Abstract
Inherited metabolic disorders (IMDs) are a heterogeneous group of rare single-gene disorders caused by enzyme defects that disrupt biochemical and metabolic pathways. Acute metabolic decompensation is a medical emergency that can be fatal if untreated. It can be triggered by catabolic stressors such as fasting, infection, surgery, pain, bleeding, or exposure to anesthetic agents. Traumatic injury in particular can precipitate IMD life-threatening crises. The risks of complicating surgical interventions can be reduced by careful perioperative management of fluids, nutrition, and medications under the guidance of a biochemical genetics specialist. Management of IMD metabolic emergencies is often complex, and trauma and surgical providers may only have limited access to specific protocols. Without prompt recognition and treatment, patients with IMDs are at high risk of poor outcomes in trauma or surgical settings. Optimal management requires early consultation with a metabolic specialist and a coordinated multidisciplinary team. This review highlights key principles and resources to enhance recognition and management of IMD metabolic crises for trauma and surgical teams.
Keywords
Introduction
Common Categories of Small-Molecule Inherited Metabolic Disorders

The clinical manifestations of IMD are variable and can involve multiple organ systems to cause problems in children and adults.5,6 In a subset of IMDs, symptoms can be managed and sometimes prevented but lifelong therapy is typically required. 5 Clinical features can generally be divided into acute and chronic. In general, small-molecule IMDs can precipitate rapid, life-threatening crises under catabolic stress, such as traumatic injury or surgical intervention. 7 In contrast, large-molecule IMDs typically follow a more chronic, progressive course and are less likely to cause sudden metabolic collapse in the acute perioperative setting. 7 Accordingly, this review focuses on small-molecule IMDs that predispose patients to acute metabolic decompensation.
Common Perioperative Triggers of Acute Metabolic Decompensation
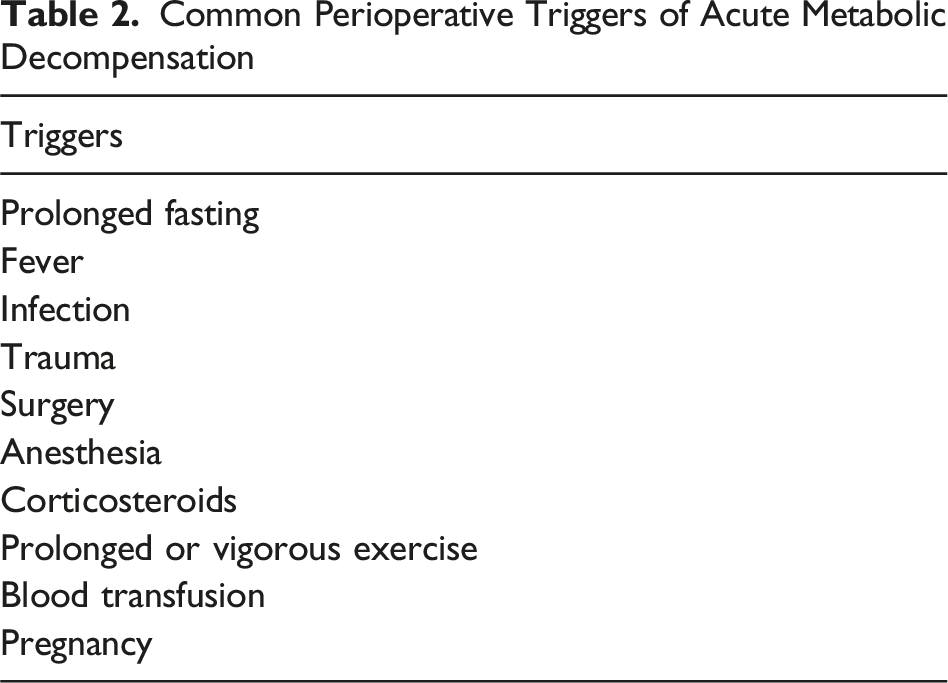
Management of metabolic emergencies in IMDs can be complicated, and access to protocols may be difficult for surgical providers to obtain. Importantly, surgical management of traumatic injuries without access to such protocols can put patients with IMDs at high risk of poor outcomes.13,14 Prompt recognition and timely intervention under the guidance of a metabolic specialist can be lifesaving. Optimal outcomes may require a coordinated, synergistic multidisciplinary approach that includes trauma surgery, medical and biochemical genetics, nephrology, pharmacy, dieticians, and intensive care providers. This review aims to improve the awareness and resources needed by surgical teams by highlighting the recognition and management of metabolic emergencies in IMDs. While this review introduces commonly used management approaches and frequently used medication and fluid doses, these should always be tailored to the individual case and carefully reviewed in collaboration with a biochemical geneticist and the surgical team.
Trauma and Perioperative Considerations
Acute metabolic decompensation of IMDs can be fatal if treatment is delayed. For IMD patients, an acute physical stressor—such as prolonged fasting, fever, infection, and traumatic injury—can initiate a cascade of metabolic disturbances that rapidly perturb metabolic homeostasis (Table 2).7-9,12,13 Muscle breakdown, commonly seen in crush injuries or major orthopedic trauma, can cause the dangerous release of proteins and amino acids into the circulation. Additionally, the physiologic stress response to trauma induces a catabolic state that increases endogenous protein breakdown. 15 Required surgical intervention can also exacerbate catabolism due to the combined effects of anesthesia, fasting, tissue injury, and systemic stress.9,12 Combinations of these factors can precipitate catabolic crises and lead to rapid neurologic deterioration, cerebral edema, and death. 7
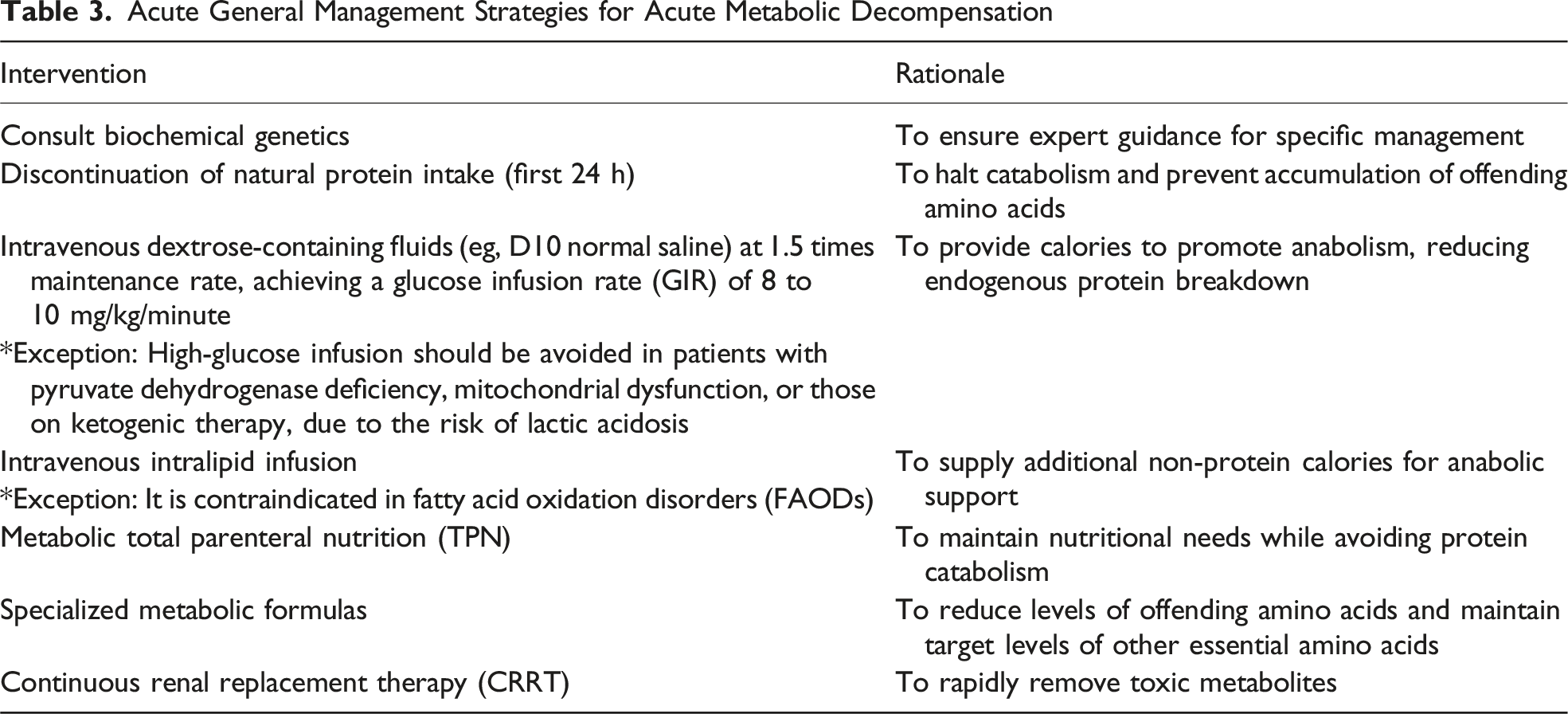
Acute General Management Strategies for Acute Metabolic Decompensation
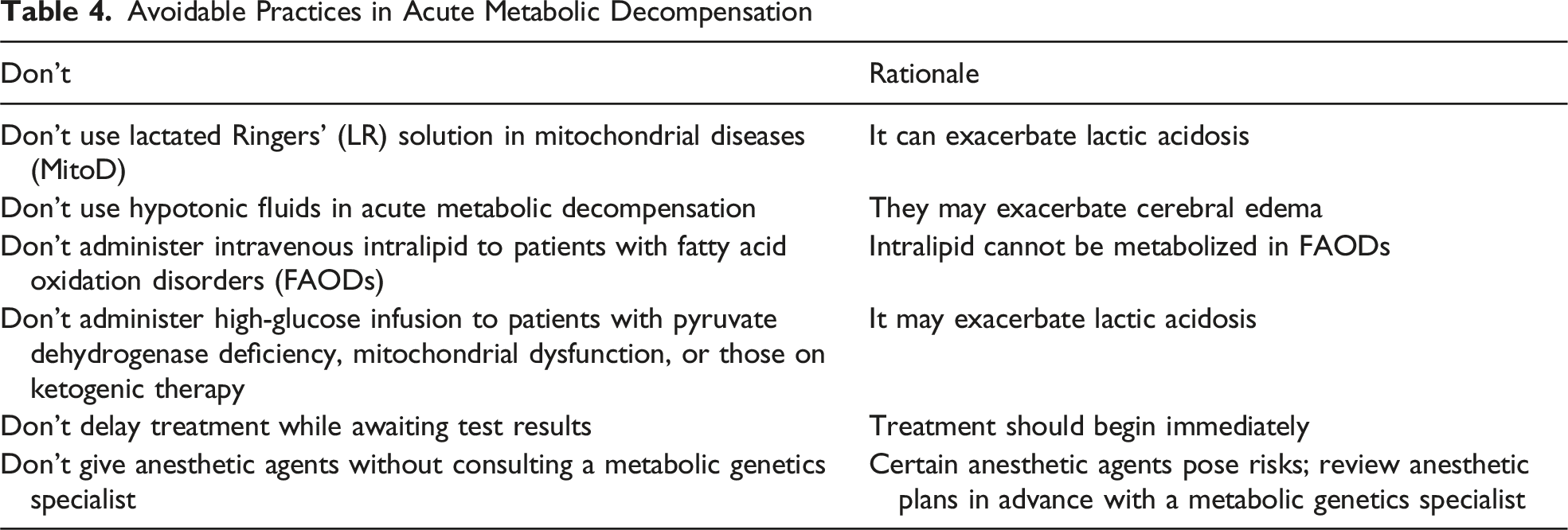
Avoidable Practices in Acute Metabolic Decompensation
Another part of the acute care of IMDs is the rapid removal of toxic metabolites (Table 3). Continuous renal replacement therapy (CRRT) is an effective way to urgently clear toxic metabolites, but it requires early nephrology consultation and placement of a vascular catheter for access. 29 In addition, disorder-specific medications that enhance elimination of toxic metabolites (eg, nitrogen scavengers for hyperammonemia) should be initiated if clinically indicated.22,25 Since pain and associated sympathetic activation can be stressors that accelerate catabolism, effective pain management is an important component of care. Labored breathing and tachypnea can also increase catabolism. Therefore, intubation with ventilation support should not be delayed, accompanied by adequate sedation and analgesia when clinically indicated.9,13
Initial Workup for Acute Metabolic Decompensation in Traumatic Injury

Patients with IMD are at increased risk of anesthesia-related complications. Careful preoperative consultation with an anesthesiologist with a metabolic genetics provider is crucial (Table 3).9,13 In general, preoperative fasting should be minimized, and adequate glucose should be administered to prevent catabolism. Dextrose-containing intravenous fluids (eg, D10 normal saline) are preferred, and LR solution should be avoided—particularly in MitoD—because of its potential to exacerbate lactic acidosis (Table 4).26,27 While a higher infusion rate (eg, 1.5 times the maintenance rate) of D10 normal saline or D10 half normal saline is generally ideal to achieve an adequate GIR, fluid management should be tailored on an IMD-specific basis. For example, some IMDs cause cardiomyopathy and heat failure in OAs as well as cerebral edema in MSUD. To avoid volume overload, a more concentrated dextrose-containing fluid (eg, D12.5 or D15 normal saline) at a relatively slower rate may be needed.
21
In patients with pyruvate dehydrogenase deficiency, mitochondrial dysfunction, or those on ketogenic therapy, high-glucose infusion should be avoided because it may exacerbate lactic acidosis (Table 4).26,27 While dextrose fluids containing up to 10% dextrose can be infused through peripheral intravenous catheters, higher concentrations of dextrose require a central intravenous catheter.
31
That said, emergent dextrose infusion should not be delayed. Anesthetic agents must be selected with caution due to their potential adverse effects in metabolic disorders, and anesthetic plans should be discussed in advance with an anesthesiologist and a metabolic genetics specialist (Table 4).9,13 For instance, propofol is contraindicated in both MitoD and FAODs.26,27,32 Corticosteroids should also be used cautiously because they promote catabolism and may precipitate metabolic crisis (Table 2).
33
Absorption of blood extravasated into tissues can trigger decompensation of some IMDs because of absorbed protein (Table 2).
11
Communication with a metabolic specialist during surgery along with serial laboratory monitoring can help optimize fluid and medication management. A suggested management algorithm of traumatic injury in patients with IMD is summarized in Figure 1.21-24,30 Algorithm for Management of Traumatic Injury in Patients With Inherited Metabolic Disorders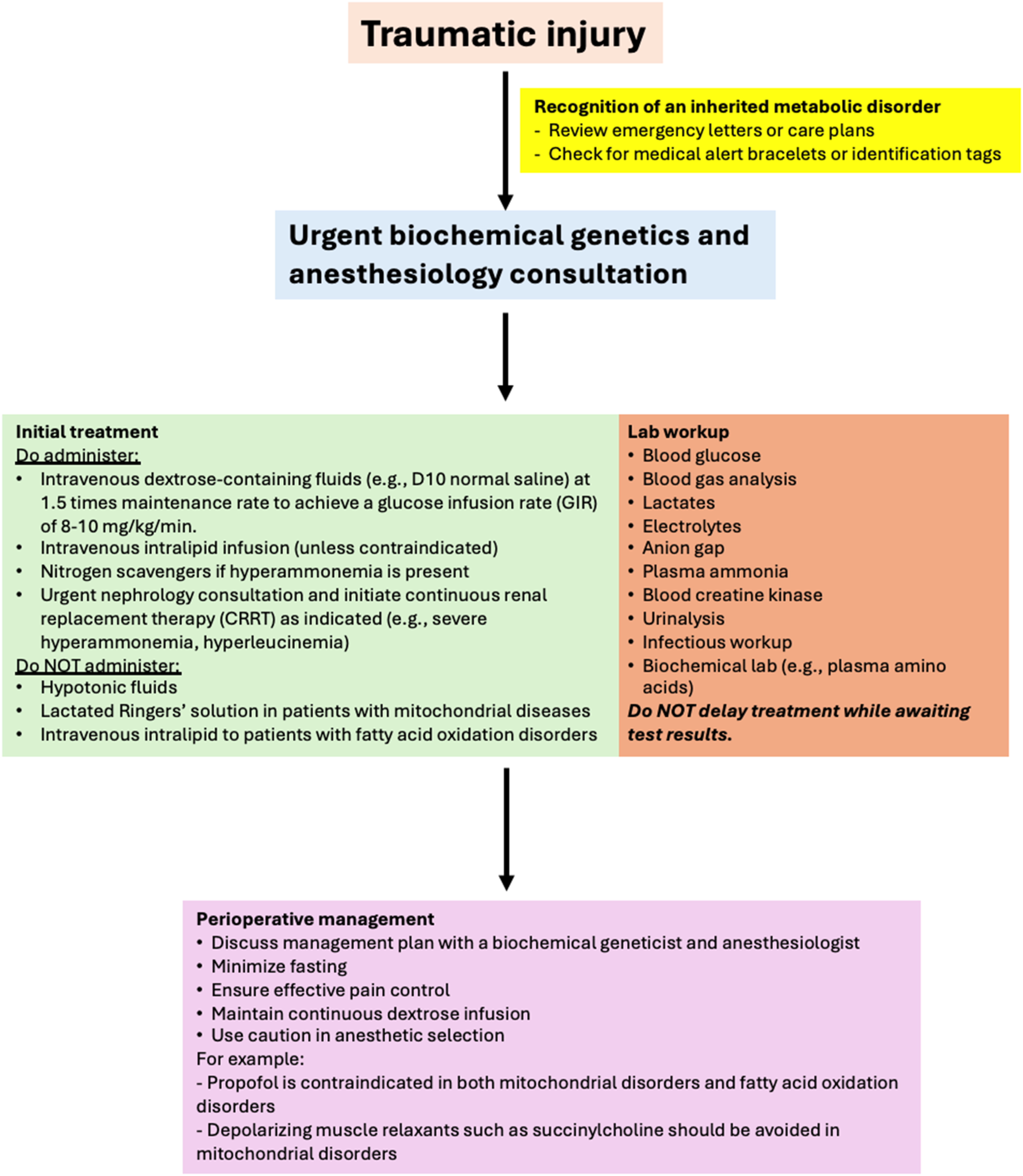
Specific Scenarios
Fatty Acid Oxidation Disorders (FAODs)
Fatty acid oxidation disorders result from defects in either mitochondrial β-oxidation or the fatty acid transport through the carnitine transport pathway (Table 1).34,35 Depending on the subtypes—short-chain, medium-chain, or long-chain—common clinical manifestations include non- or hypoketotic hypoglycemia, skeletal and cardiac muscle involvement (eg, muscle cramps, rhabdomyolysis, cardiomyopathy, and arrhythmia), and, more rarely, encephalopathy.34,36,37
Because individuals with FAODs cannot utilize ketone bodies as an essential energy source during catabolic stress such as fasting, infection, and surgery. Fatty acid oxidation disorders can cause non- or hypoketotic hypoglycemia (Table 2). High-energy-dependent organs, including skeletal and cardiac muscle, and the brain, can be particularly affected in FAODs.34,37 To promote anabolism and prevent hypoglycemia, it is essential to administer a higher GIR using dextrose-containing fluids (eg, D10 normal saline) (Table 3).21,37 Importantly, intralipids are contraindicated in FAODs (Table 4). Carnitine supplementation may be beneficial when plasma carnitine levels are deficient, to prevent secondary carnitine deficiency by enhancing renal clearance of accumulated levels of abnormal acylcarnitine compounds.21,37 However, intravenous carnitine administration can be contraindicated in long chain FAODs because of its potential to precipitate serious cardiac complications, including life-threatening arrhythmias. 38 In cases of rhabdomyolysis, aggressive hydration is indicated to prevent renal injury. 21 Conversely, in patients with evidence of heart failure, fluid volume must be minimized to avoid volume overload. 21 Anesthesia with propofol should be avoided in FAODs because propofol contains long-chain fatty acids, which cannot be adequately metabolized and increases the risk of propofol infusion syndrome (PRIS). 32
Urea Cycle Disorders (UCDs)
The urea cycle is responsible for ammonia detoxication (Table 1). In UCDs, defects in any of the enzymes involved lead to impaired ammonia clearance, resulting in hyperammonemia, particularly under catabolic stress. 25 Elevated ammonia level can precipitate acute encephalopathy, irreversible cerebral damage, coma, and even death. 25 Respiratory alkalosis can be an early indicator of UCDs, in contrast to the metabolic acidosis commonly observed in OAs, which may also present with hyperammonemia (Table 5).20,25 In UCDs, elevated ammonia levels cause respiratory alkalosis from hyperventilation triggered by stimulation of the central respiratory drive in the brain. To prevent irreversible neurological injury and cerebral edema, it is crucial to reduce plasma ammonia level without delay.
In patients with UCDs presenting with acute hyperammonemia, all protein intake should be initially withheld for 24 h to minimize catabolism, and dextrose-containing intravenous fluids should be initiated to promote anabolism (Table 3).20,22,25,39 Continuous renal replacement therapy is a key intervention for rapid removal of increased levels of ammonia. 29 In addition, nitrogen scavenger therapy (eg, Ammonul, a combination of sodium phenylacetate and sodium benzoate) is a cornerstone of medical management in acute hyperammonemia. These nitrogen scavengers lower ammonia levels by phenylacetate conjugating with glutamine to form phenylacetylglutamine and benzoate conjugating with glycine to form hippurate.22,40 Since these conjugated products are excreted in the urine, adequate renal function is essential for their efficacy. Importantly, nitrogen scavengers are specifically indicated for UCDs and should only be used under the supervision of a metabolic geneticist because nausea and vomiting, hypokalemia, and hypernatremia can be common side effects. Prophylactic use of antiemetics (eg, ondansetron) is recommended before boluses of these nitrogen scavengers.22,40 The high sodium load in Ammonul increases the risk of hypernatremia. Hypokalemia is also commonly observed and requires close potassium monitoring and supplementation. Also, phenylacetate can cause hepatotoxicity. Arginine is an essential amino acid that participates in the final step of the urea cycle, where it is converted by arginase into urea and ornithine. Arginine supplementation is used in treating most UCD crises to help preserve residual urea cycle activity, except in arginase deficiency, in which it is contraindicated (Table 1). 25 As a precursor of nitric oxide, arginine can promote vasodilation and lead to hypotension; so that it should be used with caution in patients who are hemodynamically unstable.
Organic Acidemias (OAs)
Organic acidemias are characterized by the accumulation of toxic organic acid metabolites and increased urinary excretion of organic acids (Table 1). Clinically, these disorders typically present with anion gap metabolic acidosis, ketosis, and hyperammonemia.18,19,23,41 The clinical spectrum of OAs is broad, ranging from encephalopathy, seizures, developmental delay, failure to thrive, and to systemic complications such as cardiomyopathy, arrhythmia, pancreatitis, pancytopenia, and osteoporosis.18,19
The key management of OAs is to prevent endogenous catabolism and promote anabolism (Table 3). Dextrose-containing intravenous fluids should be initiated and when a higher GIR is required, lactate levels should be monitored because lactate can accumulate due to secondary mitochondrial dysfunction caused by a potential interference with tricarboxylic acid (TCA) cycle entry and inhibition of pyruvate dehydrogenase by toxic metabolites. 18 In such cases, insulin infusion may be needed to maintain normoglycemia.18,19,23,41 Intralipid may be necessary as an alternative calorie source.18,19,23 If metabolic acidosis is pronounced, intravenous bicarbonate therapy may also be needed. However, excess bicarbonate administration should be avoided because it may cause hypernatremia and cerebral edema, especially in patients with hyperammonemia.23,28
Similar to UCD hyperammonemia may occur in OAs (Tables 1 and 5). The underlying mechanism involves the accumulation of CoA derivatives of organic acids, which inhibit the synthesis of N-acetylglutamate (NAG)—the essential allosteric activator of carbamoyl synthetase 1 (CPS1) in the liver.25,42 Impaired NAG formation consequently reduces CPS1 activity, leading to impaired ureagenesis and ammonia accumulation.25,42 For the treatment of hyperammonemia in OA, N-carbamylglutamine (carglumic acid), a structural analog to NAG synthetase, can serve as an obligate allosteric activator of CPS1. By restoring CPS1 activity, carglumic acid can facilitate ammonia detoxication. Similar to UCD, nitrogen scavengers are also effective for hyperammonemia in OA.18,19
Maple Syrup Urine Disorder (MSUD)
Maple syrup urine disorder is an autosomal recessive AA caused by a deficiency in the branched chain alpha-ketoacid dehydrogenase complex, which breaks down branched-chain amino acids (BCAAs) (Table 1).43,44 In patients with MSUD, impaired degradation of BCAAs (isoleucine, leucine, and valine) can cause accumulation of neurotoxic levels of leucine and coma. Without prompt treatment, acute metabolic decompensation in MSUD triggered by trauma or other factors can lead to cerebral edema and death due to the toxic accumulation of leucine.24,43
The primary goal of managing MSUD is to halt catabolism and promote anabolism to decrease dangerous leucine levels (Table 3).24,45 This is done by withholding natural protein to limit increases in BCAAs, especially leucine. It is critical to provide calories to prevent further catabolism by using dextrose-containing intravenous fluids (eg, D10 normal saline), intralipid, metabolic total parenteral nutrition, and specialized metabolic formulas with appropriate levels of BCAAs.24,45 Dangerously high leucine levels can be lowered rapidly though CRRT which requires timely nephrology consultation and placement of a vascular catheter for CRRT access.24,29
Mitochondrial Diseases (MitoD)
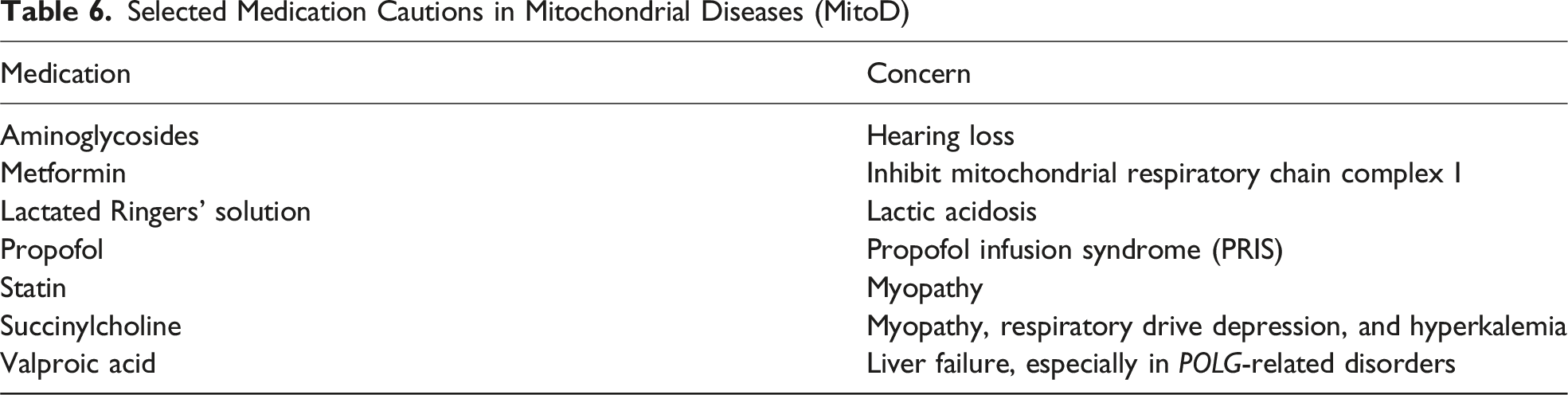
MitoD are a group of IMDs caused by mitochondrial dysfunction (Table 1). Because mitochondria are the “powerhouses” of the cell, generating cellular energy, patients with MitoD are unable to utilize energy efficiently, resulting in multisystemic manifestations. Lactic acidosis is common because impaired conversion of pyruvate to acetyl-CoA limits entry into the TCA cycle, leading to lactate accumulation and a shift toward anaerobic metabolism.26,27
Selected Medication Cautions in Mitochondrial Diseases (MitoD)
Useful Website Resources for Trauma and Surgical Providers
Management of metabolic emergencies in IMDs is complex, and trauma and surgical providers may have limited access to appropriate protocols. Immediate consultation with a metabolic specialist is essential to guide treatment. However, the following free online resources are valuable for understanding and reviewing the management of metabolic emergencies in IMDs.
New England Consortium of Metabolic Programs (https://www.newenglandconsortium.org/)
Established in 1998, this consortium aims to disseminate information on IMDs to health care professionals across all levels involved in treating individuals with IMDs.
47
Its projects include
GeneReviews (https://www.ncbi.nlm.nih.gov/books/NBK1116/)
It contains expert-authored, peer-reviewed online articles that describe specific inherited disorders. 48 Many IMDs are included with tables with acute metabolic decompensation in IMDs.
International Mito Patients (https://mitopatients.org/list-of-medicines/)
This group shares best practice, information, and knowledge on MitoD. 46 The website contains a list of medications that should be used with caution in MitoD.
OrphanAnaesthesia (https://www.orphananesthesia.eu/en/news.html)
This is an open-access database created in 2005 by the Scientific Working Group of Pediatric Anesthesia of the German Society of Anesthesiology and Intensive Care Medicine. 49 It provides peer-reviewed, freely available, and concise recommendations for anesthesiologists on how to plan and deliver anesthesia in patients with rare diseases, including IMDs.
Conclusion
This review highlights the importance of recognizing and managing IMDs in the setting of traumatic injuries and acute surgery. Raising awareness within trauma teams, along with a better understanding of the general principles of metabolic emergency management, is essential. Early recognition and timely intervention guided by a metabolic specialist and a coordinated, multidisciplinary approach are critical to optimizing outcomes for these patients.
Footnotes
Ethical Considerations
The relevant institutional review board waived the need for ethical approval.
Author Contributions
Conception and study design: YF and JAP. Literature review: YF, RJT, and JAP. Drafting of the manuscript: YF and JAP. Critical revision: All authors critically reviewed and accepted the manuscript submitted.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
