Abstract
Background
Gastric neuroendocrine tumors (gNETs) are rare malignancies with distinct biological behavior compared with gastric adenocarcinoma. Data directly comparing surgical and oncologic outcomes, including lymph node yield, recurrence pattern, and survival, remain limited.
Methods
A retrospective review of 285 gastrectomies (2014-2024) identified 20 gNET and 265 adenocarcinoma cases. After excluding palliative procedures and mixed histology, propensity-score matching (1:2.5; age, sex, BMI, and comorbidities) yielded 18 gNET and 45 adenocarcinoma patients. Outcomes included lymph node harvest, nodal metastasis, lymph node ratio (LNR), recurrence pattern, disease-free survival (DFS), and overall survival (OS).
Results
Median lymph node yield was lower in gNET than in adenocarcinoma (18 vs 28; P = 0.004), and the overall rate of nodal metastasis did not differ between groups (44.4% vs 44.4%; P = 1.00), although the nodal metastatic burden was significantly lower in gNET as reflected by a lower median number of positive nodes and lymph node ratio (LNR 0.05 vs 0.17; P = 0.008), with no gNET patient exhibiting pN3 disease. DFS was comparable (21.4 vs 18.7 months; P = 0.617), whereas OS favored gNET (45.3 vs 27.4 months; P = 0.045). Peritoneal recurrence was markedly less frequent in gNET (16.7% vs 81.3%; P = 0.003), while hepatic relapse predominated. Perioperative morbidity and 90-day mortality were similar.
Conclusion
Compared with adenocarcinoma, gastric neuroendocrine tumors show reduced nodal metastatic burden, lower peritoneal dissemination, and improved overall survival. These findings support biology-adapted lymphadenectomy and surveillance for gNET, although validation in larger cohorts is required.
Introduction
Gastric neuroendocrine tumors (gNETs) arise from enterochromaffin-like (ECL) cells of the gastric mucosa and represent a biologically and clinically distinct subset of gastric neoplasms compared to adenocarcinoma.1,2 Their incidence has increased markedly over the past 2 decades, largely due to improved endoscopic surveillance, refined histopathologic techniques, and evolving WHO classification systems.2,3
In this context, because evidence-based surgical guidelines for gastric neuroendocrine tumors remain limited, operative management, including the extent of lymphadenectomy—is frequently extrapolated from gastric adenocarcinoma paradigms, despite known biologic differences.
gNETs are traditionally categorized into three major clinicopathologic types: Type I, associated with autoimmune atrophic gastritis and hypergastrinemia; Type II, linked to MEN1 or Zollinger-Ellison syndrome; and Type III, sporadic, gastrin-independent, and often more aggressive.3-5 Type I and II gNETs are typically multifocal, small, and limited to the mucosa or submucosa, making them amenable to endoscopic resection.6,7 In contrast, Type III gNETs tend to present as solitary, invasive lesions with a higher risk of nodal or distant metastases, often requiring formal gastrectomy with lymphadenectomy.8-10
The ENETS and NCCN guidelines have emphasized that the surgical management of gNETs should be individualized according to tumor type, grade, and depth of invasion, rather than being automatically modeled after gastric adenocarcinoma.4-6,9,11 This distinction stems from the well-recognized biological differences between gNET and adenocarcinoma: gNETs more commonly disseminate via hematogenous or lymphatic routes, whereas adenocarcinoma frequently spreads transcoelomically with peritoneal seeding.8,12
Nevertheless, in clinical practice, the extent of lymphadenectomy, recurrence surveillance strategy, and adjuvant therapy for gNETs continue to be extrapolated from adenocarcinoma protocols in routine practice. Robust comparative surgical outcome data remain scarce, as most published series are small, single-institution cohorts without adequate matching for patient comorbidities or operative approach.13-15
To address this gap, we conducted a propensity-matched cohort study comparing gNETs and gastric adenocarcinomas undergoing curative-intent gastrectomy. We aimed to assess differences in lymph node harvest, nodal metastasis burden, recurrence patterns, disease-free survival (DFS), and overall survival (OS), and to evaluate whether applying adenocarcinoma-based surgical paradigms to gNETs results in distinct lymph node burden, recurrence patterns, and survival outcomes.
While prior studies have described clinicopathologic features and outcomes of gastric neuroendocrine tumors, no contemporary analyses have directly tested whether adenocarcinoma-derived surgical paradigms—particularly the extent of lymphadenectomy—are appropriate when applied to this pathology.
Methods
Study Design and Patient Selection
This retrospective cohort study included all patients who underwent curative-intent gastrectomy for gastric malignancy at a tertiary referral center between January 2014 and July 2024. Exclusion criteria included neuroendocrine carcinoma (NEC), mixed adenoneuroendocrine carcinoma (MANEC/MiNEN), palliative procedures, and follow-up <30 days. Final analysis compared patients with gastric neuroendocrine tumors (gNETs) vs gastric adenocarcinoma.
Data Collection
Clinical, operative, and pathological data were obtained from electronic records and a prospectively maintained departmental database. Variables included demographics (age, sex, BMI, and comorbidities), ASA score, tumor location, histologic type, WHO grade, Ki-67 index, surgical procedure, extent of lymphadenectomy (D1 vs D2), total and metastatic lymph nodes, lymph node ratio (LNR), postoperative complications, recurrence, and survival outcomes.
Extent of Lymphadenectomy
The extent of lymphadenectomy for gastric neuroendocrine tumors was determined according to a biology- and risk-adapted strategy based on current ENETS and NCCN recommendations. Upfront surgical resection was performed for tumors >20 mm, lesions with suspected muscularis propria invasion on preoperative imaging or EUS, or tumors exhibiting high-risk features on biopsy (eg, higher-grade lymphovascular invasion). In patients with well-differentiated, clinically node-negative disease, a limited gastrectomy with formal D1 lymphadenectomy and regional nodal sampling was considered adequate. D2 lymphadenectomy and/or total gastrectomy was selectively performed in cases with radiologic suspicion of nodal involvement, higher-grade tumors, or as a completion procedure when final pathology demonstrated lymphatic spread, acknowledging that high-level evidence supporting routine extended dissection in gNET is lacking.
In contrast, D2 lymphadenectomy constituted standard practice for resectable gastric adenocarcinoma at our institution. Thus, differences in D1 vs D2 distribution reflect intentional, guideline-driven tailoring rather than variability in surgical thoroughness.
Histopathologic grading followed the 2019 WHO classification of digestive system tumors, 2 and tumor staging was based on the ENETS TNM framework for foregut neuroendocrine neoplasms. 1 Immunohistochemistry for chromogranin A and synaptophysin confirmed neuroendocrine differentiation.
Definitions and Outcomes
• Major complications were defined as Clavien-Dindo grade ≥III. • Ninety-day mortality encompassed all-cause death within 90 days postoperatively. • Disease-Free Survival (DFS) was defined from the date of surgery to the first recurrence or disease-related death. • Overall Survival (OS) was defined from the date of surgery to death from any cause or last follow-up. • Recurrence pattern was classified as peritoneal, hepatic, nodal, or other distant sites.
Propensity-Score Matching (PSM)
To minimize baseline selection bias, patients with gNET were matched to adenocarcinoma patients using nearest-neighbor matching (ratio 1:2.5, caliper 0.30) on age, sex, BMI, and major comorbidities (hypertension, diabetes, ischemic heart disease, chronic kidney disease, COPD, and cerebrovascular disease). An SMD <0.20 was accepted as adequate balance. The extent of lymphadenectomy was intentionally not included as a matching variable, as it reflected tumor biology-driven surgical decision-making and constituted part of the exposure of interest rather than a confounder.
Follow-Up Protocol
Patients were followed at 3 to 6-month intervals for the first 2 years, then annually, with clinical evaluation, tumor markers, and contrast-enhanced CT imaging. Recurrence was confirmed radiologically or histologically.
Statistical Analysis
Categorical variables were compared using χ2 or Fisher’s exact test, and continuous variables using the Student’s t-test or Mann-Whitney U test (depending on Shapiro-Wilk normality). Survival curves were plotted via Kaplan-Meier analysis and compared using the log-rank test. P < 0.05 was considered significant. Analyses were performed with SPSS v29.0 (IBM Corp.) and R v4.3.3.
Ethical Approval
The institutional ethics committee approved the study (0651-20-TLV). Individual patient consent was waived in accordance with national regulations due to the retrospective, anonymized design.
Results
Study Cohort
Baseline Characteristics of Patients With Gastric NET vs Matched Adenocarcinoma (1:2.5 PSM)
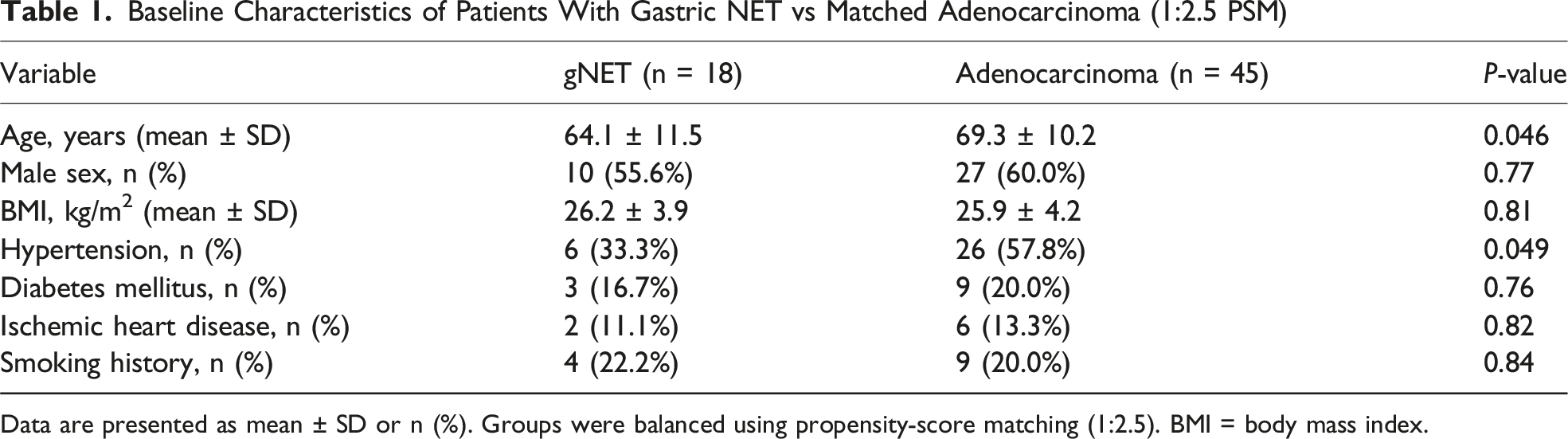
Data are presented as mean ± SD or n (%). Groups were balanced using propensity-score matching (1:2.5). BMI = body mass index.
After exclusion of palliative procedures, NEC/MiNEN histology, and incomplete follow-up, 18 gNET patients were eligible and matched with 45 adenocarcinoma patients (1:2.5 ratio). Baseline demographics and comorbidities were well balanced except for slightly older age and more hypertension in the adenocarcinoma cohort.
Tumor Characteristics
Pathologic Features and Lymph Node Status After Matching
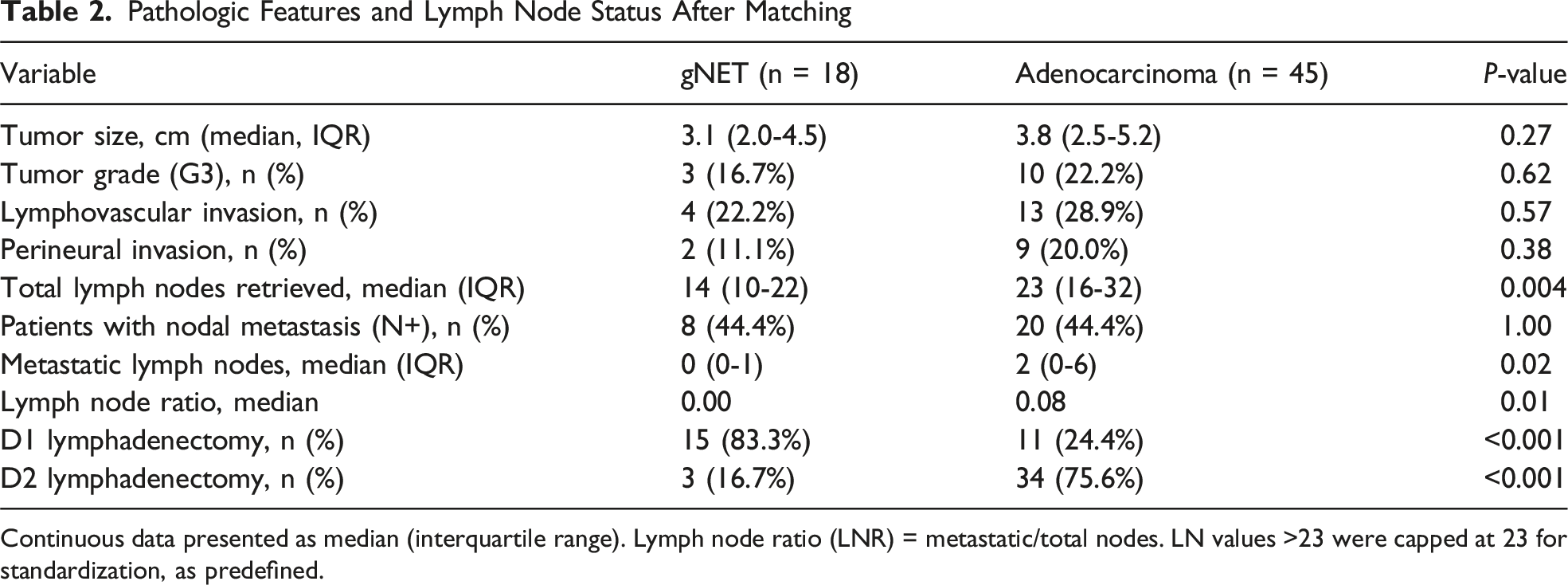
Continuous data presented as median (interquartile range). Lymph node ratio (LNR) = metastatic/total nodes. LN values >23 were capped at 23 for standardization, as predefined.
Lymph Node Harvest and Metastatic Burden
The median lymph node yield was significantly lower for gNET (median 18 [IQR 13-26]) than for adenocarcinoma (28 [IQR 22-41]; P = 0.004), consistent with the more frequent use of D1 lymphadenectomy in gNET. In the propensity-matched cohort, nodal metastases were present in 44.4% of both gNET and adenocarcinoma patients (P = 1.00). However, gNETs demonstrated a significantly lower nodal metastatic burden, reflected by fewer positive lymph nodes and a lower lymph node ratio (median LNR 0.05 vs 0.17; P = 0.008), and no gNET patient exhibited pN3 disease.
Perioperative Outcomes
Major postoperative complications (Clavien-Dindo grade ≥ III) occurred in 11.1% of gNET patients and 15.6% of adenocarcinoma patients (P = 0.64). Reoperation rates were comparable (5.6% vs 6.7%, P = 0.87), as were ICU admissions (16.7% vs 22.2%, P = 0.59). Median postoperative length of stay did not differ between groups (13 [IQR 10-17] vs 14 [IQR 11-18] days, P = 0.72). Thirty-day mortality was 0% in the gNET group and 2.2% in the adenocarcinoma group (P = 0.48), while 90-day mortality was similar (5.6% vs 6.7%, P = 0.87). Readmission within 30 days occurred in 5.6% of gNET patients and 8.9% of adenocarcinoma patients (P = 0.67).
Recurrence and Survival
Short-Term and Long-Term Oncological Outcomes (Matched Cohort: 18 gNET vs 45 Adenocarcinoma)
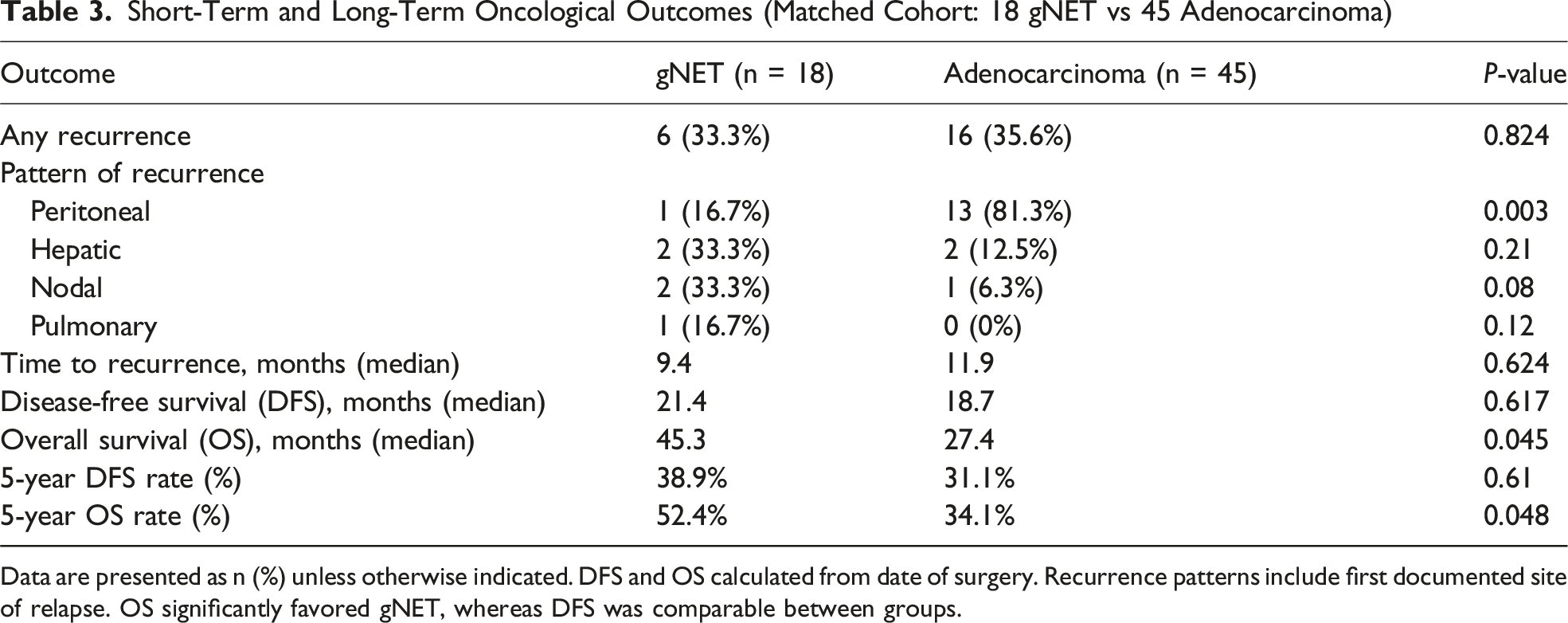
Data are presented as n (%) unless otherwise indicated. DFS and OS calculated from date of surgery. Recurrence patterns include first documented site of relapse. OS significantly favored gNET, whereas DFS was comparable between groups.
Discussion
This propensity-matched study demonstrates that gastrectomy for well-differentiated gastric neuroendocrine tumors achieves similar perioperative safety and DFS to gastric adenocarcinoma, yet provides superior OS and markedly reduced peritoneal recurrence. These findings reaffirm the biological divergence between gNET and adenocarcinoma.2,4-6,8,10,11,13
Tumor Biology and Grading
Contrary to the older belief that most surgically resected gNETs are aggressive Type III lesions,3,8 the majority here were WHO Grade 1-2 with low Ki-67 (≈2 %), paralleling recent large-scale analyses.7,13,16 High-grade (G3) cases were rare. These data reinforce the prognostic relevance of histologic grade and proliferative index over the classic Type I-III scheme.2,7,9,10
Extent and Impact of Tailored Lymphadenectomy
Our selective approach—D1 lymphadenectomy for well-differentiated, low-risk tumors and D2 dissection for higher-risk or node-suspected disease—mirrors current ENETS and NCCN recommendations emphasizing biology-adapted surgery rather than uniform extrapolation from gastric adenocarcinoma paradigms.4-6,11 Accordingly, the higher proportion of D1 dissections in the gNET cohort reflects deliberate, guideline-driven risk stratification rather than less meticulous nodal clearance. Despite fewer D2 procedures, lymph node yields exceeded 15 nodes in most gNET cases, satisfying AJCC criteria for adequate pathologic staging and indicating oncologically thorough surgery.1,2,4,16
Current recommendations support limited resection with regional lymph node sampling for well-differentiated, clinically node-negative tumors, while reserving D2 lymphadenectomy or completion gastrectomy for cases with radiologic or histologic evidence of nodal involvement or higher-grade disease, acknowledging the lack of high-level evidence for routine extended dissection in gNET.4-6,11 In this context, the significantly lower lymph node ratio and reduced nodal metastatic burden observed in gNET likely reflect true biological behavior rather than systematic understaging, consistent with multicenter registry and SEER analyses.16,17 This biology-adapted surgical strategy may also contribute to the distinct recurrence pattern, characterized by rare peritoneal dissemination and a predominance of hematogenous spread, further supporting tailored rather than adenocarcinoma-based oncologic paradigms.
Patterns of Recurrence
Our findings confirm that gNET rarely exhibits peritoneal carcinomatosis, consistent with its lymphatic/hematogenous dissemination pattern.8,12 In contrast, adenocarcinoma commonly spreads transcoelomically, explaining the survival gap despite similar DFS. Similar recurrence distributions have been reported by Hirasawa et al and Li et al.15,16
Survival Implications
Although DFS was similar, OS was superior in gNET, likely due to its indolent course, better tolerance of recurrence, and availability of targeted or somatostatin-analog therapy.4,7,9,18 These results align with national registry data showing longer median survival for resected gNETs vs gastric adenocarcinoma.16,17
Operative Outcomes
Major morbidity and mortality rates mirrored those in adenocarcinoma, supporting that gastrectomy for gNET is oncologically safe in experienced centers.10,13,15
Clinical Implications
1. Tailored lymphadenectomy: D1 is adequate for well-differentiated, localized gNET without nodal suspicion; D2 reserved for higher-grade or invasive lesions.4-6,11,16 2. Surveillance focus: Hepatic and nodal imaging should predominate, given rare peritoneal relapse.8,12 3. Adjuvant therapy: The role remains undefined; ongoing trials suggest molecular and peptide-receptor–based strategies.7,9,18
Limitations
This single-center retrospective study is limited by the small sample size and the potential for residual confounding despite propensity-score matching. Accordingly, the present analysis should be interpreted as hypothesis-generating rather than as providing definitive guidance for operative strategy in gastric neuroendocrine tumors. In addition, Type I-II gNETs managed endoscopically were not included, and therefore, the findings primarily apply to surgically treated, predominantly Type III lesions. Nevertheless, the relatively long follow-up, uniform surgical approach, and centralized histopathologic review strengthen the internal validity of the observations.19,20
Conclusions
In this matched cohort, gastrectomy for gNET was associated with a lower nodal metastatic burden, a markedly reduced rate of peritoneal recurrence, and improved overall survival compared with gastric adenocarcinoma, despite similar disease-free survival. Given the retrospective, single-center design and limited sample size, these observations should be regarded as hypothesis-generating. While they require validation in larger, multi-institutional studies, the data suggest that biological differences between gNET and adenocarcinoma may justify future investigation of tailored surgical and surveillance strategies rather than uniform extrapolation from adenocarcinoma paradigms Figure 1. Kaplan-Meier survival analysis for gastric neuroendocrine tumor (gNET) and gastric adenocarcinoma. (A) Disease-free survival (DFS) did not differ significantly between groups (median 21.4 vs 18.7 months; P = 0.617); (B) Overall survival (OS) was significantly longer in gNET (45.3 vs 27.4 months; P = 0.045). Log-rank test used for comparisons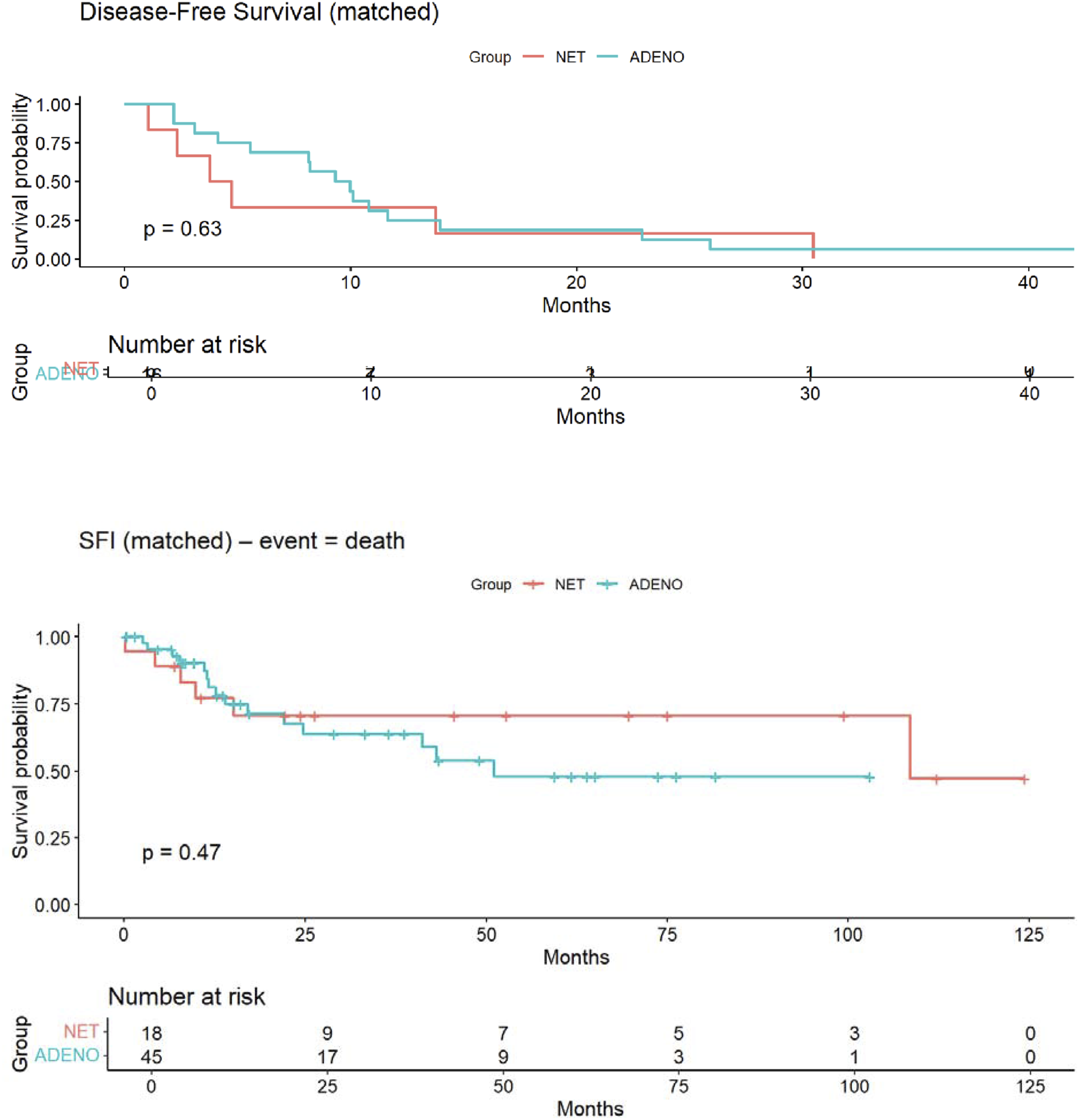
Footnotes
Ethical Considerations
The study was approved by the Tel Aviv Sourasky Medical Center Institutional Review Board (Approval No. 0651-20-TLV). Given the retrospective design and anonymized data collection, the requirement for informed consent was waived.
Author Contributions
Concept and study design: TIW, YL
Data collection and curation: TIW, YL
Statistical analysis: LO, FK
Clinical oversight: EO, BS, RG, IW, GL
Manuscript drafting: TIW, YL
Critical revision and approval of the final version: All authors
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data sets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of AI and AI-Assisted Technologies in the Writing Process
During the preparation of this work, the authors used ChatGPT and Grammarly for language refinement, consistency checking, and reference organization. The authors reviewed and edited all generated content and take full responsibility for the scientific accuracy and integrity of the final manuscript.
