Abstract
Objectives
Endovascular Aortic Repair (EVAR) is the preferred treatment for symptomatic and asymptomatic abdominal aortic aneurysms (AAA) meeting anatomical criteria. This study compares outcomes of EVAR for symptomatic AAA (ruptured and non-ruptured) performed under general anesthesia (GA) vs. local anesthesia (LA).
Methods
Using the Society for Vascular Surgery Vascular Quality Initiative (SVS-VQI) registry (2003-2021), patients undergoing EVAR were divided into Group I (symptomatic, non-ruptured AAA) and Group II (ruptured AAA), then subdivided by anesthesia type: Ia (LA), Ib (GA), IIa (LA), IIb (GA). Primary outcomes were 30-day and in-hospital mortality, length of stay (LOS), and discharge disposition. Secondary outcomes included ICU stay and systemic complications.
Results
Among 8710 patients (Group I: n = 5310; Group II: n = 3400), LA was used in 6.2% of symptomatic, non-ruptured cases (Ia) and 13.4% of ruptured cases (IIa). Group Ia patients were older, White, Medicare-insured, and had higher CHF prevalence compared to group Ib. Group IIa patients had higher hospital transfer rates and family AAA history compared to IIb. In ruptured AAA, LA was associated with significantly lower mortality compared to GA (30-day: 12.3% vs 20.5%, OR 0.50, P < 0.001; in-hospital: 10.3% vs 19.1%, OR 0.42, P < 0.001). Secondary outcomes favored LA, including shorter ICU stay (3.6 ± 5.2 vs 4.9 ± 7.2 days), lower dysrhythmia (9.0% vs 12.4%), and stroke rates (0.9% vs 2.4%). High-volume hospitals performed more EVAR under LA for ruptured AAA.
Conclusions
For ruptured AAA, EVAR under LA is associated with significantly lower mortality and reduced complications, supporting its use particularly in high-volume centers.
Keywords
Introduction
The Society for Vascular Surgery (SVS) clinical practice guidelines recommend endovascular abdominal aortic aneurysm repair (EVAR) as the first-line treatment for both symptomatic and asymptomatic patients with abdominal aortic aneurysms (AAA) who meet the anatomic criteria for endovascular repair. 1 Symptomatic patients can present with either intact AAA or ruptured AAA. Operative mortality for patients with ruptured AAA continues to be high and previously published literature has clearly shown that EVAR performed under local anesthetic (LA) in these patients is associated with favorable outcomes as compared to general anesthesia (GA). 2 Patients presenting with symptomatic, but not ruptured, AAA present a unique challenge as many of them may have undetected underlying cardiovascular and pulmonary disease and there is not enough time to perform thorough preoperative risk factor assessment and medical risk factor stratification. Inducing general anesthesia and its association with perioperative hypotension may contribute to major adverse cardiovascular events in this patient population.
The perioperative mortality for any operation for ruptured AAA is a result of a two-hit process. The first hit is hypotension due to sudden loss of massive amounts of blood from the ruptured AAA. The second insult is the hypotension during the operation which activates an inflammatory cascade which results in multisystem organ failure and mortality. 3 General anesthesia may contribute to this two-hit phenomenon and worsen the systemic effects of multisystem organ failure and increase mortality. The second-hit phenomenon is believed to be worse in patients who are elderly and frail. 4 Local anesthesia does offer an alternative to GA for EVAR and avoids peripheral vasodilatory effects of GA and hence reduces the risk of perioperative hypotension associated with induction of GA. It is particularly an attractive option for patients who are elderly and frail.4,5 Patients who have been diagnosed with symptomatic AAA but have not ruptured their aneurysm are at a high risk for suffering from aneurysm rupture and dying if they are not treated expeditiously. The shorter time available for patient risk assessment, perioperative risk optimization and operative planning are a few of the factors associated with high mortality in symptomatic AAA patients. 6 This study investigates the impact of LA for EVAR, for both ruptured and symptomatic non-ruptured AAA’s, on mortality by analyzing the data from the Society for Vascular Surgery’s Vascular Quality Initiative (SVS-VQI) database.
Methods
Data Source
This retrospective cohort study utilized data from the Society for Vascular Surgery Vascular Quality Initiative (SVS-VQI) registry for EVAR, a prospectively maintained data set encompassing 14 major vascular surgery procedures from nearly 800 centers across the United States and Canada. The VQI database provides comprehensive information on patient demographics, procedural characteristics, and outcomes for patients from 2003 to 2021. The study was approved by Institutional Review Board (Penn State Health IRB No. 00025656).
Study Population
The study population of patients undergoing EVAR for symptomatic AAA was categorized into two primary groups based on the indication of EVAR: symptomatic, non-ruptured AAA (Group I) vs ruptured AAA (Group II). Each group was further subdivided into two sub-groups (a and b), based on the type of anesthesia used for the operation: local anesthesia (LA) (a), which involved local anesthetics with varying amounts of intravenous conscious sedation but no airway support, and general anesthesia (GA) (b), which involved the use of a laryngeal mask airway or intubation.
Inclusion criteria encompassed all patients undergoing EVAR during the study period, while patients who met exclusion criteria: missing urgency information, missing anesthesia information, those under 18 years old, surgical years 2020 and 2021 during the COVID pandemic, and those whose procedures were converted to open aortic repair, as it is assumed that all open AAA repairs are performed under GA were excluded (Figure 1). Study Population and Exclusion Criteria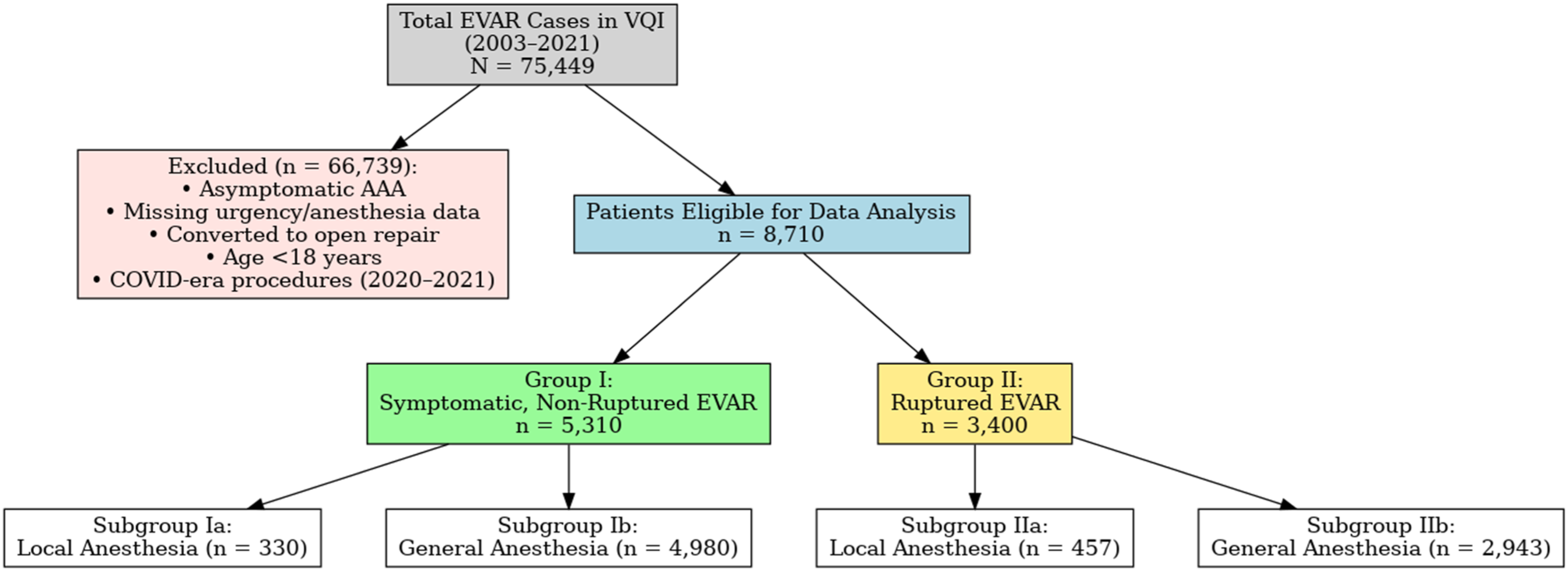
Patient Variables, Medical Center, and Surgeon Volumes
The study assessed and compared patient demographics, procedural characteristics, and outcomes between the two anesthesia groups within both groups. Variables analyzed included age, sex, transfer status, race, type of insurance, body mass index (BMI), living status, functional status, preoperative medications (such as aspirin, P2Y12 antagonists, statins, and others), and medical and surgical history (including hypertension, coronary artery disease, congestive heart failure, smoking history, diabetes, and various types of prior interventions). Additionally, hospital and surgeon volumes were stratified into low, medium, and high categories, based on case volume tertiles.
Outcomes
Outcomes of interest were categorized into primary and secondary outcomes. Primary outcomes included 30-day mortality, in-hospital mortality, hospital length of stay (LOS), and discharge destination (home vs non-home). Secondary outcomes consisted of ICU length of stay, myocardial infarction, dysrhythmia, congestive heart failure, stroke, Major Adverse Cardiovascular Events (MACE), on a ventilator, pneumonia, increase in baseline creatinine, and dialysis.
Statistical Analysis
Statistical analyses were performed using Stata 18/IC. Differences in categorical patient characteristics were evaluated using the Pearson chi-square test, while continuous variables were compared using the Welch test. Frequencies and percentages were reported for categorical variables and means, and standard deviations were provided for continuous variables. Statistical significance was set at a P-value of less than 0.05. For significant variables identified in univariate analysis for patient characteristics, hospital and surgeon volume, and study outcomes, multivariable-adjusted logistic regression was employed for ruptured EVAResults were presented as adjusted odds ratios and 95% confidence intervals.
Results
Patient Characteristics
Univariate Analysis of Preoperative Variables for Patients Undergoing EVAR for Ruptured and Non-ruptured Abdominal Aortic Aneurysms Under General and Local Anesthesia (GA vs LA)
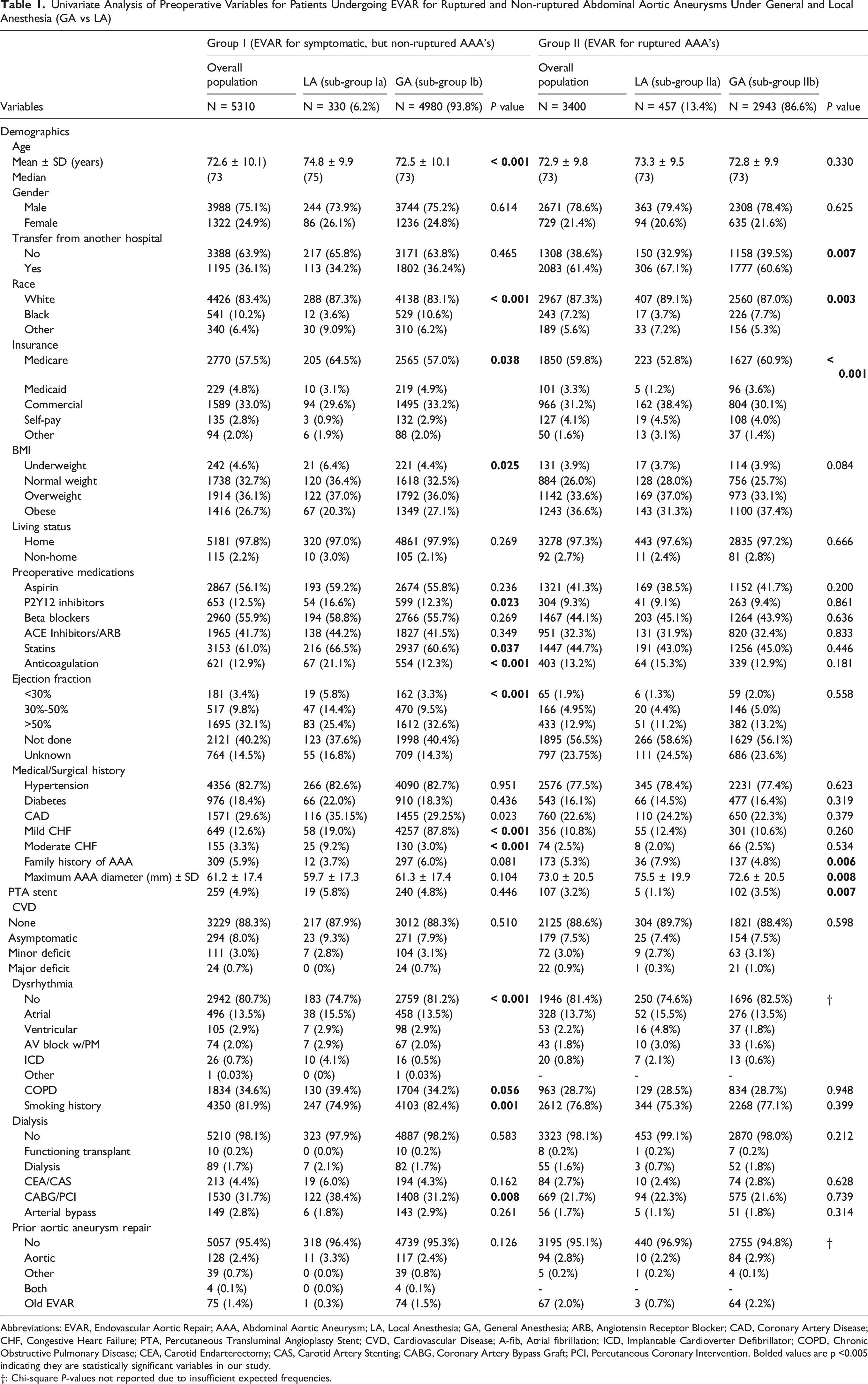
Abbreviations: EVAR, Endovascular Aortic Repair; AAA, Abdominal Aortic Aneurysm; LA, Local Anesthesia; GA, General Anesthesia; ARB, Angiotensin Receptor Blocker; CAD, Coronary Artery Disease; CHF, Congestive Heart Failure; PTA, Percutaneous Transluminal Angioplasty Stent; CVD, Cardiovascular Disease; A-fib, Atrial fibrillation; ICD, Implantable Cardioverter Defibrillator; COPD, Chronic Obstructive Pulmonary Disease; CEA, Carotid Endarterectomy; CAS, Carotid Artery Stenting; CABG, Coronary Artery Bypass Graft; PCI, Percutaneous Coronary Intervention. Bolded values are p <0.005 indicating they are statistically significant variables in our study.
†: Chi-square P-values not reported due to insufficient expected frequencies.
Symptomatic Non-ruptured AAA
For Group I patients (EVAR for symptomatic, non-ruptured AAA’s), following variables were found to be statistically significant between two sub-groups (Sub-Group Ia: LA vs Sub-Group IIa: GA): mean age (74.8 ± 9.9 yrs vs 72.5 ± 10.1 yrs), Black race (3.6% vs 10.6%), Medicare insurance status (64.5% vs 57%), obesity (20.3% vs 27.1%), P2Y12 inhibitors (16.6% vs 12.3%), statin use (66.5% vs 60.6%), anticoagulation (21.1% vs 12.3%), ejection fraction <30% (5.8% vs 3.3%), mild CHF (19% vs 87.8%), moderate CHF (9.2% vs 3%), no dysrhythmia (74.7% vs 81.2%), COPD (39.4% vs 34.2%), smoking history (74.9% vs 82.4%) and history of CABG/PCI (38.4% vs 31.2%).
Ruptured AAA
For Group II patients (EVAR for ruptured AAA’s), following variables were found to be statistically significant between two sub-groups (Sub-Group IIa: LA, Sub-Group IIb: GA): transfer from another hospital (Sub-Group IIa: 67.1% vs Sub-Group IIb: 60.6%), Black race (Sub-Group IIa: 3.7% vs Sub-Group IIb: 7.7%), Medicare insurance (Sub-Group IIa: 52.8% vs Sub-Group IIb: 60.9%), family history of AAA (Sub-Group IIa: 7.9% vs Sub-Group IIB: 4.8%), maximum aortic diameter (Sub-Group IIa: 75.5 ± 19.9 mm vs Sub-Group IIb: 72.6 ± 20.5 mm), PTA stent (Sub-Group IIa: 1.1% vs Sub-Group IIb: 3.5%) and no dysrhythmia (Sub-Group IIa: 74.6% vs Sub-Group IIb: 82.5%).
Hospital and Surgeon Volume
Univariate Analysis of Hospital Volume and Surgeon Volume for Patients Undergoing EVAR Under Local Anesthesia Versus General Anesthesia
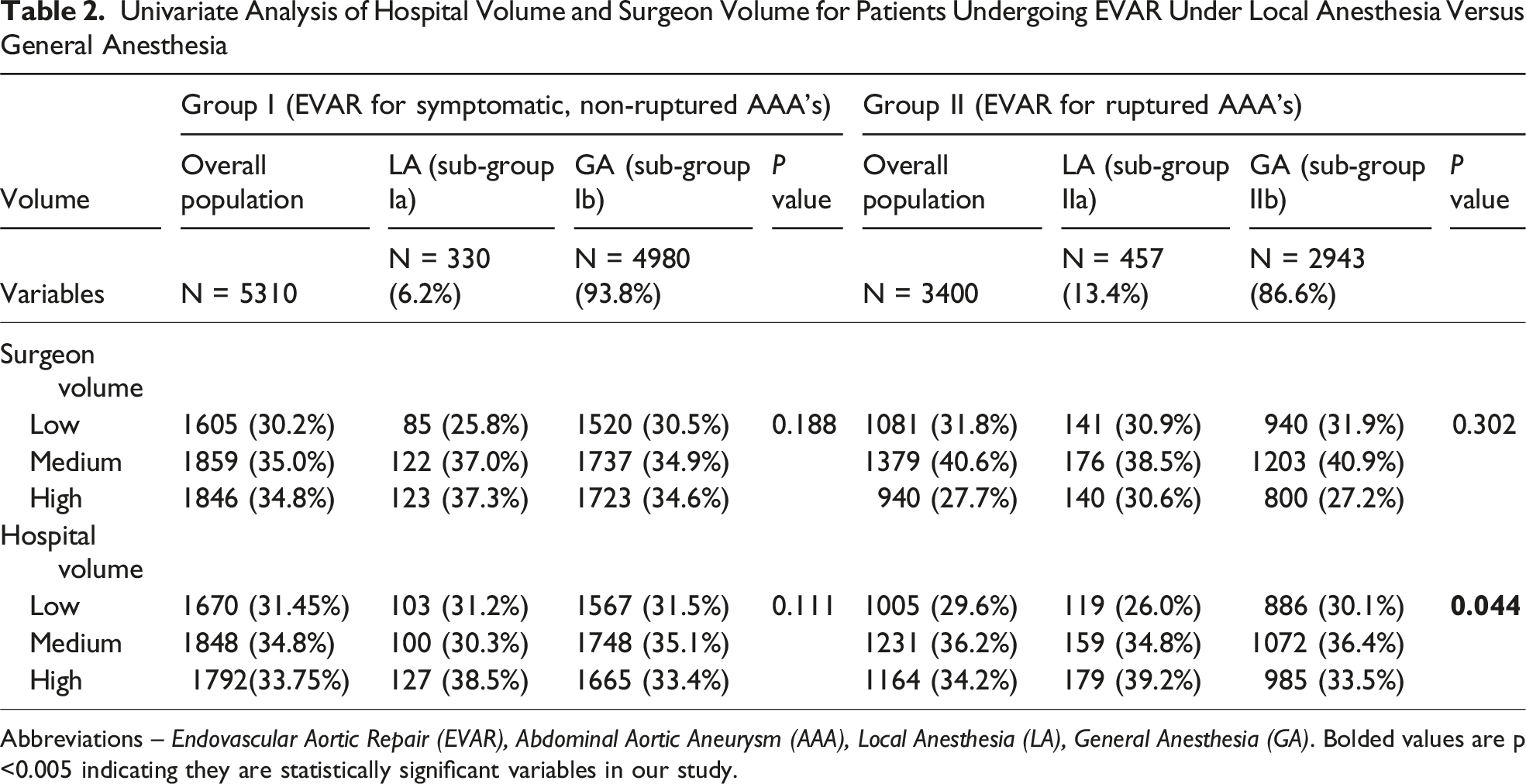
Abbreviations – Endovascular Aortic Repair (EVAR), Abdominal Aortic Aneurysm (AAA), Local Anesthesia (LA), General Anesthesia (GA). Bolded values are p <0.005 indicating they are statistically significant variables in our study.
Primary and Secondary Study Outcomes
There was no difference in mortality for patients undergoing EVAR under GA vs LA for symptomatic, non-ruptured AAA operations. Patients undergoing EVAR for ruptured AAA under GA (Sub-Group IIb) had an increased 30-day mortality as compared to those patients who underwent EVAR for ruptured AAA under LA (Sub-Group IIa): (20.5% vs 12.3%; P < .001) (Figure 2). Similarly, in-hospital mortality was also higher for GA (Sub-Group IIb) as compared to (Sub-Group IIa) (19.1% vs 10.3%, P < .001). Patients undergoing EVAR for ruptured AAA under LA (Sub-Group IIa) were more likely to be discharged to non-home locations (70.2%) vs those undergoing EVAR under GA (Sub-Group IIb) (65.3%; P = 0.051). As compared to patients undergoing EVAR for ruptured AAA under GA (Sub-Group IIb), those undergoing EVAR for ruptured AAA under LA (Sub-Group IIa) had lower length of ICU stay (3.6 days vs 4.9 days) and lower incidence of dysrhythmias (9% vs 12.4%), stroke (0.9% vs 2.4%) and ventilator associated pneumonia (8.4% vs 13.8%) (all P < .0.5). Secondary outcomes are summarized in Table 3. Kaplan-Meier Survival Curves for patients Undergoing EVAR for Ruptured AAA with local Anesthesia (red) or general Anesthesia (Blue). Log-rank Test Shows Significant difference Between the study Groups (P < .001) Univariate Analysis of Postoperative Outcomes for Patients Undergoing LA and GA During EVAR Abbreviations– LA, Local Anesthesia; GA, General Anesthesia; LOS, Length of Stay, SD, Standard Deviation, CHF, Congestive Heart Failure, MACE, Major Adverse Cardiovascular Event. Bolded values are p <0.005 indicating they are statistically significant variables in our study.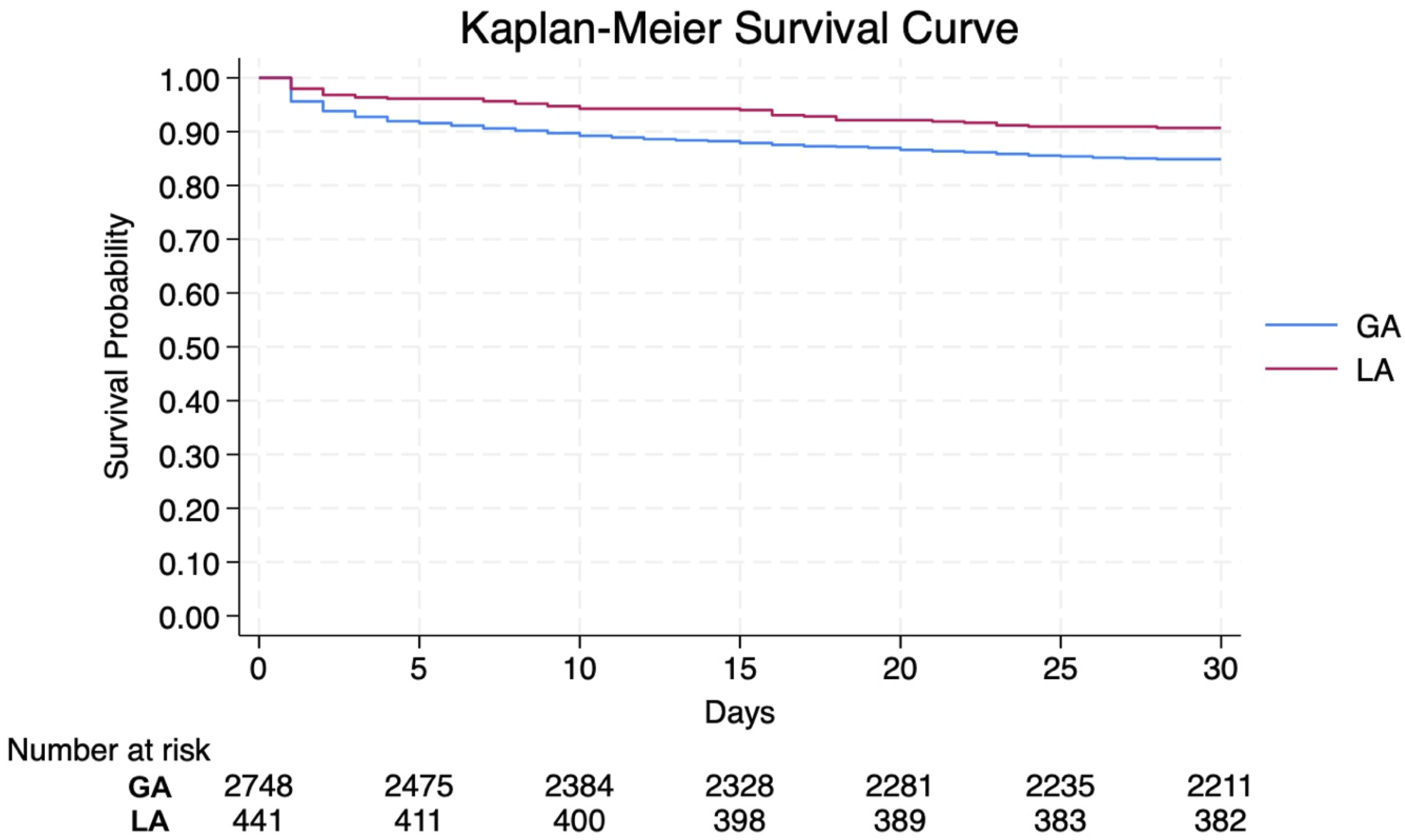
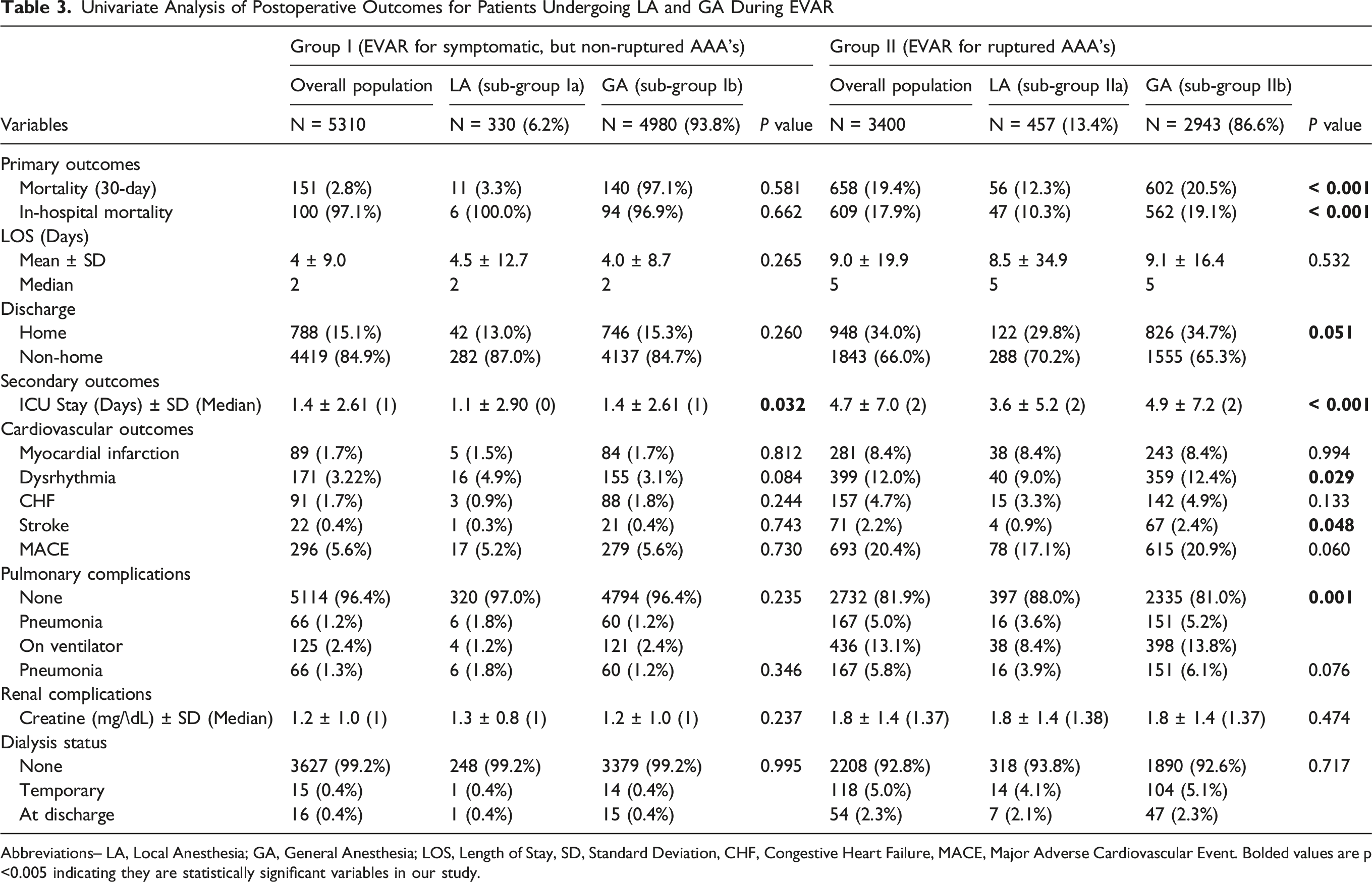
Risk-Adjusted Multivariable Sub-analysis of In-Hospital Mortality, 30-Day Mortality and Home Discharge for Patients Undergoing EVAR for Ruptured AAA under LA
Risk-Adjusted Multivariable Analysis for In-Hospital Mortality, Home Discharge, and 30-Day Mortality for Patients Undergoing EVARs for Ruptured AAA Under Local Anesthesia
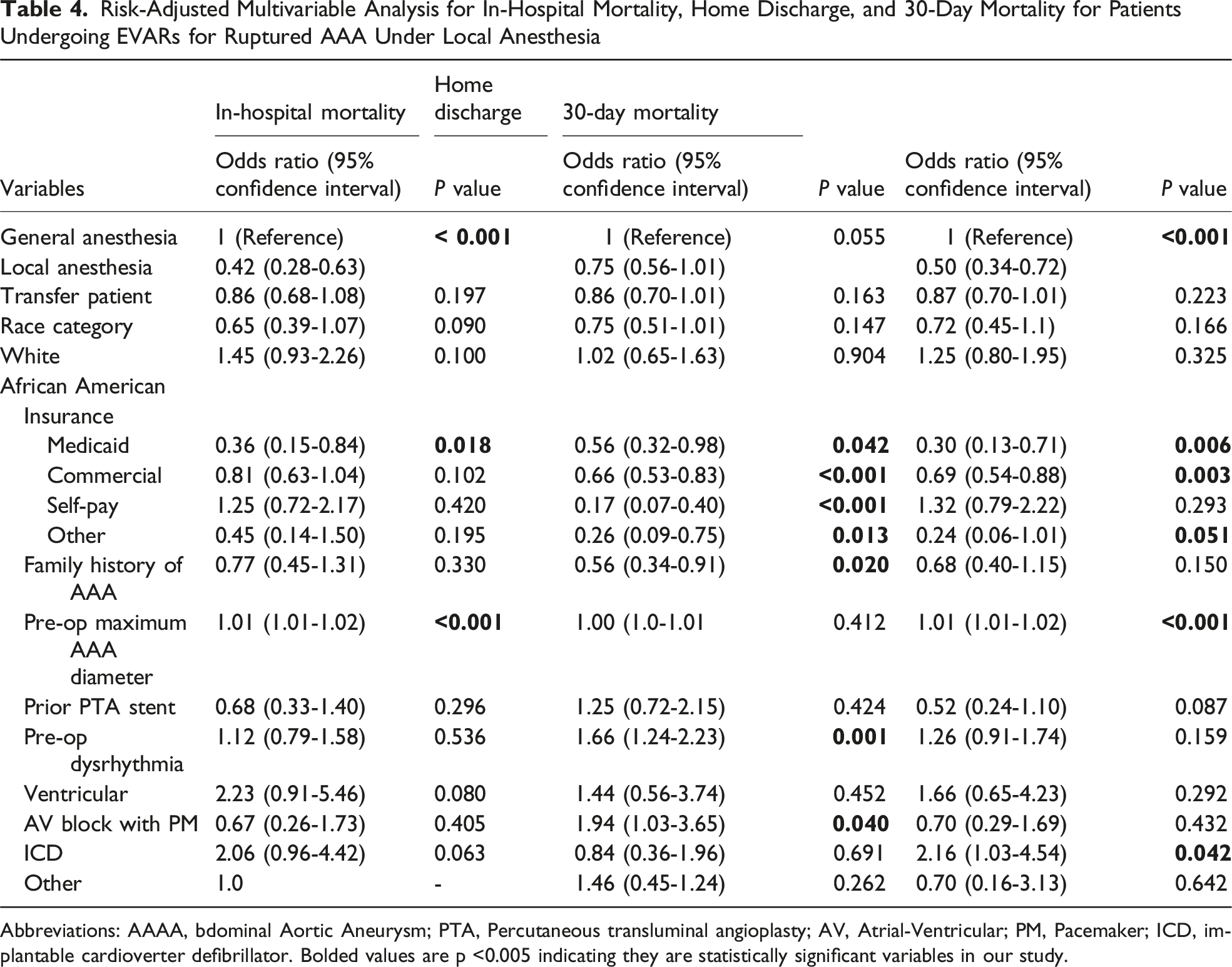
Abbreviations: AAAA, bdominal Aortic Aneurysm; PTA, Percutaneous transluminal angioplasty; AV, Atrial-Ventricular; PM, Pacemaker; ICD, implantable cardioverter defibrillator. Bolded values are p <0.005 indicating they are statistically significant variables in our study.
Discussion
This retrospective analysis of a large national database evaluates the impact of anesthesia type on outcomes in EVAR for symptomatic AAAs, comparing symptomatic, non-ruptured and ruptured cases. We first assessed patient characteristics for those receiving LA or GA as well as if hospital volume was associated with anesthesia type. Patients undergoing EVAR under LA were often older and more medically complex, hwile LA was more commonly used in high-volume centers. Importantly, ruptured AAAs treated under LA had significantly decreased 30-day mortality and in-hospital mortality than those under GA.. Multivariable analysis further confirmed GA’s association with elevated mortality, highlighting anesthesia choice as a critical factor in EVAR outcomes for ruptured AAAs.
The benefits of LA compared to GA have been extensively discussed in literature, particularly in comparison of patient outcomes.7,8 LA has been shown to be a safe alternative to GA for EVAR, especially among elderly and frail patients. 5 Studies have shown that prolonged EVAR procedures performed under LA result in reduced cardiac complications, primarily through decreased rates of MI. 9 Use of LA has also been associated with shorter operative times and decreased LOS compared to GA. 10 Furthermore, LA has shown decreased mortality in emergent EVAR cases, and shorter hospital and ICU length of stays with decreased vasopressor requirements and improved hemodynamic stability postoperatively.7,8 These findings align with the findings of the current analysis, highlighting the perioperative benefits offered by LA, particularly in high-risk patients undergoing EVAR. By avoiding intubation and mechanical ventilation, LA can reduce the risk of postoperative complications such as pneumonia and prolonged ventilatory support. The shorter ICU stays observed in patients under LA suggests a faster recovery time with fewer postoperative complications in those that have more preoperative risk factors. While past studies have largely underscored the use of LA in symptomatic, non-ruptured and ruptured EVAR cases, this analysis is unique, as it specifically analyzes only those patients who presented with symptomatic AAAs and compares the outcomes of patients with both ruptured and non-ruptured AAA’s. The findings that for patients presenting with symptomatic but non-ruptured AAAs, those undergoing the procedure under LA were more likely to be older, have ejection fraction of <50% and have moderate CHF suggest that these patients were considered high risk for developing perioperative morbidity and mortality and due to lack of time needed for a thorough preoperative assessment, were most likely deemed too high risk to proceed with surgery under GA.
Overall, patients undergoing EVAR under LA for symptomatic EVAR had lower morbidity and mortality when compared to those who underwent EVAR under GA is reassuring that despite being considered high risk, undergoing EVAR under LA was associated with favorable morbidity and mortality.
The analysis finds that the patients undergoing EVAR to treat ruptured AAAs were more likely to undergo the procedure under LA, if it was performed at a high-volume hospital. Of all EVARs for ruptured AAAs, 39% were performed under LA at the high-volume centers, reflecting availability of resources and the comfort level of surgical teams in busier health centers. A similar trend was observed in the retrospective analysis of the UK National Vascular Registry which showed that the centers with high volume of aortic surgery were more likely to offer EVAR under LA for patients presenting with ruptured AAA’s. 11 Phillips et al. assessed the open and EVAR outcomes reported in the literature from European countries concluded an inverse relationship between the hospital volume of aortic surgery and the postoperative outcomes. 12 On the contrary, this finding was not consistent when studied on Medicare beneficiaries in the US, which showed only significant association of hospital volume with postoperative mortality after open AAA repair but minimal relationship in EVAR patients. 13 The same analysis also showed that the hospitals which perform less than 9 EVARs in a year have the highest incidence of postoperative complications. The results from the current analysis align with the findings of UK National Vascular Registry and the systematic review by Phillips et al. suggesting that patients undergoing EVAR for ruptured AAA were more likely to undergo the procedure under LA if performed at a high-volume aortic center. High-volume aortic centers generally have large number of resources, and the teams likely have high level of comfort in performing these procedures under LA. Although no specific minimal number of EVAR cases has been universally agreed upon to achieve acceptable perioperative mortality rates, it seems likely that the teams at centers with high aortic volumes will continue to have increased degree of comfort in performing these procedures under LA and having improved postoperative outcomes.
This study has inherent limitations due to its retrospective design, which introduces potential biases. Although adjustments were made for various confounders through multivariate analyses, unidentified confounding variables may still influence the results. The VQI registry, while comprehensive, is still limited by its predetermined variables, restricting the scope of analysis. Secondary procedures such as laparotomy for abdominal compartment syndrome after the index EVAR is not available, therefore limits delineating factors associated with morbidity and mortality in the ruptured AAA group. In addition, pre-operative hemodynamics is not captured for our study groups and is an important limitation because it could bias anesthesia selection. It is known that EVAR performed off the indications of use (IFU) do worse than on-IFU and may be a pre-operative risk factor. Although, VQI captures some anatomic fields such as maximum aortic diameter, we did not encode IFU compliance in our study groups and there for a limitation IFU in complications. Selection bias is also possible, given that the VQI is a self-reported database. Therefore, as with most database-driven studies, these findings should be interpreted cautiously, with clinical judgment guiding real-world application. Despite these limitations, this study’s strengths are significant. Leveraging a large, diverse sample from the VQI database, it includes a comprehensive array of pre- and postoperative variables, enhancing the generalizability of the findings to a broader patient population. The VQI database’s accessibility to most surgeons and hospitals for evaluating outcomes and risk factors in vascular procedures bolsters the robustness of this analysis. Notably, the observed associations between local anesthesia use and improved outcomes in ruptured EVAR cases contribute valuable insights to the ongoing discussion on optimal anesthetic strategies in vascular surgery.
Conclusions
LA is associated with reduced 30-day and in-hospital mortality, improved systemic outcomes, and shorter ICU stays compared to GA in patients undergoing EVAR for ruptured AAA. Mortality was significantly lower in the LA group both at 30 days and in-hospital. Additionally, LA was linked to a reduced incidence of stroke and decreased ventilator support in ruptured AAAs. However, no significant differences were observed between the two anesthesia types for symptomatic, non-ruptured AAAs. These findings highlight important modifiable and non-modifiable risk factors in EVAR for ruptured and non-ruptured AAAs, suggesting that patient selection based on anesthesia type could improve outcomes in symptomatic AAA cases. These results can contribute to a patient selective algorithm to decrease adverse outcomes for patients undergoing EVAR for symptomatic AAA’s.
Footnotes
Author Note
Faizaan Aziz is a Sophomore at University of Michigan, Ann Arbor.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
