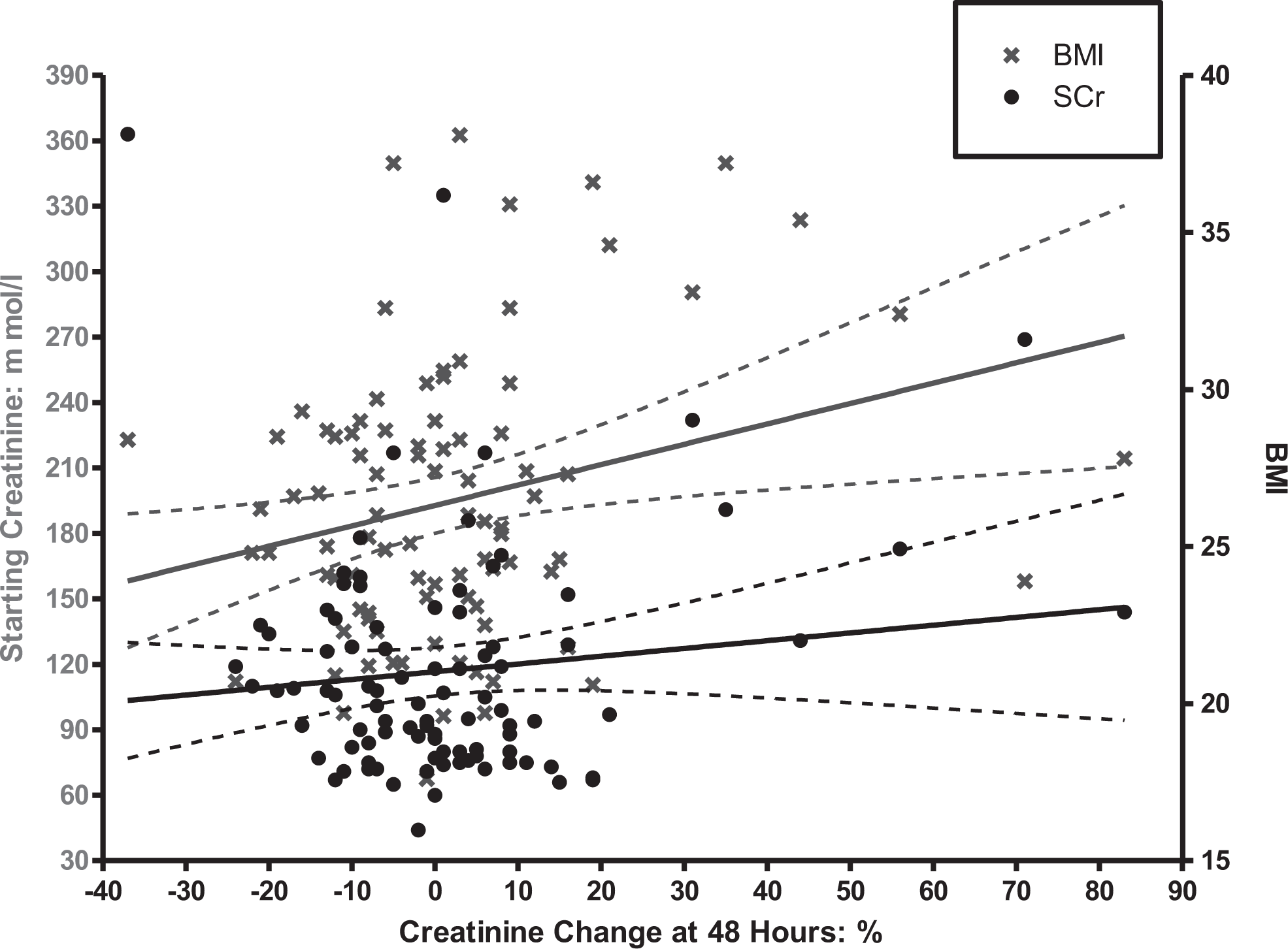
Creatinine change at 48 hours plotted against BMI and baseline SCr. BMI indicates body mass index; SCr, serum creatinine.
Logistic regression confirmed BMI to be a risk factor for CIN (P = .012). Furthermore, of the obese patients (BMI >30), 4 of 17 developed CIN compared with 2 of 77 for the nonobese (P = .0092: Fisher exact test).
Every patient with CIN had a raised baseline SCr — which in our hospital corresponds to an SCr >120 mmol/L (1.32 mg/dL) for males and >97 mmol/L (1.07 mg/dL) for females. Given that 38 patients had raised SCr before administration of RCM (ie, baseline), this means in the CIN group, 100% had raised SCr, whereas only 36% (32/88) in the non-CIN group had a raised SCr (P = .0034: Fisher exact test).
In terms of CIN and change in CKD stages, 3 of the 17 patients with BMI >30 had CIN and deterioration in CKD stage 48 hours post-intervention (P = .039). However, only 2 of 77 patients with BMI of <30 developed CIN and deterioration in CKD. One patient had CIN as per our criteria but did not show any change in CKD stage, which remained at 4.
Discussion
To our knowledge, this study is the first to identify obesity as a risk factor for CIN. One can only speculate as to what the reasons might be: perhaps contrast or nephrotoxic metabolites are held within body fat for longer — it is well known that volumes of distribution, binding, and elimination of drugs are unpredictable in obese patients. 26 The uncertainty is not helped by the fact that we do not fully understand the mechanism involved in CIN. Theories focus on outer medullary hypoxia with subsequent oxidative stress and repair, perhaps through the generation of free oxygen radicals. 3,27,28
We confirmed that pre-existing renal impairment is also a risk factor for CIN. Our group has previously shown that vascular patients with normal SCr have a significant level of occult renal impairment. 29 However, in the context of this relatively small study, there is reassurance that only patients with a raised SCr developed CIN. Of the patients with raised SCr, 6 (16%) of 37 had CIN, which is consistent with other studies. 30,31
Surprisingly, RCM dose and diabetes were not identified as risk factors. This would go against some of the previous work in the literature, though with only 30 patients identified as having diabetes, any comment on diabetes may be limited by the small numbers involved. Furthermore, diabetes is sometimes only identified as a risk factor when combined with renal impairment. 4 Part of the explanation may lie in the use of intravenous hydration pre- and post-RCM to dilute the impact of RCM dose and mitigate against the tendency for diabetic patients, in particular, to get dehydrated. We do note that patients with diabetes on metformin need particular care with respect to the potential development of lactic acidosis, and the Contrast Media Safety Committee of the European Society of Urogenital Radiology states that patients with an estimated glomerular filtration rate <30 mL/min/1.73 m2 should withhold metformin for 48 hours and only restart metformin if the renal function has not changed significantly. 32
We did not vary the volume and protocol of hydration for weight, renal function, or heart failure status for practical reasons of ensuring protocol adherence, which may be a limitation, though none of our patients had significant renal or heart failure. More recently, the value of prophylactic hydration has been questioned. 33 Furthermore, there is some discussion in the literature suggesting that the RCM dose issue may be specifically related to coronary interventions where bolus doses of contrast directly enter the renal arterial circulation. 34,35 Although diabetes is associated with obesity, in this study, of the 4 obese patients with CIN, only 1 had diabetes (type 2). In contrast, of the 2 nonobese patients, both were diabetic. Obviously, such an analysis is limited by the small number of patients. Our study is relatively small, and we acknowledge that a retrospective study did not find such a link with obesity. 36 However, our study was prospective and was focused on predominantly intra-arterial rather than intravenous contrast administration. Certainly, the findings with regard to obesity need to be repeated in a larger study. However, if obesity is indeed a risk factor for CIN, the implications are considerable.
The incidence of obesity — defined by a BMI of ≥30 — is rising throughout the Western world in particular, 37 with up to a third of UK adults affected by 2010. 38 Obesity is also associated with the development of many disease processes such as type 2 diabetes mellitus, hypertension, atherosclerotic diseases, and cancer. 39 –41 Furthermore, obesity is associated with reduced quality of life and life expectancy. 42,43 In regard to renal disease, obesity has been shown to be a risk factor in its development. 44 Furthermore, the metabolic syndrome—central obesity, dyslipidemia, hypertension, and impaired glucose tolerance—is associated with both renal disease and mortality with renal failure. 45,46
Given the small size of the studied population, we were unable to investigate several potential factors in CIN such as drug therapy and the presence of the metabolic syndrome. There is evidence in the cardiology literature of potential benefits of statin therapy and even loading, but this may be hard to study now 21,22 as patients with PAD should all be on high-dose statin therapy (eg. atorvastatin 80 mg) as per all the established national and international guidelines. 47,48 Certainly, it would seem prudent to ensure this is the case prior to intra-arterial contrast studies.
The main results of this study are 3-fold. Firstly, many patients undergo RCM examinations without having renal function checked postprocedure. This is especially the case with patients undergoing day-case investigations. We feel that renal function-impaired (SCr above the normal range) patients must have post-RCM SCr checked. Other patients who could be identified as being at risk and therefore considered for monitoring include obese patients, diabetic patients, and those receiving large RCM doses. Secondly, all patients must be hydrated adequately. Our belief is the only way to ensure this is with the use of intravenous hydration pre- and post-RCM administration. Thirdly, using CKD staging to define a real clinical end point, our study shows a significant correlation between obesity and decline in CKD stage in the CIN group of patients. Hence, this reinforces the view that there is a clinical importance to the effect of RCM on renal function in obese patients.
Footnotes
Authors’ Note
All authors contributed to (1) substantial contributions to conception and design, or acquisition of data, or analysis and interpretation of data; (2) drafting the article or revising it critically for important intellectual content; and (3) final approval of the version to be published.
Acknowledgments
The authors thank Dr Richard Morris from the Department of Primary Care & Population Science, University College London, for his statistical advice in analyzing the data. The authors also thank Ms Sarah Louth who assisted with the data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
